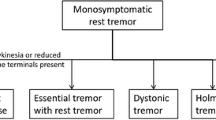
This review addresses tremor, which is among the maladaptive and difficult-to-treat symptoms of Parkinson’s disease (PD). Along with classical resting tremor, patients with PD may experience postural and kinetic types of tremor, reflecting the multimodal mechanism of its formation, which involves multiple neurotransmitter systems. Unpredictable therapeutic responses and mixed responses to levodopa also reflect the roles of multiple underlying pathophysiological processes. Among medication-based methods for tremor correction, preference is given to dopamine receptor agonists because of their wide range of pharmaceutical effects and high efficacy in relation to the major motor and various non-motor manifestations. Evidence of the efficacy of advanced neurosurgical and non-invasive treatment methods is not always convincing; there have been no large-scale comparative studies assessing their efficacy in patients with tremor-dominant forms of PD.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. J. Armstrong and M. S. Okun, “Diagnosis and treatment of parkinson disease: A review,” JAMA, 323, 548–560 (2020), https://doi.org/10.1001/jama.2019.22360(2020).
Parkinson’s Disease, Secondary Parkinsonism and Other Diseases Manifested by Parkinsonism Syndrome. Clinical Guidelines (2021).
H. Zach, M. Dirkx, B. R. Bloem, et al., “The clinical evaluation of Parkinson’s tremor,” J. Parkinsons Dis., 5, 471–474 (2015);https://doi.org/10.3233/JPD-150650.
J. Pasquini, R. Ceravolo, Z. Qamhawi, et al., “Progression of tremor in early stages of Parkinson’s disease: A clinical and neuroimaging study,” Brain, 141, 811–821 (2018), https://doi.org/10.1093/brain/awx376.
S. Kipfer, “Resting Tremor in Parkinson Disease,” Arch. Neurol., 68, 1037–1041. https://doi.org/10.1001/archneurol.2011.147(2011).
Z. A. Zalyalova, “Tremor phenotypes of Parkinson’s disease,” in: Parkinson’s Disease and Movement Disorders. A Guide for Doctors Based on Materials from the II National Congress, S. N. Illarioshkin and O. S. Levin (eds.), Moscow (2011), pp. 5–59.
A. Gironell, B. Pascual-Sedano, I. Aracil, et al., “Tremor types in Parkinson disease: A descriptive study using a new classifi cation,” Parkinsons Dis., 56, No. 7, 45–50 (2018), https://doi.org/10.1155/2018/4327597.
D. Aleksovski, D. Miljkovic, D. Bravi, et al., “Disease progression in Parkinson subtypes: the PPMI dataset,” Neurol. Sci., 39, No. 7, 1971–1976 (2018), https://doi.org/10.1007/s10072-018-3522-z.
O. S. Levin and V. K. Datieva, “ Tremor in Parkinson’s disease: Phenomenological features and treatment,” Sovrem. Ter. Psikhiatrii Nevrol., O. S. 3, No. 6, 14–19 (2014).
S. N. Illarioshkin and I. A. Ivanova-Smolenskaya (eds.), Tremor Hyperkinesis: Guidelines for Doctors (Manual Series “Movement Disorders”), Atmosfera Press, Moscow (2011).
A. M. Madelein van der Stouwe, F. Nieuwhof, and R. C. Helmich, “Tremor pathophysiology: lessons from neuroimaging,” Curr. Opin. Neurol., 33, No. 4, 474–481 (2020), https://doi.org/10.1097/WCO.0000000000000829.
M. Hallett, “Tremor: pathophysiology,” Parkinsonism Relat. Disord., 20, Suppl. 1, S118–S122 (2014), https://doi.org/10.1016/S1353-8020(13)70029-4.
R. C. Helmich and M. F. Dirkx, “Pathophysiology and management of Parkinsonian tremor,” Semin. Neurol., 37, 127–134 (2017), https://doi.org/10.1055/s-0037-1601558(2017).
