Abstract
Background Potentially inappropriate medications (PIMs) are often found in high proportion among the elderly population. The STOPP criteria have been suggested to detect more PIMs in European elderly than the Beers criteria. Objective This study aimed to determine the prevalence of PIMs and potential prescribing omissions (PPOs) in a sample of Portuguese nursing homes residents. Setting Four elderly facilities in mainland Portugal Method A descriptive cross-sectional study was used. Elderly polypharmacy patients were included in the study and their medication (registered in patient clinical records) analysed using the Beers (2012 original version and 2008 version adapted to Portugal), STOPP (Screening Tool of Older Person’s Prescriptions) and START (Screening Tool to Alert doctors to Right Treatment) criteria. Data were analysed using univariate and bivariate descriptive statistics, considering a confidence interval of 95 %. Main outcome measures: Prevalence of PIMs and PPOs. Results The sample included 161 individuals, with a mean age of 84.7 years (SD = 6.35), 68.9 % being female. A total of 807 PIMs and 90 PPOs were identified through the application of the three set of criteria. The prevalence of PIMs using the most recent version of the Beers criteria was 85.1 and 42.1 % for independent and dependent of diagnosis, respectively. The Portuguese adaptation of this same tool indicated a lower prevalence of PIMs, 60.3 and 16.7 %, respectively. The prevalence of PIMs using the STOPP criteria was 75.4 %, whilst the prevalence of PPOs, using START, was 42.9 %. There were significant differences in the mean number of PIMs detected depending on the tool used. (p < 0.001). Conclusions The application of the studied criteria in an elderly sample enabled the identification of a notable amount of PIMs and PPOs, indicating there is room for improving the quality of care. The variation in prevalence indicates careful choice of the tool is a prerequisite for engaging in medication review. Using START/STOPP criteria enabled a more holistic approach to the quality of prescribing in the elderly, highlighting low levels of cardiovascular risk prevention and abuse of psychotropic drugs, aside with system failures largely preventable by electronic prescribing and alert generation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Impacts on Practice
-
The prescription pattern in Portuguese polypharmacy elderly highlights low levels of cardiovascular risk prevention and abuse of psychotropic drugs, suggesting there is room for improving the quality of pharmacotherapy.
-
Pharmacists are competent at reviewing medication. Therefore, nursing homes could benefit from the integration of a pharmacotherapy specialist in the team, a role typically to be assigned to the pharmacist.
-
Pharmacists should be aware that the tool chosen to engage in medication review largely impacts on results obtained.
Introduction
For decades the Portuguese population has been ageing rapidly. This phenomenon is characterized by a decline in birth and death rates and more recently aggravated by the emigration of the younger population [1].
The elderly, currently representing 20 % of the population of Portugal, have often demonstrated a high comorbidity index, frequently leading to polypharmacy, a good predictor of adverse drug reactions [2, 3] leading to an increase in hospitalizations and, in some cases, death. Polypharmacy is also known to lead to the use of potentially inappropriate medications (PIMs) [4–7]. In an effort to guide pharmacists in the detection of such problems, explicit criteria for medication review has been developed, making use of several screening tools. The Beers criteria is the most widely known and used in a number of scientific publications [8–10].
The Beers criteria was initially developed by the American Geriatrics Society in 1991 and was the first explicit criteria for PIMs detection to be published [11]. Since then, over 500 observational studies have been conducted worldwide making use of the criteria to identify PIMs. The original list has undergone four revisions, the latest in 2015 [11–15]. The 2012 version had a total of 53 medications organized according to physiological systems and pharmacotherapeutic classes.
Since Beers criteria was developed to accommodate the US health system, its applicability to European countries is quite limited. Focusing on the available medications in Portugal, Soares and colleagues have adapted the 2003 Beers criteria to Portuguese health care standards, enabling its use in the Portuguese population [16]. The adaptation process focused on the comparative analysis of PIM identified by Beers and medications available in the Portuguese market, which has led to the removal of 34 medications from the initial list because they were unavailable to the public. Conversely, specifications on substances available in the Portuguese market according to the National Formulary were made, whenever the original tool described the pharmacotherapeutic class only, in a vague or generalized manner (e.g. aceclofenac in the class NSAIDs). However, to our knowledge, this criteria has not been implemented in Portugal.
