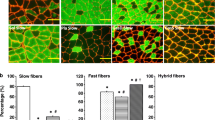
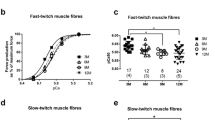
We report here a comparative study of activity in signal pathways and gene expression in the red (RGM) and white (WGM) parts of the gastrocnemius muscle in rats after series of short (1 sec) tetanic contractions evoked by stimulation of the motor nerve at a frequency of 100 Hz and an amplitude sufficient to activate all the motor units of the muscle. At 2 h after stimulation, WGM showed more marked increases in the level of ERK1/2 phosphorylation than RGM, though increases in AMPK phosphorylation were no different. Furthermore, the increases in MyoD and myogenin mRNA in WGM were significantly greater than those in RGM, while the effects of stimulation on expression of the IGF-1, MaFbx, and MuRF genes were weak and similar in WGM and RGM. There was also an increase in the content of myostatin mRNA in RGM. Thus, glycolytic muscle fibers in WGM display more marked regulatory hypertrophic-type shifts than the oxidative muscle fibers making up RGM.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
A. A. Borzykh, D. K. Gainullina, I. V. Kuz’min, et al., “Comparative analysis of gene expression in locomotor muscles and diaphragm in rats,” Ros. Fiziol. Zh., 98, No. 12, 1587–1594 (2012).
D. G. Allen, G. D. Lamb, and H. Westerblad, “Skeletal muscle fatigue: cellular mechanisms,” Physiol. Rev., 88, No. 1, 287–332 (2008).
P. H. Atherton, J. M. Higginson, J. Singh, and H. Wackerhage, “Concentrations of signal transduction proteins exercise and insulin responses in rat extensor digitorum longus and soleus muscles,” Mol. Cell Biochem., 261, No. 1–2, 111–116 (2004).
M. Badier, C. Guillot, C. Danger, et al., “M-wave changes after highand low-frequency electrically induced fatigue in different muscles,” Muscle Nerve, 22, No. 4, 488–496 (1999).
J. D. Bartlett, J. C. Hwa, T. S. Jeong, et al., “Matched work high-intensity interval and continuous running induce similar increases in PGC-Ialpha mRNA, AMPK, p38, and p53 phosphorylation in human skeletal muscle,” J. Appl. Physiol., 112, 1135–1143 (2012).
S. M. Baylor and S. Hollingworth, “Intracellular calcium movements during excitation-contraction coupling in mammalian slowtwitch and fast-twitch muscle fibers,” J. Gen. Physiol., 139, No. 4, 261–272 (2012).
D. Bloemberg and J. Quadrilatero, “Rapid determination of myosin heavy chain expression in rat, mouse, and human skeletal muscle using multicolor immunofluorescence analysis,” PLoS One, 7, No. 4, e35273–e35284 (2012).
C. J. Carlson, F. W. Booth, and S. E. Gordon, “Skeletal muscle myostatin mRNA expression is fiber-type specific and increases during hindlimb unloading,” Am. J. Physiol., 277, No. 2, R601–R606 (1999).
N. M. Cermak, T. Snijders, B. R. McKay, et al., “Eccentric exercise increases satellite cell content in type II muscle fibers,” J. Med. Sci. Sports Exerc., 45, No. 2, 230–237 (2013).
M. D. Delp and C. Duan, “Composition and size of type I, IIA, IID/X, and IIB fibers and citrate synthase activity of rat muscle,” J. Appl. Physiol., 80, No. 1, 261–270 (1996).
V. C. Foletta, L. J. White, A. E. Larsen, et al., “The role and regulation of MAFbx/atrogin-1 and MuRF1 in skeletal muscle atrophy,” Pflügers Arch., 461, No. 3, 325–335 (2011).
E. L. Glynn, C. S. Fry, M. J. Drummond, et al., “Muscle protein breakdown has a minor role in the protein anabolic response to essential amino acid and carbohydrate intake following resistance exercise,” Am. J. Physiol. Regul. Integr. Comp. Physiol., 299, No. 2, R533–R540 (2010).
M. D. Gomes, S. H. Lecker, R. T. Jagoe, et al., “Atrogin-1, a muscle-specific F-box protein highly expressed during muscle atrophy,” Proc. Natl. Acad. Sci. USA, 98, No. 25, 14,440–14,445 (2001).
K. Gundersen, “Excitation-transcription coupling in skeletal muscle: the molecular pathways of exercise,” Biol. Rev. Camb. Philos. Soc., 86, No. 3, 564–600 (2011).
K. M. Heinemeier, J. L. Olesen, F. Haddad, et al., “Effect of unloading followed by reloading on expression of collagen and related growth factors in rat tendon and muscle,” J. Appl. Physiol., 106, No. 1, 178–186 (2009).
S. M. Hughes, J. M. Taylor, S. J. Tapscott, et al., “Selective accumulation of MyoD and myogenin mRNAs in fast and slow adult skeletal muscle is controlled by innervation and hormones,” Development, 118, No. 4, 1137–1147 (1993).
E. Hultman and P. L. Greenhaff, “Skeletal muscle energy metabolism and fatigue during intense exercise in man,” Sci. Prog., 75, No. 298, 361–370 (1991).
