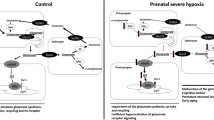
Prenatal hypoxia impairs brain formation, leading to the development of cognitive deficit in the postnatal period. The probable causes of this deficit may be linked with derangement of the functioning of the dorsal hippocampus and areas of the neocortex involved in its afferentation, particularly the entorhinal cortex, whose projection neurons innervate field CA1. The aim of the present work was to assess the effects of prenatal hypoxia on formation of nervous tissues in the entorhinal cortex during early ontogeny in rats subjected to prenatal hypoxia on days 14 or 18 of embryogenesis. In vivo labeling of neuroblasts formed in embryos at the moment of exposure to hypoxia with 3’-ethinyl-5-deoxyuridine showed that depending on the time of exposure, hypoxia impaired the formation and migration of neuroblasts to the lower (hypoxia on day 14 of pregnancy) or upper (hypoxia on day 18) layers of the entorhinal cortex. Hypoxia on E14 but not E18 led to a reduction in the number of neurons in the entorhinal cortex in rat pups in the first month of postnatal ontogeny. This reduction is evidence of neuron death. Impairment to the process of neurogenesis affected the formation of projection pyramidal neurons but had no effect on the population of inhibitory interneurons. Electron microscopy revealed pathological changes to neurons in the entorhinal cortex of rat pups on postnatal day 20 (cytoplasmic organelle lysis or hyperchromatosis). Hypoxia on day 18 of pregnancy produced no changes in cell composition or pyramidal neuron death in the postnatal period in the offspring. Prenatal hypoxia on day E14 evidently leads to impairment to the radial migration of neuroblasts in the entorhinal cortex and increases elimination of projection neurons in early postnatal ontogeny. Projection neuron death in the entorhinal cortex can lead to impairment to afferentation of hippocampal neurons. The selective action of prenatal hypoxia on the excitatory neuron population of the entorhinal cortex of the rat cerebral cortex may lead to disturbance of the balance between excitatory and inhibitory processes during subsequent development.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
D. Rice and S. Baron, “Critical periods of vulnerability for the developing nervous system: evidence from humans and animal models,” Environ. Health Perspect., 108, No. 3, 511–533 (2000).
P. Rakic, “Specification of cerebral cortical areas,” Science, 241, No. 4862, 170–176 (1988).
S. A. Bayer, “Development of the hippocampal region in the rat. II. Morphogenesis during embryonic and early postnatal life,” J. Comp. Neurol., 190, No. 1–2, 115–134 (1980).
S. A. Bayer, “Development of the hippocampal region in the rat. I. Neurogenesis examined with 3H -thymidine autoradiography,” J. Comp. Neurol., 190, No. 1-2, 87–114 (1980).
G. Kempermann, S. Jessberger, B. Steiner, and G. Kronenberg, “Milestones of neuronal development in the adult hippocampus,” Trends Neurosci., 27, No. 8, 447–452 (2004), https://doi.org/10.1016/j.tins.2004.05.013.
D. S. Vasilyev, N. L. Tumanova, and I. A. Zhuravin, “Change of the neocortex nervous tissue in rat ontogenesis after hypoxia at various times of embryogenesis,” Zh. Evolyuts. Biokhim. Fiziol., 44, 258–267 (2008).
D. S. Vasilev, N. M. Dubrovskaya, N. L. Tumanova, and I. A. Zhuravin, “Prenatal hypoxia in different periods of embryogenesis differentially affects cell migration, neuronal plasticity and rat behavior in postnatal ontogenesis Front,” Neuroscientist, 10, 126 (2016), https://doi.org/10.3389/fnins.2016.00126.
I. A. Zhuravin, N. L. Tumanova, and D. S. Vasilev, “Structural changes in hippocampal nervous tissue in rat ontogenesis after prenatal hypoxia,” Zh. Evolyuts. Biokhim. Fiziol., 45, No. 1, 138–140 (2009).