Recently, a European based tool has been developed, the STOPP (Screening Tool of Older Person’s Prescriptions) and START (Screening Tool to Alert Doctors to Right Treatment) criteria and an increasing number of studies using this tool have been undertaken in various settings [3, 17–19]. This tool has been valued for its simplicity and systematization of PIMs and consideration of omitted medications for achieving the optimal therapy. The STOPP criteria, based on physiological systems, consist of eight categories that correspond to 65 items associated to PIMs, whereas START criteria consist of 22 items that are relative to potential prescribing omissions (PPOs); the latter has also been subdivided into physiological systems. Several studies have indicated that the STOPP criteria, compared to the Beers criteria, make the PIM identification process easier and have shown greater efficacy in identifying PIMs [4]. The STOPP–START tool has already been adapted in a number of European countries but not in Portugal [20–22]. Adaptation of tools is considered important because, even within Europe, where centralised processes of medicine approval exist, the availability of medications in national markets depends heavily on coverage schemes set by national regulatory agencies. Additionally, the adaptation process should also ensure that the names of medications are easily recognised (e.g. paracetamol or acetaminophen) and that issued lists do not contradict existing prescribing guidelines.
Aim of the study
The aims of the present study were to assess and compare the prevalence of potentially inappropriate medications and potential prescribing omissions detected in a sample of Portuguese nursing home residents using three different tools. The main hypothesis was that there would be differences in the prevalence detected depending on the tool used. Additionally, we hypothesised that our sample would have results (using one of the international tools) comparable to studies undertaken in similar settings.
Ethics approval
Analysis of patient medical records was authorized by the clinical directors. Ethics committee approval was obtained from “Comissão de Ética para a Investigação nas Áreas de Saúde Humana e Bem-Estar da Universidade de Évora”.
Method
A descriptive cross-sectional study was used, where a convenience sample of four nursing homes were invited to participate. Two of them were located in the region of Lisboa and Vale do Tejo and the others in the region of Alentejo.
Population and sample
The population (n = 224) considered for the study were the residents in these four facilities meeting the eligibility criteria defined as follows:
Inclusion criteria: elderly residents (aged ≥ 65); on polypharmacy (using ≥ 5 medicines).
Exclusion criteria: living temporarily in the facility (predicted stay ≤ 3 months); residents with total independence (outpatients, i.e., people who use the nursing home as an adult day-care center).
Data collection
Data was collected for three consecutive months in 2014. Variables collected from the patients’ medical record included: socio-demographics, diagnoses, past medical history, laboratory results, biomarkers taken at the facility and instituted therapy (active ingredient, index date and daily dosage). Whenever information was missing or was unclear, the responsible professional care giver (doctor or nurse) was contacted.
Screening tools used
Medication review was initially conducted by two practising pharmacists and then checked by two clinical pharmacists and one general practitioner. Three tools were used to guide the process: the 2012 American version of the Beers criteria [14], the Portuguese adapted Beers criteria [16] and the original STOPP and START criteria [3]. Both versions of the Beers criteria target potentially inappropriate medication independent of the diagnosis (PIMs-ID) and dependent of diagnosis (referred as “considering diagnosis”, PIMs-CD), which were analysed and described separately. The STOPP criteria may only be applicable when the diagnoses are known. Therefore, whenever the completeness of patients’ clinical record was considered insufficient to accurately classify diagnoses, patients were excluded from sub-analysis focusing on PIMs-CD.
Statistical analysis
Data was analysed in SPSS, version 21, using univariate and bivariate descriptive statistics (Chi square, Wilcoxon and Friedman Tests), considering a significance level of 5 %. To determine the prevalence of PIMs, the following formula was used:
During data analysis one of the facilities presented a fair amount of incomplete clinical records, although medication charts were considered exhaustive. Therefore, the authors only used this facility to apply criteria independent of diagnosis (PIMs-ID) and it was excluded for all remaining analysis.
Results
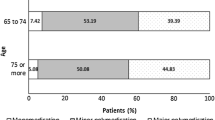
All four nursing homes invited agreed to participate in the study. From the initial 224 patients, 63 patients were excluded following the application process of the eligibility criteria. The final sample consisted of 161 polypharmacy elderly with a mean age of 84.7 years (SD = 6.35; {65–98}), where women prevailed over men (n = 111; 68.9 %). The total number of medications prescribed and analysed in the overall sample was 1618. The mean number of medications taken by the patients in the study was 10.1 (SD = 3.89), ranging from 5 to 28. Daily number of doses varied from 2 to 44 (M = 12.3; SD = 5.76) and patients presented a mean of 4.1 comorbidities (SD = 2.14).