R. C. Helmich, “The cerebral basis of Parkinsonian tremor: A network perspective,” Mov. Dis., 33, No. 7, 219–231 (2018), https://doi.org/10.1002/mds.27224.
M. F. Dirkx and M. Bologna, “The pathophysiology of Parkinson’s disease tremor,” J. Neurol. Sci., 435, 120196 (2022), https://doi.org/10.1016/j.jns.2022.120196.
L. Timmermann, J. Gross, M. Dirks, et al., “The cerebral oscillatory network of parkinsonian resting tremor,” Brain, 126, 199–212 (2003), https://doi.org/10.1093/brain/awg022.
M. F. Dirkx, H. den Ouden, et al., “The cerebral network of parkinson’s tremor: An effective connectivity fMRI study,” J. Neurosci., 36, 5362–5372 (2016), https://doi.org/10.1523/JNEUROSCI.3634-15.2016.
M. F. Dirkx, H. E. M. den Ouden, E. Aarts, et al., “Dopamine controls Parkinson’s tremor by inhibiting the cerebellar thalamus,” Brain, 140, 721–734 (2017), https://doi.org/10.1093/brain/aww331.
N. I. Kremer, R. W. J. Pauwels, N. G. Pozzi, et al., “Deep brain stimulation for tremor: Update on long-term outcomes, target considerations and future directions,” J. Clin. Med., 10, No. 5, 23–29 (2021), https://doi.org/10.3390/jcm10163468.
R. C. Helmich, M. Hallett, G. Deuschl, et al., “Cerebral causes and consequences of parkinsonian resting tremor: A tale of two circuits?” Brain, 135, No. 8, 3206–3226 (2012), https://doi.org/10.1093/brain/aws023.
C. Duval, J. F. Daneault, W. D. Hutchison, et al., “A brain network model explaining tremor in Parkinson’s disease,” Neurobiol. Dis., 85, 49–59 (2016), https://doi.org/10.1016/j.nbd.2015.10.009.
R. C. Helmich, M. J. R. Janssen, W. J. G. Oyen, et al., “Pallidal dysfunction drives a cerebellothalamic circuit into Parkinson tremor,” Ann. Neurol., 69, 269–281 (2011), https://doi.org/10.1002/ana.22361.
C. F. Underwood and L. C. Parr-Brownlie, “Primary motor cortex in Parkinson’s disease: Functional changes and opportunities for neurostimulation,” Neurobiol. Dis., 147, No. 53, 45–49 (2021), https://doi.org/10.1016/j.nbd.2020.105159.
R. G. Burciu and D. E. Vaillancourt, “imaging of motor cortex physiology in Parkinson’s disease,” Mov. Dis., 33, 1688–1699 (2018), https://doi.org/10.1002/mds.102.
Z. Ni, A. D. Pinto, A. E. Lang, et al., “Involvement of the cerebellothalamocortical pathway in Parkinson disease,” Ann. Neurol., 68, 816–824 (2010), https://doi.org/10.1002/ana.22221.
M. K. Lu, S. M. Chiou, U. Ziemann, et al., “Resetting tremor by single and paired transcranial magnetic stimulation in Parkinson’s disease and essential tremor,” Clin. Neurophysiol., 126, 2330–2336 (2015), https://doi.org/10.1016/j.clinph.2015.02.010.
K. A. Jellinger, “Neuropathology of sporadic Parkinson’s disease: Evaluation and changes of concepts,” Mov. Dis., 27, 8–30 (2012), https://doi.org/10.1002/mds.23795.
W. Paulus and K. Jellinger, “The neuropathologic basis of different clinical subgroups of Parkinson’s disease,” J. Neuropathol. Exp. Neurol., 50, No. 6, 743–755 (1991), https://doi.org/10.1097/00005072-199111000-00006.
I. U. Isaias, G. Marotta, G. Pezzoli, et al., “Enhanced catecholamine transporter binding in the locus coeruleus of patients with early Parkinson disease,” BMC Neurol., 11, No. 6, 12–19 (2011), https://doi.org/10.1186/1471-2377-11-88.
Z. Qamhawi, D. Towey, B. Shah, et al., “Clinical correlates of raphe serotonergic dysfunction in early Parkinson’s disease,” Brain, 138, 2964–2973 (2015), https://doi.org/10.1093/brain/awv215.
V. Caretti, D. Stoffers, A. Winogrodzka, et al., “Loss of thalamic serotonin transporters in early drug-naïve Parkinson’s disease patients is associated with tremor: An [123I] β-CIT SPECT study,” J. Neural. Transm., 115, 721–729 (2008), https://doi.org/10.1007/s00702-007-0015-2.
S. Perez-Lloret and F. J. Barrantes, “Defi cits in cholinergic neurotransmission and their clinical correlates in Parkinson’s disease,” Parkinsons Dis., 34, No. 2, 34–39 (2016), https://doi.org/10.1038/npjparkd.2016.1.
A. Lenka and J. Jankovic, “Tremor syndromes: An updated Review,” Front. Neurol., 12, No. 7, 1–17 (2021), https://doi.org/10.3389/fneur.2021.684835.
C. W. Hess and S. L. Pullman, “Tremor: Clinical phenomenology and assessment techniques,” Tremor Other Hyperkin. Mov. (N.Y.), 2, 1–15 (2012), https://doi.org/10.5334/tohm.115.
M. F. Dirkx, H. Zach, B. R. Bloem, et al., “The nature of postural tremor in Parkinson disease,” Neurology, 90, e1095–102 (2018), https://doi.org/10.1212/WNL.0000000000005215.
D. K. Gupta et al., “prevalence and relationship of rest tremor and action tremor in Parkinson’s disease,” Tremor Other Hyperkin. Mov. (N.Y.), 10, No. 1, 1–7 (2020) (2006) (2006), https://doi.org/10.5334/tohm.552.
V. L. Golubev, “Treatment algorithm for the early stages of Parkinson’s disease,” Atmosfera. Nervnye Bolezni, 12, No. 4, 9–12.
G. Tedeschi, “Tremor in Parkinson disease: acute response to oral levodopa,” Ital. J. Neurol. Sci., 11, No. 3, 259–263 (1990), https://doi.org/10.1007/BF02333855.
B. S. Connolly and A. E. Lang, “Pharmacological treatment of Parkinson disease: a review,” JAMA, 311, No. 16, 1670–1683 (2014), https://doi.org/10.1001/jama.2014.
A. Gigante, “Action tremor in Parkinson’s disease: frequency and relationship to motor and non-motor signs,” Eur. J. Neurol., 22, No. 2, 223–228 (2015), https://doi.org/10.1111/ene.12583.
K. A. Josephs, J. Y. Matsumoto, and J. E. Ahlskog, “Benign tremulous parkinsonism,” Arch. Neurol., 63, No. 3, 354–357 (2006), https://doi.org/10.1001/archneur.63.3.354.
T. G. Govorova, T. E. Popova, and A. A. Tappakhov, “Electrophysiological features of tremor in essential tremor and Parkinson’s disease,” in: Botkin Readings. All-Russian Therapeutic Congress with International Participation (2019), pp. 56–57.
G. T. Stebbins, “How to identify tremor dominant and postural instability/ gait diffi culty groups with the Movement Disorder Society Unifi ed Parkinson’s Disease Rating Scale: comparison with the Unifi ed Parkinson’s Disease Rating Scale,” Mov. Dis., 28, No. 5, 668–670 (2013), https://doi.org/10.1002/mds.25383.
R. M. A. De Bie, C. E. Clarke, A. J. Espay, et al., “Initiation of pharmacological therapy in Parkinson’s disease: when, why, and how,” Lancet Neurol., 19, 452–461 (2020), https://doi.org/10.1016/S1474-4422(20)30036-3.
