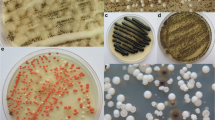
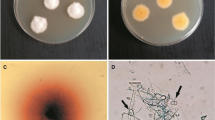
Abstract
Exophiala dermatitidis is an ascomycetous black yeast from the order Chaetothyriales. Its growth characteristics include the polymorphic life cycle, ability to grow at high and low temperatures, at a wide pH range, survival at high concentrations of NaCl, and survival at high UV and radioactive radiation. Exophiala dermatitidis causes deep or localized phaeohyphomycosis in immuno-compromised people worldwide and is regularly encountered in the lungs of cystic fibrosis patients. Regardless of numerous ecological studies worldwide, little is known about its natural habitat or the possible infection routes. The present review summarizes the published data on its frequency of occurrence in nature and in man-made habitats. We additionally confirmed its presence with culture-depending methods from a variety of habitats, such as glacial meltwater, mineral water, mineral-rich salt-pan mud, dishwashers, kitchens and different environments polluted with aromatic hydrocarbons. In conclusion, the frequency of its recovery was the highest in man-made indoor habitats, connected to water sources, and exposed to occasional high temperatures and oxidative stress.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The ascomycetous black yeast Exophiala dermatitidis (Kano) de Hoog is one of cca. 40 species within the genus Exophiala (Herpotrichiellaceae, Chaetothyriales). Molecular studies connected the anamorph species with the teleomorph genus Capronia [1]. Numerous species of the genus Exophiala are opportunistic pathogens of human and warm-blood animals [2,3,4,5,6,7], while some are causative disease agents of cold-blooded animals [8]. New species of Exophiala continue to be described, in particular linked to polluted environment [9], human homes and human mycoses [10].
Exophiala dermatitidis is a phenotypically plastic, physiologically versatile, extremotolerant fungus. It is polymorphic since it grows as yeasts, hyphae or meristematic clumps [11], colonies range from olivaceous brown to black [12]. The fungus is able to degrade many substrates, it is frequently present in environments rich in aromatic hydrocarbons [13] suggesting its ability to assimilate them, which still needs to be demonstrated. It grows in a temperature range from 4 to 47 °C, survives a wide pH range and tolerates up to 3 M NaCl [12, 14, 15]. In spite of its adaptability, isolations of E. dermatitidis from nature were only rare and sporadic. It is known as an opportunistic pathogen of humans, currently classified under the Biological safety level 2 [16]. Exophiala dermatitidis was regularly reported as a causative agent of skin and subcutaneous infections [17], systemic infections [18], catheter-related [19] and respiratory tract infections [20], especially in immuno-compromised patients. It is regularly observed as a pulmonary colonizer of patients with cystic fibrosis [21], a disorder characterized by exceptionally mucous and salty lungs. Rare cases of brain infection, located primarily in East Asia, were also reported [22].
Exophiala dermatitidis has been occasionally retrieved from nature, from tropical rainforests [23] to glaciers [24]. Its occurrence increased considerably in human-made environments rich in aromatic hydrocarbons [13] and with high humidity, such as steam baths [25], bathrooms [26, 27], kitchens [28] and dishwashers [14, 29]. After its recent detection in drinking water [30] and hot dishwasher aerosols [28], it was speculated that water serves as a transmission vector from nature indoors, while high temperatures, plastic materials and oxidative agents enrich its presence both indoors and in household appliances, increasing the risk of human exposure. The isolation techniques are reviewed in Table 1.
The present study is thus a review of published findings on the occurrence of E. dermatitidis in habitats outdoors and indoors, supplemented with yet unpublished data. The summarized ecological data might unravel infection routes of this fungus and enable a better estimate of the potential risk of infections in predisposed people.
Exophiala dermatitidis as a Causative Agent of Human Infections
The genus Exophiala contains cca. 40 species, 17 of which were recognized as opportunistic human pathogens. The most common is E. dermatitidis [31], representing 30% of all Exophiala infections in the USA [17]. It caused mainly deep infections of lungs and abdomen (19%) and rarely of brain, ears or eyes. Sporadically it caused cutaneous (6%) and subcutaneous (2%) phaeohyphomycosis [17]. The species was involved globally in similar infections [32, 33], subcutaneous phaeohyphomycosis prevailing in (sub)tropical climate zones [34]. Other localized infections are keratitis [35, 36], otitis [37], peritonitis [38, 39], pneumonia [20, 40] and other pulmonary infections [41,42,43], in particular in cystic fibrosis patients [21, 44,45,46]. Localized systemic infections are usually related to predisposing factors such as cystic fibrosis, bronchiectasis, diabetes mellitus, catheterization, rheumatoid arthritis and leukaemia [19, 22, 47, 48].