PIMs identified by the 2012 Beers’ criteria
The application of the original Beers’ criteria identified 256 medications considered as PIMs-ID, representing 15.8 % of the total medications being used (n = 1618). Prior to the application of the Beers criteria considering diagnosis, 35 patients were excluded from the analysis; therefore the analysis pertains to 126 patients and 1315 medications when referring to PIMs-CD. In this category, this same tool detected 145 PIMs-CD, representing 9.0 % of the medications used (n = 1315). The number of patients having one or more PIMs was 137 and 53, respectively for PIMs-ID and PIMs-CD, corresponding to 85.0 and 30.6 % of the patients analysed. The majority of patients presented one or two PIMs-ID and PIMs-CD (Table 1).
The medications most commonly found when using the original Beers’ criteria were short-acting benzodiazepines in high dosages, long-acting benzodiazepines, and anti-psychotics independent of diagnosis. When applying Beers’ criteria while considering the patient diagnosis, medication potentially leading to patient falls or injuries were the most commonly found, immediately followed by those potentially aggravating cognitive impairment (Table 2). In short, medication acting on the central nervous system (CNS) were responsible for the majority of PIMs found in both cases.
PIMs identified by the Portuguese version of the Beers’ criteria
Application of the Portuguese version of the Beers’ criteria to the same sample identified a considerably smaller number of PIMs. Among the 1618 medications analysed, 158 were PIMs-ID (9.8 %) and only 34 PIMs-CD (2.6 %). As with the original tool, the vast majority of patients presented only one or two PIMs (Table 1). The most commonly found PIMs-ID were short-acting benzodiazepines, laxatives, muscle relaxants and first generation anti-histaminic medications. However, when considering the diagnosis, results were more in-line with those found in the original version, with the majority of problems resulting from the use of medication acting on the CNS as well as medication inappropriate for depressed patients (Table 2).
PIMs identified by STOPP criteria
The STOPP criteria does not consider the inappropriateness of medications independent of diagnosis. Therefore, the results found using this tool can only be compared to those “considering diagnosis” of both versions of Beer’s criteria. The STOPP criteria led to the identification of 214 PIMs among the medications analysed (16.3 %), which was considerably higher than those found by the other tools. Three quarters of patients had one or more PIMs according to these criteria (n = 95; 75.4 %). Again, most patients presented either one or two PIMs (Table 1). The most commonly found PIMs that accounted for 45.8 % of the total PIM, were duplicate pharmacotherapeutic classes where antidepressants, benzodiazepines and laxatives played an important part. A notable proportion of PIMs (18.7 %) were also attributable to medications targeting the central nervous system and psychotropic medications followed by those that adversely affected the stability or mobility of the patients and cardiovascular system medications (Table 3).
When comparing the median number of PIMs using the three different tools, we found a significant difference in their ability of detection (p < 0.001). This finding included the independent of diagnosis tools (i.e., comparing the two versions of Beers criteria) and the considering diagnosis tools, (i.e., comparing the three tools—Table 1).
Potential prescribing omissions identified by START criteria
The application of the START criteria allowed the identification of 90 PPOs, corresponding to 6.8 % of medications analysed. These PPOs occurred in 54 patients representing 42.9 % of the analysed sample. The majority of the sample presented only one PPO (57.4 %) (Table 1).
The more common prescribing omissions were detected in the endocrine system medications (n = 32; 35.6 %), attributed to the absence of antiplatelet therapy, followed by the absence of statin therapy. Omissions in the musculoskeletal system medications were also common (n = 22; 24.4 %), attributed to the absence of bisphosphonates, calcium and vitamin D. The third most common prescribing omission derived from the cardiovascular system, again with the absence of statin therapy playing an important role (Table 3).
Discussion
The main findings of the study indicate that among polypharmacy elderly a notable proportion of potentially inappropriate medications are used. According to the screening tools used, the prevalence of PIMs varies significantly. Analysing medications independently of medical diagnosis and the updated Beers criteria resulted in significantly more PIMs detected in comparison with the Portuguese adaptation of a former version of this tool. Previous research has identified the 2012 Beers criteria as the tool resulting in the detection of more PIMs, which is consistent with our results [23]. However, assuming the Portuguese version to be more valid to the sample under analysis, the findings that over 60.0 % of patients had one or more PIMs among their daily medication is worrisome. These values were two times higher than those previously reported in Portugal, albeit in the outpatient setting [24].