R. Balestrino and A. H. V. Schapira, “Parkinson disease,” Eur. J. Neurol., 27, No. 8, 27–42 (2020), https://doi.org/10.1111/ene.14108.
T. Pringsheim, G. S. Day, D. B. Smith, et al., “Dopaminergic therapy for motor symptoms in early Parkinson disease practice Guideline summary,” Neurology, 97, 942–957 (2021), https://doi.org/10.1212/WNL.0000000000012868.
J. Nonnekes, M. H. M. Timmer, N. M. de Vries, et al., “Unmasking levodopa resistance in Parkinson’s disease,” Mov. Dis., 31, 1602–1609 (2016), https://doi.org/10.1002/mds.26712.
L. E. Heusinkveld, M. L. Hacker, M. Turchan, et al., “Impact of tremor on patients with early stage Parkinson’s disease,” Front. Neurol., 9, 34–39 (2018), https://doi.org/10.3389/fneur.2018.00628.
H. Zach, M. Dirkx, J. W. Pasman, et al., “The patient’s perspective: The effect of levodopa on Parkinson symptoms,” Parkinsonism Relat. Disord., 35, 48–54 (2017), https://doi.org/10.1016/j.parkreldis.2016.11.015.
H. Zach, M. F. Dirkx, D. Roth, et al., “Dopamine-responsive and dopamine-resistant resting tremor in Parkinson disease,” Neurology, 95, No. 11, e1461–e1470 (2020), https://doi.org/10.1212/WNL.0000000000010316.
M. Beckers, B. R. Bloem, and M. M. Verbeek, “Mechanisms of peripheral levodopa resistance in Parkinson’s disease,” NPJ Parkin son’s Dis., 8, No. 1, 56–61 (2022), https://doi.org/10.1038/s41531-022-00321-y.
J. Jankovic and E. K. Tan, “Parkinson’s disease: Etiopathogenesis and treatment,” J. Neurol. Neurosurg. Psychiatr., 91, 795–808 (2020), https://doi.org/10.1136/jnnp-2019-322338.
A. Maillet, S. Thobois, V. Fraix, et al., “Neural substrates of levodopa-responsive gait disorders and freezing in advanced Parkinson’s disease: a kinesthetic imagery approach,” Hum. Brain Mapp., 36, No. 3, 959–980 (2015), https://doi.org/10.1002/hbm.22679.
N. V. Titova, E. A. Katunina, R. T. Tairova, et al., ”Pharmacoresistant tremor in Parkinson’s disease and essential tremor,” Zh. Nevrol. Psikhiatr., 122, No. 10, 24–30 (2022), https://doi.org/10.17116/jnevro202212210124.
H. V. Gupta, K. E. Lyons, N. Wachter, et al., “Long term response to levodopa in Parkinson’s disease,” J. Parkinsons Dis., 9, No. 3, 525–529 (2019), https://doi.org/10.3233/JPD-191633.
M. P. Caligiuri and J. B. Lohr, “Worsening of postural tremor in patients with levodopa-induced dyskinesia: a quantitative analysis,” Clin. Neuropharmacol., 16, No. 3, 244–250 (1993), https://doi.org/10.1097/00002826-199306000-00008.
R. R. Bogdanov, “Features of drug therapies for the initial manifestations of Parkinson’s disease,” Nervnye Bolezni, No. 1, 2–5 (2013).
O. Pogarell, T. Gasser, J. J. van Hilten, et al., “Pramipexole in patients with Parkinson’s disease and marked drug resistant tremor: a randomised, double blind, placebo controlled multicentre study,” J Neurol. Neurosurg. Psychiatry, 72, No. 6, 713–720 (2002), https://doi.org/10.1136/jnnp.72.6.713.
R. J. Elble, “Tremor and dopamine agonists,” Neurology, 58, No. 4, Suppl. 1, S57–S62 (2002), https://doi.org/10.1212/WNL.58.suppl_1.S57.
