Abstract
Memory impairment is the most common symptom in patients with Alzheimer’s disease. The purpose of this study is to evaluate the memory enhancing effects of P7C3, a recently identified compound with robust proneurogenic and neuroprotective effects, on the cognitive impairment induced by scopolamine, a muscarinic acetylcholine receptor antagonist. Different behavior tests including the Y-maze, Morris water maze, and passive avoidance tests were performed to measure cognitive functions. Scopolamine significantly decreased the spontaneous alternation and step-through latency of C57BL/6J mice in Y-maze test and passive avoidance test, whereas increased the time of mice spent to find the hidden platform in Morris water maze test. Importantly, intraperitoneal administration of P7C3 effectively reversed those Scopolamine-induced cognitive impairments in C57BL/6J mice. Furthermore, P7C3 treatment significantly enhanced the level of brain-derived neurotrophic factor (BDNF) signaling pathway in the cortex and hippocampus, and the usage of selective BDNF signaling inhibitor fully blocked the anti-amnesic effects of P7C3. Therefore, these findings suggest that P7C3 could improve the scopolamine-induced learning and memory impairment possibly through activation of BDNF signaling pathway, thereby exhibiting a cognition-enhancing potential.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alzheimer’s disease (AD), the most common type of dementia is a progressive neurodegenerative disorder characterized by a decline in cognitive function and memory impairment [1, 2]. Characteristic neuropathological features of AD are accumulation of amyloid plaques, formation of neurofibrillary tangles, induction of oxidative stress and inflammatory responses, and disturbance in neurotransmission [3]. Particularly, the impairment of memory and cognitive dysfunction in AD is associated with cholinergic (acetylcholine-producing) hypofunction by increasing the acetyl-cholinesterase (AchE) activity [4–6]. Conversely, the major treatment for AD is AchE inhibitors, which improve the levels of acetylcholine by delaying its degradation at cholinergic synapses [7, 8]. However, due to the limitation of current medications for treating AD such as relatively low efficacy, severe adverse effects for the long-term use, and in-effectiveness in the late stage of AD [9], it is very necessary to develop novel and safe anti-amnesic compounds with neuroprotective properties. In this regard, one promising new candidate of interest in this study is P7C3.
Previous studies have showed that learning and memory is controlled by a lot of proteins and signaling pathways, especially the BDNF signaling pathway. BDNF is a neurotrophic factor closely involved in memory consolidation, and the dysfunction of BDNF disrupts the hippocampus-dependent memory formation [10, 11]. CREB is a transcription factor coupled to the activation of BDNF [12], and plays important roles in many developmentally regulated processes, such as cell survival, hippocampal neurogenesis and long-term memory [13–16].
P7C3 is an identified proneurogenic and neuroprotective aminopropyl carbazole [17]. Pieper et al. [17] reported that the intraperitoneal injection of P7C3 could significantly enhance the hippocampal neurogenesis at a dose of 20 mg/kg, while hippocampal neurogenesis is closely correlated with learning and memory [18, 19]. By now, this compound has already been demonstrated to treat several brain disorders in animal models, including the Parkinson’s disease, amyotrophic lateral sclerosis and traumatic brain injury [20–22]. Here, we speculated that P7C3 may have anti-amnesic effects and exhibit a preventive/therapeutic potential for treating AD.
Scopolamine is a muscarinic acetylcholine receptor (mAChR) antagonist which impairs learning acquisition and short-term memory in rodents and humans [23–26]. The scopolamine-induced amnesia has been widely used to generate experimental animal models for the screening of anti-amnesic drugs. In this study, we have utilized the scopolamine model to assess the anti-amnesic effects of P7C3, and furthermore, the actions of P7C3 were extended to the molecular levels by examining the expression of brain-derived neurotrophic factor (BDNF) signaling pathway in the hippocampus and cortex.
Materials and Methods
Animals
Adult male C57BL/6J mice (8–10 weeks old) were obtained from the Experimental Animal Center of Medical College, Nantong University. Before used, mice were housed 5 per cage under standard conditions (12 h light/dark cycle; lights on from 07:00 to 19:00; 23 ± 1 °C ambient temperature; 55 ± 10 % relative humidity) for 1 week with free access to food and water. Each experimental group consisted of 15 mice. Behavioral experiments were carried out during the light phase. The experiment procedures involving animals and their care were conducted in compliance with the National Institutes of Health Guide for Care and Use of Laboratory Animals and with the European Communities Council Directive of 24 November 1986 (86/609/EEC).
