Abstract
Spinal paragangliomas are extremely rare tumors, most frequently involving the cauda equina and the filum terminale. We aimed to investigate the clinical manifestations, radiological features, management, and follow-up data of primary spinal paraganglioma. We present the clinical data and long-term outcomes from a consecutive surgical series of 19 patients with pathologically diagnosed spinal paragangliomas. All of the patients had undergone surgical resection. Pre- and postoperative magnetic resonance imaging was performed and follow-up data and neurological functional assessment are presented and discussed. The mean age at diagnosis was 47.7 years, with a significant male predominance. The primary clinical symptoms were low back pain and sciatica. Magnetic resonance images (MRI) showed characteristic signs that help differentiate paragangliomas from other spinal tumors, including a “salt & pepper” sign, serpiginous flow void, and a peripheral hypointense rim. Also, a well-encapsulated appearance can be found intraoperatively. During a mean follow-up period of 62.1 months, remnant tumor progression was noted on MRI in three patients with incomplete resection. Pain symptoms were relieved immediately after surgical intervention, while motor and sphincter dysfunction were much slower to improve. Differential diagnosis of paraganglioma based on MR images alone is challenging, but the presence of specific characteristic features provides suggestive clues; however, accurate diagnosis depends on pathological criteria. Despite the benign course, gross total resection is ideal, given an increased risk of recurrence in situ. Timely recognition and surgical treatment should be emphasized to avoid progressive neurological deficits.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Paragangliomas are neoplasms originating from the sympathetic or parasympathetic autonomic nervous system, which can be found in adrenal and extra-adrenal locations [1]. Extra-adrenal paragangliomas are rare and can occur throughout the body, most commonly in the carotid bodies and the jugular glomus [2, 3]. Primary spinal paragangliomas are extremely rare, most frequently involving the cauda equina and the filum terminale [2–5]. Paragangliomas are generally considered to be slow-growing tumors, corresponding histologically to World Health Organization (WHO) grade I.
To our knowledge, few clinical reports of primary spinal paraganglioma have been published since the earliest description of paraganglioma of the filum terminale in 1972 [6]. Therefore, the relevant clinical characteristics, management, and outcomes of primary spinal paraganglioma remain obscure, with a lack of studies including a large case series from a single center [2]. We present the clinical data and long-term outcomes from a consecutive surgical series of 19 patients with pathologically diagnosed spinal paragangliomas.
Methods
Materials
Between 2001 and 2014, 218 patients were pathologically diagnosed with paragangliomas in our institution, with 20 cases of primary spinal paragangliomas and the remaining with tumors in the jugular foramen region. One patient was excluded from the study because of a pathological diagnosis of gangliocytic paraganglioma; therefore, a consecutive surgical series of 19 patients with spinal paragangliomas were included. Paragangliomas were diagnosed based on pathological criteria, and reassessed by two neuropathologists, independently. Perioperative magnetic resonance imaging (MRI) with gadolinium-contrast enhancement was performed as standard radiological investigation. We recorded data related to the clinical characteristics, radiological features, pathological findings, treatment, and follow-up outcomes.
Treatment
With intraoperative neurophysiologic monitoring of somatosensory and motor-evoked potentials, all patients underwent surgical resection via the posterior midline approach, and no evoked potentials were lost during the operation. Based on intraoperative findings and postoperative MR images, gross total resection (GTR) was achieved as removal with surgical margins, which were grossly and microscopically tumor-free. The extent of resection was confirmed by postoperative MR imaging and no patients received other adjuvant treatments. For immunohistochemistry, we recorded the percentage of Ki-67-positive cells and degree of staining for synaptophysin, chromogranin A, glial fibrillary acidic protein, neuron-specific enolase, epithelial membrane antigen, creatine kinase, vimentin, and S-100 protein.