P. Navan, L. J. Findley, et al., “Double-blind, single-dose, cross-over study of the effects of pramipexole, pergolide, and placebo on rest tremor and UPDRS part III in Parkinson’s disease,” Mov. Disord., 18, No. 2, 176–180 (2003), https://doi.org/10.1002/mds.10320.
F. Carbone, A. Djamshidian, K. Seppi, et al., “Apomorphine for Parkinson’s Disease: Effi cacy and safety of current and new formulations,” CNS Drugs, 33, 905–918 (2019), https://doi.org/10.1007/s40263-019-00661-z.
P. A. Kempster, J. P. Frankel, G. M. Stern, et al., “Comparison of motor response to apomorphine and levodopa in Parkinson’s disease,” J. Neurol. Neurosurg. Psychiatr., 53, 1004–1007 (1990), https://doi.org/10.1136/jnnp.53.11.1004.
M. J. Millan, “From the cell to the clinic: A comparative review of the partial D2/D3 receptor agonist and α2-adrenoceptor antagonist, piribedil, in the treatment of Parkinson disease,” Pharmacol. Ther., 128, No. 2, 229–273 (2010), https://doi.org/10.1016/j.pharmthera.2010.06.002.
A. Gobert, B. Di Cara, L. Cistarelli, et al., “Piribedil enhances frontocortical and hippocampal release of acetylcholine in freely moving rats by blockade of α2a-adrenoceptors: A dialysis comparison to talipexole and quinelorane in the absence of acetylcholinesterase inhibitors,” J. Pharmacol. Exp. Ther., 305, No. 1, 338–346 (2003), https://doi.org/10.1124/jpet.102.046383.
V. G. Evidente, R. P. Esteban, F. M. Domingo, et al., “Piribedil as an adjunct to levodopa in advanced Parkinson’s disease: the Asian experience,” Parkinsonism Relat. Disord., 10, No. 2, 117–121 (2003), https://doi.org/10.1016/s1353-8020(03)00096-8.
A. A. Pilipovich and V. L. Golubev, “Potential of using piribedil from the point of view of evidence-based medicine,” Ross. Med. Zh., 26, No. 12–1, 39–43 (2018).
N. V. Fedorova, E. G. Artem’eva, Chigir’, I. P., et al., “Use of pronoran (piribedil) for Parkinson’s disease,” Zh. Nevrol. Psikhiatr., 103, No. 9, 71–72 (2003).
P. Rondot and M. Ziegler, “Activity and acceptability of piribedil in Parkinson’s disease: a multicenter study,” J. Neurol., 239, Suppl. 1, S28–S34 (year).
G. Mentenopolous, Z. Katsarou, S. Bostantjopoulou, et al., “Piribedil therapy in Parkinson’s disease,” Clin. Neuropharm., 12, 23–28 (1989).
Y. Evrard, “Piribedil, a dopaminergic agonist,” JAMA, 34, No. 8, 16–20 (1991).
J. C. Corvol, F. Artaud, F. Cormier-Dequaire, et al., “Longitudinal analysis of impulse control disorders in Parkinson disease,” Neurology, 91, e189–201 (2018), https://doi.org/10.1212/WNL.0000000000005816.
R. Djaldetti, R. Mosberg-Galili, H. Sroka, et al., “Camptocormia (bent spine) in patients with Parkinson’s disease characterization and possible pathogenesis of an unusual phenomenon,” Mov. Dis., 14, No. 3, 443–447 (1999), https://doi.org/10.1002/1531-8257(199905)14:3<443::aid-mds1009>3.0.co;2-g.
M. Suzuki, T. Hirai, Y. Ito, et al., “Pramipexole-induced antecollis in Parkinson’s disease,” J. Neurol. Sci., 264, No. 1–2, 195–197 (2008), https://doi.org/10.1016/j.jns.2007.08.008.
R. G. Holloway, I. Shoulson, S. Fahn, et al., “Pramipexole vs levodopa as initial treatment for Parkinson disease: a 4-year randomized controlled trial,” Arch. Neurol., 61, No. 7, 1044–1053 (2004), https://doi.org/10.1001/archneur.61.7.1044.
O. Rascol, D. J. Brooks, A. D. Korczyn, et al., “A fi ve-year study of the incidence of dyskinesia in patients with early Parkinson’s disease who were treated with ropinirole or levodopa,” N. Engl. J. Med., 342, No. 20, 1484–1491 (2000), https://doi.org/10.1056/NEJM200005183422004.
C. Marras, K. R. Chaudhuri, N. Titova, et al., “Therapy of Parkinson’s disease subtypes,” Neurotherapeutics, 17, 1366–1377 (2020), https://doi.org/10.1007/s13311-020-00894-7.
R. Katzenschlager, C. Sampaio, J. Costa, et al., “Anticholinergics for symptomatic management of Parkinson’s disease,” Cochrane Database Syst. Rev., 3, CD003735 (2002), https://doi.org/10.1002/14651858.CD003735.
Y. Saeedi, M. Ghadimi, M. Rohani, et al., “Impact of anticholinergic drugs withdrawal on motor function in patients with Parkinson’s disease,” Clin. Neurol. Neurosurg., 202, No. 3, 106480 (2021), https://doi.org/10.1016/j.clineuro.2021.106480.
J. J. Van Hilten, C. C. Ramaker, R. Stowe, et al., “Bromocriptine versus levodopa in early Parkinson’s disease,” Cochrane Database Syst. Rev., 2007, No. 4, CD002258 (2007), https://doi.org/10.1002/14651858.CD002258.pub2.
R. Krishna, M. Ali, and A. A. Moustafa, “Effects of combined MAO-B inhibitors and levodopa vs monotherapy in Parkinson’s disease,” Front. Aging Neurosci., 6, 1–9 (2014), https://doi.org/10.3389/fnagi.2014.00180.
M. F. Lew, “Rasagiline treatment effects on parkinsonian tremor,” Int. J. Neurosci., 123, 859–865 (2013), https://doi.org/10.3109/00207454.2013.812085.
F. Stocchi and J. M. Rabey, “Effect of rasagiline as adjunct therapy to levodopa on severity of OFF in Parkinson’s disease,” Eur. J. Neurol., 18, No. 12, 1373–1378 (2011), https://doi.org/10.1111/j.1468-1331.2011.03512.x.
Parkinson Study Group, “Low-dose clozapine for the treatment of drug-induced psychosis in Parkinson’s disease,” N. Engl. J. Med., 340, No. 10, 757–763 (1999), https://doi.org/10.1056/NEJM199903113401003.
T. K. Yaw, S. H. Fox, and A. E. Lang, “Clozapine in parkinsonian rest tremor: A review of outcomes, adverse reactions, and possible mechanisms of action,” Mov. Disord. Clin. Pract., 3, No. 6, 116–124 (2016), https://doi.org/10.1002/mdc3.12266.
R. M. Trosch, J. H. Friedman, M. C. Lannon, et al., “Clozapine use in Parkinson’s disease: A retrospective analysis of a large multicentered clinical experience,” Mov. Dis., 13, 377–382 (1998), https://doi.org/10.1002/mds.870130302.
A. A. Thomas and J. H. Friedman, “Current use of clozapine in Parkinson disease and related disorders,” Clin. Neuropharmacol., 33, No. 1, 14–16 (2010), https://doi.org/10.1097/WNF.0b013e3181c47168.
N. J. Crosby, K. Deane, and C. E. Clarke, “Beta-blocker therapy for tremor in Parkinson’s disease,” Cochrane Database Syst. Rev., CD003361 (2003), https://doi.org/10.1002/14651858.CD003361.
S. H. Fox, “Non-dopaminergic treatments for motor control in Parkinson’s disease,” Drugs, 73, 1405–1415 (2013), https://doi.org/10.1007/s40265-013-0105-4.