Drugs and Treatments
P7C3 (purity >99 %), donepezil and scopolamine were purchased from Sigma (St. Louis, MO, USA). K252a was purchased from Alomone Laboratories (Jerusalem, Israel). P7C3 was dissolved in 5 % dextrose (pH 7.0) with 2.5 % DMSO and 10 % Cremaphor EL [17]. Donepezil, scopolamine and K252a were dissolved in the same vehicle of P7C3. The dosages of P7C3 (10, 20 mg/kg), donepezil (5 mg/kg), K252a (25 μg/kg) and scopolamine (1 mg/kg) were chosen based on previous reports [17, 25, 27]. All these compounds were administered intraperitoneally (i.p.) in a volume of 10 ml/kg. Control animals were administered with the corresponding vehicle also in the same volume.
Passive Avoidance Task
The passive avoidance performance was carried out in identical illuminated and non-illuminated boxes (20 × 20 × 20 cm). The illuminated compartment contained a 100 W bulb, and the non-illuminated compartment was equipped with an electrifiable grid floor. The two chambers were connected by an automatically operated guillotine door (5 × 5 cm). Each test involved two separate trials, a training trial and a test trial. For the training trial, a mouse was gently placed in the illuminated compartment, and after 30 s of familiarization, the light was on and the door between the two compartments was opened. When the mouse entered the dark compartment, the door automatically closed and a 0.5 mA electrical shock of 3 s in duration was delivered through the stainless steel rods. The time taken for the mouse to enter the non-illuminated chamber was recorded as the step-through latency. A test trial was performed 24 h after the training trial, the mouse was again placed in the illuminated compartment, and the time to enter the non-illuminated chamber after door opening was measured again without electric foot shock. The step-through latency was recorded up to 300 s. If a mouse did not enter the non-illuminated compartment within 300 s, the mouse was removed and assigned a latency score of 300 s.
Y-maze Test
Y-Maze is used as a measure of immediate spatial working memory which is form of short-term memory [25]. Three arms of the Y-maze (41 cm long, 25 cm high, and 10 cm wide) were positioned at an equal angle. Each mouse was placed at the end of one arm and allowed to navigate freely for an 8-min session. During the 8 min period, the sequence (e.g., ABC, BCA, CAB) and number of arm entries were recorded manually for each mouse. The Spontaneous alternation behavior was defined as entries into all three arms on consecutive choices (i.e., ABC, CAB, or BCA but not ABA). Maze arms were thoroughly cleaned between tasks to remove residual odors. The percentage (%) of spontaneous alternation behavior was defined according to the following equation: % alternation = [(number of alternations)/(total arm entries − 2)] × 100.
Morris Water Maze
The apparatus consisted of a circular pool with a diameter and height 100 and 45 cm, respectively. The pool was filled to a depth of 30 cm with water containing 500 ml of milk (25 ± 1 °C). A white platform (8 cm in diameter) was submerged 1 cm below the water surface. The pool was divided into four equal quadrants and each quadrant was marked by a different visual cue. The platform was randomly placed in one quadrant for the duration of the experiment. On the first day, each mouse was allowed a 120 s habituation session in the pool without the platform. During the four subsequent days, each mouse was given four 120 s learning trials per day with the platform in place. The time interval between each trials session was 1 min. For each learning trial, the mouse was placed into the water facing the pool wall in one of the pool quadrants. The entry point was changed in a different order for each day. The escape latency, the time taken to find the submerged platform, was recorded using a video camera-based Ethovision System (Noldus, Wageningen, The Netherlands) for each trial. When a mouse located the platform, it was permitted to remain on it for 20 s. If a mouse was unable to locate the platform within 120 s, it was led to the platform and allowed to rest for 60 s, and the escape latency in these cases was recorded as 120 s. On the fifth day, all the mice were subjected to a probe trial session in which the platform was removed from the pool. Mice were allowed to swim for 60 s to search for the removed platform, and a record was kept of the swimming time in the pool quadrant where the platform had been previously placed.