Follow-up data
Follow-up data for all but two patients were obtained during individual office visits. The patient’s neurological function and the severity of associated myelopathy was evaluated according to the Japanese Orthopedic Association score designed for lumbosacral disorders (total score of 29 points) [7, 8]. This functional assessment and MR imaging were performed perioperatively, at discharge, at 3 months after surgery, semi-annually or annually thereafter, and at the last clinical visit.
Results
Clinical presentation
A total of 218 patients were diagnosed with paragangliomas based on postoperative histology. Of these, 198 (90.8 %) had paragangliomas in the jugular foramen; one (0.5 %) was diagnosed as having spinal gangliocytic paraganglioma; and 19 (8.7 %) patients with spinal paragangliomas were finally included in the study (Table 1). Patients included seven females and 12 males, with a mean age of 47.7 ± 15.3 years (range, 20–75 years). The duration of symptoms preceding the initial diagnosis ranged from 1 month to 5 years (mean, 21.4 months) and symptoms included low back pain (15 cases, 78.9 %), sciatica (16 cases, 84.2 %), motor deficits (12 cases, 63.2 %), sensory disturbance (10 cases, 52.6 %), and sphincter dysfunction (five cases, 26.3 %). Hypertension was found in four patients, although no significant blood pressure changes were noted perioperatively and during follow-up.
Radiological manifestations
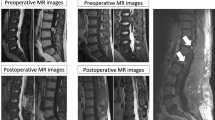
Preoperative MRI was available for all patients. The suspected diagnosis preoperatively included ependymoma, schwannoma, teratoma, meningioma, and hemangioblastoma. All tumors were located at the level of the cauda equina or the filum terminale and the mean dimension of the lesions in the maximum axis was 49.3 mm (range, 17–112 mm). Solid tumors showed homogeneous isointense signal on T1-weighted MR images (T1WI), and isointense or slightly hyperintense signal on T2-weighted images (T2WI). After gadolinium diethylene triamine pentaacetic acid administration, the tumor showed significant homogeneous enhancement. Cystic changes were found in two patients because of intratumoral hemorrhage. The preoperative MR appearance of the paragangliomas appeared similar to other solid tumors; however, certain characteristic findings were observed. A salt-and-pepper appearance, which is considered a characteristic feature on T2WI because of the hypervascular structure, was observed in eight cases (42.1 %, Fig. 1); a peripheral hypointense rim related to hemosiderin content was found in nine cases (47.3 %); and serpiginous flow voids were noted in 11 cases (78.9 %, Fig. 2). No significant peritumoral edema or associated syringomyelia was noted.
Magnetic resonance images demonstrating cystic-solid lesions with intratumoral hemorrhage (arrowheads) in two patients. The solid tumors showed homogeneous isointense signals on sagittal T1-weighted images (a, d) and isointense or slightly hyperintense signal on sagittal T2-weighted images (b, e) with significant homogeneous enhancement after gadolinium diethylene triamine pentaacetic acid administration (c, f). A “salt & pepper” appearance because of the rich vascularization of paragangliomas was noted (arrows)
Magnetic resonance images of two patients showing lesions. Homogeneous isointense signal on sagittal T1-weighted images (a, d); slightly hyperintense signal on sagittal T2-weighted images (b, e); and significant homogeneous enhancement after gadolinium diethylene triamine pentaacetic acid administration (c, f). A peripheral hypointense rim related to hemosiderin (arrowheads) and serpiginous flow voids and feeding arteries (arrows) were also observed
Treatment and immunohistological findings
No patient underwent preoperative embolization or radiosurgery and all patients underwent laminotomy and microsurgical tumor removal via a posterior approach. After the dura mater was opened in the dorsal midline, a grayish mass with a thin fibrous capsule was identified. The feeding arteries were coagulated, and the tumor was then resected en-bloc or piecemeal according to the relationship of the tumor and nerve roots. Every attempt was made to remove the tumor en-bloc because piecemeal removal could lead to extensive bleeding. Paragangliomas are hypervascular but intraoperative bleeding was effectively controlled even without prior endovascular embolization. The lesions were found attached to the cauda equina to varying degrees in 15 cases, and to the filum terminale in four cases. The interface between the tumor and the nerve root was separated with microdissectors and forceps and in 14 cases, and GTR of the tumor with a well-demarcated dissection plane was achieved. However, in the remaining five cases, the tumor was extremely adherent, and subtotal resection (STR) was elected to preserve the nerve roots. Three patients underwent reoperation because of recurrences in situ detected on MRI during follow-up, and ultimately GTR was achieved by sacrificing the nerve rootlets in these patients. We encountered no intraoperative blood pressure or heart rate changes suggesting potential catecholamine secretion and no patients received adjuvant radiotherapy or chemotherapy.
