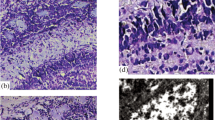
Analysis of the age-related dynamics of olfactory behavior in the odor preference and food search testsshowed that all male Wistar rats, regardless of age, preferred valerian essential oil, whose components have the properties of pheromones in rodents, when given a selection of eight essential oils; young rats displayed better food-seeking results than adult and old animals. Acute prenatal hypoxia (PH) on E14 (7% O2 for 3 h) led to impairment of the valerian odor preference at all ages studied and to decreased productivity of food searches. Neurodegenerative processes were seen in the piriform cortex after PH, with reductions in the number of neurons and increases in glial elements. We have previously observed these changes in the entorhinal cortex and hippocampus, but not in the olfactory bulbs. This suggests that PH-induced decreases in olfactory function in rats may result from impairments to the formation of the central elements of the analyzer during the first months of postnatal ontogeny.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Akers, K. G., Kushner, S. A., Leslie, A. T., et al., “Fetal alcohol exposure leads to abnormal olfactory bulb development and impaired odor discrimination in adult mice,” Mol. Brain, 4, 29 (2011), http://www.molecularbrain.com/content/4/1/29.
Apfelbach, R., Russ, D., and Slotnick, B. M., “Ontogenetic changes in odor sensitivity, olfactory receptor area and olfactory receptor density in the rat,” Chem. Senses, 16, 209–218 (1991), https://doi.org/10.1093/chemse/16.3.209.
Arzi, A. and Sobel, N., “Olfactory perception as a compass for olfactory neural maps,” Trends Cogn. Sci., 15, 537–545 (2011), 10/1016/j.tics.2011.09.007.
Barnes, K., Doherty, S., and Turner, A. J., “Endopeptidase-24.11 is the integral membrane peptidase initiating degradation of somatostatin in the hippocampus,” J. Neurochem., 64, 1826–1832 (1995), 10/1046/j.1471-4159.1995.64041826.x.
Barresi, M., Ciurleo, R., Giacoppo, S., et al., “Evaluation of olfactory dysfunction in neurodegenerative diseases,” J. Neurol. Sci, 323, No. 1–2, 16–24 (2012), 10/1016/j.jns.2012.08.028.
Belyakov, V. I., Makeeva, Yu. V., and Gribanov, I. A., “Effects of different odorants on the features of behavioral reactions in rats,” Evraz. Soyuz Uchenykh, 4, No. 13, 6–11 (2015), 10.31618/esu.2411-6467.8.53.1.
Bianchi, P., Bettini, S., Guidi, S., et al., “Age-related impairment of olfactory bulb neurogenesis in the Ts65Dn mouse model of Down syndrome,” Exp. Neurol., 251, 1 (2014), https://doi.org/10.1016/j.expneurol.2013.10.018.
Boryakova, E. E., Gladysheva, O. S., and Krylov, V. N., “Age-related dynamics of olfactory sensitivity in female laboratory mice to the odor of isovaleric acid,” Sens. Sistemy, 21, No. 4, 341–346 (2007).
Brennan, P. A. and Kendrick, K. M., “Mammalian social odours: attraction and individual recognition,” Philos. Trans. R. Soc. Lond. B Biol. Sci., 361, No. 1476, 2061–2078 (2006), https://doi.org/10.1098/rstb.2006.1931.
Bsteh, G., Hegen, H., Ladstätter, F., et al., “Change of olfactory function as a marker of infl ammatory activity and disability progression in MS,” Mult. Scler., 25, No. 2, 267–274 (2019), https://doi.org/10.1177/1352458517745724.
Carnemolla, S. E., Hsieh, J. W., Sipione, R., et al., “Olfactory dysfunction in frontotemporal dementia and psychiatric disorders: A systematic review,” Neurosci. Biobehav. Rev., 118, 588–611 (2020), https://doi.org/10.1016/j.neubiorev.2020.08.002.
Doty, R. L., “Olfactory dysfunction in Parkinson disease,” Nat. Dev. Neurol., 8, 329 (2012), https://doi.org/10.1038/nrneurol.2012.80.
Dubrovskaya, N. M. and Zhuravin, I. A., “Ontogenetic features of behavior in rats exposed to hypoxia on days 14 or 18 of embryogenesis,” Zh. Vyssh. Nerv. Deyat., 58, No. 5, 616–625 (2009), https://doi.org/10.1007/s11055-009-9235-2.