I. A. Zhuravin, N. M. Dubrovskaya, D. S. Vasilev, et al., “Prenatal hypoxia produces memory deficits associated with impairment of long-term synaptic plasticity in young rats,” Neurobiol. Learn. Mem., 164, 1–10 (2019), https://doi.org/10.1016/j.nlm.2019.107066.
C. Perez-Cruz, M. W. Nolte, M. M. van Gaalen, et al., “Reduced spine density in specific regions of CA1 pyramidal neurons in two transgenic mouse models of Alzheimer’s disease,” J. Neurosci., 31, No. 10, 3926–3934 (2011), https://doi.org/10.1523/JNEUROSCI.6142-10.2011.
S. A. Bayer and J. Altman, “Neurogenetic timetables in the telencephalon,” in: The Rat Nervous System, Elsevier, Academic Press (2004), 3rd ed., pp. 28–36.
G. Paxinos and C. Watson, The Rat Brain in Stereotaxic Coordinates, Elsevier Inc. (2007), 6th ed.
D. S. Vasilev, N. L. Tumanova, A. A. Kovalenko, et al., “Neuroinflammatory processes affect structural abnormalities in the amygdala in the lithium-pilocarpine model of epilepsy in rats,” Ros. Fiziol. Zh., 105, No. 6, 694–706 (2019), https://doi.org/10.1134/S0869813919060098.
N. L. Tumanova, D. S. Vasilev, N. M. Dubrovskaya, and I. A. Zhuravin, “Ultrastructural alterations in the sensorimotor cortex upon delayed development of motor behavior in early ontogenesis of rats exposed to the prenatal hypoxia,” Cell Tiss. Biol., 12, No. 5, 419–425 (2018), https://doi.org/10.1134/S1990519X18050097.
E. S. Ang, Jr., V. Gluncic, A. Duque, et al., “Prenatal exposure to ultrasound waves impacts neuronal migration in mice,” Proc. Natl. Acad. Sci. USA, 103, No. 34, 12903–10 (2006), https://doi.org/10.1073/pnas.0605294103.
M. W. Miller, “Effects of alcohol on the generation and migration of cerebral cortical neurons,” Science, 233, No. 4770, 1308–1311 (1986).
M. P. Aronne, T. Guadagnoli, P. Fontanet, et al., “Effects of prenatal ethanol exposure on rat brain radial glia and neuroblast migration,” Exp. Neurol., 229, 364–371 (2011), https://doi.org/10.1016/j.expneurol.2011.03.002.
D. S. Kalinina, D. S. Vasilev, A. V. Volnova, et al., “Age dynamics of electrocorticogram and 4-aminopyridine-induced seizures in rats undergoing prenatal hypoxia,” Dev. Neurosci., 41, No. 1–2, 56–66 (2019), https://doi.org/10.1159/000497224.
I. A. Zhuravin, N. L. Tumanova, and D. S. Vasilyev, “Changes in mechanisms of brain adaptation in postnatal ontogenesis of rats after prenatal hypoxia,” Dokl. Akad. Nauk, 425, 123–125 (2009).
R. Okubo-Suzuki, D. Okada, M. Sekiguchi, and K. Inokuchi, “Synaptopodin maintains the neural activity-dependent enlargement of dendritic spines in hippocampal neurons,” J. Mol. Cell. Neurosci., 38, No. 2, 266–76 (2008), https://doi.org/10.1016/j.mcn.2008.03.001.
M. H. Golan, R. Mane, G. Molczadzki, et al., “Impaired migration signaling in the hippocampus following prenatal hypoxia,” Neuropharmacology, 57, 511–522 (2009), https://doi.org/10.1016/j.neuropharm.2009.07.028.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Rossiiskii Fiziologicheskii Zhurnal imeni I. M. Sechenova, Vol. 106, No. 10, pp. 1278–1288, October, 2020.
Rights and permissions
About this article
Cite this article
Vasilev, D.S., Tumanova, N.L. & Kalinina, D.S. Prenatal Hypoxia Leads to Impaired Formation of Nervous Tissue in the Entorhinal Area of the Cerebral Cortex in Rats. Neurosci Behav Physi 51, 681–686 (2021). https://doi.org/10.1007/s11055-021-01121-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-021-01121-2