Histopathological examination showed a typical nesting “Zellballen” pattern with abundant interstitial vascularity with tumor cells forming lobules and the nests separated by cellular cords or fibrovascular septa (Fig. 3). Immunohistochemical staining showed that all of the available cases were positive for synaptophysin, chromogranin A, and neuron-specific enolase, but negative for glial fibrillary acidic protein, epithelial membrane antigen, and creatine kinase. Also, vimentin was significantly positive in 11 patients (58 %), and S-100 protein was positive in 16 (84 %). The positive expression rate for Ki-67 was <10 % (Table 2).
Histopathological photomicrographs of paraganglioma. Images show typical nesting “Zellballen” pattern with abundant interstitial vascularity (a, arrowheads). The tumor cells formed lobules and the nests were separated by cellular cords or fibrovascular septa (b, arrows) (hematoxylin and eosin stain, original magnification: ×100)
Postoperative course and functional evaluation
The postoperative course was uneventful in this cohort, with no significant complications noted and no mortality or metastasis. Among the five patients who underwent incomplete tumor removal, three were admitted to our institution for reoperations because of residual tumor regrowth and GTR was ultimately achieved. The mean interval of recurrence was 8.8 months. During the mean follow-up period of 62.1 months (standard deviation = 37.4, range 11–139), no recurrence after GTR was observed, and no evidence of residual tumor regrowth was shown on MRI in the remaining two patients following STR. Postoperatively, all patients experienced improved symptoms to varying degrees. The low back pain and sciatica was relieved in all patients immediately after operation, while the neurological functional recovery was relatively difficult. According to the JOA scores, two patients experienced complete remission following surgery; significant efficacy was achieved in nine patients; and operations were effective in the remaining eight patients. At the last follow-up, there were no complaints of pain, and paresthesia was partially relieved in six patients. No significant improvement in motor or sphincter function was observed, but one patient showed slightly improved lower extremity weakness. No significant blood pressure changes were noted during follow-up.
Discussion
Epidemiology and clinical features
Paragangliomas are relatively rare tumors that develop from the paraganglia of the autonomic nervous system [1]. They can be found in adrenal and extra-adrenal tissues, and the extra-adrenal paragangliomas can be divided into sympathetic and parasympathetic types. The sympathetic paragangliomas are usually secretory and produce catecholamines [9, 10], while parasympathetic paragangliomas tend to be non-secretory [4]. As previously reported, paragangliomas in the central nervous system are predominantly found at the bifurcation of the common carotid artery and in the middle ear regions, occupying approximately 80–90 % of all paragangliomas. These are predominantly parasympathetic without remarkable symptoms [11]. Spinal paragangliomas are extremely rare, and they are usually located in the lumbar and sacral regions [2, 4], but precise data about the prevalence of spinal paragangliomas remain undetermined [3, 4]. In the current study, intraspinal paragangliomas constituted approximately 9 % of all paragangliomas confirmed pathologically and all of the tumors presented as intradural, extramedullary tumors at the level of the cauda equina and the filum terminale, which is consistent with previous reports. The male to female ratio was 1.7:1, similar to previous reports of male predominance [1]. No distinct age distribution was noted. The average duration of symptoms preceding diagnosis was 21.4 months, which is significantly shorter than reported [12]. No significant correlation was observed between the duration of symptoms and tumor dimension.