Dutriez, I., Salès, N., Fournié-Zaluski, M. C., and Roques, B. P., “Pre- and post-natal ontogeny of neutral endopeptidase 24-11 (‘enkephalinase ‘) studied by in vitro autoradiography in the rat,” Experientia, 48, 290 (1992), https://doi.org/10.1007/BF01930479.
Franks, K. M. and Isaacson, J. S., “Synapse-specifi c downregulation of NMDA receptors by early experience: a critical period for plasticity of sensory input to olfactory cortex,” Neuron, 47, 101–114 (2005), https://doi.org/10.1016/j.neuron.2005.05.024.
Ghosh, S., Larson, S. D., Hefzi, H., et al., “Sensory maps in the olfactory cortex defi ned by long-range viral tracing of single neurons,” Nature, 472, 217 (2011), https://doi.org/10.1038/nature09945.
Gulyaeva, N. V., Bobkova, N. V., Kolosova, N. G., et al., “Molecularcellular mechanisms of the pathogenesis of the sporadic form of Alzheimer’s disease: in vivo studies in rodent models,” Biokhimiya, 82, 1427–1443 (2017).
Koralnik, I. J. and Tyler, K. L., “COVID-19: A global threat to the nervous system,” Ann. Neurol., 88, 1 (2020), https://doi.org/10.1002/ana.25807.
Liu, Y.-H., Lai, W.-S., Tsay, H.-J., et al., “Effects of maternal immune activation on adult neurogenesis in the subventricular zone–olfactory bulb pathway and olfactory discrimination,” Schizophr. Res., 151, No. 1–3, 1–11 (2013).
Mel’nik, S. A., Gladysheva, O. S., and Krylov, V. N., “Age-related changes in olfactory sensitivity in male mice to the odor of isovaleric acid,” Sens. Sistemy, 23, No. 2, 151–155 (2009).
Mel’nik, S. A., Gladysheva, O. S., and Krylov, V. N., “Effects of prior action of isovaleric acid vapor on olfactory sensitivity in male house mice,” Sens. Sistemy, 26, No. 1, 52–56 (2012).
Morozova, A. Yu., Arutyunyan, A. V., Morozova, P. Y., et al., “Effect of prenatal hypoxia on activity of the soluble forms of cholinesterases in rat brain structures during early postnatal ontogenesis,” J. Evol. Biochem. Physiol., 56, No. 6, 531–540 (2020), https://doi.org/10.1134/S002209302006006X.
Mori, K. and Sakano, H., “How is the olfactory map formed and interpreted in the mammalian brain?” Annu. Rev. Neurosci., 34, 467–499 (2011), https://doi.org/10.1146/annurev-neuro-112210-112917.
Murphy, C., “Olfactory and other sensory impairments in Alzheimer disease,” Nat. Dev. Neurol., 15, 11–24 (2019), https://doi.org/10.1038/s41582-018-0097-5.
Nalivaeva, N. N., Belyaev, N. D., Lewis, D. I., et al., “Effect of sodium valproate administration on brain neprilysin expression and memory in rats,” J. Mol. Neurosci., 46, 569–577 (2012), https://doi.org/10.1007/s12031-011-9644-x.
Nalivaeva, N. N. and Turner, A. J., “Targeting amyloid clearance in Alzheimer’s disease as a therapeutic strategy,” Br. J. Pharmacol., 176, 3447–3463 (2019), https://doi.org/10.1111/bph.14593.
Nedogreeva, O. A., Stepanichev, M. Yu., and Gulyaeva, N. V., “Removal of the olfactory bulbs in mice leads to changes in emotional behavior,” Zh. Vyssh. Nerv. Deyat., 70, No. 1, 104–114 (2020), 10.31857/S0044467720010116.
Niimura, Y., “Evolutionary dynamics of olfactory receptor genes in chordates: interaction between environments and genomic contents,” Hum. Genomics, 4, No. 2, 107–118 (2009), https://doi.org/10.1186/1479-7364-4-2-107.
Nocera, S., Simon, A., Fiquet, O., et al., “Somatostatin serves a modulatory role in the mouse olfactory bulb: Neuroanatomical and behavioral evidence,” Front. Behav. Neurosci., 13, 61 (2019), https://doi.org/10.3389/fnbeh.2019.00061.
Parvand, M. and Rankin, C. H., “Is there a shared etiology of olfactory impairments in normal aging and neurodegenerative disease?” J. Alzheimers Dis., 73, 1–21 (2020), https://doi.org/10.3233/JAD-190636.
Paxinos, G. and Watson, C., The Rat Brain in Stereotaxic Coordinates, Academic Press-Elsevier Inc., Amsterdam (2007), 6th ed.