Western Blotting Analysis
The experiment was conducted as we have described [27]. The mice used for performing Morris water maze were sacrificed 24 h after the probe test. Bilateral hippocampi were rapidly dissected and homogenized in lyses buffer [50 mM Tris–HCl, pH 7.4; 1 mM EDTA; 100 mM NaCl; 20 mM NaF; 3 mM Na3VO4; 1 mM PMSF with 1 % (v/v) Nonidet P-40; and protease inhibitor cocktail], and then kept on ice for 30 min. The homogenate was centrifuged at 12,000×g for 15 min, and supernatants were then collected. Protein concentration was estimated by Coomassie blue protein-binding assay (Jiancheng Institute of Biological Engineering, Nanjing, China). After denaturation, 30 μg of protein samples were separated by 10 % SDS/PAGE gel and then transferred to nitrocellulose membranes (Bio-Rad, Hercules, CA, USA). After blocking with 5 % nonfat dried milk powder/Tris-buffered saline Tween-20 (TBST) for 1 h, membranes were incubated overnight at 4 °C with primary antibodies to cAMP response element-binding protein (CREB; 1:500; Cell Signaling, MA, USA), phospho-CREB-ser133 (pCREB; 1:500; Cell Signaling, MA, USA); brain derived neurotrophic factor (BDNF; 1:500; Epitomics, CA, USA), glyceraldehydes-3-phosphate dehydrogenase (GAPDH; 1:1000; Santa Cruz, CA, USA). The antigen–antibody complexes were visualized with goat anti-rabbit or goat anti-mice horseradish peroxidase-conjugated secondary antibodies (1:2000; Santa Cruz, CA, USA) by using enhanced chemiluminescence (ECL; Pierce, Rockford, IL, USA). The optical density of the bands was determined using Optiquant software (Packard Instruments BV, Groningen, Netherlands).
Statistical Analysis
All analyses were performed using SPSS 13.0 software (SPSS Inc., USA) and data are presented as mean ± SEM. Differences between mean values were evaluated using one-way analysis of variance (ANOVA), and post hoc tests were performed using LSD test. p < 0.05 was considered statistically significant.
Results
Effects of P7C3 on the Scopolamine-Induced Memory Impairments
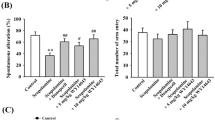
We first tested the effect of P7C3 on the scopolamine-induced memory deficit using the step-through passive avoidance task which is largely dependent on long-term memory. P7C3, donepezil or vehicle was administrated 60 min prior to the training trial, and scopolamine was injected 30 min after the drug administration. Donepezil was used as a positive control. Data are summarized in Fig. 1b, it was found that there was no significant difference in step-through latency time among experimental groups in the training trial. However, in the test trial, the step-through latency of scopolamine-treated (1 mg/kg, i.p.) mice was significantly lower than that of vehicle-treated control mice (n = 12, p < 0.01 vs. Control), and interestingly, the reduced step-through latency induced by scopolamine was fully reversed by P7C3, especially at 20 mg/kg (n = 11, p < 0.01 vs. Scopolamine), similar to donepezil. Then the effects of P7C3 on the spontaneous alternation behavior were examined using the Y-maze task. P7C3, donepezil or vehicle was administrated 60 min prior to the task, and scopolamine was injected 30 min after the drug administration. As shown in Fig. 1c, the spontaneous alternation of scopolamine-treated mice was significantly lower than that of vehicle-treated control mice (n = 12, p < 0.01 vs. Control), and similar to donepezil, this change was also restored by P7C3 at both the 10 mg/kg and 20 mg/kg (n = 10–11, p < 0.01 vs. Scopolamine). However, the number of arm entries was similar across all experimental groups (Fig. 1c), indicating that P7C3 did not affect the general locomotor activity of animals.