Spinal paragangliomas can present with symptoms of radiculopathy or slowly progressive spinal cord compression. Similar to previous reports, low back pain and sciatica were the most common clinical presentations in the present study, and sensory-motor and sphincter dysfunction can develop along with clinical progression [1, 5]. Very few previously reported cases presented with sympathetic secretory symptoms related to catecholamine and in our case series, no significant blood pressure or heart rate changes were noted peri-operatively and during follow-up, ruling out a diagnosis of secreting paraganglioma.
Diagnosis and clinical considerations
Preoperative diagnosis of paragangliomas based on MRI features alone is challenging because of their rarity [1, 5]. On MRI, paragangliomas are generally hypointense to isointense on T1WI and isointense to hyperintense on T2WI, and contrast-enhanced T1WI homogeneity delineates the tumor [4, 13]. It should be noted that the following potential characteristics can assist the diagnosis of paraganglioma: a “salt & pepper” appearance on T2WI because of the rich vascularization of paragangliomas, serpiginous flow void, peripheral hypointense rim related to hemosiderin, and a location at the level of the cauda equina or the filum terminale [1, 3–5]. Differential diagnoses include ependymoma, schwannoma, meningioma, teratoma, and hemangioma [1]. Intratumoral hemorrhage can also be found in sporadic cases, posing greater challenges for diagnosis [14].
A definitive diagnosis depends on pathological examination, which shows a unique “Zellballen” pattern as a solid mass of paraganglion-type nests or cords [2, 3, 5]. Immunohistochemical staining can assist the diagnosis [15] and positivity for S-100 protein, which has a specific high positive rate in low-grade tumors, and a low-level positive expression rate of Ki-67 may indicate benign spinal paragangliomas [2, 4, 12].
Therapeutic options and surgical outcomes
Paragangliomas are considered slow-growing benign tumors, corresponding to WHO grade I, histologically. While less than 1 % of paragangliomas are reported with aggressive behavior in extremely rare cases [2, 16–18], and they are susceptible to long-term control by total resection, GTR is the ideal treatment. Considering the hypervascular nature of paragangliomas, preoperative embolization is advocated for jugular foramen paragangliomas in some reports [14, 19, 20]. In our experience, spinal paragangliomas are removable with manageable intraoperative bleeding, and preoperative embolization may not be necessary. As they are most often located in the intradural, extramedullary compartment, and the masses are well encapsulated with a distinct vascular pedicle intraoperatively, total en-bloc resection should be attempted [1, 4, 5]. The main technical problem preventing GTR is dense adhesion to nerve roots [21]. If GTR is not possible, STR can be effective for pain relief and neurological functional improvement [15]. With meticulous operation and elaborate care, the postoperative course can be uneventful, and complications such as infection, pressure ulcers, and venous thrombosis can be avoided.
Pain symptoms can resolve completely with operation, but neurological functional recovery is prolonged. After complete excision, no focal recurrence or metastasis was observed during the follow-up period in our study, while three of the five patients undergoing incomplete removal were readmitted because of in situ recurrence. Our institutional experience supports that spinal paragangliomas have a benign course and a favorable prognosis [22]. However, the natural history of spinal paragangliomas remains unclear because of a lack of large-sample dynamic observations. Considering the relatively short preoperative clinical course and recurrence interval, timely surgery and GTR should be emphasized to avoid severe progressive neurological deficits and to reduce the risk of residual tumor recurrence [21, 23].