When considering diagnosis, the STOPP criteria resulted in the identification of an even higher number of PIMs, as previously reported [4, 25]. The difference detected between tools led us to confirm our hypothesis and conclude that the tool does in fact matter.
It is important to define prevalence when such studies are undertaken because this concept may be interpreted differently, resulting in enormous differences in the literature. In the present study, the prevalence was estimated when considering patients with one or more PIMs as the unit of analysis. However, if one considers medications as the unit of analysis, this should be labelled a proportion and not prevalence, although it has been referred to as a prevalence at the drug level [25]. Using the estimate of the 2012 Beers criteria, the present study identified PIMs in 85.0 % of patients. However, the proportion of medications considered as PIMs in the sample of medications analysed was 15.8 %. Likewise, using the STOPP criteria, the proportion of medication identified as PIMs was 16.3 % which is similar to the previous reports where the authors have made a distinction of concepts [25]. The lowest proportion was detected when applying the Portuguese version of the Beers criteria (2.6 %), with which no comparisons were possible since there are no previous studies using this tool.
In general, the proportions of PIMs reported in this study are higher than expected, regardless of the screening tool used. This finding led us to discard our hypothesis that we would find results similar to previous studies.
While many studies focus on the detection of PIMs reporting their prevalence, perhaps more important is the detailed information that will enable clinicians to implement a quality control regimen for their prescribing pattern adopted for this particular group of vulnerable patients. The inclusion of the START criteria in this study was determinant to achieve this goal. In fact, analysing all screening tools used, it is apparent that the prescribing pattern is consistent with national periodical reports issued by the Health Ministry. In fact, the Portuguese population has been described as needing to increase lipid lowering medications to prevent cardiovascular disease, a major cause of morbidity and mortality in Portugal [26]. Such has been verified in the present study, where the START criteria predominantly identified low cardiovascular risk prevention, characterised by the missing statin and antiplatelet therapy. Another important group missing in this particular tool was the musculoskeletal system medications. The bisphosphonates consumption in the overall Portuguese population has been increasing, unlike calcium and vitamin D supplements, which per se has not led to a decrease in hip fractures [27]. Conversely, CNS medicines, namely benzodiazepines and medications that adversely affect the fallers, were among the top 5 PIMs, having potential consequences on the frail elderly. Another aggravating factor was the detection of duplicate drug classes at the top of the list, which was mainly represented by benzodiazepines and antidepressants. The average consumption of these two drug classes in Portugal is higher than in most European countries [28].
The choice of the screening tool does matter
Our results clearly show that, depending on the tool chosen, the proportion of PIMs detected vary widely. [4] The main reasons are that the rational for tool development may differ and the years they were issued may also vary. As such, the Portuguese Beers criteria considers laxatives as a PIM, a class not considered in the other screening tools, whereas the American Beers criteria, considers antipsychotics as a PIM, a class considered in STOPP but not in the Portuguese Beers criteria. Additionally, the STOPP criteria considers additional classes not based on systems, such as “duplication of therapy” or “medication that adversely affect people that are prone to falling”. This criteria may be seen as relevant when considering the case of less developed countries, where essentially administrative errors are not prevented by the use of online prescribing or other alternative safety measures, leading to undetected duplications, which is probably the reason why our results are so high using STOPP. However, the use of these categories also leads to categories not mutually exclusive, which results in the same mistake being quantified twice, an incorrect procedure in our view.
The original Beers criteria emerged from a need to improve patient care, particularly in nursing homes. This tool has later been used in the context of professional development [29], and has also been adapted to become part of the quality indicators for assessing nursing homes [30]. The National Committee for Quality Assurance then adapted the concept of PIM into high-risk medications so that it could become part of HEDIS (Healthcare Effectiveness Data and Information Set), used to measure performance and quality of care. In Portugal, as in many other countries, continuity of patient care is a concern and policy measures such as these could be used to foster practice improvement and ultimately result in a better and safer quality of life for the patient.