In some cases E. dermatitidis showed remarkable neurotropism, acting as a true pathogen, infecting mainly healthy individuals [34]. It colonized the brain tissues as cerebral metastases, with a high mortality rate [18, 22, 49, 50], with sometimes severe secondary cutaneous mutilation [51,52,53,54]. Brain infections were reported mainly from Asia [55, 56]. Today, it seems probable that at least part of these cases concerned humans with rare inherited innate immune disorders, such as CARD9 or STAT1, previously unknown [57], although it remains puzzling why so many such cases are observed in East Asia. Cases of meningitis in the USA were due to epidural injection of steroids into cerebrospinal fluid which proved to be contaminated with E. dermatitidis [58]. Its presence was reported also in stool samples of otherwise healthy individuals in very low frequencies (0.5–1.2%) during periods of severe diarrhoea [25, 59].
Two probable routes of infection are noted in black fungi: via traumatic implantation in the skin, or via inhalation. Disseminated infections may have a pulmonary onset, but the severe (sub)cutaneous are secondary, i.e. emerging via the blood. Conversely, cutaneous infection with cerebral metastasis has been reported [60]. A possible connection between clinical and environmental isolates via trauma was confirmed in an AFLP analysis of 285 strains from global sampling study [61]. Infection routes for neurotropic infections remain unknown [22].
Exophiala dermatitidis in Terrestrial and Water-Related Natural Environments
Although E. dermatitidis was relatively frequently isolated from clinical specimens around the globe, its main natural habitat(s) remained unknown. Sporadically it has been isolated from plant material in Brazil [62], from tropical fruits in Thailand [23], from faeces of frugivorous birds, hornbills and flying foxes in Thailand [23], and from the cuticle of leaf-cutting ants in Brazil [63]. Based on these data it was for long speculated that tropical rainforests are its main natural source.
Its presence has later been noted in sea water and in fresh water-related environments, such as deep-sea sediments in India [64], deep-sea hydrothermal vents in Portugal [65], beach sand in Greece [66], natural hot springs in Thailand [23] and in glaciers in Italy and on Antarctica [24, 67] (Table 1).
All environments listed above have a strong connection with low-nutrient water. To confirm this hypothesis we additionally sampled different water sources in Slovenia: melt water from the Triglav glacier, 5 natural springs of mineral water, 10 sea water samples from the Adriatic Sea and 18 samples of brine from the salterns. We also analysed 24 samples of saltern sediments in contact with brine (mineral mud from salterns or fango), used for therapeutic skin treatments. Exophiala dermatitidis was recovered with a low frequency from glacial melt water, once from mineral water and once from fango, while samples of Adriatic Sea water and brine were negative (Table 2). Although frequencies are low, positive sampling sites were invariably oligotrophic.
Exophiala dermatitidis is Enriched in Environments Polluted with Aromatic Hydrocarbons
Order Chaetothyriales harbours numerous etiologic agents of chromoblastomycosis, phaeohyphomycosis and other diseases of vertebrate hosts, which range from mild cutaneous to fatal cerebral or disseminated infections and affect humans and cold-blooded animals. Chaetothyriales also comprise polyextremotolerant species with aquatic, rock-inhabiting, ant-associated and mycoparasitic lifestyles. Members of nearly all Chaetothyriales clades can degrade toxic monoaromates and tolerate or sometimes even prefer tannin-rich material, aromates and etheric oils, gasoline and mine waste rich in heavy metals, suggesting a high degree of versatile extremotolerance and tendency to pathogenicity. A recent study that compared genomes of 23 species within the Chaetothyriales [68] revealed a reduction of genes for carbohydrate degrading enzymes, expansion in certain protein degrading enzyme families, in alcohol dehydrogenase domains and in the trichothecene efflux pump. Analysis of cytochrome p450 genes (CYPs) which play a fundamental role in primary, secondary and xenobiotic metabolism and in a large number of detoxification reactions as well as in the metabolism of specific xenobiotics revealed an extraordinary p450 expansion, indicating that some black yeasts are among the Ascomycota species with the highest number of CYPs. PHA (2-hydroxy phenylacetate) and HGD (homogentisate 1,2-dioxygenase) genes involved in aromatic compound metabolism were organized in a syntenic cluster with additional conserved genes coding for hypothetical proteins, an MFS transporter, a trehalose-6-phosphate hydrolase (T6P-hydrolase) and a fumarylacetoacetase, which have been linked to protection against heat, freezing, starvation, dehydration and desiccation stress and pathogenicity [68].
