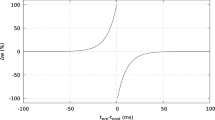
Spike timing-dependent plasticity (STDP) plays an important role in sculpting information-storing circuits in the hippocampus, since motor learning and memory are thought to be closely linked with this classical plasticity. To further understand the information delivery in a hippocampus circuit, we build a computational model to study the potential role of linear changes in the synaptic weight and synaptic number. Several key results have been obtained: (i) Changes in the synaptic weight and numbers lead to different long-term modifications; (ii) the first paired spiking from two neurons significantly influences the adjusted subsequent paired spiking; the pre-post spiking pair strengthens the following paired spiking; however, the post-pre spiking pair depresses the subsequent spiking; (iii) when the synaptic weight and synaptic numbers are changed, the interval of the first spiking pair may undergo reduction, and (iv) when we stimulate a stellate neuron weakly or decrease the capacitance of the CA1 pyramidal neuron, LTP is more easily produced than LTD; on the contrary, LTD is more easily produced in an opposite situation; increase in the synaptic numbers can promote activation of the CA1 pyramidal neuron.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Reference
T. V. Bliss and T. Lomo, “Long-lasting potentiation of synaptic transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path,” J. Physiol., 232, 331-356 (1973).
R. C. Malenka and S. A. Siegelbaum, Synaptic Plasticity, Johns Hopkins Univ. Press (2001).
S. J. Martin, P. D. Grimwood, and R. G. Morris, “Synaptic plasticity and memory: an evaluation of the hypothesis,” Annu. Rev. Neurosci., 23, 649-711 (2000).
Y. Dan and M. M. Poo, “Spike timing-dependent plasticity: from synapse to perception,” Physiol. Rev., 86, 1033-1048 (2006).
W. B. Levy and O. Steward, “Temporal contiguity requirements for long-term associative potentiation/depression in the hippocampus,” Neuroscience, 8, No. 4, 791-797 (1983).
G. Q. Bi and M. M. Poo, “Synaptic modifications in cultured hippocampal neurons: dependence on spike timing, synaptic strength, and postsynaptic cell type,” J. Neurosci., 18, No. 24, 10464-10472 (1998).
C. Natalia and Y. Dan, “Spike timing-dependent plasticity: A Hebbian learning rule,” Annu. Rev. Neurosci., 31, 25-46 (2008).
C. Vassilis, C. Stuart, and P. G. Bruce, “Encoding and retrieval in a model of the hippocampal CA1 microcircuit,” Hippocampus, 20, 423-446 (2010).
M. Royeck, M. T. Horstmann, S. Remy, et al., “Role of axonal NaV1.6 sodium channels in action potential initiation of CA1 pyramidal neurons,” J. Neurophysiol., 100, No. 4, 2361-2380 (2008).
H. Peter, E. Daniel, B. Angela, et al., “Distinct classes of pyramidal cells exhibit mutually exclusive firing patterns in hippocampal area CA3,” Hippocampus, 18, No. 4, 411-424 (2008).
W. M. Yamada, C. Koch, and P. R. Adams, Multiple Channels and Calcium Dynamics, MIT Press, Cambridge (1987).
M. Migliore and G. M. Shepherd, “Dendritic action potentials connect distributed dendrodendritic microcircuits,” J. Comput. Neurosci., 24, 207-221 (2008).
R. Michel, M. T. Horstmann, R. Stefan, et al., “Role of axonal Na V 1.6 sodium channels in action potential initiation of CA1 pyramidal neurons,” J. Physiol., 4, 2361-2380 (2008).
L. Wang and S. Q. Liu, “Neural circuit and its functional roles in cerebellar cortex,” Neurosci. Bull., 27, 3, 173-184 (2011).
J. R. Hughes, “Post-tetanic potentiation,” Physiol. Rev., 38, No. 1, 91-113 (1958).
T. D. Joshua, T. David, and A. S. Steven, “A role for synaptic inputs at distal dendrites: Instructive signals for hippocampal long-term plasticity,” Neuron, 56, 866-879 (2007).
J. Tim, R. Alex, L. K. William, and S. Nelson, “Conditional dendritic spike propagation following distal synaptic activation of hippocampal CA1 pyramidal neurons,” Nat. Neurosci., 8, 1667-1676 (2005).
R. R. Clarke and J. P. Stephen, “Spatial segregation of neuronal calcium signals encodes different forms of LTP in rat hippocampus,” J. Physiol., 570, No. 1, 97-111 (2006).
T. Jarsky, A. Roxin, W. L. Kath, and N. Spruston, “Conditional dendritic spike propagation following distal synaptic activation of hippocampal CA1 pyramidal neurons,” Nat. Neurosci., 8, No. 12, 1667-1676 (2005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ren, H.X., Liu, S.Q., Zhang, X.C. et al. Spike Timing-Dependent Plasticity in the CA1 Pyramidal Neuron in a Modeled Hippocampal Circuit. Neurophysiology 47, 264–270 (2015). https://doi.org/10.1007/s11062-015-9532-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11062-015-9532-z