Memory enhancing effects of P7C3 in the passive avoidance test and Y-maze test. a The molecular structure of P7C3. b Protective effects of P7C3 on the learning and memory deficit caused by scopolamine in the passive avoidance test. c Promoting effects of P7C3 on the scopolamine-decreased spontaneous alternation in the Y-maze test, while the total number of arm entries was similar between all groups. Data are expressed as mean ± SEM (n = 10–12); **p < 0.01 versus control; # p < 0.05, ## p < 0.01 versus scopolamine group. Comparison was made by one-way ANOVA followed by post-hoc LSD test
We further investigated the effects of P7C3 on the scopolamine-induced spatial memory impairments using the Morris water maze task. P7C3, donepezil or vehicle was administrated 60 min prior to the first learning trial of each training day, and scopolamine was injected 30 min after the drug administration. As shown in Fig. 2a, the mice in the vehicle-treated control group readily learned and memorized the location of the submerged hidden platform during the four training days, which was reached within 30 s by the last day of the training period. In contrast, the scopolamine-treated mice exhibited significantly longer escape latency than control mice (n = 12, p < 0.01 vs. Control). However, we found that 10 mg/kg P7C3 treatment significantly reduced the escape latency of scopolamine-treated mice starting from the second day of the training period (n = 12, p < 0.01 vs. Scopolamine), and 20 mg/kg P7C3-treated group exhibited nearly similar escape latency to the control group during the whole training period. On the day following the training period, the probe test was performed. The swimming time within the target quadrant in the scopolamine-treated group were significantly less than those in the vehicle-treated control group (Fig. 2b, n = 12, p < 0.01 vs. Control), and this change was also fully reversed by either P7C3 or donepezil treatment (Fig. 2b, n = 10–11, p < 0.01 vs. Scopolamine). Together, these data suggest that P7C3 could rescue the scopolamine-induced memory impairments.
Memory promoting activities of P7C3 in the Morris water maze test. a Effects of P7C3 on the scopolamine-induced spatial memory impairment which was represented by mean escape latency during the training trials in Morris water-maze test. b Comparison of swimming time spent in the target quadrant during the probe test in Morris water-maze test. Data are expressed as mean ± SEM (n = 11–12); **p < 0.01 versus Control; # p < 0.05, ## p < 0.01 versus scopolamine group. Comparison was made by one-way ANOVA followed by post-hoc LSD test
In addition, the memory enhancing effects of P7C3 were investigated in naive mice. However, it was found that P7C3 treatment did not promote the memory of naive mice in the passive avoidance task, Y-maze task or Morris water maze task (Fig. 3, n = 12), indicating that the effects of P7C3 were limited to amnesic mice.
P7C3 produces no significant memory enhancing effects in naive mice. a Effects of P7C3 on the memory of naive mice in the passive avoidance test. P7C3 was administrated 60 min before the training trial. b Effects of P7C3 on the memory of naive mice in the Y-maze test. P7C3 was administrated 60 min prior to the test. c Effects of P7C3 on the memory of naive mice in the Morris water maze test. P7C3 was injected 60 min prior to the first learning trial of each training day. Data are expressed as mean ± SEM (n = 12). Comparison was made by one-way ANOVA followed by post-hoc LSD test
Effects of P7C3 on the Expression of BDNF Signaling Pathway in the Brain
To further investigate the molecular mechanisms underlying the memory enhancing effects of P7C3, we conducted western blot experiments to examine the expression of BDNF and activation of CREB in the cortex and hippocampus. As shown in Fig. 4a, b, both the hippocampal and cortical BDNF expression in the P7C3-treated group were significantly higher than that in the scopolamine-treated group (n = 7, p < 0.01 vs. Scopolamine). Similarly, the scopolamine-decreased expression of p-CREB in the hippocampus and cortex was also markedly restored by P7C3 administration, especially at 20 mg/kg (Fig. 4a, b, n = 7, p < 0.01 vs. Scopolamine). These results suggest that the anti-amnesic effects of P7C3 may due to the activation of BDNF signaling pathway in the hippocampus and cortex.
P7C3 treatment reverses the scopolamine-induced decrease of BDNF signaling pathway in the hippocampus and cortex. a Representative western blotting of BDNF/GAPDH and p-CREB/CREB showed that P7C3 treatment restored the scopolamine-induced decrease of BDNF and p-CREB expression in the hippocampus. Scopolamine + P7C3-treated mice displayed significantly higher BDNF and p-CREB bands than Scopolamine-treated mice. b In parallel to hippocampus, P7C3 administration also restored the scopolamine-induced inhibition of BDNF and p-CREB expression in the cortex. Data are expressed as mean ± SEM (n = 5); **p < 0.01 versus control; # p < 0.05, ## p < 0.01 versus CSDS+ vehicle group. Comparison was made by one-way ANOVA followed by post-hoc LSD test
Attenuation of P7C3’s Anti-amnesic Effects by the BDNF Signaling Pathway Blockade
To determine whether the BDNF signaling pathway is required for the P7C3-induced anti-amnesic effects, the potent pharmacological inhibitor of BDNF receptor TrkB, K252a, was used [28–30]. Mice were firstly injected with K252a, then P7C3 (after 30 min), and lastly scopolamine (after 1 h). Behavioral tests were then performed. Data are summarized in Fig. 5a. The passive avoidance task results showed that the step-through latency of P7C3+ K252a+ scopolamine-treated mice was significantly lower than that of P7C3+ scopolamine-treated mice, and nearly the same level to that of scopolamine-treated mice. Similarly, the Y-maze task results showed that the spontaneous alternation of P7C3+ K252a+ scopolamine-treated mice was also significantly lower than that of P7C3+ scopolamine-treated mice, and nearly the same level to that of scopolamine-treated mice (Fig. 5b). Furthermore, the Morris water maze task results revealed that the escape latency of P7C3+ K252a+ scopolamine-treated mice was significantly longer than that of P7C3+ scopolamine-treated mice (Fig. 6a), and the swimming time within the target quadrant in the P7C3+ K252a+ scopolamine group were significantly less than that in the P7C3+ scopolamine group (Fig. 6b). Together, these results suggest that the BDNF system is involved in the anti-amnesic effects of P7C3.
