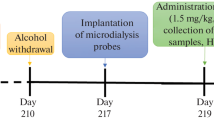
A group of 20 rats was subjected to long-lasting alcoholization (90-day-long forced consumption of 10% aqueous ethanol solution, ES). Every day at 17.00, the ES volume consumed by the animals per day was measured, and the level of ketone bodies in the urine was estimated in points using a semiquantitative quick ketone test. Then, rats were divided into two equivalent groups, experimental and control ones (n = 10 in each group). During the subsequent three-day-long main stage of the experiment, animals of the experimental group were preliminarily (at 17.00 of the preceding day) subjected to peroral administration of 1.0 ml of solution of unithiol, i.e., an agent neutralizing ketone bodies. Within three days of the mentioned period, the level of ketonuria was estimated within a 9.00–17.00 observation period with one-hour-long intervals. This time, free access of rats to water or ES was provided. We found that rats of the experimental group consumed more than one-third of the daily ES norm within the first hour of observation; i.e., within the interval where the level of ketonuria was minimum (0.17 points, on average). After this, the amount of the consumed ES decreased, while the level of ketonuria increased significantly in a parallel manner. In control rats throughout the observation period, hourly consumption of ES and the level of ketonuria demonstrated no significant dynamics. We hypothesize that there is a causal relationship between the ketosis level and the behavioral reaction of alcohol consumption (the lower the ketosis, the higher the consumption). Under conditions of alcoholization resulting in the development of hypoglycemia, ketone bodies begin to be used in the brain as an energy substrate, and the brain becomes dependent on the level of ketonemia to a significant extent. These level is a rather important factor determining alcohol addiction; direct factors initiating a situational drive for alcohol consumption are hypoketonemia episodes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
J. R. Mertens, A. H. Kline-Simon, K. L. Delucchi, et al., “Ten-year stability of remission in private alcohol and drug outpatient treatment: non-problem users versus abstainers,” Drug Alcohol Depend., 125, Nos. 1-2, 67-74 (2012).
B. Tabakoff and P. L. Hoffman, “The neurobiology of alcohol consumption and alcoholism: an integrative history,” Pharmacol. Biochem. Behav., No. 113, 20-37 (2013).
N. D. Volkow, J. S. Fowler, J. Logan, et al., “Effects of modafinil on dopamine and dopamine transporters in the male human brain: clinical implications,” JAMA, 301, No. 11, 1148-1154 (2009).
M. T. Fernández López, M. D. García Bargo, M. T. Rivero Luis, et al., “Alcoholic ketoacidosis and reversible neurological complications due to hypophosphatemia,” Nutr. Hosp., 27, No. 3, 936-939 (2012).
Z. Zhao, M. Yu, D. Crabb, at al., “Ethanol-induced alterations in fatty acid-related lipids in serum and tissues in mice,” Alcohol. Clin. Exp. Res., 35, No. 2, 229-234 (2011).
N. Paquot, J. De Flines, and A. J. Scheen, “Alcoholism, an addiction leading to multiple somatic complications,” Rev. Med. Liege, 68, No. 5-6, 272-280 (2013).
A. K. Bortnikova and T. I. Panova, “Peculiarities of utilization of glucose by brain tissues of alcoholdependent rats,” Neurophysiology, 46, No. 3, 229-235 (2014).
A. K. Bortnikova and T. I. Panova, “Ethanol craving in alcoholized rats and the glycemia level and ketosis intensity,” Laborat. Diagnost. East. Eur., No. 1 (9), 52-61 (2014).
A. V. Droblenkov and N. R. Karelina, “Activation of programmed cell death and degenerative changes of neurons of mesocorticolimbic dopaminergic system as a possible cause of inherited alcohol addiction,” Morfologiya, 141, No. 1, 16-22 (2012).
W. A. Koss, R. N. Sadowski, L. K. Sherrill, et al., “Effects of ethanol during adolescence on the number of neurons and glia in the medial prefrontal cortex and basolateral amygdala of adult male and female rats,” Brain Res., No. 1466, 24-32 (2012).
M. H. Chen and C. A. Cheng, “Alcoholic ketoacidosis coincides with acute Marchiafava-Bignami disease,” Am. J. Emerg. Med., 30, No. 9, e7-8 (2012).
H. Jain, S. Beriwal, and S. Singh, “Alcohol induced ketoacidosis, severe hypoglycemia and irreversible encephalopathy,” Med. Sci. Monit., 8, No. 11, CS 77-79 (2012).
C. Distel, S. Jacobson, and P. M. Tille, “Alcohol induced diabetic ketoacidosis exacerbated by an acute respiratory infection with Klebsiella pneumonie,” Clin. Lab. Sci., 26, No. 2, 68-71 (2013).
J. B. Dwyer and K. Tamama, “Ketoacidosis and trace amounts of isopropanol in a chronic alcoholic patient,” Clin. Chim. Acta, No. 415, 245-249 (2013).
H. Meier, S. Gschwend, S. Raimondi, and C. Giambarba, “Ethanol, sugar, acid and coma,” Praxis, 100, No. 13, 797-799 (2011).
O. K. Napreyenko, L. V. Zhyvotovs’ka, N. Yu. Petrina, and l. V. Rakhman, Narcology, Zdorov’ya, Kyiv (2011).
J. L. Parai, S. Kodikara, C. M. Milroy, and M. S. Pollanen, “Alcoholism and the Armanni-Ebstein lesion,” Forensic. Sci. Med. Pathol., 8, No. 1, 19-22 (2012).
M. D. Mashkovskii, Medicinal Means, 15th ed., Publ. House Novaya Volna, Moscow (2005)
A. V. Gudkov, D. V. Titov, A. V. Tsareva, et a., Technique of modeling of the hyperactive bladder, Patent RF No. 2496148. 20.10.2013. (12.05.2012).
Yu. Ye. Lyakh, V. G. Gur\yanov, V. N. Khomenko, and O. A. Panchenko, Elements of conputer biostatistics: analysis of information in biology, medicine, and pharmacia. Statistical software MedStat,” Ye. Papakitsa (Publisher), Donetsk (2006).
I. P. Zapandyuk, V. I. Zapadnyuk, Ye. A. Zakharia, and B. V. Zapadnyuk, Laboratory Animals. Breeding, keeping, and use in experiments,” Vyshcha Shkola, Kyiv (1983).
V. O. Akhpolova, Pecularities of the development of renal manifestations of lead intoxication in rats under conditions of modified calcium homeostasis, Author’s abstaract if Thesis. Cand. Med. Sci., Vladikavkaz (2011) http://dis.podelise.ru/text/index-41893.html
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Panova, T.I., Bortnikova, A.K. Ketosis Level as a Factor Determining Addictive Behavior of Alcoholized Rats. Neurophysiology 48, 252–258 (2016). https://doi.org/10.1007/s11062-016-9596-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11062-016-9596-4