In accordance with the observations above, aromatic hydrocarbons, such as oil, gasoline and components of different detergents which are toxic for most microorganisms, can serve as a substrate for enrichment of E. dermatitidis [13, 26, 69]. A number of related Exophiala species (particularly E. xenobiotica and E. oligosperma) can assimilate alkylbenzenes (i.e. toluene, ethylbenzene and styrene) as the sole source of carbon and energy. This uncommon metabolic feature among eukaryotes has been investigated for bioremediation purposes, e.g. the treatment of polluted air in biofilters, however, with concerns regarding the potential biohazard of enriching and aerosolizing black yeast cells [70].
Although a recent study [71] showed that only a single strain out of 9 tested E. dermatitidis assimilated toluene, while all 9 strains did not grow on hexadecane and polychlorinated biphenyl 126 (PCB126) as the sole carbon and energy sources, the reviewed ecological data show higher frequency of occurrence of E. dermatitidis in habitats with aromatic hydrocarbons. For example, creosote-treated railway sleepers revealed a higher frequency of E. dermatitidis (3–10%) in comparison with environments without aromatic hydrocarbons (Table 1) [23, 29, 62, 72], especially in humid and temperate climates [73]; Exophiala dermatitidis was also found in crude oil in Britain [74]. Additionally we sampled in Slovenia 8 handles of petrol pipes and 11 cement floors of gas stations polluted with oil or gasoline, 19 rubbers on car tank reservoirs, 21 sinks for waste water in carwashes and 3 railway sleepers, which were in contact with sea water. Only the railway sleepers were negative for E. dermatitidis, while we successfully recovered it with frequencies between 5 and 18% from all other listed habitats (Table 2).
Exophiala dermatitidis Mostly Invades Indoor Environments
In developed countries, tap water system represents the connection between the natural water environment and the indoor environment, where water is used for drinking, housework and hygiene. Presence of Exophiala species in biofilms at domestic water taps was thoroughly investigated in Germany [75]. Although this study recovered many different Exophiala species, primarily E. lecanii-corni, no strains of E. dermatitidis were obtained. However, a recent study revealed the presence of E. dermatitidis in 12.5% of sampled Slovenian groundwater samples and in 6% of tap water samples (Table 1) [30]. This study showed the importance of chemical characteristics of water samples exposing the concentration of calcium and magnesium ions, and nitrate as the crucial factors with positive influence on the fungal presence in water [30]. These results might explain the differences between the two studies, suggesting that areas with hard water are more likely colonized with black yeasts [30]. This is in accordance with previous study [11], showing that higher calcium concentrations favour polarized yeast/hyphal growth, whereas lower values induced multicellular and meristematic growth in E. dermatitidis [11].
In indoor environments E. dermatitidis was isolated primarily from niches in regular contact with water. Its first indoor isolation was from a humidifier [76]. Twenty years later it was detected in steam baths in Slovenia and in The Netherlands [25], later also in Austria and Thailand [23, 59], with a higher frequency of occurrence than in any natural sample. The study reported high temperature (40–42 °C) as the decisive parameter and this matches with prevalence of E. dermatitidis in (sub)tropical climates [61, 72]. Temperatures in bathrooms are lower than in saunas, and frequencies are concomitantly lower (Table 1): E. dermatitidis has been isolated from bathrooms in Japan [26] and Turkey [27].
Highest frequencies of E. dermatitidis in indoor environments are steam baths and dishwashers, where high temperatures, water and changing pH combine. Dishwashers are characterized by the presence of plastic elements, silicon or rubber seals, which might act as an enrichment factor or trapping surfaces. The enriching could be due to the presence of aromatic compounds present in the added surfactants (i.e. linear alkylbenzene sulphonate or LAS), related to alkylbenzenes, as well as to slowly released aromatics (e.g. bisphenols) from the rubber seals. Additionally, rugose and hydrophobic rubber and plastic surfaces offer favourable surface conditions for the promotion of biofilm formation.
