Abstract
Cut stumps are the legacy of forest harvesting and allow for the estimation of carbon loss from the ecosystem. The objective of this study was to estimate aboveground carbon storage and the impact of forest harvesting based on measurements of cut stumps in pine forests within the Área de Protección de Flora y Fauna Nevado de Toluca (APFFNT) in the state of México. A total of 1621 circular 0.1-ha sampling plots were established in a 12,924 ha pine forest (1.25% sampling intensity). Biomass was calculated using conventional volumetric equations incorporating species-specific wood densities. Aboveground carbon storage in the biomass was stratified by forest type including dense forest (≥ 336 trees), semi-dense forest (< 335 and > 150 trees), fragmented forest (< 150 and > 20 trees), and stands with isolated trees (< 20 trees). In the 12, 924 ha of forest dominated by Pinus hartwegii, 1,695,004 MgC was contained in aboveground biomass. Data on forest harvesting obtained by analysis of recent cut stumps allowed for the estimation of the removal of 42,701 MgC. In addition, we accounted for carbon in wind thrown trees of 14,904 MgC, some of which were removed over time in harvests. Total loss of carbon from the forest corresponded to 211,218 MgCO2 per year. In the APFFNT, forests dominated by pine with high biomass (P. hartwegii and P. montezumae) are being replaced by those with lower biomass (Alnus jorullensis). Changes in these communities will result in lower carbon capture in the APFFNT.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The carbon (C) cycle comprises fluxes that depend on physiochemical and biogeochemical processes, such as photosynthesis, plant respiration and soil heterotrophic relationships (Cao and Woodward 1998). C sinks and natural sources regulate these fluxes. The oceans, the atmosphere, and forests are examples of C sinks; while plant respiration, decomposition of organic matter, and fires are examples of sources of C (Falkowski et al. 2000). The function of terrestrial ecosystems has been affected in the past 200 years by an imbalance in the C cycle, reflected by an increase in atmospheric CO2 concentration from 280 to 400 ppm as of 2015 (Dlugokencky et al. 2016). The principle causes of this increase have been emissions of CO2 from the use of fossil fuels and the impact of changes in land use (IPCC 2007). Furthermore, deforestation is the second largest source of anthropogenic greenhouse gas emissions (Gullison et al. 2007).
Vegetation acts as a C sink and mitigates increases in atmospheric CO2 (Brown 2002), however the CO2 stored in vegetation can return to the atmosphere through the rate of respiration from plants, decomposition or occurrence of wildfire. Plant communities can be a source of C due to natural disturbances or human activities (Rojo et al. 2003). The loss of forest cover generates impacts at different scales. At landscape scale, it includes the degradation of soils, the loss of ability of biological systems to support human needs and the increased vulnerability of regions in situations of climatic disturbances (Lambin et al. 2003). At local level, it causes a decrease both in material goods and in the environmental services provided by the forest (MEA 2003), among them the C storage and capture (Lambin et al. 2003).
One of the strategies involved in decreasing the rate of loss in forest cover in Mexico is the establishment of Protected Natural Areas. The legal definition of these protected areas and their creation through presidential decree calls for special protection that promotes the storage of C in forests over the long term (Rojas-García 2008). The General Law of Ecological Equilibrium and Environmental Protection establish the activities permitted in such protected areas, between them protection, management, restoration, research, cultural and leisure. The National Commission of Protected Natural Areas administers 176 federal units representing 25.4 million ha. Protected areas in central México are present in mountainous areas where most previous studies have been on the response of forests to environmental changes and the growing areas of timber harvesting (Villers and Trejo 1998). One of the most utilized practices since the 1980s is the mitigation of environmental degradation through reforestation programs. These programs have mainly focused on conservation impacts than on forest harvesting processes, particularly, on the appropriate site selection for planted tree species (Rescala 2009).
The Forest Inventory of the State of Mexico indicates that 48% of the state is covered by forests, with 95% of these forests being temperate with different degrees of disturbance (PROBOSQUE 2010). Collado and Serrato (2009) estimated that, from 1900 to 2001, there was a 53% loss in forest cover in the state, which has caused temporary halts to logging because of deforestation and problems in the forest industry. The loss of temperate forests has had negative effects including soil degradation, reduction in watershed services, loss of diversity and increased pest outbreaks (GEM 1999).
The Intergovernmental Panel on Climate Change (IPCC) identified five C storage areas in plant communities: live aboveground biomass, live belowground biomass, dead aboveground biomass, leaf litter and soil. The storage of C in dead aboveground biomass includes a wide variety of stem types and diameter sizes that includes standing dead trees and fallen logs, stumps, fallen branches and roots (IPCC 2003).
Dead wood is a key component in the flow of energy and nutrients, it is fuel for potential wildfires and is one of the most important C storage components in natural forests (Baker et al. 2007; Chao et al. 2009). When a tree dies of natural causes or it is harvested for timber, its C enters one of the three reservoirs: woody debris, wood products, or the atmosphere (GOFC-GOLD 2010). The duration of C storage in dead wood in natural forests depends on the rates of mortality and decomposition (Palace et al. 2007).
Cut stumps are the evidence of forest harvesting, allowing for the estimation of C loss from the ecosystem. From them, it is possible to estimate volume, biomass, and amount of C removed from an ecosystem and allows for calculations of C emissions from timber harvesting. The Área de Protección de Flora y Fauna Nevado de Toluca (APFFNT), in the state of Mexico presented changes in land use that converted forested areas to agricultural land. This conversion occurred in spite of the fact that some areas were not suitable for agriculture. Furthermore, evidence of illegal harvesting was found, such practice can be responsible for the rise of wildfire probabilities, forest fragmentation and for the reduction of pest and disease outbreaks resistance (CONANP 2017). Due to the above, the objective of this study was to estimate aboveground C storage in forest trees and to assess the impact of forest harvesting based on measurements of cut stumps in pine forests inside the APFFNT.