However, an issue with all these tools is that they have been described as valid [14, 31], but their validation depends mostly on literature review followed by consensus development processes and can therefore be questioned. There seems to be some awareness of these limitations, judging by the frequently issued updates of these screening tools; others have opted to develop new criteria from scratch [32, 33]. Validation is a complex process, which involves ensuring various psychometric properties [34], including the ability to predict real harm to the patient by the medicine prescribed [35] and agreement with other external measures [36]. To our knowledge, this has never been fully achieved, perhaps because validation must follow development and revisions emerge prior to the final validation process is complete.
Strengths and weaknesses
This study is important for various reasons. The most important is perhaps the fact that it makes a detailed comparison of the findings using three separate screening tools and demonstrating that results are affected by the measurement chosen, with obvious practice implications. For instance, using the American version of Beers criteria, our results highlight the use of antipsychotics, whereas when we consider the Portuguese version, laxatives appear to be the most predominant; when using STOPP/START criteria, duplications were the most frequent, suggesting safety measures are lacking in these facilities. Secondly, the study was the first to test a worldwide screening tool adapted for Portugal, making results more robust, indicating that validation of originally American or English tools should be a prerequisite for all non-English-speaking countries engaging in medication review. The drawback is that a Portuguese version of STOPP/START criteria is not yet available which has lead us to use the original version. Moreover, it includes a target patient sample that is growing in Portugal and indeed in many countries worldwide, where efforts should be focused on the quality of care. We believe this work provides an important contribution to the development of stronger bonds between health care professionals that are involved with elderly and their medications. This study was also able to demonstrate the advantage of using various tools when engaging in medication review and it became clear that one should also consider missing medication therapy in addition to PIMs. This enabled a global picture of the quality of medication therapy that when applied to individual patient care may help guide future interventions.
Nonetheless, there were some limitations in this study, namely the inability to overcome the number of incomplete medical records in one of the participating facilities. However, the way the analysis was undertaken minimised this problem, and was considered a better approach than restricting analysis to part of the criteria, as reported elsewhere [31]. It can be considered an ethical dilemma to detect these PIMs and PPOs and not act in response to the findings. Therefore, the situations and findings that were brought to light during our data gathering that we considered clinically relevant have been reported. The drawback was that no feedback was requested, making quantification of eventual changes impossible. Nonetheless, this study was not designed to have an intervention for which the impact ought to be measured.
Conclusion
The prevalence of PIMs in the studied sample was relatively high, regardless of the tool used, but with significant differences. The application of the Portuguese adaptation of Beers criteria resulted in a significantly smaller proportion of PIMs detected, suggesting this tool, although more appropriate for National studies as all medications on the list are, in fact, available on the market, the pharmacological reasoning behind the list may be out-of-date. Our data also shows that using the START/STOPP criteria enabled the detection of a significantly higher proportion of PIMs, but more important, a more holistic approach to the quality of prescribing to the elderly. In particular, the pattern of prescribing to Portuguese polypharmacy elderly highlights low levels of cardiovascular risk prevention and abuse of psychotropic medications, suggesting there are potential interventions to be developed for improving rational and safe drug use that should be monitored for quality improvement using the same tool. Clinicians’ focus should be put on these therapeutic groups, where administrative safety issues are best detected by STOPP, abuse of psychotropic medicines by the American Beers and preventive medications are better assessed by START.
References
PORDATA. Indicadores de envelhecimento em Portugal. http://www.pordata.pt/Portugal/Indicadores+de+envelhecimento-526. Assessed 10 Oct 2014.
Borges EP, Morgado M, Macedo AF. Prescribing omissions in elderly patients admitted to a stroke unit: descriptive study using START criteria. Int J Clin Pharm. 2012;34(3):481–9.
Gallagher P, Barry PJ, O´Mahony D. Inappropriate prescribing in the elderly. J Clin Pharm Ther. 2007;32(2):113–21.
Ryan C, O’Mahony D, Kennedy J, Weedle P, Byrne S. Potentially inappropriate prescribing in an Irish elderly population in primary care. Br J Clin Pharmacol. 2009;68(6):936–47.
San-José A, Agustí A, Vidal X, Formiga F, López-Soto A, Fernández-Moyano A, et al. Inappropriate prescribing to older patients admitted to hospital: a comparison of different tools of misprescribing and underprescribing. Eur J Intern Med. 2014;25(8):710–6.
Liu CL, Peng LN, Chen YT, Lin MH, Liu LK, Chen LK. Potentially inappropriate prescribing (IP) for elderly medical inpatients in Taiwan: a hospital-based study. Arch Gerontol Geriatr. 2012;55(1):148–51.
Retamal CG, Fernández RG, Espínola SF, Serrato AR, Ordóñez MA, Marín VP. Prevalence of potentially inappropriate medication in hospitalized elderly patients by using explicit criteria. Farm Hosp. 2014;38(4):305–16.
Hill-Taylor B, Sketris I, Hayden J, Byrne S, O’Sullivan D, Christie R. Application of the STOPP/START criteria: a systematic review of the prevalence of potentially inappropriate prescribing in older adults, and evidence of clinical, humanistic and economic impact. J Clin Pharm and Ther. 2013;38(5):360–72.
Gillespie U, Alassaad A, Hammarlund-Udenaes M, Mörlin C, Henrohn D, Bertilsson M, Melhus H. Effects of pharmacists’ interventions on appropriateness of prescribing and evaluation of the instruments’ (MAI, STOPP and STARTs’) ability to predict hospitalization–analyses from a randomized controlled trial. PLoS ONE. 2013;8(5):e62401.
Karandikar YS, Chaudhari SR, Dalal NP, Sharma M, Pandit VA. Inappropriate prescribing in the elderly: a comparison of two validated screening tools. JCGG. 2013;4(4):109–14.
Beers MH, Ouslander J, Rollingher I, Reuben D, Brooks J, Beck J. Explicit criteria for determining in—appropriate medication use in nursing homes. Arch Intern Med. 1991;151(9):1825–32.
Beers MH. Explicit criteria for determining potentially inappropriate medication use by the elderly. Arch Intern Med. 1997;157(14):1531–6.
Fick DM, Cooper JW, Wade WE, Waller JL, Maclean JR, Beers MH. Updating the Beers criteria for potentially inappropriate medication use in older adults. Arch Intern Med. 2003;163(22):2716–24.
Campanelli CM. American Geriatrics Society updated beers criteria for potentially inappropriate medication use in older adults: American Geriatrics Society 2012 Beers Criteria Update Expert Panel. J Am Geriatr Soc. 2012;60(4):616–31.
American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society. Updated Beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;2015(63):2227–46.
Soares MA, Fernandez-Llimós F, Lança C, Cabrita J, Morais JA. Operacionalização para Portugal: Critérios de Beers de Medicamentos Inapropriados nos Doentes Idosos. Acta Med Port. 2008;21(5):441–52.
Cahir C, Moriarty F, Teljeur C, Fahey T, Bennett K. Potentially inappropriate prescribing and vulnerability and hospitalization in older community-dwelling patients. Ann Pharmacother. 2014;48(12):1546–54.
Hudhra K, García-Caballos M, Jucja B, Casado-Fernández E, Espigares-Rodriguez E, Bueno-Cavanillas A. Frequency of potentially inappropriate prescriptions in older people at discharge according to Beers and STOPP criteria. Int J Clin Pharm. 2014;36(3):596–603.
Miquel MD, Cuervo MS, Silveira ED, Machuca IS, González-Blazquez S, Errasquin BM, Cruz-Jentoft AJ. Potentially inappropriate drug prescription in older subjects across health care settings. Eur Geriatr Med. 2010;1(1):9–14.
Lang PO, Hasso Y, Belmin J, Pavot I, Bayens JP, Vogt-Ferrier N, Gallagher P, O’Mahony D, Michel JP. STOPP-START: adaptation of a French language screening tool for detecting inappropriate prescriptions in older people. Can J Public Health. 2009;100(6):426–31 [Article in French].
Delgado Silveira E, Montero Errasquín B, Muñoz-Garcia M, Vélez-Díaz-Pallarés M, Lozano Montoya I, Sánchez-Castellano C, et al. Improving drug prescribing in the elderly: a new edition of STOPP/START criteria. Esp Geriatr Gerontol. 2015;50(2):89–96 [Article in Spanish].
Vermeulen Windsant-van den Tweel AM, Verduijn MM, Derijks HJ, van Marum RJ. Detection of inappropriate medication use in the elderly; will the STOPP and START criteria become the new Dutch standards? Ned Tijdschr Geneeskd. 2012;156(40):A5076 [Article in Dutch].