Blockade of BDNF signaling by K252a abolishes the anti-amnesic actions of P7C3 in the passive avoidance test and Y-maze test. a K252a pretreatment 30 min before P7C3 administration prevented the P7C3-induced increase in the step-through latency of scopolamine-treated mice in passive avoidance test. b K252a pretreatment also abolished the P7C3-induced increase in the spontaneous alternation of scopolamine-treated mice in Y-maze test, while the total number of arm entries was similar between all groups. Data are expressed as mean ± SEM (n = 11–12); **p < 0.01 versus Control; # p < 0.05, ## p < 0.01 versus scopolamine group. Comparison was made by one-way ANOVA followed by post-hoc LSD test
Blockade of BDNF signaling by K252a abolishes the anti-amnesic effects of P7C3 in the Morris water maze test. a K252a pretreatment 30 min before P7C3 administration prevented the P7C3-induced decrease in the mean escape latency of scopolamine-treated mice during the training trials. b K252a pretreatment also abolished the effects of P7C3 on the scopolamine-treated mice in the probe test. Data are expressed as mean ± SEM (n = 11–12); **p < 0.01 versus Control; # p < 0.05, ## p < 0.01 versus scopolamine group. Comparison was made by one-way ANOVA followed by post-hoc LSD test
Discussion
In this study, the major findings of this study are as follows. First, P7C3 produces anti-amnesic effects in multiple animal models screening for anti-amnesic activity, including the passive avoidance task, Y-maze test and Morris water maze. Second, the anti-amnesic effects of P7C3 require the BDNF signaling pathway, since it could be prevented by selective inhibition of BDNF system in the hippocampus and cortex. Together, these data identify a novel function of P7C3 suggesting it could be developed as a potential treatment for AD.
AD is the most common form of age-related neurodegenerative disorder that is characterized with an insidious loss of memory, associated functional decline, and behavioral disturbances [31]. Cholinergic deficit is a major feature that is associated with memory loss and cognitive dysfunction in AD [4–6], and so it is well known that the anti-cholinergic agent scopolamine-induced memory deficits are similar to those found in age-related senile central nervous system dysfunction. Therefore, scopolamine challenge could serve as a useful tool for selecting compounds with therapeutic potential for treating AD. The compound, P7C3, was selected in our study by virtue of the knowledge that it demonstrates robust proneurogenic activity as neurogenesis is closely involved in learning and memory [17, 19, 32], and this compound also has neuroprotective effects while AD is always accompanied with neuronal loss [33–35]. P7C3 is orally bioavailable, endowed with a relatively long half life, capable of crossing the blood brain barrier, and safely tolerated by mice [17]. Here, we investigated whether P7C3 has anti-AD potential in the scopolamine-induced memory impairment animal model, and conducted a series of behavior tests. Firstly, in the passive avoidance test, scopolamine treatment reduced the step-through latency, which was effectively restored by intraperitoneal injection of P7C3. Secondly, in the Y-maze test, the scopolamine-treated mice showed decreased spontaneous alternation compared with that of vehicle-treated mice, which was significantly improved by P7C3 pretreatment. Lastly, in the Morris water-maze task, the scopolamine-treated animals took longer time to find the platform than those animals in the control group, and P7C3+ scopolamine-treated animals easily found the location of the hidden platform. Collectively, these data strongly suggest that P7C3 could be a novel anti-AD compound.
