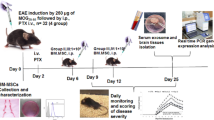
Abstract
Background
Multiple sclerosis (MS) is a complex autoimmune disease that affects the central nervous system, causing inflammation, demyelination, and neurodegeneration. Understanding the dysregulation of Tregs, dynamic cells involved in autoimmunity, is crucial in comprehending diseases like MS. However, the role of lymphocyte-activation gene 3 (Lag-3) in MS remains unclear.
Methods
In this study, we explore the potential of exosomes derived from human umbilical cord mesenchymal stem cells (hUMSCs-Exs) as an immune modulator in experimental autoimmune encephalomyelitis (EAE), a model for MS.
Results
Using flow cytometry, our research findings indicate that groups receiving treatment with hUMSC-Exs revealed a significant increase in Lag-3 expression on Foxp3 + CD4 + T cells. Furthermore, cell proliferation conducted on spleen tissue samples from EAE mice using the CFSE method exposed to hUMSC-Exs yielded relevant results.
Conclusions
These results suggest that hUMSCs-Exs could be a promising anti-inflammatory agent to regulate T-cell responses in EAE and other autoimmune diseases. However, further research is necessary to fully understand the underlying mechanisms and Lag-3’s precise role in these conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Multiple sclerosis (MS) is a chronic inflammatory disorder of the central nervous system (CNS) and spinal cord. It is characterized by several typical clinical findings, including muscle weakness, visual impairment, and an unstable gait [1] The precise triggers of the disease are not fully understood, but immunological, genetic, and environmental factors are potential contributors [2]. Abnormal activation of T cells against self-antigens, such as myelin basic protein, is believed to play a role in disease progression [3]. Current therapeutic interventions for MS primarily focus on anti-inflammatory drugs, but their side effects and limited efficacy in some patients highlight the need for new treatment approaches [4].
Mesenchymal stem cells (MSCs) have emerged as promising candidates for the treatment of autoimmune diseases, including MS, due to their unique anti-inflammatory properties [5, 6]. MSCs can decrease the inflammatory response through cell-to-cell contact or secretion of anti-inflammatory mediators. Additionally, MSCs can produce macrovesicles (MVs) and exosomes (Exs), which have various sizes ranging from 30 to 150 nm and contain proteins, lipids, and miRNAs with diverse biological effects [7, 8]. MSCs-Exs possess immunomodulatory properties similar to their parental cells and can downregulate both innate and adaptive immunity through regulatory miRNAs and epigenetic regulation [9, 10]. Regulatory T cells (Tregs) play a critical role in maintaining self-tolerance, and their dysfunction has been observed in MS patients [11]. Tregs exert their immunosuppressive function through cell-to-cell contact and the production of anti-inflammatory cytokines [12, 13]. However, high expression of co-inhibitory molecules, such as CTLA-4, PD-1, Lag-3, and Tim-3, by Tregs can contribute to their immunosuppressive function [14]. Lag-3, in particular, has been implicated in directing autoreactive Tregs in the context of autoimmunity. Studies have shown that the lack of Lag-3 on Tregs is involved in several autoimmune diseases, and antigen-specific Tregs with high levels of Lag-3 can prevent auto-reactive T cells in experimental autoimmune encephalomyelitis (EAE) mice [15, 16]. The impact of Lag-3 on Tregs and autoimmunity is indicated by studies showing that the lack of this co-inhibitory receptor on Tregs is involved in several autoimmune diseases [17]. It has been suggested that Lag-3 can change the functionality of Tregs in several ways, such as interaction with transcription factors including Foxp3, Emes, and Egr, and could also limit the proliferation and maintenance of Treg cells in autoimmune conditions [18].
Given the prominent role of co-inhibitory receptors like PD-1 and Lag-3 in maintaining self-tolerance, this study aimed to investigate whether human umbilical cord-derived MSCs-Exs (hUMSCs-Exs) can modulate the expression of Lag-3 on the surface of Tregs cells in EAE mice. This research seeks to further understand the potential immunomodulatory effects of hUMSCs-Exs and their impact on Lag-3-mediated regulation of Tregs in the context of EAE mice.
Materials and methods
Culture, and characterization of hUMSCs
Human umbilical-derived mesenchymal stem cells (hUMSCs) were obtained from the Bonyakhteh Stem Cell Technology Center in Iran. Their adipogenic and osteogenic differentiation capacity and phenotypic characterization were assessed as previously described [19]. To identify and characterize the purchased cells, the expression patterns of CD73, CD90, CD105, CD34, and HLA (all from eBioscience) were examined, and the differentiation of these cells into bone and adipocytes was also determined. Cultured MSCs at passages 2–5 were detached by trypsinization (Sigma), washed twice with FACS buffer (PBS containing 1% FCS), and stained for 30 min at 4 °C in PBS containing 1% FCS. After washing with FACS buffer, the cells were fixed with 4% paraformaldehyde and subjected to flow cytometry examination using FACS Calibur (USA).
Exosome isolation
To obtain exosomes from hUMSCs, the complete DMEM medium supplemented with 10% fetal bovine serum (FBS) was replaced by a serum-free medium when the seeded cells reached 80–90% confluency. After 48 h, the supernatant was collected and centrifuged at 300 g for 10 min to eliminate cell debris and apoptotic bodies. The resulting supernatant was then filtered with a 0.22 μm syringe filter and resuspended in kit reagent according to the manufacturer’s protocol (Invitrogen). The pelleted exosomes were harvested and stored in PBS at -80 °C.
For physical analysis, hUMSC-Exs were analyzed using electron microscopy as previously described [20]. To determine the size of the peptide-loaded exosomes, they were subjected to dynamic light scattering (DLS).
EAE induction
For EAE induction, 6 to 8-week-old C57BL/6 mice (purchased from the Pasteur Institute, Iran) were immunized subcutaneously with 300 µg of MOG (35–55) (Sigma Aldrich), supplemented with incomplete Freund’s adjuvant (CFA, Sigma Aldrich/1 mg/mL). Additionally, i.p. injections of 300 ng of pertussis toxin (Santa Cruz Biotechnology, Santa Cruz, CA, USA) were administered on days 0 and 2 post-immunization, as previously described [21]. Clinical scoring and weight assessment were conducted daily using a 0–5 scoring system as follows: 0 - no disease; 1 - decreased tail tone; 2 - hind limb weakness or partial paralysis; 3 - complete hind limb paralysis; 4 - front and hind limb paralysis; and 5 - moribund.
Treatment protocol
For the in vivo mouse experiments, all mice at day 13 post-immunization (d.p.i) were selected based on the EAE scales and randomly assigned to three groups: a normal (CTRL) group, an untreated (PBS treated) group, and a treated group receiving 30–50 µg of hUMSCs-EXs. The hUMSCs-EXs were injected intravenously (IV) through the tail in each mouse. Treatment injections were performed at 16, 19, and 22 days after d.p.i. All mice were euthanized on day 28 d.p.i.
Western blot
To date, several well-known markers, including CD63, CD81, CD9, TSG-101, and ALIX, have been assigned for the characterization of MSCs-EVs [22]. For all western blots, we used 10 µg of total exosome lysate, which was loaded onto 10% SDS denaturing polyacrylamide gels. hUMSCs-Exs are characterized by the expression of CD63, CD9, and CD81 proteins.
To perform immunoblotting, the protein concentration was determined using the Bradford Protein Assay. Samples were prepared and boiled for 5 min before loading. Proteins were separated on 8% or 15% Tris bis-acrylamide gels prepared on the same day at 120 V for 45 min. Then, the samples were transferred onto a PVDF membrane at 120 mA for 1.5 h. After transfer, the membranes were blocked in 5% w/v bovine serum albumin in Tris-buffered saline with 0.5% Tween-20 (TBS-T). The membranes were probed with primary antibodies diluted in TBS-T in 5% w/v skim milk overnight at 4 °C on a rotator.
In this study, we used the following primary antibodies: anti-CD9 (sc-13,118, Santa Cruz Biotechnology, INC), anti-CD63 (sc-5275, Santa Cruz Biotechnology, INC), anti-CD81 (sc-166,029, Santa Cruz Biotechnology, INC), and mouse anti-rabbit IgG-HRP (sc-2357, Santa Cruz Biotechnology, INC) and m-IgGκBP-HRP (sc-516,102, Santa Cruz Biotechnology, INC) both were used as secondary antibodies to determine the monoclonal antibodies. After overnight incubation, the membranes were washed 3 times for 10 min in TBS-T. HRP-conjugated secondary antibodies were applied at room temperature in 5% w/v bovine serum albumin for 1 h with rotation. After washing 3 times for 10 min in TBS-T, the membranes were visualized.
Flow cytometry analysis
For the characterization of human umbilical cord mesenchymal stem cells (hUMSCs), cells were stained with positive markers, including anti-CD73/PE, anti-CD90/FITC, and anti-CD105/PE-Cy7, as well as negative markers, including anti-CD45/FITC and anti-CD34/PE. Briefly, samples were incubated with monoclonal antibodies (mAb) for 30 min at 4 °C.
To analyze immune cells, spleen cells were harvested for flow cytometry analysis, and isolated lymphocytes were washed twice with PBS. The cells were then suspended in FACS buffer (PBS containing 0.2% FBS), washed two more times with FACS buffer, and fixated and permeabilized using fixation/permeabilization buffer (BD Pharmingen) for 30 min at 4 °C.
Next, the cells were stained with anti-CD4/FITC, Lag-3/BV421 for 30 min at room temperature, and in the case of Foxp3, anti-Foxp3/Alexa 647 (all from Biolegend) was used at 37 °C for 30 min in the dark. The cells were washed with FACS buffer and analyzed by flow cytometry (Becton Dickinson, Mountain View, CA). The data were analyzed using Flowjo software (Tree Star Inc., San Carlos, CA).
Cell proliferation assay
Flow cytometry was used to measure cell proliferation and determine the proliferation index between the different groups.
To examine cell proliferation, isolated cells were cultured in complete RPMI 1640 medium with 10% FCS (Gibco), with or without PMA/ionomycin as a mitogen, and 30 ng MOG 35–55 as a specific stimulator. Briefly, 1 × 10^6 spleen-derived immune cells were suspended with 0.5 mM CFSE (Biolegend) and incubated in the dark for 15 min at 37 °C. Then, CFSE was quenched with an equal volume of cold RPMI1640 medium containing 20% FBS. The cells were washed, re-suspended in complete RPMI1640 media, and incubated with mitogens and 30 ng of MOG35-55 for 3 days at 37 °C with 5% CO2.
Real-time PCR assay
Total RNA was extracted from the spleen isolated cells using an RNA extraction kit (favorgene, Taiwan). The quality of the RNA extracts was assessed using a Nanodrop 2000 spectrophotometer (Thermo Fisher Scientific). According to the manufacturer’s instructions, 5 µg of RNA was used for cDNA synthesis using a cDNA synthesis kit (GeneAll Biotechnology kit, Korea). SYBR Green Real-Time PCR method with the Amplicon master mix was conducted using the MIC mini thermal cycler from Bio Molecular Systems. For mRNA expression analysis, we utilized the and the primers, Metabion in Germany, listed in Table 1. To determine the relative expression of genes, we employed the 2 − ΔΔCt formula, comparing the results to the control groups.
Histopathological analysis
Brain tissue samples from both EAE and normal mice were collected in 15 ml centrifugal tubes and fixed with 4% para formaldehyde. The samples were then embedded in paraffin and sections were prepared at a thickness of 6 μm. Following the standard procedure [23], the sections were stained with hematoxylin and eosin (H&E) to examine the hippocampal area of the brain tissues for cell infiltration. All animal procedures were conducted following Urmia University of Medical Sciences guidelines on the use and care of experimental animals, and using 100 mg/Kg ketamine and 10 mg/kg Xylazine per body weight followed by cervical dislocation to confirm euthanasia. In this study we used 4 and 10 magnifications to examine the sections.
Statistical analyses
For statistical analyses, the results of the experiments were analyzed using GraphPad Prism 6.0 (GraphPad Software, Inc., La Jolla, CA, USA). One-way analysis of variance (ANOVA) and Tukey’s Multiple Comparison Test were used to determine differences between the groups. All data are presented as the mean ± SD, and a p-value of less than 0.05 was considered statistically significant (*, P < 0.05; **, P < 0.005; ***, P < 0.001).
Results
Characterization of the hUMSCs-Exs
To define the characteristic profile of human umbilical cord mesenchymal stem cells (hUMSCs) and their exosomes (hUMSCs-Exs), we assessed the levels of several positive CD markers, including CD73, CD105, and CD90, as well as negative markers for hUMSCs, including CD34 and CD45 (Fig. 1A), using flow cytometry. We also confirmed the differentiation capacity of isolated hUMSCs (Fig. 1B). After 48 h of culture in serum-free medium, exosomes were isolated using an exosome isolation kit from Invitrogen and further characterized. Transmission electron microscopy (TEM) and field emission scanning electron microscopy (FE-SEM) were used to confirm the size and morphology of the isolated exosomes (Fig. 1C&D). Western blot analysis showed that the isolated exosomes expressed CD9, CD63, and CD81 markers (Fig. 1E). Dynamic light scattering (DLS) was used to determine the size distribution of the exosomes (Fig. 1F).
Characterization of hUMSCs and hUMSCs-EXs. Flow cytometry was used to verify the expression of positive and negative surface markers of hUMSCs, including CD73, CD90, CD44, and CD105, and CD34 and CD45, respectively (A). Cultured human UMSCs were stained with Oil Red for adipogenic differentiation (B1) and Alizarin Red for osteogenic differentiation (B2). Transmission electron microscopy (TEM) analysis (C) and field emission scanning electron microscopy (FESEM) were used to determine the size distribution of purified exosomes from hUMSCs (D). To further characterize UMSCs-EXs, western blotting was performed to determine the expression of CD9, CD63, and CD81 (E), and the density of exosomes was assessed by dynamic light scattering (DLS) (F). The exosome-treated group received 50–100 µg of hUMSCs-Exs every 3 days for 3 times (iv)
HUMSC-Exs administration increased Lag-3 expression on Foxp3+/CD4 + T cells
Given the significant role of Lag-3 in the suppressive function of Tregs [16], we investigated the expression level of Lag-3 on Foxp3 + CD4 + T cells in hUMSCs-Exs treated and untreated EAE mice. The expression of Lag-3 in the hUMSCs-Ex treated group significantly increased compared with the untreated EAE and CTRL groups. (Fig. 2A&B).
Immuno-phenotyping of Treg marker expression on spleen-derived immune cells. The frequency of Lag-3-positive Tregs was analyzed by flow cytometry. Surface staining with antibodies targeting CD4, Foxp3, and CD223 (LAG-3) on Tregs at day 28 dpi was examined using flow cytometry. Lymphocytes were gated for CD4 and Foxp3 expression (A). The percentage of Lag-3-positive cells among the CD4 + and Foxp3-positive cells is depicted (B). P-values were calculated using one-way ANOVA. Error bars represent mean ± SEM. *, P < 0.05
HUMSC-derived exs downregulate the proliferation of immune cells
To measure the proliferation of T cells, we used CFSE to assess the impact of hUMSCs-Exo on specific T cell proliferation. CFSE-labeled spleen-derived immune cells were significantly decreased during the 3-day co-culture with MOG (35–55) and hUMSCs-Exs (Fig. 3).
HUMSCs-Exs modulate the expression of inflammatory genes
RT-PCR analysis of spleen isolated lymphocytes demonstrated that the expression of pro-inflammatory cytokines including IFN-γ, IL-17, and NFkB(P65), was significantly decreased following hUMSCs-Exs administration. As shown in Fig. 4A, the clinical score of the hUMSCs-Exs significantly lower than in the untreated group, and we observed that the hUMSC-Exs treated group significantly enhanced the expression of several inhibitory receptors and transcription factors on spleen-harvested immune cells, e.g., CTLA-4, Lag-3, and STAT3, (Fig. 4B).
Clinical score and real-time PCR. The clinical score of all EAE mice was monitored daily, and the results were analyzed and compared between groups (A and B). Real-time PCR was used to verify the expression of immune-related genes in the spleen-derived immune cells of the EAE and treated groups (n = 3). The relative expression of each gene in the hUMSCs-Exs treated group compared with the CTRL (Blue) and untreated group (EAE, orange) is presented. Error bars represent mean ± SEM. P-values were calculated using the student’s T-test. *P < 0.05, **P < 0.01, ***P < 0.001
Histopathological analysis and clinical outcomes
Using the H&E staining method [23], we next examined the hippocampal area of brain tissues of all mice in all groups to evaluate cell infiltration. Notably, histopathologic examination of the hippocampal area of hUMSCs-EXs treated EAE mice showed markedly reduced infiltrated cells compared with the untreated EAE mice (Fig. 5).
Tissue damage and immune cell infiltration in the brains of mice under a light microscope with H&E staining. Images show brain sections of CTRL (A), EAE (B), and UMSCs-Exs treated group (C). Brain tissue was examined using 4x (left) and 10x objective (right) images. We used three sections per group for Brain tissue analyzes. Arrows highlight the immune cell infiltration in the brain tissue. Results are presented as mean ± SEM and analyzed by ANOVA. *, P < 0.05; **, P < 0.01. (D)
Discussion
In this study, we investigated the therapeutic effects of hUMSCs-Exs and evaluated the expression of Lag-3 on Foxp3 + CD4 + T cells, which could potentially counteract the activated auto-reactive T cells in EAE mice [11, 24]. Our results indicated that the use of hUMSCs-Exs could be a powerful anti-inflammatory agent that can modify the T cell response in established EAE.
We observed that mice treated with hUMSCs-Exs showed a higher percentage of Lag-3 + Foxp3 + CD4 T cells compared to the untreated groups. Consistent with previous studies, we also found elevated expression of Lag-3 on CD4 + T cells in EAE mice [16].
Among the various co-inhibitory receptors on T cells, Lag-3 is the one with the most controversial role. It is a complex molecule that plays a crucial role in Treg function in autoimmune disorders [17]. Despite the important role of Tregs in maintaining self-tolerance [25], we have limited knowledge about the complex role of co-inhibitory receptors, particularly Lag-3, on Treg cells. Additionally, patients with MS exhibit decreased and altered Treg function, although the underlying mechanisms are still unknown [26]. Recent studies have suggested that Lag-3 may be associated with increased inflammation in autoimmune disorders, including SLE [27,28,29,30]. And decreased Lag-3 expression on Tregs could reduce pathogenic Tregs in autoimmune conditions like non-obese diabetic (NOD) mice [18]. However, other studies have demonstrated that Lag-3 deficiency in Treg cells increases the susceptibility of mice to fatal EAE [31]. Various studies indicate a strong propensity of Lag-3 to stimulate Treg formation and has been found to play a crucial role in controlling EAE, with IL-27 and IL-10 signaling [15, 32]. However, the relationship between lag-3 expression and disease severity in MS patients remains unclear [33].
By understanding the mechanisms that enhance Treg function, including the role of lag-3, new therapeutic approaches for autoimmune diseases could be developed [34, 35]. It has been observed that the inhibitory function of Lag-3 is tightly regulated by factors such as its expression levels on the cell surface and various post-translational modifications. These findings suggest that lag3 may act as a rheostat rather than a breaker of T cell activation [36]. Lag-3, unlike other co-inhibitory receptors, has unique characteristics due to its interaction with diverse ligands. Notably, it has been reported to have multiple ligands such as FGL1, Galectin-3, LSECtin, and α-synuclein in addition to MHCII. With various motifs in its cytoplasmic domain, Lag-3 plays a crucial role in controlling immune cells. Interestingly, Lag-3’s inhibitory signaling does not rely on the traditional ITIM motif but instead contains distinctive motifs like FSAL, KIEELE, and EX-repeat [33]. Particularly intriguing is the potential role of Lag-3 in Tregs during autoimmunity and its impact on diseases like multiple sclerosis [37]. To fully understand the mechanisms at play with Lag-3, further research is needed to explore factors such as ligands, tissue microenvironment, and timing of expression.
Our results indicated that the hUMSC-Exs treated group showed stronger inhibition of lymphocyte proliferation than the untreated group through an in vitro CFSE cell proliferation assay.
Additionally, we further examined the expression pattern of inhibitory receptors including Lag-3, CTLA-4, the inflammatory cytokine IFN-γ, and the transcription factor STAT3 within the groups. This was done to delineate a more pronounced inhibition of lymphocyte regulation in the group treated with hUMSC-Exs, compared to the untreated groups - an observation made through an RT-PCR assay, which indicate that hUMSCs-Exs could play a crucial role in directing CD4 + T cell behavior Moreover, histopathological examinations of the brain tissue of hUMSCs-Exs treated EAE mice provided relevant findings [38, 39].
The properties of hUMSCs-Exs such as small size, stability, non-self-renewal property, less or non-tumorigenic nature, and long-term storage capability compared to MSCs, make them an attractive and highly promising alternative for cell-free therapy and drug delivery approaches [40,41,42]. A detailed description of the specific properties of MSCs-Exs in modulating the immune system is essential for future clinical applications. Several investigations have indicated that MSCs-Exs have a potent ability to promote Treg formation [39, 43].
Conclusions
This study provides evidence that hUMSCs-Exs can modulate the immune response in EAE mice by promoting Lag-3 expression on CD4+/Foxp3 Tregs and reducing immune cell infiltrations in the hypothalamus. The downregulation of pro-inflammatory cytokines and transcription factors further supports the therapeutic potential of hUMSC-Exs in EAE mice. Although this study has not addressed the mechanisms underlying such effects, future studies are needed to reveal the exact role of MSCs-Exs in regulating immune cells. The difficulty in producing large amounts of MSCs-Exs and the lack of a universal consensus procedure for reproducibility of MSCs-Exs have compromised the clinical use of MSCs-Exs [44]. In addition to the lack of standardized isolation and characterization methods, there are other therapeutic concerns, including mechanisms of action and safety, that should be considered to encourage clinical requests.
Data availability
No datasets were generated or analysed during the current study.
References
Kuhlmann T, Antel J (2023) Multiple sclerosis: 2023 update. Free Neuropathol 4. https://doi.org/10.17879/freeneuropathology-2023-4675
Alfredsson L, Olsson T (2019) Lifestyle and environmental factors in multiple sclerosis. Cold Spring Harb Perspect Med 9(4). https://doi.org/10.1101/cshperspect.a028944
Huang W-J, Chen W-W, Zhang X (2017) Multiple sclerosis: Pathology, diagnosis and treatments. Experimental and therapeutic medicine 13. 63163–3166. https://doi.org/10.3892/etm.2017.4410
Tavazzi E, Rovaris M, La Mantia L (2014) Drug therapy for multiple sclerosis. CMAJ 186(11):833–840. https://doi.org/10.1503/cmaj.130727
Gao F, Chiu SM, Motan DAL, Zhang Z, Chen L, Ji HL, Tse HF, Fu QL, Lian Q (2016) Mesenchymal stem cells and immunomodulation: current status and future prospects. Cell Death & Disease 7(1): e2062-e2062: https://doi.org/10.1038/cddis.2015.327
Wu X, Jiang J, Gu Z, Zhang J, Chen Y, Liu X (2020) Mesenchymal stromal cell therapies: immunomodulatory properties and clinical progress. Stem Cell Res Ther 11(1):345. https://doi.org/10.1186/s13287-020-01855-9
Seo Y, Kim H-S, Hong I-S (2019) Stem cell-derived extracellular vesicles as Immunomodulatory therapeutics. Stem Cells Int 2019. https://doi.org/10.1155/2019/5126156
Qian X, An N, Ren Y, Yang C, Zhang X, Li L (2021) Immunosuppressive effects of mesenchymal stem cells-derived exosomes. Stem Cell Reviews Rep 17(2):411–427. https://doi.org/10.1007/s12015-020-10040-7
Zhou X, Xie F, Wang L, Zhang L, Zhang S, Fang M, Zhou F (2020) The function and clinical application of extracellular vesicles in innate immune regulation. Cell Mol Immunol 17(4):323–334. https://doi.org/10.1038/s41423-020-0391-1
Wang JH, Liu XL, Sun JM, Yang JH, Xu DH, Yan SS (2020) Role of mesenchymal stem cell derived extracellular vesicles in autoimmunity: a systematic review. World J Stem Cells 12(8):879–896. https://doi.org/10.4252/wjsc.v12.i8.879
Kimura K (2020) Regulatory T cells in multiple sclerosis. Clin Experimental Neuroimmunol 11(3):148. https://doi.org/10.1111/cen3.12591. 155: https://DOI:
Yang S, Fujikado N, Kolodin D, Benoist C, Mathis D (2015) Immune tolerance. Regulatory T cells generated early in life play a distinct role in maintaining self-tolerance. Science 348(6234) 589 – 94: https://DOI. https://doi.org/10.1126/science.aaa7017
McIntosh CM, Alegre M-L (2019) Teamwork by IL-10 + and IL-35 + Tregs. American. J Transplantation 19(8):2147. https://doi.org/10.1111/ajt.15511. 2147: https://DOI
Kumar P, Bhattacharya P, Prabhakar BS (2018) A comprehensive review on the role of co-signaling receptors and Treg homeostasis in autoimmunity and tumor immunity. J Autoimmun 95:77–99. https://doi.org/10.1016/j.jaut.2018.08.007
Pohar J, O’Connor R, Manfroi B, El-Behi M, Jouneau L, Boudinot P, Bunse M, Uckert W, Luka M, Ménager M, Liblau R, Anderton SM, Fillatreau S (2022) Antigen receptor-engineered Tregs inhibit CNS autoimmunity in cell therapy using nonredundant immune mechanisms in mice. European Journal of Immunology 52(8): 1335–1349: https://https://doi.org/10.1002/eji.202249845
Thaker YR, Andrews LP, Workman CJ, Vignali DAA, Sharpe AH (2018) Treg-specific LAG3 deletion reveals a key role for LAG3 in regulatory T cells to inhibit CNS autoimmunity. J Immunol 200(1Supplement):101. https://doi.org/10.4049/jimmunol.200.Supp.101.7
Gertel S, Polachek A, Elkayam O, Furer V (2022) Lymphocyte activation gene-3 (LAG-3) regulatory T cells: an evolving biomarker for treatment response in autoimmune diseases. Autoimmun rev 21(6):103085. https://doi.org/10.1016/j.autrev.2022.103085. https://DOI
Zhang Q, Chikina M, Szymczak-Workman AL, Horne W, Kolls JK, Vignali KM, Normolle D, Bettini M, Workman CJ, Vignali DAA (2017) LAG3 limits regulatory T cell proliferation and function in autoimmune diabetes. Sci Immunol 2(9). https://doi.org/10.1126/sciimmunol.aah4569
Secunda R, Vennila R, Mohanashankar AM, Rajasundari M, Jeswanth S, Surendran R (2015) Isolation, expansion and characterisation of mesenchymal stem cells from human bone marrow, adipose tissue, umbilical cord blood and matrix: a comparative study. Cytotechnology 67(5):793–807. https://doi.org/10.1007/s10616-014-9718-z
Jung MK, Mun JY (2018) Sample Preparation and Imaging of exosomes by Transmission Electron Microscopy. J Vis Exp 131https://doi.org/10.3791/56482
Huntemann N, Vogelsang A, Groeneweg L, Willison A, Herrmann AM, Meuth SG, Eichler S (2022) An optimized and validated protocol for inducing chronic experimental autoimmune encephalomyelitis in C57BL/6J mice. J Neurosci Methods 367. https://doi.org/10.1016/j.jneumeth.2021.109443. 109443: https://DOI
Yin K, Wang S, Zhao RC (2019) Exosomes from mesenchymal stem/stromal cells: a new therapeutic paradigm. Biomark Res 7(1):8. https://doi.org/10.1186/s40364-019-0159-x
Li Y, Li N, Yu X, Huang K, Zheng T, Cheng X, Zeng S, Liu X (2018) Hematoxylin and eosin staining of intact tissues via delipidation and ultrasound. Sci Rep 8(1):12259. https://doi.org/10.1038/s41598-018-30755-5
Sharma VK, Bayry J (2022) Restoration of established systemic inflammation and autoimmunity by Foxp3 + regulatory T cells. Cellular &. Mol Immunol 19(2):133–135. https://doi.org/10.1038/s41423-021-00831-4
Zhang Q, Chikina M, Szymczak-Workman AL, Horne W, Kolls JK, Vignali KM, Normolle D, Bettini M, Workman CJ, Vignali DAA (2017) LAG3 limits regulatory T cell proliferation and function in autoimmune diabetes. Sci Immunol 2(9):eaah4569. https://doi.org/10.1126/sciimmunol.aah4569
Verma ND, Lam AD, Chiu C, Tran GT, Hall BM, Hodgkinson SJ (2021) Multiple sclerosis patients have reduced resting and increased activated CD4 + CD25 + FOXP3 + T regulatory cells. Sci Rep 11(1):10476. https://doi.org/10.1038/s41598-021-88448-5
Yu S, Fujio K, Ishigaki K, Shoda H, Okamura T, Noor T, Sumitomo S, Yamamoto K (2012) Increased concentration of serum soluble LAG3 in systemic lupus erythematosus. Arthritis Res Ther 14(Suppl 1):P16. https://doi.org/10.1186/ar3617Epub 2012 Feb 9
Kato R, Sumitomo S, Tsuchida Y, Tsuchiya H, Nakachi S, Sakurai K, Hanata N, Nagafuchi Y, Kubo K, Tateishi S, Kanda H, Okamura T, Yamamoto K, Fujio K (2019) CD4 + CD25 + LAG3 + T cells with a feature of Th17 cells Associated with systemic Lupus Erythematosus Disease Activity. Front Immunol 10. https://doi.org/10.3389/fimmu.2019.01619
Lundmark F, Harbo HF, Celius EG, Saarela J, Datta P, Oturai A, Lindgren CM, Masterman T, Salter H, Hillert J (2006) Association analysis of the LAG3 and CD4 genes in multiple sclerosis in two independent populations. J Neuroimmunol 180(1):193–198. https://doi.org/10.1016/j.jneuroim.2006.08.009. https://DOI
García-Martín E, Agúndez JAG, Gómez-Tabales J, Benito-León J, Millán-Pascual J, Díaz-Sánchez M, Calleja P, Turpín-Fenoll L, Alonso-Navarro H, García-Albea E, