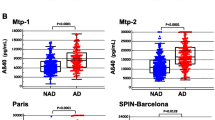
Objective. To compare levels of Aβ40, Aβ42, total tau protein, and tau protein phosphorylated at threonine 181 in cerebrospinal fl uid (CSF) with clinical diagnoses of Alzheimer’s disease (AD). Materials and methods. The study was conducted in 64 patients with diagnoses of dementia and Mini-Mental State Examination (MMSE) scores of 24 points or lower. All patients underwent lumbar puncture. Aβ40 and Aβ42 levels, the Aβ42/40 ratio, and levels of total Tau and Tau phosphorylated at threonine 181 were determined in CSF using a multiplex assay following the manufacturer’s protocol. Concentrations were expressed in pg/ml. Results. Prior clinical diagnoses of AD had been made in only three patients (5%). The study revealed increased contents of AD-typical pathological proteins in the CSF in 48 subjects (75%). The data obtained indicate that the diagnosis of AD is made 10–14 times less frequently, according to global statistics, than might be expected. The discrepancy between the clinical diagnosis and laboratory data is confi rmed by our study. Conclusions. Differences in the treatment of dementia and the development of new drugs targeting specifi c elements in the pathogenesis of different types of dementia require accurate and complete diagnosis of dementia, especially AD as the most common form of this disease.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Global Action Plan on the Public Health Response to Dementia 2017–2025, World Health Organization, Geneva (2017), Licence: CC BY-NC-SA 3.0 IGO.
Jeremic, D., Jiménez-Díaz, L., and Navarro-López, J. D., “Past, present and future of therapeutic strategies against amyloid-β peptides in Alzheimer’s disease: a systematic review,” Ageing Res. Rev., 72, 101496 (2021), https://doi.org/10.1016/j.arr.2021.101496.
MacLeod, R., Hillert, E. K., Cameron, R. T., and Baillie, G. S., “The role and therapeutic targeting of α-, and β- and γ-secretase in Alzheimer’s disease,” Future Sci. OA, 1, No. 3 (2015), https://doi.org/10.4155/fso.15.9.
Murphy, M. P., LeVine H 3rd, “Alzheimer’s disease and the amyloid-beta peptide,” J. Alzheimers Dis., 19, No. 1, 311–323 (2010), https://doi.org/10.3233/JAD-2010-1221.
Folch, J., Ettcheto, M., Petrov, D., et al., “Review of the advances in treatment for Alzheimer disease: Strategies for combating β-amyloid protein [Una revisión de los avances en la terapéutica de la enfermedad de Alzheimer: estrategia frente a la proteína β-amiloide],” Neurologia (Engl. ed.), 33, No. 1, 47–58 (2018), https://doi.org/10.1016/j.nrl.2015.03.012.
Grandal Leiros, B., Pérez Méndez, L. I., Zelaya Huerta, M. V., et al., “Prevalence and concordance between the clinical and the post-mortem diagnosis of dementia in a psychogeriatric clinic [Prevalencia y concordancia entre diagnóstico clínico y anatomopatológico de demencia en una clínica psicogeriátrica],” Neurologia (Engl. ed.), 33, No. 1, 13–17 (2018), https://doi.org/10.1016/j.nrl.2016.04.011.
Selvackadunco, S., Langford, K., Shah, Z., et al., “Comparison of clinical and neuropathological diagnoses of neurodegenerative diseases in two centres from the Brains for Dementia Research (BDR) cohort,” J. Neural Transm (Vienna), 126, No. 3, 327–337 (2019), https://doi.org/10.1007/s00702-018-01967-w.
Sengupta, U. and Kayed, R., “Amyloid β, Tau, and α-synuclein aggregates in the pathogenesis, prognosis, and therapeutics for neurodegenerative diseases,” Prog. Neurobiol., 214, 102270 (2022), https://doi.org/10.1016/j.pneurobio.2022.102270.
Sunderland, T., Linker, G., Mirza, N., et al., “Decreased beta-amyloid1-42 and increased tau levels in cerebrospinal fluid of patients with Alzheimer disease,” JAMA, 289, No. 16, 2094–2103 (2003), https://doi.org/10.1001/jama.289.16.2094.
Li, R. X., Ma, Y. H., Tan, L., and Yu, J. T., “Prospective biomarkers of Alzheimer’s disease: A systematic review and meta-analysis,” Ageing Res. Rev., 81, 101699 (2022), https://doi.org/10.1016/j.arr.2022.101699.
Mielke, M. M., Frank, R. D., Dage, J. L., et al., “Comparison of plasma phosphorylated tau species with amyloid and tau positron emission tomography, neurodegeneration, vascular pathology, and cognitive outcomes,” JAMA Neurol., 78, No. 9, 1108–1117 (2021), https://doi.org/10.1001/jamaneurol.2021.2293.
Folstein, M. F., Folstein, S. E., and McHugh, P. R., “Mini-Mental State. A practical method for grading the cognitive state of patients for the clinician,” J. Psychiatr. Res., 12, No. 3, 189–198 (1975), https://doi.org/10.1016/0022-3956(75)90026-6.
Ebenau, J. L., Timmers, T., Wesselman, L. M. P., et al., “A/T/N classification and clinical progression in subjective cognitive decline: The SCIENCe project,” Neurology, 95, No. 1, e46–e58 (2020), https://doi.org/10.1212/WNL.0000000000009724.