As a molecular mechanism underlying the anti-amnesic effects of P7C3, we showed that P7C3 treatment could enhance the BDNF signaling pathway in the hippocampus and cortex. We selected BDNF in our study as BDNF is very important for learning and memory of rodents [36]. Evidences have been accumulated that P7C3 may affect the BDNF system. For example, it has been found that P7C3 administration could rescue the reduced dendritic length and branching in the dentate gyrus of NPAS3 knockout mice [17], and BDNF plays a critical role in the growth and differentiation of neuronal dendrite [37]. P7C3 also robustly promotes the hippocampal neurogenesis, while hippocampal neurogenesis is not only correlated with learning and memory, but also closely controlled by BDNF [17, 19, 38]. Furthermore, the usage of K252a fully abolished the anti-amnesic effects of P7C3, proving that BDNF is indeed involved in this response. Interestingly, Garcia et al. reported that exogenously added BDNF evoke acetylcholine release in rodent motor nerve terminals via trkB receptors [39]. Thus we think P7C3 may promote acetylcholine release in the brain to protect against the scopolamine-induced cholinergic deficits via activating the BDNF-trkB signaling, and this needs further study. Moreover, De Jesus-Cortes et al. [20] showed that P7C3 could block the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-mediated cell death of dopaminergic neurons in the substantia nigra of adult mice, a model of Parkinson disease (PD). It is known that both the PD and AD belong to neurodegenerative disorders, and the pathogenesis of AD also included neuronal death [33–35], further implying the anti-AD potential of P7C3.
How does P7C3 affect the expression of BDNF? Wang et al. [40] reported that P7C3 produces neuroprotective effects via activating the nicotinamide phosphoribosyltransferase (NAMPT), the rate-limiting enzyme which converts nicotinamide into nicotinamide adenine dinucleotide (NAD+). Previous studies showed that the NAMPT in hippocampal and cortical excitatory neurons is critical for cognitive function [41], and lowering NAD+ levels in mouse cortical neurons led to decreased activity-dependent BDNF expression by increasing the DNA methylation of the activity-dependent BDNF promoter [42]. Therefore it is possible that P7C3 enhances the BDNF protein level by activating the biosynthesis of NAD+, and this also needs further research.
In accordance with our findings, a lot of compounds have been reported to have neuroprotective and memory enhancing effects via the BDNF-CREB signaling pathway. For example, Kim et al. found that arabinoxylan, a major component of dietary fiber in wheat, prevented the scopolamine-induced amnesia in rats possibly through activating the expression of BDNF and CREB in the cortex and hippocampus [43]. Oh et al. also reported that Angelica keiskei significantly attenuated the scopolamine-induced cognitive impairment in mice, and recovered the scopolamine-reduced phosphorylation of CREB and expression of BDNF in the hippocampus [25].
Collectively, P7C3 has wide-ranging biological effects, and many reveal positive therapeutic indexes. Our results show that P7C3 possesses anti-amnesic property through promotion of the BDNF system, providing a new insight to understand the pharmacological effects of P7C3. More importantly, this study shed light on the development of new anti-amnesic treatments with higher efficacy and fewer side effects.