In a global study involving 189 dishwashers, 28.6% of the rubber seals harboured a characteristic fungal community dominated by E. dermatitidis [14]. Later studies confirmed these observations (Table 1) [27, 28, 77] and revealed that E. dermatitidis colonizes the dishwashers interior with rubbers, drains and side nozzles being the most contaminated sites. Exophiala dermatitidis was also detected in 6% of dishwasher waste water samples [28], in hot aerosols released from the dishwashers immediately after the washing cycle, and on surfaces of kitchens with dishwashers (Table 1), indicating possible transmission routes and cyclic environmental contamination [28].
We confirmed these results by additional sampling of dishwashers, kitchen sinks and dish drying racks (Table 2). Results were comparable with previous studies. Additionally, we sampled rubber seals in kitchen refrigerators of which one positive indicated a broad thermotolerance of E. dermatitidis, and its preference for rubber and humidity.
Physiology and Polyextremotolerant Nature of Exophiala dermatitidis
The environmental occurrence of E. dermatitidis indicates its versatility. It assimilates d-glucose, d-galactose, d-xylose, L-arabinose, sucrose, maltose, α,α-trehalose, cellobiose, salicin, raffinose and melezitose. It can utilize alcohols like glycerol, meso-erythritol, xylitol, d-glucitol and ethanol, as well as ethylamine, lysine and cadaverine. It differs from other Exophiala species, being incapable to utilize nitrate, nitrite, creatine and creatinine [12]. As mentioned earlier, it can also be enriched by toxic monoaromatic hydrocarbons [13, 69].
Exophiala dermatitidis strains show three main genotypes A, B and C with rDNA ITS [78]. Genotype A is prevalent in dishwashers and the preponderant genotype on humans, B was mainly connected to the natural habitats, and C is extremely rare [14, 59]. Melanized cell walls are a unifying characteristic for black yeasts. Melanin is essential in resistance to stressful conditions [79]. Polyextremotolerant E. dermatitidis is able to sustain high dosages of UV and radioactive radiation [80, 81], and it can grow at temperatures above 40 °C, and tolerates temperatures from 4 to 47 °C [12, 14]. The same holds true for pH (2.5–12.5) and NaCl (up to 17%) [14, 77]. Its chaotolerance was confirmed in media containing NaCl, KCl, MgCl2, CaCl2, NaBr and MgSO4 at concentrations of 2.5, 2.5, 0.75, 0.5, 0.75 and 3 M, respectively [15]. Polymorphism, melanization and production of extracellular polysaccharides (EPS) are considered as the most important factors explaining its polyextremotolerant nature [5, 79].
Possible Routes of Infection
Exophiala dermatitidis is an important opportunistic pathogen able to cause a gamut of human infections. Since its natural niche is unknown, it was only sporadically isolated from natural environments, and the risk of exposure may be low. Its occurrence is enhanced by high environmental temperatures, the presence of aromatic, aliphatic and aromatic hydrocarbons, and the presence of water. Oligotrophism was suggested from its presence in low-nutrient waters of extremely differing temperatures, and salinities. Its isolation from household tap water confirms water as a route of transmission, where it is enriched in indoor niches such as bathrooms and kitchens, and even in steam baths and dishwashers. Creosote, mono- and polyaromates, rubber seals, plastics and detergents may all be favourable in strengthening its competition with susceptible co-inhabitant microorganisms (Fig. 1).
In practice, exposition risks of dry habitats such as railway ties are judged to be low as well as infections via small skin trauma. Effective transmission is expected to take place via inhaled aerosols (steam baths, shower heads and household appliances). The CF-population is the main group of patients with regular pulmonary presence of E. dermatitidis. These patients spend a lot of time indoors and also use personal aerosolizers to relief their stuffiness. Further research is needed to establish a potential connection between the presence of E. dermatitidis in indoor environments and its occurrence in the patients.
References
Haase G, Sonntag L, Melzer-Krick B, de Hoog GS. Phylogenetic inference by SSU-gene analysis of members of the Herpotrichiellaceae with special reference to human pathogenic species. Stud Mycol. 1999;43:80–97.
Badali H, Najafzadeh MJ, van Esbroeck M, et al. The clinical spectrum of Exophiala jeanselmei, with a case report and in vitro antifungal susceptibility of the species. Med Mycol. 2010;48:318–27.