Adjunctive therapy
To date, the role of radiotherapy and chemotherapy for spinal paragangliomas is undetermined. According to the literature regarding intracranial counterparts, radiotherapy cannot help tumor regression, but many published series following conventional radiotherapy report 10-year tumor control rates of 90–100 % [24–26]. Radiotherapy is effective for long-term control including stabilization and an absence of recurrence of symptoms, and absence of tumor growth and radiological signs of progression [24]. Stereotactic radiotherapy has been reported useful to irradiate small intracranial paragangliomas, achieving long-term control [27]. These authors propose radiotherapy as a first-line treatment for intracranial paragangliomas, particularly in patients with no preoperative deficits. Chemotherapy has been reported in a few sporadic cases with extra-CNS malignant paraganglioma, but the efficacy is controversial [28]. There are also a small number of studies suggesting that radiotherapy and chemotherapy have insignificant efficacy, proposing that these should be used only in patients who cannot tolerate surgery and in those with metastases [4, 18].
In our cohort with spinal paragangliomas, the primary goal was removal of the space-occupying mass and decompression; therefore we proposed surgery as the preferred treatment strategy. Considering the dull biological activity and indolent natural history, we did not recommend postoperative radiotherapy, even in the cases with STR. However, remnant tumor progression was noted on follow-up MRI in three patients who underwent incomplete resection. Therefore, a more aggressive strategy is recommended in patients with spinal paragangliomas, and postoperative adjuvant radiotherapy might be helpful when GTR is difficult to achieve [29, 30].
Conclusion
Spinal paraganglioma is an extremely rare entity, with a benign clinical course. Certain characteristics on MRI could suggest the diagnosis, including “salt & pepper” appearance, serpiginous flow void, peripheral hypointense rim, and predilection for the lumbosacral segment. En-bloc GTR is ideal. Surgery is effective for pain relief and varying degrees of neurological functional improvement. Postoperative radiotherapy may be helpful for long-term tumor control following incomplete resection. Early recognition, correct diagnosis, and timely surgical treatment are important to avoid progressive neurological deficits.
References
Caruso R, Wierzbicki V, Marrocco L, Salvati M (2006) Paragangliomas of the cauda equina. Report of one case and review of the literature. J Exp Clin Cancer Res 25:269–275
Moran CA, Rush W, Mena H (1997) Primary spinal paragangliomas: a clinicopathological and immunohistochemical study of 30 cases. Histopathology 31:167–173
Yang SY, Jin YJ, Park SH, Jahng TA, Kim HJ, Chung CK (2005) Paragangliomas in the cauda equina region: clinicopathoradiologic findings in four cases. J Neurooncol 72:49–55
Conti P, Mouchaty H, Spacca B, Buccoliero AM, Conti R (2006) Thoracic extradural paragangliomas: a case report and review of the literature. Spinal Cord 44:120–125
Sundgren P, Annertz M, Englund E, Stromblad LG, Holtas S (1999) Paragangliomas of the spinal canal. Neuroradiology 41:788–794
Lerman RI, Kaplan ES, Daman L (1972) Ganglioneuroma-paraganglioma of the intradural filum terminale. Case report. J Neurosurg 36:652–658
Nakamura M, Miyamoto K, Shimizu K (2009) Difference in evaluation of patients with low back pain using the Japanese Orthopaedic Association Score for Back Pain and the Japanese Version of the Roland-Morris Disability Questionnaire. J Orthop Sci 14:367–373
Ohtori S, Ito T, Yamashita M et al (2010) Evaluation of low back pain using the Japanese Orthopaedic Association Back Pain Evaluation Questionnaire for lumbar spinal disease in a multicenter study: differences in scores based on age, sex, and type of disease. J Orthop Sci 15:86–91
Jeffs GJ, Lee GY, Wong GT (2003) Functioning paraganglioma of the thoracic spine: case report. Neurosurgery 53:992–994; discussion 994–995