Abbreviations
- AchE:
-
Acetyl-cholinesterase
- AD:
-
Alzheimer’s disease
- ANOVA:
-
Analysis of variance
- BDNF:
-
Brain derived neurotrophic factor
- CREB:
-
cAMP response element-binding protein
- GAPDH:
-
Glyceraldehydes-3-phosphate dehydrogenase
- NAD+ :
-
Nicotinamide adenine dinucleotide
- NAMPT:
-
Nicotinamide phosphoribosyltransferase
- TrkB:
-
Tyrosine kinase B
- PD:
-
Parkinson disease
References
Schonberger SJ, Edgar PF, Kydd R, Faull RL, Cooper GJ (2001) Proteomic analysis of the brain in Alzheimer’s disease: molecular phenotype of a complex disease process. Proteomics 1:1519–1528
Brookmeyer R, Johnson E, Ziegler-Graham K, Arrighi HM (2007) Forecasting the global burden of Alzheimer’s disease. Alzheimers Dement 3:186–191
Karran E, Mercken M, De Strooper B (2011) The amyloid cascade hypothesis for Alzheimer’s disease: an appraisal for the development of therapeutics. Nat Rev Drug Discov 10:698–712
Coyle JT, Price DL, DeLong MR (1983) Alzheimer’s disease: a disorder of cortical cholinergic innervation. Science 219:1184–1190
Sugimoto H, Yamanishi Y, Iimura Y, Kawakami Y (2000) Donepezil hydrochloride (E2020) and other acetylcholinesterase inhibitors. Curr Med Chem 7:303–339
Schliebs R, Arendt T (2011) The cholinergic system in aging and neuronal degeneration. Behav Brain Res 221:555–563
Ahmed T, Gilani AH (2009) Inhibitory effect of curcuminoids on acetylcholinesterase activity and attenuation of scopolamine-induced amnesia may explain medicinal use of turmeric in Alzheimer’s disease. Pharmacol Biochem Behav 91:554–559
Parsons CG, Danysz W, Dekundy A, Pulte I (2013) Memantine and cholinesterase inhibitors: complementary mechanisms in the treatment of Alzheimer’s disease. Neurotox Res 24:358–369
Casey DA, Antimisiaris D, O’Brien J (2010) Drugs for Alzheimer’s disease: are they effective? PT 35:208–211
Alonso M, Bekinschetin P, Cammarota M, Vianna MR, Izquierdo I, Medina H (2005) Endogenous BDNF is required for long-term memory formation in the rat parietal cortex. Learn Mem 12:504–510
Bekinschtein P, Cammarota M, Katche C, Slipczuk L, Rossato JI, Goldin A et al (2008) BDNF is essential to promote persistence of long-term memory storage. Proc Natl Acad Sci USA 105:2711–2716
Shaywitz AJ, Greenberg ME (1999) CREB: a stimulus-induced transcription factor activated by a diverse array of extracellular signals. Annu Rev Biochem 68:821–861
Alberini CM (2009) Transcription factors in long-term memory and synaptic plasticity. Physiol Rev 89:121–145
Chen G, Zou X, Watanabe H, van Deursen JM, Shen J (2010) CREB binding protein is required for both short-term and long-term memory formation. J Neurosci 30:13066–13077
Dworkin S, Mantamadiotis T (2010) Targeting CREB signalling in neurogenesis. Expert Opin Ther Targets 14:869–879
Saura CA, Valero J (2011) The role of CREB signaling in Alzheimer’s disease and other cognitive disorders. Rev Neurosci 22:153–169
Pieper AA, Xie S, Capota E, Estill SJ, Zhong J, Long JM et al (2010) Discovery of a proneurogenic, neuroprotective chemical. Cell 142:39–51
Gould E, Beylin A, Tanapat P, Reeves A, Shors TJ (1999) Learning enhances adult neurogenesis in the hippocampal formation. Nat Neurosci 2:260–265
Snyder JS, Hong NS, McDonald RJ, Wojtowicz JM (2005) A role for adult neurogenesis in spatial long-term memory. Neuroscience 130:843–852
De Jesus-Cortes H, Xu P, Drawbridge J, Estill SJ, Huntington P, Tran S et al (2012) Neuroprotective efficacy of aminopropyl carbazoles in a mouse model of Parkinson disease. Proc Natl Acad Sci USA 109:17010–17015
Tesla R, Wolf HP, Xu P, Drawbridge J, Estill SJ, Huntington P et al (2012) Neuroprotective efficacy of aminopropyl carbazoles in a mouse model of amyotrophic lateral sclerosis. Proc Natl Acad Sci USA 109:17016–17021
Blaya MO, Bramlett HM, Naidoo J, Pieper AA, Dietrich WD (2014) Neuroprotective efficacy of a proneurogenic compound after traumatic brain injury. J Neurotrauma 31:476–486
Sun XL, Ito H, Masuoka T, Kamei C, Hatano T (2007) Effect of Polygala tenuifolia root extract on scopolamine-induced impairment of rat spatial cognition in an eight-arm radial maze task. Biol Pharm Bull 30:1727–1731