Morin L, Fastbom J, Laroche ML, Johnell K. Potentially inappropriate drug use in older people: a nationwide comparison of different explicit criteria for population-based estimates. Br J Clin Pharmacol. 2015;80(2):315–24.
Oliveira-Martins S, Soares MA, van Mil JWF, Cabrita J. Inappropriate drug use by Portuguese elderly outpatients—effects of the Beers criteria update. Pharm World Sci. 2006;28:296–301.
Gouveia Pinto C, Carrageta MO, Miguel LS. Cost-effectiveness of rosuvastatin in the prevention of ischemic heart disease in Portugal. Value Health. 2008;11(2):154–9.
Chang C-B, Chen J-H, Wen C-J, Kuo H-K, Lu I-S, Chiu L-S, Wu S-C, Chan D-C. Potentially inappropriate medications in geriatric outpatients with polypharmacy: application of six sets of published explicit criteria. Br J Clin Pharmacol. 2011;72(3):482–9.
Rocha O, Lunet N, Costa L, Barros H. Tratamento da osteoporose em Portugal: Tendência e variação geográfica. Acta Med Port. 2006;19:373–80.
Ravera S, Hummel SA, Stolk P, Heerdink RE, de Jong–van den Berg LTW, de Gier JJ. The use of driving impairing medicines: a European survey. Eur J Clin Pharmacol. 2009;65:1139–47.
Kojima G, Bell CL, Tamura B, Davis J, Inaba M, Masaki K. Combining quality improvement and geriatrics training: the nursing home polypharmacy outcomes project. Gerontol Geriatr Educ. 2014;35(4):395–408.
Pugh MJ, Hanlon JT, Zeber JE, et al. Assessing potentially inappropriate prescribing in the elderly Veterans Affairs population using the HEDIS 2006 quality measure. J Manag Care Pharm. 2006;12:537.
Cahir C, Bennett K, Teljeur C, Fahey T. Potentially inappropriate prescribing and adverse health outcomes in community dwelling older patients. Br J Clin Pharmacol. 2013;77(1):201–10.
O’Mahony D, O’Sullivan D, Byrne S, O’Connor MN, Ryan C, Gallagher P. STOPP/START criteria for potentially inappropriate prescribing in older people: version 2. Age Ageing. 2015;44:213–8.
Tommelein E, Petrovic M, Somers A, Mehuys E, van der Cammen T, Boussery K. Older patients’ prescriptions screening in the community pharmacy: development of the Ghent Older People’s Prescriptions community Pharmacy Screening (GheOP3S) tool. J Public Health. 2016;38(2):e158–70.
Costa FA, Duggan C, Bates I. Validation of a survey tool for use in cross-cultural studies. Pharm Pract (Internet). 2008;6(3):121–7.
Endres HG, Kaufmann-Kolle P, Steeb V, Bauer E, Böttner C, Thürmann P. Association between potentially inappropriate medication (PIM) use and risk of hospitalization in older adults: an observational study based on routine data comparing PIM use with use of PIM alternatives. PLoS ONE. 2016;11(2):e0146811. doi:10.1371/journal.pone.0146811
Bjerre LM, Halil R, Catley C, Farrel B, Hogel M, Blacks C, et al. Potentially inappropriate prescribing (PIP) in long-term care (LTC) patients: validation of the 2014 STOPP–START and 2012 Beers criteria in a LTC population—a protocol for a cross-sectional comparison of clinical and health administrative data. BMJ Open. 2015;5:e009715. doi:10.1136/bmjopen-2015009715.
Acknowledgments
The authors wish to thank the people in charge of the elderly facilities that participated in this study and their clinical directors for their full support. We also acknowledge the important contribution of Lawrence Sturm in reviewing the English language.
Funding
The study did not receive any specific funding and all costs were entirely supported by Egas Moniz, Cooperativa de Ensino, SRL, located in Campus Universitário, Quinta da Granja, Monte da Caparica, Portugal.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no financial or other relationships that might lead to a conflict of interest.
Rights and permissions
About this article
Cite this article
da Costa, F.A., Periquito, C., Carneiro, M.C. et al. Potentially inappropriate medications in a sample of Portuguese nursing home residents: Does the choice of screening tools matter?. Int J Clin Pharm 38, 1103–1111 (2016). https://doi.org/10.1007/s11096-016-0337-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11096-016-0337-y