Chen G, Luo Z, Liu T, Yang H (2011) Functioning paraganglioma of the cervical spine. Orthopedics 34:e700–e702
Destito D, Bucolo S, Florio A, Quattrocchi C (2012) Management of head and neck paragangliomas: a series of 9 cases and review of the literature. Ear Nose Throat J 91:366–375
Sonneland PR, Scheithauer BW, LeChago J, Crawford BG, Onofrio BM (1986) Paraganglioma of the cauda equina region. Clinicopathologic study of 31 cases with special reference to immunocytology and ultrastructure. Cancer 58:1720–1735
Lee KY, Oh YW, Noh HJ et al (2006) Extraadrenal paragangliomas of the body: imaging features. AJR Am J Roentgenol 187:492–504
Demircivi OF, Aydin M, Bezircioglu H, Oran I (2010) Paraganglioma of the cauda equina: a highly vascular tumour. J Clin Neurosci 17:1445–1447
Hong JY, Hur CY, Modi HN, Suh SW, Chang HY (2012) Paraganglioma in the cauda equina. A case report. Acta Orthop Belg 78:418–423
North CA, Zinreich ES, Christensen WN, North RB (1990) Multiple spinal metastases from paraganglioma. Cancer 66:2224–2228
Lau D, La Marca F, Camelo-Piragua S, Park P (2013) Metastatic paraganglioma of the spine: case report and review of the literature. Clin Neurol Neurosurg 115:1571–1574
Hamilton MA, Tait D (2000) Metastatic paraganglioma causing spinal cord compression. Br J Radiol 73:901–904
Lehmen JA, Babbel DM, Mikhitarian K, Choma TJ (2010) Paraganglioma presenting as metastatic lesion in a cervical vertebra: a case report and review of the literature. Spine (Phila Pa 1976) 35:E152–154
Makiese O, Chibbaro S, Marsella M, Tran BHP, George B (2012) Jugular foramen paragangliomas: management, outcome and avoidance of complications in a series of 75 cases. Neurosurg Rev 35:185–194; discussion 194
Fitzgerald LF, Cech DA, Goodman JC (1996) Paraganglioma of the thoracic spinal cord. Clin Neurol Neurosurg 98:183–185
Amato B, Bianco T, Compagna R et al (2014) Surgical resection of carotid body paragangliomas: 10 years of experience. Am J Surg 207:293–298
van Heerden JA, Roland CF, Carney JA, Sheps SG, Grant CS (1990) Long-term evaluation following resection of apparently benign pheochromocytoma(s)/paraganglioma(s). World J Surg 14:325–329
Tran BHP (2014) Radiotherapy for glomus jugulare paraganglioma. Eur Ann Otorhinolaryngol Head Neck Dis 131:223–226
Dupin C, Lang P, Dessard-Diana B et al (2014) Treatment of head and neck paragangliomas with external beam radiation therapy. Int J Radiat Oncol Biol Phys 89:353–359
Kiziltan HS, Ozucer B, Eris AH, Veyseller B (2014) Bilateral carotid paraganglioma: surgery and radiotherapy. Clin Med Insights Case Rep 7:53–57
Navarro MA, Maitz A, Grills IS et al (2010) Successful treatment of glomus jugulare tumours with gamma knife radiosurgery: clinical and physical aspects of management and review of the literature. Clin Transl Oncol 12:55–62
Niemeijer ND, Alblas G, van Hulsteijn LT, Dekkers OM, Corssmit EP (2014) Chemotherapy with cyclophosphamide, vincristine and dacarbazine for malignant paraganglioma and pheochromocytoma: systematic review and meta-analysis. Clin Endocrinol (Oxf) 81:642–651
Li ZJ, Lan XL, Hao FY et al (2014) Primary cerebellar paraganglioma: a pediatric case report and review of the literature. Pediatr Neurol 50:303–306
Suarez C, Rodrigo JP, Mendenhall WM et al (2014) Carotid body paragangliomas: a systematic study on management with surgery and radiotherapy. Eur Arch Otorhinolaryngol 271:23–34
Acknowledgments
We thank all of the patients who trusted us with their care, and all of the physicians and staff who helped in this study.
Compliance with Ethical Standards
The authors declare that they have no conflict of interest. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, C., Li, G., Fang, J. et al. Clinical characteristics and surgical outcomes of primary spinal paragangliomas. J Neurooncol 122, 539–547 (2015). https://doi.org/10.1007/s11060-015-1742-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-015-1742-0