Roesch, M. R., Stalnaker, T. A., and Schoenbaum, G., “Associative encoding in anterior piriform cortex versus orbitofrontal cortex during odor discrimination and reversal learning,” Cereb. Cortex, 17, No. 3, 643–652 (2007), https://doi.org/10.1093/cercor/bhk009.
Saiz-Sanchez, D., Ubeda-Bañon, I., de la Rosa-Prieto, C., et al., “Somatostatin, Tau, and β-amyloid within the anterior olfactory nucleus in Alzheimer disease,” Exp. Neurol., 223, 347 (2010), https://doi.org/10.1016/j.expneurol.2009.06.010.
Sarnat, H. B. and Flores-Sarnat, L., “Development of the human olfactory system,” Handb. Clin. Neurol., 164, 29–45 (2019), https://doi.org/10.1016/B978-0-444-63855-7.00003-4.
Sokolov, V. E. and Voznesenskaya, V. V., “The role of early olfactory experience in individual recognition in gray rats,” Dokl. Akad. Nauk., 348, No. 5, 140–142 (1996).
Sosulski, D. L., Lissitsyna Bloom, M., Cutforth, T., et al., “Distinct representations of olfactory information in different cortical centres,” Nature, 472, 213 (2011), https://doi.org/10.1038/nature09868.
Sun, S., Li, T., Davies, H., et al., “Altered morphologies and functions of the olfactory bulb and hippocampus induced by miR-30c,” Front. Neurosci., 10, 207 (2016), https://doi.org/10.3389/fnins.2016.00207.
Suzuki, J. and Osumi, N., “Neural crest and placode contributions to olfactory development,” Curr. Top. Dev. Biol., 111, 351–74 (2015), https://doi.org/10.1016/bs.ctdb.2014.11.010.
Takahashi, H., Yoshihara, S., and Tsuboi, A., “The functional role of olfactory bulb granule cell subtypes derived from embryonic and postnatal neurogenesis,” Front. Mol. Neurosci., 11, 229 (2018), https://doi.org/10.3389/fnmol.2018.00229.
Tumanova, N. L., Vasilev, D. S., Dubrovskaya, N. M., et al., “Effects of prenatal hypoxia on the cytoarchitectonics and ultrastructural organization of brain areas associated with olfactory function in rats,” Tsitologiya, 71, No. 2, 163–175 (2021), 10.31857/S0041377121020085.
Tyul’kova, E. I., Vataeva, L. A., Samoilov, M. O., and Otellin, V. A., “Mechanisms forming brain reactions to the actions of hyperbaric hypoxia at different times of the prenatal period of development in rats,” Zh. Akursh. Zhensk. Bolez., 59, No. 4, (2010).
Vasilev, D. S., Dubrovskaya, N. M., Tumanova, N. L., and Zhuravin, I. A., “Prenatal hypoxia in different periods of embryogenesis differentially affects cell migration, neuronal plasticity, and rat behavior in postnatal ontogenesis,” Front. Neurosci., 10, 126 (2016), https://doi.org/10.3389/fnins.2016.00126.
Vasilev, D. S., Dubrovskaya, N. M., Zhuravin, I. A., and Nalivaeva, N. N., “Developmental profi le of brain neprilysin expression correlates with olfactory behaviour of rats,” J. Mol. Neurosci., (2021), https://doi.org/10.1007/s12031-020-01786-3.
Vasilev, D. S., Tumanova, N. L., and Kalinina, D. S., “Prenatal hypoxia leads to impaired formation of nervous tissue in the entorhinal area of the rat cerebral cortex,” Ros. Fiziol. Zh., 106, No. 10, 1278–1288 (2020), 10.31857/S086981392010012X.
Vasilev, D. S., Tumanova, N. L., and Zhuravin, I. A., “Structural changes in the nervous tissue of the neocortex during ontogeny of rats after hypoxia at different periods of ontogeny,” Zh. Evol. Biokhim. Fiziol., 44, No. 3, 258–266 (2008).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is dedicated to the memory of Doctor of Biological Sciences Igor Aleksandrovich Zhuravin
Translated from Zhurnal Vysshei Nervnoi Deyatel’nosti imeni I. P. Pavlova, Vol. 71, No. 3, pp. 415–427, May–June, 2021.
Rights and permissions
About this article
Cite this article
Dubrovskaya, N.M., Vasilev, D.S., Tumanova, N.L. et al. Prenatal Hypoxia Impairs Olfactory Function in Postnatal Ontogeny in Rats. Neurosci Behav Physi 52, 262–270 (2022). https://doi.org/10.1007/s11055-022-01233-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-022-01233-3