Badali H, Chander J, Bayat M, et al. Multiple subcutaneous cysts due to Exophiala spinifera in an immunocompetent patient. Med Mycol. 2012;50:207–13.
Morio F, Berre JY, Garcia-Hermoso D, et al. Phaeohyphomycosis due to Exophiala xenobiotica as a cause of fungal arthritis in an HIV-infected patient. Med Mycol. 2012;50:513–7.
Pattanaprichakul P, Bunyaratavej S, Leeyaphan C, et al. An unusual case of eumycetoma caused by Exophiala jeanselmei after a sea urchin injury. Mycoses. 2013;56:491–4.
Woo PC, Ngan AH, Tsang CC, et al. Clinical spectrum of Exophiala infections and a novel Exophiala species, Exophiala hongkongensis. J Clin Microbiol. 2013;51:260–7.
Najafzadeh MJ, Suh MK, Lee MH, et al. Subcutaneous phaeohyphomycosis caused by Exophiala equina, with susceptibility to eight antifungal drugs. J Med Microbiol. 2013;62:797–800.
de Hoog GS, Vicente VA, Najafzadeh MJ, et al. Waterborne Exophiala species causing disease in cold-blooded animals. Persoonia. 2011;27:46–72.
Seyedmousavi S, Badali H, Chlebicki A, et al. Exophiala sideris, a novel black yeast isolated from environments polluted with toxic alkyl benzenes and arsenic. Fungal Biol. 2011;115:1030–7.
Borman AM, Fraser M, Szekely A, Larcombe DE, Johnson EM. Rapid identification of clinically relevant members of the genus Exophiala by matrix-assisted laser desorption ionization-time of flight mass spectrometry and description of two novel species, Exophiala campbellii and Exophiala lavatrina. J Clin Microbiol. 2017;55:1162–76.
Karuppayil SM, Szaniszlo PJ. Importance of calcium to the regulation of polymorphism in Wangiella (Exophiala) dermatitidis. Med Mycol. 1997;35:379–88.
de Hoog GS, Haase G. Nutritional physiology and selective isolation of Exophiala dermatitidis. Antonie Van Leeuwenhoek. 1993;64:17–26.
Prenafeta-Boldú XF, Summerbell R, de Hoog GS. Fungi growing on aromatic hydrocarbons: biotechnology’s unexpected encounter with biohazard? FEMS Microbiol Rev. 2006;30:109–30.
Zalar P, Novak M, de Hoog GS, Gunde-Cimerman N. Dishwashers—a man-made ecological niche accommodating human opportunistic fungal pathogens. Fungal Biol. 2011;115:997–1007.
Zajc J, Džeroski S, Kocev D, et al. Chaophilic or chaotolerant fungi: a new category of extremophiles? Front Microbiol. 2014;5:1–15.
de Hoog GS, Guarro J, Gené J, Figueras MJ. Atlas of clinical fungi. 3rd ed. Centraalbureau Voor Schimmelcultures/Univeesitat Rovira I Virgili; 2009.
Zeng JS, Sutton DA, Fothergill AW, et al. Spectrum of clinically relevant Exophiala species in the United States. J Clin Microbiol. 2007;45:3713–20.
Hiruma M, Kawada A, Ohata H, et al. Systemic phaeohyphomycosis caused by Exophiala dermatitidis. Mycoses. 1993;36:1–7.
Nachman S, Alpan O, Malowitz R, Spitzer ED. Catheter-associated fungemia due to Wangiella (Exophiala) dermatitidis. J Clin Microbiol. 1996;34:1011–3.
Cohen ZY, Stead W. Exophiala pneumonia presenting with a cough productive of black sputum. Case Rep Infect Dis. 2015;2015:821049.
Kondori N, Gilljam M, Lindblad A, et al. High rate of Exophiala dermatitidis recovery in the airway of patients with cystic fibrosis is associated with pancreatic insufficiency. J Clin Microbiol. 2011;49:1004–9.
Horré R, de Hoog GS. Primary cerebral infections by melanized fungi: a review. Stud Mycol. 1999;43:176–93.
Sudhadham M, Prakitsin S, Sivichai S, et al. The neurotropic black yeast Exophiala dermatitidis has a possible origin in the tropical rain forest. Stud Mycol. 2008;61:145–55.
Branda E, Turchetti B, Diolaiuti G, et al. Yeast and yeast-like diversity in the southernmost glacier of Europe (Calderone Glacier, Apennines, Italy). FEMS Microbiol Ecol. 2010;72:354–69.
Matos T, de Hoog GS, de Boer AG, de Crom I, Haase G. High prevalence of the neurotrope Exophiala dermatitidis and related oligotrophic black yeasts in sauna facilities. Mycoses. 2002;45:373–7.
Hamada N, Abe N. Physiological characteristics of 13 common fungal species in bathrooms. Mycoscience. 2009;50:421–9.
Döğen A, Kaplan E, Oksüz Z, et al. Dishwashers are a major source of human opportunistic yeast-like fungi in indoor environments in Mersin, Turkey. Med Mycol. 2013;51:493–8.
Zupančič J, Novak Babič M, Zalar P, Gunde-Cimerman N. The black yeast Exophiala dermatitidis and other selected opportunistic human fungal pathogens spread from dishwashers to kitchens. PLoS ONE. 2016;11(2):e0148166.
Gümral R, Tümgör A, Saraçlı MA, et al. Black yeast diversity on creosoted railway sleepers changes with ambient climatic conditions. Microb Ecol. 2014;68:699–707.
Novak Babič M, Zalar P, Ženko B, Džeroski S, Gunde-Cimerman N. Yeasts and yeast-like fungi in tap water and groundwater, and their transmission to household appliances. Fungal Ecol. 2016;20:30–9.
Anaissie EJ, McGinnis RM, Pfaller AM. Clinical mycology. 2nd ed. Philadelphia: Churchill Livingstone Elsevier; 2009.
Russo JP, Raffaeli R, Ingratta SM, Rafti P, Mestroni S. Cutaneous and subcutaneous phaeohyphomycosis. Skinmed. 2010;8:366–9.
Park KY, Kim HK, Suh MK, Seo SJ. Unusual presentation of onychomycosis caused by Exophiala (Wangiella) dermatitidis. Clin Exp Dermatol. 2011;36:418–9.
Richardson DM, Warnock WD. Fungal infection: diagnosis and management. UK: Wiley-Blackwell; 2012. p. 383–90.
Banaoudia F, Assouline M, Pouliquen Y, Bouvet A, Guého E. Exophiala (Wangiella) dermatitidis keratitis after keratoplasty. Med Mycol. 1999;37:53–6.
Pastel SR, Hammersmith KM, Rapuano CJ, Cohen EJ. Exophiala dermatitidis keratitis after laser in situ keratomileusis. J Cataract Refract Surg. 2006;32:681–4.
Kerkmann ML, Piontek K, Mitze H, Haase G. Isolation of Exophiala (Wangiella) dermatitidis in a case of otitis externa. Clin Infect Dis. 1999;36:241–7.
Vlassopoulos D, Kouppari G, Arvanitis D, et al. Wangiella dermatitidis peritonitis in a CAPD patient. Perit Dial Int. 2001;39:2261–6.
Greig J, Harkness M, Taylor P, et al. Peritonitis due to the dermatiaceous mold Exophiala dermatitidis complicating continuous ambulatory peritoneal dialysis. Clin Microbiol Infect. 2003;9:713–5.
Kusenbach G, Skopnik H, Haase G, Friedrichs F, Dohmen H. Exophiala dermatitidis pneumonia in cystic fibrosis. Eur J Pediatr. 1992;151:344–6.
Mukaino T, Koga T, Oshita Y, et al. Exophiala dermatitidis infection in non-cystic fibrosis bronchiectasis. Respir Med. 2006;100:2069–71.
Taj-Aldeen SJ, El Shafie S, Alsoub H, Eldeeb Y, de Hoog GS. Isolation of Exophiala dermatitidis from endotracheal aspirate of a cancer patient. Mycoses. 2006;49:504–9.
Ozawa Y, Suda T, Kaida Y, et al. A case of bronchial infection of Wangiella dermatitidis. Nihon Kokyuki Gakkai Zasshi. 2007;45:907–11.
Haase G, Skopnik H, Kusenbach G. Exophiala dermatitidis infection in cystic-fibrosis. Lancet. 1990;336:188–9.
Diemert D, Kunimoto D, Sand C, Rennie R. Sputum isolation of Wangiella dermatitidis in patients with cystic fibrosis. Scand J Infect Dis. 2001;33:777–9.
Horré R, Schaal KP, Siekmeier R, et al. Isolation of fungi, especially Exophiala dermatitidis, in patients suffering from cystic fibrosis. A prospective study. Respiration. 2004;71:360–6.
Kabel PJ, Illy KE, Holl RA, Buiting AGM, Wintermans RGF. Nosocomial intravascular infection with Exophiala dermatitidis. Lancet. 1994;344:1167–8.
Myoken Y, Sugata T, Fujita Y, et al. Successful treatment of invasive stomatitis due to Exophiala dermatitidis in a patient with acute myeloid leukemia. J Oral Pathol Med. 2003;32:51–4.
Kenney RT, Kwon-Chung KJ, Waytes AT, et al. Successful treatment of systemic Exophiala dermatitidis infection in a patient with chronic granulomatous disease. Clin Infect Dis. 1992;14:235–42.
Matsumoto T, Matsuda T, McGinnis MR, Ajello L. Clinical and mycological spectra of Wangiella dermatitidis infections. Mycoses. 1993;36:145–55.
Fukushiro R, Kagawa S, Nishiyama S, Takahashi H. Un cas de chromoblastomycose cutanée avec métastase cérébrale mortelle. Presse Méd. 1957;65:2142.
Mori A, Morikawi H, Akagi M. A case of chromoblastomycosis due to Hormiscium dermatitidis. Skin Res. 1961;3:158.
Naka W, Fukuda T, Nishikawa T, et al. Cerebral Exophiala dermatitidis infection in a patient with a history of cutaneous Exophiala jeanselmei infection (in Japanese). 14th Kanto Med Mycol Conf.; 1993. p. 14.
Ikeda K, Kunisaki T, Abe H, et al. Chromoblastomycosis occurring with cerebral symptoms without cutaneous lesions (in Japanese). Sogo Rinsho. 1973;22:1463–71.
Chang LC, Kim D-S, Park JD, et al. Acute cerebral phaeohyphomycosis due to Wangiella dermatitidis accompanied by cerebrospinal fluid eosinophilia. J Clin Microbiol. 2000;38:1965–6.
Uijthof JMJ, de Hoog GS, de Cock AWAM, Takeo K, Nishimura K. Pathogenicity of strains of the black yeast Exophiala (Wangiella) dermatitidis: an evaluation based on polymerase chain reaction. Mycoses. 1994;37:235–42.
Lanternier F, Barbati E, Meinzer U, et al. Inherited CARD9 deficiency in 2 unrelated patients with invasive Exophiala infection. J Infect Dis. 2014;211:1241–50.
CDC - Centers for Disease Control and Prevention. Exophiala infection from contaminated injectable steroids prepared by a compounding pharmacy. Morb Mortal Wkly Rep. 2002;51:1109–12.
de Hoog GS, Matos T, Sudhadham M, Luijsterburg FK, Haase G. Intestinal prevalence of the neurotropic black yeast Exophiala (Wangiella) dermatitidis in healthy and impaired individuals. Mycoses. 2005;48:142–5.
Pedrozo CPM, de Azevedo S, Gomes RR, et al. Fonsecaea pugnacius, a novel agent of disseminated chromoblastomycosis. J Clin Microbiol. 2015;53:2674–85.
Sudhadham M, van den Ende AHG, Sihanonth P, et al. Elucidation of distribution patterns and possible infection routes of the neurotropic black yeast Exophiala dermatitidis using AFLP. Fungal Biol. 2011;115:1051–65.
Vicente AV, Attili-Angelis D, Pie MR, et al. Environmental isolation of black yeast-like fungi involved in human infection. Stud Mycol. 2008;61:137–44.
Duarte APM, Attili-Angelis D, Baron NC, Forti LC, Pagnocca FC. Leaf-cutting ants: an unexpected microenvironment holding human opportunistic black fungi. Antonie Van Leeuwenhoek. 2014;106:465–73.
Singh P, Raghukumar C, Verma P, Shouche Y. Phylogenetic diversity of culturable fungi from the deep-sea sediments of the Central Indian Basin and their growth characteristics. Fungal Divers. 2010;40:89–102.
Gadanho M, Sampaio JP. Occurrence and diversity of yeasts in the mid-Atlantic ridge hydrothermal fields near the Azores Archipelago. Microb Ecol. 2005;50:408–17.
Efstratiou M, Velegraki A. Recovery of melanized yeasts from Eastern Mediterranean beach sand associated with the prevailing geochemical and marine flora patterns. Med Mycol. 2009;48:1–3.
Vishniac HS. Yeast biodiversity in the Antarctic. In: Gabor P, Carlos Augusto R, editors. The yeast handbook. Biodiversity and ecophysiology of yeasts. Berlin: Springer; 2006. p. 419–40.
Teixeira MM, Moreno LF, Stielow BJ, et al. Exploring the genomic diversity of black yeasts and relatives (Chaetothyriales, Ascomycota). Stud Mycol. 2017;86:1–28.
Zhao J, Zeng J, de Hoog GS, Attili-Angelis D, Prenafeta-Boldú FX. Isolation and identification of black yeasts by enrichment on atmospheres of monoaromatic hydrocarbons. Microb Ecol. 2010;60:149–56.
Prenafeta-Boldú FX, Guivernau M, Gallastegui G, et al. Fungal/bacterial interactions during the biodegradation of TEX hydrocarbons (toluene, ethylbenzene and p-xylene) in gas biofilters operated under xerophilic conditions. FEMS Microbiol Ecol. 2012;80:722–34.
Blasi B, Poyntner C, Rudavsky T, et al. Pathogenic yet environmentally friendly? Black fungal candidates for bioremediation of pollutants. Geomicrobiol J. 2016;33:308–17.
Döğen A, Kaplan E, Ilkit M, de Hoog GS. Massive contamination of Exophiala dermatitidis and E. phaeomuriformis in railway stations in subtropical Turkey. Mycopathologia. 2013;175:381–6.
Yazdanparast SA, Mohseni S, De Hoog GS, et al. Consistent high prevalence of Exophiala dermatitidis, a neurotropic opportunist, on railway sleepers. J Mycol Med. 2017. doi:10.1016/j.mycmed.2017.01.007.
UKNCC. UKNCC biological resource: properties, maintenance and management. 1st ed. London: The UK National Culture Collection; 2001.
Heinrichs G, Hübner I, Schmidt KC, de Hoog GS, Haase G. Analysis of black fungal biofilms occurring at domestic water taps (I): compositional analysis using Tag-encoded FLX amplicon pyrosequencing. Mycopathologia. 2013;175:387–97.
Nishimura K, Miyaji M. Studies on a saprophyte of Exophiala dermatitidis isolated from a humidifier. Mycopathologia. 1982;77:173–81.
Gümral R, Özhak-Baysan B, Tümgör A, et al. Dishwashers provide a selective extreme environment for human opportunistic yeast-like fungi. Fungal Divers. 2016;76:1–9.
Matos T, Haase G, Gerrits van den Ende AHG, de Hoog GS. Molecular diversity of oligotrophic and neurotropic members of the black yeast genus Exophiala, with accent on E. dermatitidis. Antonie Van Leeuwenhoek. 2003;83:293–303.
Sterflinger K. Black yeasts and meristematic fungi: ecology, diversity and identification. In: Rosa AC, Gábor P, editors. Biodiversity and ecophysiology of yeasts. Berlin: Springer; 2006. p. 501–14.
Yurlova NA, de Hoog GS. Exopolysaccharides and capsules in human pathogenic Exophiala species. Mycoses. 2002;45:443–8.
Dadachova E, Bryan RA, Huang X, et al. Ionizing radiation changes the electronic properties of melanin and enhances the growth of melanized fungi. PLoS ONE. 2007;2(5):e457.
Kazanas N. Pathogenicity of a fungus resembling Wangiella dermatitidis isolated from edible mushrooms. Appl Environ Microbiol. 1986;51:261–7.
Acknowledgements
Authors kindly acknowledge everybody who helped to collect the samples. We also thank the Ministry of Higher Education, Science and Technology of the Republic of Slovenia in corporation with Slovenia Research Agency for financially supporting Jerneja Zupančič as a Young Researcher (Grant No. 382228-1/2013), as well as the Slovenian Research Agency ARRS for providing financial support to the Infrastructural Centre Mycosmo, MRIC UL (Grant P2-0103).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All other authors declare no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Babič, M.N., Zupančič, J., Gunde-Cimerman, N. et al. Ecology of the Human Opportunistic Black Yeast Exophiala dermatitidis Indicates Preference for Human-Made Habitats. Mycopathologia 183, 201–212 (2018). https://doi.org/10.1007/s11046-017-0134-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11046-017-0134-8