J. Marjama-Lyons and W. Koller, “Tremor-predominant Parkinson’s disease. Approaches to treatment,” Drugs Aging, 16, No. 4, 273–278 (2000), https://doi.org/10.2165/00002512-200016040-00003.
A. H. Abusrair, W. Elsekaily, and Bohlega S., “Tremor in Parkinson’s disease: From pathophysiology to advanced therapies,” Tremor Other Hyperkinet. Mov., 12, 29 (2022), https://doi.org/10.5334/tohm.712.
R. M. Trosch and S. L. Pullman, “Botulinum toxin A injections for the treatment of hand tremors,” Mov. Dis., 9, No. 6, 601–609 (1994), https://doi.org/10.1002/mds.870090604.
S. O. Mittal and S. Pandey, “Botulinum toxin for the treatment of tremor,” J. Neurol. Sci., 435, 120203 (2022), https://doi.org/10.1016/j.jns.2022.120203.
S. O. Mittal, M. Jog, J. Lee, et al., “Novel botulinum toxin injection protocols for parkinson tremor and essential tremor – the Yale technique and sensor-based kinematics procedure for safe and effective treatment,” Tremor Other Hyperkinet. Mov., 10, 1–8 (2020), https://doi.org/10.5334/tohm.582.
N. Niemann and J. Jankovic, “Botulinum toxin for the treatment of hand tremor,” Toxins (Basel), 10, 1–10 (2018), https://doi.org/10.3390/toxins10070299.
O. Samotus, J. Lee, and M. Jog, “Long-term tremor therapy for Parkinson and essential tremor with sensor-guided botulinum toxin type A injections,” PLoS One, 12, 1–19 (2017), https://doi.org/10.1371/journal.pone.0178670.
P. S. Larson, “Deep brain stimulation for movement disorders,” Neurotherapeutics, 11, No. 3, 465–474 (2014), https://doi.org/10.1007/s13311-014-0274-1.
E. Y. Uc and K. A. Follett, “Deep brain stimulation in movement disorders,” Semin. Neurol., 27, No. 2, 170–182 (2007), https://doi.org/10.1055/s-2007-971175.
T. M. Herrington, J. J. Cheng, and E. N. Eskandar, “Mechanisms of deep brain stimulation,” J. Neurophysiol., 115, No. 12, 19–38 (2016), https://doi.org/10.1152/jn.00281.2015.
S. Chiken and A. Nambu, “Mechanism of deep brain stimulation: Inhibition, excitation, or disruption?” Neuroscientist, 22, No. 8, 313–322 (2016), https://doi.org/10.1177/1073858415581986.
Z. Mao, Z. Ling, L. Pan, et al., “Comparison of effi cacy of deep brain stimulation of different targets in Parkinson’s disease: A network meta-analysis,” Front. Aging Neurosci., 10, No. 8, 1–8 (2019), https://doi.org/10.3389/fnagi.2019.00023.
C. R. Honey, C. Hamani, S. K. Kalia, et al., “Deep brain stimulation target selection for Parkinson’s disease,” Can. J. Neurol. Sci., 44, 3–8 (2017), https://doi.org/10.1017/cjn.2016.22.
J. K. Wong, V. T. Viswanathan, K. S. Nozile-Firth, et al., “STN versus GPi deep brain stimulation for action and rest tremor in Parkinson’s disease,” Front. Hum. Neurosci., 14, 578615 (2020), https://doi.org/10.3389/fnhum.2020.578615.
Z. Lin, X. Zhang, L. Wang, et al., “Revisiting the L-dopa response as a predictor of motor outcomes after deep brain stimulation in Parkinson’s disease,” Front. Hum. Neurosci., 15, No. 5, 1–10 (2021), https://doi.org/10.3389/fnhum.2021.604433.