Methods
Study area
The APFFNT is located in the state of Mexico, which includes the Nevado de Toluca volcano. The altitudinal range is 3000–4680 m asl. The protected area is located in the southern region of the state, comprising a surface of 53,590 ha. It was declared a national park in 1936 with the objective of safeguarding its scenic quality and hydrological importance (SEMARNAT 2013).
Andisols are the predominant soil type, covering 90% of the park, as well as feozem, regosol, cambisol, fluvisol and leptosol types, which are the products of extrusive igneous rocks of Tertiary and Quaternary origins (Sotelo et al. 2010). The instability of the soil structure, along with deforestation, overgrazing, steep slopes and torrential rain events, can generate intense soil erosions (Sánchez-Jasso et al. 2013).
The Nevado de Toluca volcano presents a vertical thermal gradient. Above the 3700 m altitude, the climate is cold with a summer rainfall regime (E(T)Hwig). Below this altitudinal elevation and up to 2800 m, the climate surrounding the volcano is subhumid, semi-cold with rainfall occurring in the summer (C(E)wig) (García 1981).
Sampling methods
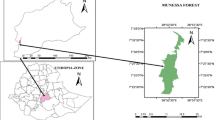
The heterogeneity of the terrain and climate in the park were considered in developing a sampling protocol that included estimates of species diversity, tree sizes and tree conditions. Circular sampling plots of 0.1 ha (17.86 m in radius) were distributed systematically to each 200 m along transects, following the elevation contours separated by of 100 m, by above 3400 m. The distribution of plots allowing include changes in forest structure associated with climatic variation due to the elevation (Mayer and Ott 1991) (Fig. 1).
The 1621 sampling plots were established in 12,924 ha of pine forest occurring between August 2016 and August 2017. The sampling intensity recommended for the surface is 0.44% (Dauber 1995), in the present study a 1.25% was reached.
Plots were located using topographic maps and the center of UTM coordinates, and the altitude (m) was recorded with a GPS Rhino® 650 Garmin. For each sampling plot, we recorded the slope and aspect with a compass and clinometer (Tandem Suunto).
The trees that had ≥ 7.5 cm diameter breast height (DBH, 1.30 m aboveground) were measured for DBH (cm), total height (m), and condition (e.g., presence of parasitic plants, disease). For standing dead trees or trees fallen in the past year, we observed the species, DBH (cm) and total height (m). The stumps that conserved their structural integrity, presence of resins and sawdust, lack of understory vegetation around or evidence of damage by felling of the surrounding trees, were measured. The diameter of the cutting surface (dt, 0.30 m above the ground) was recorded.
We additionally observed dominant tree species, percentage cover of herbaceous and shrub species, and any evidence of recent fire. The relative dominance of species in each plot by percent basal area was estimated (Kent and Coker 1992). The density of forests was defined by Endara et al. (2013), in dense forest (≥ 336 trees), semi-dense forest (150–335), fragmented forest (21–150), and isolated trees (≤ 20) (10% of the density over all plots).
Estimation of C storage in forest biomass
The stemwood volume in each measured individual tree was calculated using species-specific volumetric equations, developed by the National Forest and Soils Inventory (CONAFOR 2012):
where V = volume (m3); DBH = diameter at breast height (cm); HT = total height (m) and a(n) = species-specific coefficients.
To convert volume to biomass, it is necessary to include basic wood density that allows for conversion of wet weight to dry weight (Brown et al. 1989). According to the recommendation of Brown and Lugo (1984) in the calculation of individual tree biomass, a multiplier of 1.3 for broad-leaved trees and 1.18 for conifers, the stem expansion factor (SEF), to include branch and foliar biomass is:
where B = biomass (kg), V = volume (m3); D = wood density (kg/m3), SEF =stem expansion factor (index). Table 1 shows the estimated wood density values for each species in mountain forests according to Markwar and Meck (Echenique and Díaz 1972).
The C content of individual trees based on biomass, which corresponds to 45–50% of dry weight, the most conservative value was used in this study (45%) (IPCC 2003).
Estimation of C loss due to timber harvesting
We used an equation developed in Mexico to estimate the DBH of pine trees based on measurements of the diameter of the stump (dt) (cm) (Corral et al. 2007):
Once the DBH was known (cm), the HT (m) was estimated by means of a correlation of the data of 30,599 individual trees measured (R2 = 0.849):
to determine the volume of the wood (m3), and later the biomass, following the procedure of the live trees.
The values of the V, B and C content per sampling plot was obtained by adding the individual values of each individual tree in 0.1 ha. An average value of volume, biomass and C content of each of the sampling plots was obtained. Finally extrapolated per hectare into m3 ha−1 for the volume, megagrams for biomass (Mg ha−1) and megagramos of C (MgC ha−1), considering the type of vegetation and the density of trees (Padmakumar et al. 2018). Subsequently, a conversion was made based on the molecular C equivalent (44/12), to estimate C emissions.
Results
Relative dominance and tree density
The relative density of natural pine forests within the 12,924 ha area included five monospecific communities of pine and 16 pine-dominated communities of the total 22 plant communities. Table 2 shows the basal area and density in plant communities representative of the four classes of forest density. In this study, we included areas with isolated trees that corresponded to 10% or less of the mean total density or basal area.