Wang Q, Sun LH, Jia W, Liu XM, Dang HX, Mai WL et al (2010) Comparison of ginsenosides Rg1 and Rb1 for their effects on improving scopolamine-induced learning and memory impairment in mice. Phytother Res 24:1748–1754
Oh SR, Kim SJ, Kim DH, Ryu JH, Ahn EM, Jung JW (2013) Angelica keiskei ameliorates scopolamine-induced memory impairments in mice. Biol Pharm Bull 36:82–88
Park SJ, Ahn YJ, Oh SR, Lee Y, Kwon G, Woo H et al (2014) Amyrin attenuates scopolamine-induced cognitive impairment in mice. Biol Pharm Bull 37:1207–1213
Jiang B, Xiong Z, Yang J, Wang W, Wang Y, Hu ZL et al (2012) Antidepressant-like effects of ginsenoside Rg1 are due to activation of the BDNF signalling pathway and neurogenesis in the hippocampus. Br J Pharmacol 166:1872–1887
Tapley P, Lamballe F, Barbacid M (1992) K252a is a selective inhibitor of the tyrosine protein kinase activity of the trk family of oncogenes and neurotrophin receptors. Oncogene 7:371–381
Yan HC, Qu HD, Sun LR, Li SJ, Cao X, Fang YY et al (2010) Fuzi polysaccharide-1 produces antidepressant-like effects in mice. Int J Neuropsychopharmacol 13:623–633
Zhu XH, Yan HC, Zhang J, Qu HD, Qiu XS, Chen L et al (2010) Intermittent hypoxia promotes hippocampal neurogenesis and produces antidepressant-like effects in adult rats. J Neurosci 30:12653–12663
Bullock R (2002) New drugs for Alzheimer’s disease and other dementias. Br J Psychiatry 180:135–139
Deng W, Aimone JB, Gage FH (2010) New neurons and new memories: how does adult hippocampal neurogenesis affect learning and memory? Nat Rev Neurosci 11:339–350
Carter J, Lippa CF (2001) Beta-amyloid, neuronal death and Alzheimer’s disease. Curr Mol Med 1:733–737
Niikura T, Tajima H, Kita Y (2006) Neuronal cell death in Alzheimer’s disease and a neuroprotective factor, humanin. Curr Neuropharmacol 4:139–147
Donev R, Kolev M, Millet B, Thome J (2009) Neuronal death in Alzheimer’s disease and therapeutic opportunities. J Cell Mol Med 13:4329–4348
Yamada K, Mizuno M, Nabeshima T (2002) Role for brain-derived neurotrophic factor in learning and memory. Life Sci 70:735–744
Tongiorgi E, Righi M, Cattaneo A (1997) Activity-dependent dendritic targeting of BDNF and TrkB mRNAs in hippocampal neurons. J Neurosci 17:9492–9505
Lee J, Duan W, Mattson MP (2002) Evidence that brain-derived neurotrophic factor is required for basal neurogenesis and mediates, in part, the enhancement of neurogenesis by dietary restriction in the hippocampus of adult mice. J Neurochem 82:1367–1375
Garcia N, Tomas M, Santafe M, Besalduch N, Lanuza MA, Tomas J (2010) The interaction between tropomyosin-related kinase B receptors and presynaptic muscarinic receptors modulates transmitter release in adult rodent motor nerve terminals. J Neurosci 30:16514–16522
Wang G, Han T, Nijhawan D, Theodoropoulos P, Naidoo J, Yadavalli S et al (2014) P7C3 neuroprotective chemicals function by activating the rate-limiting enzyme in NAD salvage. Cell 158:1324–1334
Stein LR, Wozniak DF, Dearborn JT, Kubota S, Apte RS, Izumi Y et al (2014) Expression of Nampt in hippocampal and cortical excitatory neurons is critical for cognitive function. J Neurosci 34:5800–5815
Chang J, Zhang B, Health H, Galjart N, Wang X, Milbrandt J (2010) Nicotinamide adenine dinucleotide (NAD)-regulated DNA methylation alters CCCTC-binding factor (CTCF)/cohesin binding and transcription at the BDNF locus. Proc Natl Acad Sci USA 107:21836–21841
Kim CY, Lee GY, Park GH, Lee J, Jang JH (2014) Protective effect of arabinoxylan against scopolamine-induced learning and memory impairment. Biomol Ther (Seoul) 22:467–473
Acknowledgments
This work was supported by the Provincial Natural Science Foundation of Jiangsu Province (China) to Dr. Bo Jiang (No. 14KJB310013), and A Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
None.
Additional information
Bo Jiang and Lu Song are equally contributed to this paper.
Rights and permissions
About this article
Cite this article
Jiang, B., Song, L., Huang, C. et al. P7C3 Attenuates the Scopolamine-Induced Memory Impairments in C57BL/6J Mice. Neurochem Res 41, 1010–1019 (2016). https://doi.org/10.1007/s11064-015-1783-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-015-1783-y