Hansen, E. O., Dias, N. S., Burgos, I. C. B., et al., “Millipore xMap® Luminex (HATMAG-68K, An accurate and cost-effective method for evaluating Alzheimer’s Biomarkers in cerebrospinal fluid,” Front. Psychiatry, 12, 716686 (2021), https://doi.org/10.3389/fpsyt.2021.716686.
Constantinides, V. C., Boufi dou, F., Bourbouli, M., et al., “Application of the AT(N) and other CSF classification systems in behavioral variant frontotemporal dementia,” Diagnostics (Basel), 13, No. 3, 332 (2023), https://doi.org/10.3390/diagnostics13030332.
Jack CR Jr, Bennett, D. A., Blennow, K., et al., “A/T/N: An unbiased descriptive classification scheme for Alzheimer disease biomarkers,” Neurology, 87, No. 5, 539–547 (2016), https://doi.org/10.1212/WNL.0000000000002923.
Milà-Alomà, M., Salvadó, G., Gispert, J. D., et al., “Amyloid beta, tau, synaptic, neurodegeneration, and glial biomarkers in the preclinical stage of the Alzheimer’s continuum,” Alzheimers Dement., 16, No. 10, 1358–1371 (2020), https://doi.org/10.1002/alz.12131.
Bogolepova, A. N., Vasenina, E. E., Gomzyakova, N. A., et al., “Clinical guidelines “Cognitive disorders in elderly and senile patients,” Zh. Nevrol. Psikhiatr., 121, No. 10-3, 6–137 (2021), https://doi.org/10.17116/jnevro20211211036.
Hasani, S. A., Mayeli, M., Salehi, M. A., and Barzegar Parizi, R., “A systematic review of the association between amyloid-β and τ pathology with functional connectivity alterations in the Alzheimer dementia spectrum utilizing PET scan and rsfMRI,” Dement.Geriatr. Cogn. Disord. Extra, 11, No. 2, 78–90 (2021), https://doi.org/10.1159/000516164.
Resnick, S. M. and Sojkova, J., “Amyloid imaging and memory change for prediction of cognitive impairment,” Alzheimers Res. Ther., 3, No. 1, 3 (2011), https://doi.org/10.1186/alzrt62.
Jellinger, K. A. and Attems, J., “Prevalence and pathology of vascular dementia in the oldest-old,” J. Alzheimers Dis., 21, No. 4, 1283–1293 (2010), https://doi.org/10.3233/jad-2010-100603.
Levin, O. S. and Trusova, N. A., “Vascular risk factors for Alzheimer’s disease,” Zh. Nevrol. Psikhiatr., 113, No. 7, Iss. 2, 3–12 (2013).
Zakharov, V. V., “Differential diagnosis of cognitive impairments,” Effekt. Farmakoter. Nevrol., 3, 25 (2014).
Fierini, F., “Mixed dementia: Neglected clinical entity or nosographic artifi ce?” J. Neurol. Sci., 410, 116662 (2020), https://doi.org/10.1016/j.jns.2019.116662.
Apátiga-Pérez, R., Soto-Rojas, L. O., Campa-Córdoba, B. B., et al., “Neurovascular dysfunction and vascular amyloid accumulation as early events in Alzheimer’s disease,” Metab. Brain. Dis., 37, No. 1, 39–50 (2022), https://doi.org/10.1007/s11011-021-00814-4.
Badimon, A., Torrente, D., and Norris, E. H., “Vascular dysfunction in Alzheimer’s disease: Alterations in the plasma contact and fi brinolytic systems,” Int. J. Mol. Sci., 24, No. 8, 7046 (2023), https://doi.org/10.3390/ijms24087046.
Hort, J., O’Brien, J. T., Gainotti, G., et al., “EFNS guidelines for the diagnosis and management of Alzheimer’s disease,” Eur. J. Neurol., 17, No. 10, 1236–1248 (2010), https://doi.org/10.1111/j.1468-1331.2010.03040.x.
Gavrilova, S. I., Kolykhalov, I. V., Kulik, A. S., et al., “Experience im the clinical use of Memantal in patients with moderate and severe Alzheimer’s disease,” Zh. Nevrol. Psikhiatr., 116, No. 2, 52–57 (2016), https://doi.org/10.17116/jnevro20161162152-57.
Olivares, D., Deshpande, V. K., Shi, Y., et al., “N-methyl D-aspartate (NMDA) receptor antagonists and memantine treatment for Alzheimer’s disease, vascular dementia and Parkinson’s disease,” Curr. Alzheimer Res., 9, No. 6, 746–758 (2012), https://doi.org/10.2174/156720512801322564.
Kokkinou, M., Beishon, L. C., Smailagic, N., et al., “Plasma and cerebrospinal fluid ABeta42 for the differential diagnosis of Alzheimer’s disease dementia in participants diagnosed with any de mentia subtype in a specialist care setting,” Cochrane Database Syst. Rev., 2, No. 2, CD010945 (2021), https://doi.org/10.1002/14651858.CD010945.pub2.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Nevrologii i Psikhiatrii imeni S. S. Korsakova, Vol. 124, No. 1, pp. 121–127, January, 2024.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zorkina, Y.A., Morozova, I.O., Abramova, O.V. et al. Use of Modern Classification Systems for the Complex Diagnostics of Alzheimer’s Disease. Neurosci Behav Physi 54, 623–629 (2024). https://doi.org/10.1007/s11055-024-01637-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-024-01637-3