The evaluation of the inventory data allowed the identification of dendrometric characteristics of the forests that grow in the Nevado deToluca, which are described below. P. hartwegii forests occur on the slopes of the volcano from 3300 to 4375 m in altitude. The forests are monospecific from 3700 m up to the tree line. At lower elevations, forests are dominated by A. jorullensis, A. religiosa and P. montezumae. Individuals in this community have an average height of 18 m and an average DBH of 26 cm; however, DBH, height and the DBH to height ratio show a similar behavior along the altitudinal gradient (Fig. 2). An increase of these three characteristics is observed at 3000 m and at the elevation ranging from 3700 to 4100 m asl. Variation in tree size and allometry is attributable to environmental conditions such as temperature, precipitation, stone content in soil, and aspect. The shrub stratum has low density with a few individuals of Penstemon gentianoides (Kunth) Poir., Eupatorium glabratum Kunth and Baccharis conferta Kunth, Cirsium nivale (Kunth), Robinsonecio gerberifolius (Sch. Bip. ex Hemsl.) T.M.Barkley and Janovec Sch. Bip. The herbaceous plant stratum is represented by grass species, such as Muhlenbergia quadridentata (Kunth) Trin., M. macroura (Kunth) Hitchc. and Festuca tolucensis Kunth. Semi-woody shrubs, such as Eryngium proteaeflorum F. Delaroche and Lupinus aschenbornii S. Schauer in highly disturbed sites, are much less common.
A. jorullensis forests occur in stands frequently mixed with A. religiosa or Pinus. This community occurs between 3200 and 3500 m. Trees had an average diameter of 30 cm and varied in height from 5 to 30 m. Roldana angulifolia (DC.)H. Rob. and Brettell is dominant in the shrub stratum; and in the herbaceous stratum Astragalus guatemalensis Hemsl. and Stellaria cuspidata Willd. ex Schltdl are the most common species.
A. religiosa forests are distributed in pure stands or mixed stands with Pinus between 2800 and 3500 m asl and have a mean DBH of 30 cm and a height between 15 and 45 m. These forests occur on sites that are sometimes totally covered in moss and have a shrub stratum dominated by Senecio cinerarioides Kunth., Baccharis conferta Kunth, Ageratina glabrata (Kunth) R. M. King and H. Rob., Asplenium monanthes L. and Fuchsia thymifolia Kunth. In the herbaceous stratum, common species include Festuca amplissima Rupr., Castilleja arvensis Schltdl. and Cham., Salvia elegans Vahl., Senecio callosus Sch. Bip. and Vicia pulchella Kunth. They occur in pure stands or in mixed stands with pine species or in association with pine and fir.
Pinus montezumae forests are distributed in isolated stands between 3000 and 3200 m in the center of the protected area. Trees in this forest are 15–30 m in height and have an average DBH of 30 cm. Inside these forests include species of Pinus, as well as Q. laurina, A. religiosa, A. xalapensis, A. jorullensis, S. cana and B. cordata. The shrub stratum is dominated by Arctostaphylos pungens Kunth, Roldana barba-johannis (DC.) H.Rob. and Brettell and Pernettya prostrata (Cav.) DC. In the herbaceolus stratum Muhlenbergia macroura, Penstemon campanulatus (Cav.) Willd., Lupinus montanus Kunth, Castilleja tenuiflora Benth., Salvia laevis Benth., Cirsium subuliforme Ownbey and Senecio sinuatus Gilib are common.
Wood volume and biomass
Table 3 describes wood volume and aboveground biomass in seven major plant communities and four density classes in the protected area. Wood volume and aboveground biomass presented a similar behavior along the altitudinal gradient. This behavior of the variables is more closely relate to the basal area than to the density of trees (Fig. 3).
Density, wood volume and aboveground biomass of live trees of Pinus hartwegii in an altitudinal gradient. Density represented with bars; wood volume (m3 ha−1) represented with triangular markers and aboveground biomass (Mg ha−1) represented with quadrangular markers. Basal area (m2 ha−2) represented with a continuous black line shows on the secondary axis
Table 4 shows the number of stumps, the estimated volume and the aboveground biomass present in cut trees. Forests that were the most exploited were dominated by pine regardless of density class. Despite P. montezumae having the lowest density in forests of Nevado de Toluca, it is the most harvested specie, due to the stem size and wood density. In P. hartwegii stands, there was an average of 18.69 Mg of aboveground biomass harvested in all density categories. In P. hartwegii–A. religiosa and A. jorullensis-P. hartwegii forests, P. hartwegii stems were the most abundant, followed by those of A. religiosa. In general, A. jorullensis trees were not preferentially harvested due to the low density of the wood.
Impact of harvesting on C storage
Table 5 shows the mean C storage in situ aboveground biomass and C removed due to harvesting in the seven communities and four density classes in high mountain forests.
Figure 4 shows the distribution of C in aboveground forest biomass in the protected area. A total of 1,695,004 MgC was stored in the 12,924 ha area (mean 131.1 MgC ha−1). The harvesting data is in agreement with the survey of recently cut stumps, which allows for a defensible estimate in which 2.5% of C storage was removed from the protected area during the year of sampling (42,701 MgC). In addition, there was an estimated 14,903 MgC in fallen trees because of windthrow, although some of these trees had been removed during harvesting. Estimates are that 211,218 MgCO2 were emitted during the year of the study.
Discussion
Storage of C in montane forests
Forest ecosystems are often seen as potentially large C sinks and plant biomass providers (Augusto et al. 2015). Improved knowledge of carbon stocks and fluxes is need in order to understand the current state of the C cycle and how it might evolve with changing land use and climatic conditions (Hollinger 2008).