A. Rughani, J. M. Schwalb, Sidiropoulos C., et al., “congress of neurological surgeons systematic review and evidence-based guideline on subthalamic nucleus and globus pallidus internus deep brain stimulation for the treatment of patients with Parkinson’s disease: Executive summary,” Neurosurgery, 82, No. 6, 753–756 (2018), https://doi.org/10.1093/neuros/nyy037.
H. Walters and B. B. Shah, “Focused ultrasound and other lesioning therapies in movement disorders,” Curr. Neurol. Neurosci. Rep., 19, 66–71 (2019), https://doi.org/10.1007/s11910-019-0975-2.
C. Ohye, Y. Higuchi, T. Shibazaki, et al., “Gamma knife thalamotomy for Parkinson disease and essential tremor: a prospective multicenter study,” Neurosurgery, 70, No. 3, 526–536 (2012), https://doi.org/10.1227/NEU.0b013e3182350893.
D. Berg and J. Corvol, “European Academy of Neurology/Movement Disorder Society – European Section Guideline on the Treatment of Parkinson’s Disease,” Invasive Ther., 37, 1360–1374 (2022), https://doi.org/10.1002/mds.29066.
Z. Lin, C. Zhang, D. Li, et al., “Preoperative Levodopa response and deep brain stimulation effects on motor outcomes in Parkinson’s disease: A systematic review,” Mov. Disord. Clin. Pract., 9, No. 2, 140–155 (2021), https://doi.org/10.1002/mdc3.13379.
V. Krishna, F. Sammartino, R. Cosgrove, et al., “Predictors of outcomes after focused ultrasound thalamotomy,” Neurosurgery, 87, 229–237 (2020), https://doi.org/10.1093/neuros/nyz417.
A. E. Bond, B. B. Shah, D. S. Huss, et al., “Safety and effi cacy of focused ultrasound thalamotomy for patients with medication-refractory, tremor-dominant Parkinson disease: A randomized clinical trial,” JAMA Neurol., 74, No. 9, 1412–1418 (2017) (2021) (2021), https://doi.org/10.1001/jamaneurol.2017.3098.
R. M. Galimova, D. K. Krekotin, and O. V. Kachemaeva, et al., “Focused ultrasound therapy under magnetic resonance navigation control of motor disorders in Parkinson’s disease and essential tremor,” in: IX All-Russian Congress of Neurosurgeons. Collection of Abstracts, Moscow (2021), pp. 91–92.
R. Martinez-Fernandez, S. Mahendran, J. A. Pineda-Pardo, et al., “Bilateral staged magnetic resonance-guided focused ultrasound thalamotomy for the treatment of essential tremor: a case series study,” J Neurol. Neurosurg. Psychiatry, 92, 927–931 (2021), https://doi.org/10.1136/jnnp-2020-325278.
Y. Xu, Q. He, M. Wang, et al., “Safety and effi cacy of magnetic resonance imaging-guided focused ultrasound neurosurgery for Parkinson’s disease: a systematic review,” Neurosurg. Rev., 44, 115–127 (2021), https://doi.org/10.1007/s10143-019-01216-y.
M. A. Chalah, J. P. Lefaucheur, and S. S. Ayache, “Non-invasive central and peripheral stimulation: New hope for essential tremor?” Front. Neurosci., 9, 1–11 (2015), https://doi.org/10.3389/fnins.2015.00440.
L. Meng, M. Jin, X. Zhu, et al., “Peripherical electrical stimulation for parkinsonian tremor: A systematic review,” Front. Aging Neurosci., 14, 1–11 (2022), https://doi.org/10.3389/fnagi.2022.795454.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Nevrologii i Psikhiatrii imeni S. S. Korsakova, Vol. 124, No. 4, pp. 28–35, April, 2024.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zalyalova, Z.A., Katunina, E.A., Pokhabov, D.V. et al. Tremor-Dominant Form of Parkinson’s Disease. Neurosci Behav Physi 54, 809–816 (2024). https://doi.org/10.1007/s11055-024-01685-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-024-01685-9