M. Ishido, K. Kami, and M. Masuhara, “Localization of MyoD, myogenin and cell cycle regulatory factors in hypertrophying rat skeletal muscles,” Acta Physiol. Scand., 180, No. 3, 281–289 (2004).
M. Karalaki, S. Fili, A. Philippou, and M. Koutsilieris, “Muscle regeneration: cellular and molecular events,” In Vivo, 23, No. 5, 779–796 (2009).
I. Kim, D. Yang, X. Tang, and J. L. Carroll, “Reference gene validation for qPCR in rat carotid body during postnatal development,” BMC Res. Notes, 4, 440–448 (2011).
B. J. Krawiec, G. J. Nystrom, R. A. Frost, et al., “AMP-activated protein kinase agonists increase mRNA content of the muscle-specific ubiquitin ligases MAFbx and MuRF1 in C2C12 cells,” Am. J. Physiol. Endocrinol. Metab., 292, No. 6, E1555–E1567 (2007).
R. S. Lee-Young, B. J. Canny, D. E. Myers, and G. K. McDonnell, “AMPK activation is fiber type specific in human skeletal muscle: effects of exercise and short-term exercise training,” J. Appl. Physiol., 107, No. 1, 283–289 (2009).
B. R. MacIntosh, S. P. Esau, R. J. Holash, and J. R. Fletcher, “Procedures for rat in situ skeletal muscle contractile properties,” J. Vis. Exp., 56, e3167 (2011).
A. Matsakas, C. Bozzo, N. Cacciani, et al., “Effect of swimming on myostatin expression in white and red gastrocnemius muscle and in cardiac muscle of rats,” Exp. Physiol., 6, 983–994 (2006).
T. Moritani, M. Muro, and A. Kijima, “Electromechanical changes during electrically induced and maximal voluntary contractions: electrophysiologic responses of different muscle fiber types during stimulated contractions,” Exp. Neurol., 88, No. 3, 471–483 (1985).
G. A. Nader and K. A. Esser, “Intracellular signaling specificity in skeletal muscle in response to different modes of exercise,” J. Appl. Physiol. (1985), 90, No. 5, 1936–1942 (2001).
A. S. Pimenta, R. H. Lambertucci, R. Gorjão, et al., “Effect of a single session of electrical stimulation on activity and expression of citrate synthase and antioxidant enzymes in rat soleus muscle,” Eur. J. Appl. Physiol. 102, No. 1, 119–126 (2007).
D. Popov, R. Zinovkin, E. Karger, et al., “Effects of continuous and intermittent aerobic exercise upon mRNA expression of metabolic genes in human skeletal muscle,” J. Sports Med. Phys. Fitness, 54, No. 3, 362–369 (2014).
A. C. Ronda, A. Vasconsuelo, and R. Boland, “Extracellular-regulated kinase and p38 mitogen-activated protein kinases are involved in the antiapoptotic action of 17beta-estradiol in skeletal muscle cells,” J. Endocrinol., 206, No. 2, 235–246 (2010).
D. G. Sale, “Influence of exercise and training on motor unit activation,” Exerc. Sport Sci. Rev., 15, 95–151 (1987).
H. Shi, J. M. Scheffler, J. M. Pleitner, et al., “Modulation of skeletal muscle fiber type by mitogen-activated protein kinase signaling,” FASEB J., 22, No. 8, 2990–3000 (2008).
A. Tsutaki, R. Ogasawara, K. Kobayashi, et al., “Effect of intermittent low-frequency electrical stimulation on the rat gastrocnemius muscle,” Biomed. Res. Int., 2013, 480620 (2013).
T. Van Wessel, A. de Haan, W. J. van der Laarse, and R. T. Jaspers, “The muscle fiber type-fiber size paradox: hypertrophy or oxidative metabolism?” Eur. J. Appl. Physiol., 110, No. 4, 665–694 (2010).
C. Wretman, U. Widegren, A. Lionikas, et al., “Differential activation of mitogen-activated protein kinase signalling pathways by isometric contractions in isolated slow- and fast-twitch rat skeletal muscle,” Acta Physiol. Scand., 70, No. 1, 45–49 (2000).
X. Yang, A. Wei, Y. Liu, et al., “IGF-1 protects retinal ganglion cells from hypoxia-induced apoptosis by activating the Erk-1/2 and Akt pathways,” Mol. Vis., 19, 1901–1912 (2013).
N. Zanou and P. Gailly, “Skeletal muscle hypertrophy and regeneration: interplay between the myogenic regulatory factors (MRFs) and insulin-like growth factors (IGFs) pathways,” Cell Mol. Life Sci., 70, No. 21, 4117–4130 (2013).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Rossiiskii Fiziologicheskii Zhurnal imeni I. M. Sechenova, Vol. 101, No. 11, pp. 1289–1298, November, 2015.
Rights and permissions
About this article
Cite this article
Borzykh, A.A., Kuz’min, I.V., Lysenko, E.A. et al. Measures of Growth Processes and Myogenesis in Glycolytic and Oxidative Muscle Fibers in Rats after Indirect Electrical Stimulation. Neurosci Behav Physi 47, 352–358 (2017). https://doi.org/10.1007/s11055-017-0404-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-017-0404-4