Trees in temperate forests in México have a large economic importance and are the base of the forest industry in the country. However, the pursuit of suitable climates and fertile soils for agriculture are causing severe deforestation and degradation in this ecosystem (Challenger 1998).
Rzedowski (1978) calculated that between 50 and 67% of the original areas of these forests have been converted to other uses and now cover only 13% of the nation (Flores-Villela and Gerez 1994). These ecosystems are among the best represented within the national system of protected natural areas but, the majority of these areas are near large cities where they have been subjected to large human impacts, such as the case with the protected area of this study (Challenger 2003).
The Nevado de Toluca protected areas have been subjected to intensive human disturbances, such as potato farming and cattle ranching in recent years that have extended to elevations higher than 3700 m. The vegetation that develops in the APFFNT is the result of its geographic and altitudinal position conjugated with the characteristics of relief, geology, edaphic conditions and climate. Currently, it is possible to observe a mosaic of plant communities that vary with different tree densities within the protected area.
The high sampling intensity used in this study allowed the characterization of aboveground C storage in montane forests where composition, density, and dominance of species can change rapidly over small changes in altitude (Vázquez and Givnish 1998; Sánchez-González and López-Mata 2003). The inventory also permitted us to understand important aspects of forest communities, particularly, the distribution of resources among different species through indirect estimate of biomass, such as plant cover and basal area (Mueller-Dombois and Ellenberg 1974; Whittaker 1975). The density classes of the high mountain forests proposed in this study agree with the measurements made on tree densities reported in other studies in the Nevado de Toluca (Villers et al. 1998; Endara et al. 2013).
The magnitude of C storage in the APFFNT is very important because it contains 1,695,004 MgC, although this storage had been diminished in recent years by 42,701 MgC due to forest harvesting. In multi-ages P. hartwegii forests, the application of thinning of an intensity not greater than 30% of the initial density (expressed in number of trees per hectare and the uniformly distributed cuttings by age classes) is recommended. In this way, a sustained yield of 4.74 m3 ha−1 year−1 would be guarantee (Sarukhán and Franco 1981).
It is important to establish strategies into management program of APFFNT that promotes the preservation, restoration and health of forest communities in the natural protected area (Rojas-García 2008). It was found in this work that the extraction of trees is the same in all the stand density categories (37.6 m3 ha−1 year−1). It is suggest that the thinning practices be carried out every 10 years, preferably in the dense and semi-dense stands of P. hartwegii. In this way, it is possible to achieve the highest annual yields in timber volume and thereby increase the C sequestration.
Further studies have measured the storage of C in the forests of P. hartwegii in different parts of mountainous Mexico, these data agree with the estimates made in this study, where on average in dense forests (268 individuals ha−1) 129.51 MgC ha−1 has been stored. In the La Malinche National Park 111.35 MgC ha−1 were estimated in a dense forest (320 individuals ha−1; Rojas-García 2008). For a similar type of dense forest at the Perote Natonal Park, Mendoza and Galicia (2010) reported an average C content of 146.3 MgC ha−1.
Ecological impact of timber harvesting and implications for C storage
The estimation of aboveground biomass allows, first, to know the status of the forest at a given time to analyze how management can affect some aspect of the ecosystem, including the timber extraction (Vilanova et al. 2010). Second, if there is information collected in the long term, it is possible to evaluate changes and compare them in different periods (WWF 2004).
De Jong et al. (2010) noted that montane forests represent the large C storage area in Mexico. The wood volume contained in these forests is among the lowest in the country, but growth and regeneration is among the highest, although the continued development largely depends on management conditions (Beaman 1962; Anaya et al. 1980).
The extraction of timber, firewood and non-wood products in the foothills has occurred from the middle elevation foothills to the high elevation mountains with negative environmental impacts on the protected area. Along with this, weather variables (wind, frost, etc.), the impact of pests and diseases, and the occurrence of wildfires are factors that have been important in the modification and fragmentation of forest cover (SEMARNAT 2013). During March 2016, the eleventh winter storm and cold front number 45 from northern Mexico caused strong winds in central country, reaching 80 km/h. The strong winds brought down numerous trees in the P. hartwegii forest. These trees were measured and their aboveground biomass on the ground corresponds to14,903 MgC.
The politico-administrative condition of the Nevado de Toluca Protected Area and the lack of protection and control mechanisms lead to clandestine logging of trees for diverse products by unauthorized traders. Some of the common uses of these products are for furniture and construction wood, charcoal, and posts, the problem has extended to felling of large trees with diameters > 35 cm and heights of 20 m (Franco et al. 2006).
The loss of forest cover and tree density has not been homogeneous with the protected area. According to Endara (2010), the majority of trees harvested for all species are those less than 30 cm in diameter and used for firewood. Pinus forests are the most subjected to disturbance and have been the target of reforestation campaigns, often planted in mixtures with cedar (Cupressus spp.) (Endara 2007). The current major sources of damage and continuing threat to these forests is from pasture burning, conversion to agriculture, overgrazing, and overharvesting because of its important as a wood resource to the nation.
A. jorullensis is a successional plant community that often occurs in ecotonal boundaries mixed with pine and fir at elevations around 3500 m. A. jorullensis forests have few harvestable trees because of its value for artisanal wood. Species has a rapid growth rate and is increasing in abundance because of forest fragmentation and colonization of abandoned farmland (Endara 2010). It is possible that these forests have occupied area where P. hartwegii forests were harvested, burned areas or through long-term displacement at higher altitudes. The establishment of A. jorullensis is also associated with natural selection in montane forests because it is a nitrogen-fixing species (Tobita et al. 2016), that promotes regeneration of forests (Kumar and Ram 2005).
Species in montane ecosystems tolerate dramatic climate variation over short distances (Körner 2007) and displacement of species induced by climate change possible will cause changes in these communities with elevation (Hartl-Meier et al. 2014). Within the APFFNT, there has been a displacement of species with higher biomass (forests of P. hartwegii and P. montezumae) by those with lower biomass (A. jorullensis). This change could negatively affect CO2 capture in this protected area.
Monitoring of logging is essential in all of the protected areas within the country, because extraction estimates are based on land use change determined through remote sensing (Rojas-García 2008). The inventory measurements carried out in the present work allowed us to estimate that 162,230 m3 of wood volume were extracted in a year. Data on logging is necessary to calculate emissions of greenhouse gases attributed to land use change and forestry (De Jong et al. 2010).
Initiatives such as REDD + (Reducing Emissions from Deforestation and Forest Degradation and improving carbon stocks) are important efforts aimed at combating climate change. However, for the effective implementation of such mechanisms, accurate estimation and monitoring of aboveground biomass and associated C reserves in forests is first required (Salimon et al. 2011).
Reports on emissions of greenhouse gases, include C, are part of an international, state, and municipal agreement. It is imperative to carry out research directed to identify the factors that affect C storage and biodiversity both at the national and subnational levels (Armenteras et al. 2015), including protected natural areas as the Nevado de Toluca. During 2013, the Municipality of Toluca declared that it was not possible to evaluate the C emissions at the local level due to a lack of data of forest extraction and C storage (H Ayuntamiento de Toluca 2013). Therefore, the results of this work can contribute to the greenhouse gas inventories at the municipal, state and national scales.
Conclusions
Quantification of forest harvesting is necessary to calculate greenhouse gas emissions in the categories of Land Use Change and Silviculture at the local, state and national levels. It is estimated that 211,218 Mg CO2 per year is emitted due to harvesting and natural treefalls in the Área de Protección de Flora y Fauna Nevado de Toluca, in the state of México.
C storage in dense forest (≥ 336 trees), semi-dense forest (150–335), fragmented forest (20–149), and stands with isolated trees (≤ 20), was estimated in 1621 circular plots of 1000 m2 in pine forest. The large sampling effort allowed for an accurate estimation of C storage in montane ecosystems in Mexico. The montane forests of the APFFNT store 1,695,004 MgC in aboveground forests in the 12,924 ha.
Pinus trees are the primary one harvested in Mexico, followed by A. religiosa. Generally, A. jorullensis trees are not often harvested because their wood is not dense and is relatively weak. This harvesting creates a negative C balance in the ecosystem as a result of the unregulated extraction of 42,701 MgC during an year.
References
Aguilar S, Abundiz L, Barajas J (2001) Comparación de la gravedad específica y características anatómicas de la madera de dos comunidades vegetales en México. An Inst Biol UNAM Ser Bot 72:171–185
Anaya L, Hernández S, Madrigal S (1980) La vegetación y los suelos de un transecto altitudinal del declive occidental del Iztaccíhuatl. Bol. Téc. 65. INIFAP, México. https://books.google.com.mx/books/about/La_vegetacion_y_los_suelos_de_un_transec.html?id=J6rAMAAACAAJ&redir_esc=y. Accessed 30 Aug 2017
Armenteras D, Rodríguez N, Retana J (2015) National and regional relationships of carbon storage and tropical biodiversity. Biol Conserv 192:378–386. https://doi.org/10.1016/j.biocon.2015.10.014
Augusto L, Achat D, Bakker M, Bernier F, Bert D, Danjon F, Khlifa R, Meredieu C, Trichet P (2015) Biomass and nutrients in tree root systems–sustainable harvesting of an intensively managed Pinus pinaster (Ait.) planted forest. GCB Bioenergy 7:231–243. https://doi.org/10.1111/gcbb.12127
Baker TR, Honorio Coronado EN, Phillips OL, Martin J, van Der Heijden GMF, García M, Silva Espejo J (2007) Low stocks of coarse woody debris in a southwest Amazonian forest. Oecologia 152:495–504. https://doi.org/10.1007/s00442-007-0667-5
Bárcenas G, Zárate R, Salomón I, Martínez J, Ordóñez V (2007) Propiedades físicas y mecánicas de la madera de seis especies forestales usadas como sombra en cafetales del centro del estado de Veracruz. Memorias del VIII Congreso Mexicano de Recursos Forestales. ISBN: 968-7469-81-1
Beaman J (1962) The timberlines of Iztaccihuatl and Popocatepetl, Mexico. Ecology 43:377–385. https://doi.org/10.2307/1933367
Brown S (2002) Measuring carbon in forests: current status and future challenges. Environ Pollut. 116: 363–372. https://www.winrock.org/wp-content/uploads/2016/03/2002ForestCarbon.pdf. Accessed 12 May 2017
Brown S, Lugo A (1984) Biomass of tropical forests: a new estimate based on forest volumes. Science 223:1290–1293. https://doi.org/10.1126/science.223.4642.1290
Brown S, Gillespie A, Lugo A (1989) Biomass estimation methods for tropical forests with applications to forest inventory data. For Sci 35:881–902. https://doi.org/10.1126/science.223.4642.1290
Cao M, Woodward FI (1998) Dynamic responses of terrestrial ecosystem carbon cycling to global climate change. Nature 393:249–252. https://doi.org/10.1038/30460
Challenger A (1998) Utilización y conservación de los ecosistemas terrestres de México: pasado, presente y futuro. Comisión Nacional para el Uso y Conocimiento de la Biodiversidad, Instituto de Biología de la UNAM y Agrupación Sierra Madre S.C., México
Challenger A (2003) Conceptos generales acerca de los ecosistemas templados de montaña de México y su estado de conservación. In: Sánchez O, Vega E, Peters E, Monroy-Vilchis O (eds) Conservación de ecosistemas templados de montaña en México, Instituto Nacional de Ecología. México. https://es.scribd.com/document/121903498/Conservacion-de-Ecosistemas-Templados. Accessed 1 Dec 2016
Chao K-J, Phillips OL, Baker TR, Peacock J, Lopez-Gonzalez G, Vasquez R, Monteagudo A, Torres-Lezama A (2009) After trees die: quantities and determinants of necromass across Amazonia. Biogeosciences 6:1615–1626. https://doi.org/10.5194/bg-6-1615-2009
Collado E, Serrato J (2009) El sector forestal en la actualidad. In: Ceballos G, List R, Garduño G, López-Cano R, Muñozcano-Quintanar MJ, Collado E, Elvin-San Román J (eds) Diversidad biológica del Estado de México. Estudio de Estado, Biblioteca Mexiquense del Bicentenario. Colección Mayor, Estado de México, pp 331–335. http://www.biodiversidad.gob.mx/region/EEB/pdf/EEB_EDOMEX_baja.pdf. Accessed 11 Aug 2017
CONAFOR, Comisión Nacional Forestal (2012) Inventario Nacional Forestal y de Suelos Informe de Resultados 2004-2009. Anexo 7. Coordinación General de Planeación e Información-Gerencia de Inventario Forestal y Geomática de la Comisión Nacional Forestal. Jalisco, México. http://www.cnf.gob.mx:8090/snif/portal/infys/temas/resultados-2004-2009. Accessed 22 Oct 2016
CONANP, Comisión Nacional de Áreas Naturales Protegidas (2017) Programa de manejo Área de Protección de Flora y Fauna Nevado de Toluca. Work document. Toluca, México. https://www.gob.mx/conanp/articulos/programa-de-manejo-del-area?idiom=es. Accessed 26 July 2017
Corral JJ, Barrio M, Aguirre O, Diéguez U (2007) Use of stump diameter to estimate diameter at breast height and tree volume for major pine species in El Salto, Durango (Mexico). Forestry 80:29–40. https://doi.org/10.1093/forestry/cpl048
Dauber E (1995) Guía práctica y teórica para el diseño de un inventario forestal de reconocimiento. BOLFOR: Proyecto de Manejo Forestal Sostenible. Organización de las Naciones Unidas para la Alimentación y la Agricultura. Bolivia. http://pdf.usaid.gov/pdf_docs/Pnabx164.pdf. Accessed 11 Oct 2017
De Jong B, Anaya C, Masera O, Olguín M, Paz F, Etchevers J, Martínez R, Guerrero G, Balbontín C (2010) Greenhouse gas emissions between 1993 and 2002 from land-use change and forestry in Mexico. For Ecol Manag 260:1689–1701. https://doi.org/10.1016/j.foreco.2010.08.011
Dlugokencky EJ, Hall BD, Crotwell MJ, Montzka SA, Dutton G, Mühle J, Elkins JW (2016) Atmospheric composition. Long-lived greenhouse gases. In State of the Climate in 2015. Bull Am Meteorol Soc 97:S44–S46
Echenique R, Díaz G (1972) Algunas características tecnológicas de la madera de 11 especies mexicanas. Boletín Técnico Num. 27. INIF. México
Endara A (2007) Estructura forestal de Pinus hartwegii en el Parque Nacional Nevado de Toluca. Master Thesis, Universidad Autónoma del Estado de México, México. http://sistemabibliotecario.uaemex.mx/janium-bin/titulos_autoridad.pl?Id=20180228153729
Endara A (2010) Análisis de los procesos de recuperación en el bosque templado del Parque Nacional Nevado de Toluca. Doctoral Thesis, Universidad Autónoma del Estado de México, México. http://sistemabibliotecario.uaemex.mx/janium-bin/detalle.pl?Id=20180228153343
Endara A, Calderón R, Nava G, Franco S (2013) Analysis of fragmentation processes in high-mountain forests of the centre of Mexico. Am J Plant Sci 4:697–704. https://doi.org/10.4236/ajps.2013.43A088
Falkowski P, Scholes RJ, Boyle E, Canadell J, Canfield D, Elser J, Gruber N, Hibbard K, Högberg P, Linder S, Mackenzie FT, Moore B III, Pedersen T, Rosenthal Y, Seitzinger S, Smetacek V, Steffen W (2000) The global carbon cycle: a test of our knowledge of Earth as a system. Science 290:291–296. https://doi.org/10.1126/science.290.5490.291
Flores-Villela O, Gerez P (1994) Biodiversidad y conservación en México: vertebrados, vegetación y uso del suelo. Comisión Nacional para el Cocimiento y Uso de la Biodiversidad y Universidad Nacional Autónoma de México, México. http://www.biodiversidad.gob.mx/publicaciones/librosDig/pdf/biodiversidadConservacion.pdf. Accessed 11 May 2017
Franco S, Regil H, Ordoñez J (2006) Dinámica de perturbación-recuperación de las zonas forestales en el Parque Nacional Nevado de Toluca. Madera y Bosques 12:17–28
Fuentes M (1998) Propiedades tecnológicas de las maderas mexicanas de importancia en la construcción. Rev Chapingo Ser Cie 4:221–229
García E (1981) Modificaciones al sistema de clasificación climática de Köeppen. Instituto de Geografía, Universidad Nacional Autónoma de México. D. F., México. http://www.igeograf.unam.mx/sigg/utilidades/docs/pdfs/publicaciones/geo_siglo21/serie_lib/modific_al_sis.pdf. Accessed 19 June 2016
GEM, Gobierno del Estado de México (1999) Programa de Ordenamiento Ecológico del Territorio del Estado de México, Secretaría de Ecología, Toluca, México
GOFC-GOLD, Global Observation of Forest and Land Cover Dynamics (2010) Reducing greenhouse gas emission from deforestation and degradation in developing countries: a sourcebook of methods and procedures for monitoring, measuring and reporting. GOFC-GOLD report version COP14-2. Natural Resources Canada. https://unfccc.int/files/methods_science/redd/methodologies/other/application/pdf/gofc-gold_redd_sourcebook_version_july_2009_cop14_2.pdf. Accessed 21 July 2017
Gullison RE, Frumhoff PC, Canadell JG, Field CB, Nepstad DC, Hayhoe K, Avissar R, Curran LM, Friedlingstein P, Jones CD, Nobre C (2007) Tropical forests and climate policy. Science 316:985–986. https://doi.org/10.1126/science.1136163
H Ayuntamiento de Toluca (2013) Plan de Acción Climática Municipal Toluca. Toluca, México. http://ieecc.edomex.gob.mx/sites/ieecc.edomex.gob.mx/files/files/PACMUN/PACMUN-TOLUCA.pdf. Accessed 18 Feb 2017
Hartl-Meier C, Dittmar C, Zang C, Rothe A (2014) Mountain forest growth response to climate change in the Northern Limestone Alps. Trees 28:819–829. https://doi.org/10.1007/s00468-014-0994-1
Hollinger D (2008) Defining a landscape-scale monitoring tier for the North American Carbon Program. In: Hoover C (ed) Field measurements for forest carbon monitoring a landscape-scale approach. Springer, Dordrecht, p 240. https://www.springer.com/us/book/9781402085055
Honorato J, Meraz R (2002) Caracterización tecnológica de algunas especies de coníferas de la región de El Salto, en Pueblo Nuevo, Durango. Proyecto CONAFOR. México. http://conafor.gob.mx/biblioteca/foros/Madera/CARACTERIZACION_TECNOLOGICA_DE_ESPECIES_DE_CONIFERAS.PDF. Accessed 15 Jan 2017
IPCC, Intergovernmental Panel on Climate Change (2003) Orientación sobre las buenas prácticas para uso de la tierra, cambio de uso de la tierra y silvicultura (UTCUTS). Organización Meteorológica Mundial, Génova
IPCC, Intergovernmental Panel on Climate Change (2007) Cambio Climático 2007: Impactos y Vulnerabilidad. Cambridge University Press, Reino Unido
Kent M, Coker P (1992) Vegetation description and analysis. A practical approach. Wiley, New York
Körner C (2007) The use of ‘altitude’ in ecological research. Trends Ecol Evol 22:569–574. https://doi.org/10.1016/j.tree.2007.09.006
Kumar A, Ram J (2005) Anthropogenic disturbances and plant biodiversity in forests of Uttaranchal, central Himalaya. Biodivers Conserv 14:309–331. https://doi.org/10.1007/s10531-004-5047-4
Lambin E, Geist H, Lepers E (2003) Dynamics of land-use and land-cover change in tropical regions. Annu Rev Environ Resour 28:205–241. https://doi.org/10.1146/annurev.energy.28.050302.105459
Mayer H, Ott E (1991) Gebirgswaldbau, Schutzwaldpflege. Ein waldbaulicher Beitrag zur Landschaftso¨kologie und zum Umweltschutz. Gustav Fischer Verlag, Stuttgart, New York
Mendoza A, Galicia L (2010) Aboveground and belowground biomass and carbon pools in highland temperate forest landscape in central Mexico. Forestry 83(5):497–506. https://doi.org/10.1093/forestry/cpq032
MEA, Millenium Ecosystem Assessment (2003) Ecosystem and human well-being: a framework for assessment. Island Press, Washington. https://millenniumassessment.org/en/Framework.html
Mueller-Dombois D, Ellenberg H (1974) Aims and methods of vegetation ecology. Wiley, New York
Padmakumar B, Sreekanth NP, Shanthiprabha V, Paul J, Sreedharan K, Augustine T, Jayasooryan KK, Rameshan M, Mohan M, Ramasamy EV, Thomas AP (2018) Tree biomass and carbon density estimation in the tropical dry forest of Southern Western Ghats, India. iForest 11:534–541. https://doi.org/10.3832/ifor2190-011
Palace M, Keller M, Asner GP, Silva JNM, Passos C (2007) Necromass in undisturbed and logged forests in the Brazilian Amazon. For Ecol Manag 238:309–318. https://doi.org/10.1016/j.foreco.2006.10.026
PROBOSQUE, Protectora de Bosques del Estado de México (2010) Inventario Forestal 2010. Toluca, Estado de México, México. https://www.yumpu.com/es/document/view/37081053/inventario-forestal-2010-gobierno-del-estado-de-mexico. Accessed 16 Apr 2017
Rescala J (2009) Historia del sector forestal. In: Ceballos G, List R, Garduño G, López-Cano R, Muñozcano-Quintanar MJ, Collado E, Elvin-San Román J (eds) Diversidad biológica del Estado de México. Estudio de Estado, Biblioteca Mexiquense del Bicentenario. Colección Mayor, Estado de México, pp 319–331. http://www.biodiversidad.gob.mx/region/EEB/pdf/EEB_EDOMEX_baja.pdf. Accessed 14 Mar 2017
Rodríguez S, Torres M (1995) Estudio de la composición química de la madera de ciprés (Cupressus lusitanica) en relación al fuste. Ciencia e investigación forestal 9:47–56
Rojas-García F (2008) Consideraciones para el balance de carbono: evaluación del movimiento de biomasa en el Parque Nacional Malinche. Master Thesis, Universidad Nacional Autónoma de México, México. http://132.248.9.195/ptd2008/septiembre/0632062/Index.html. Accessed 7 Nov 2016
Rojas-García F, Villers L (2005) Comparación de dos métodos para estimar la densidad de la madera de Pinus hartwegii Lindl. del Volcán La Malinche. Madera y Bosques 11:63–71
Rojas-García F, Villers L (2008) Estimación de la biomasa forestal del Parque Nacional Malinche: Tlaxcala-Puebla. Rev Ciencia Forestal en México 33:59–86
Rojo MGE, Mata JJ, Velásquez MA (2003) Las masas forestales como sumideros de CO2 ante un cambio climático global. Rev Chapingo S Cien For Amb 9:57–67
Rzedowski J (1978) Vegetación de México. Limusa, Mexico City
Salimon CI, Putz FE, Menezes-Filho L, Anderson A, Silveira M, Brown IF, Oliveira LC (2011) Estimating statewide biomass carbon stocks for a REDD plan in Acre, Brazil. For Ecol Manag 262:555–560. https://doi.org/10.1016/j.foreco.2011.04.025
Sánchez-González A, López-Mata L (2003) Clasificación y ordenación de la vegetación del norte de la Sierra Nevada, a lo largo de un gradiente altitudinal. Anal Inst Biol UNAM Ser Bot 74:47–71
Sánchez-Jasso J, Aguilar-Miguel X, Medina-Castro J, Sierra-Domínguez G (2013) Riqueza específica de vertebrados en un bosque reforestado del Parque Nacional Nevado de Toluca, México. Rev Mex Biodiv 84:360–373. https://doi.org/10.7550/rmb.29473
Sarukhán J, Franco M (1981) Un modelo de simulación de la productividad forestal de un bosque de pino. SARH. Subsecretaría Forestal y de la Fauna, México
SEMARNAT, Secretaría de Medio Ambiente y Recursos Naturales (2013) Decreto que reforma, deroga y adiciona diversas disposiciones del diverso publicado el 25 de enero de 1936, por el que se declaró Parque Nacional la montaña denominada “Nevado de Toluca”. Octubre 01 2013. https://www.gob.mx/conanp/documentos/decreto-que-reforma-deroga-y-adiciona-diversas-disposiciones-del-diverso-por-el-que-se-declaro-parque-nacional-el-nevado-de-toluca. Accessed 10 July 2016
Sotelo E, González E, Cruz G, Moreno F, Ochoa S (2010) La clasificación FAO-WRB y los suelos del Estado de México. Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias, México, D.F. www.inifap.gob.mx/circe/Documents/publime/Publicaciones%20FAO.pdf. Accessed 20 Aug 2017
Tobita H, Yazaki K, Harayama H, Kitao M (2016) Responses of symbiotic N2 fixation in Alnus species to the projected elevated CO2 environment. Trees 30:523–537. https://doi.org/10.1007/s00468-015-1297-x
Vázquez GA, Givnish TJ (1998) Altitudinal gradients in tropical forest composition structure and diversity in the Sierra de Manantlán. J Ecol 86:999–1020. https://doi.org/10.1046/j.1365-2745.1998.00325.x
Vilanova E, Ramírez H, Torres A (2010) El almacenamiento de carbono en la biomasa aérea como indicador del impacto del aprovechamiento de maderas en la Reserva Forestal Imataca, Venezuela. Interciencia 35(9):659–665
Villers L, Trejo I (1998) El impacto del cambio climático en los bosques y áreas naturales protegidas de México. Interciencia 23:10–19. https://doi.org/10.1016/S0959-3780(98)00012-0
Villers L, García L, López-Blanco J (1998) Evaluación de los bosques templados en México: una aplicación en el Parque Nacional Nevado de Toluca. Investigaciones Geográficas 36:7–19
Whittaker RH (1975) Communities and ecosystems. MacMillan, New York
WWF, World Wide Fund for Nature Centroamérica (2004) Monitoreo Ecológico del Manejo Forestal en el Trópico Húmedo: Una Guía para Operadores Forestales y Certificadores con Énfasis en Bosques de Alto Valor para la Conservación. San José, Costa Rica. 124 p. http://wwf.panda.org/?133221/Monitoreo-ecologico-del-manejo-forestal-en-el-tropico-humedo-Una-guia-para-operadores-forestales-y-certificadores-con-enfasis-en-Bosques-de-Alto-Valor-para-la-Conservacion. Accessed 12 May 2016
Acknowledgements
The study was funded by the Proyecto CONAFOR-UAEMex 3668/2014E. We express our gratitude to the two Reviewers and the Associate Editor, whose comments and suggestions greatly improve our work. Especially thank Xareni Pacheco for her recommendations to our manuscript. In addition, we would like to acknowledge of the High Mountain Group who assisted with sampling intensive of the forest inventory.
Author information
Authors and Affiliations
Contributions
FRG contributed to conceive the study, data analysis, and discussion of results and wrote the manuscript, TSF contributed to discussion and writing, SVL contributed with the cartographic processing, AREA guided the development of the fieldwork and cabinet methodology and contributed to writing at discussion.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rojas-García, F., Fredericksen, T.S., Vazquez Lozada, S. et al. Impact of timber harvesting on carbon storage in montane forests of central Mexico. New Forests 50, 1043–1061 (2019). https://doi.org/10.1007/s11056-019-09714-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11056-019-09714-z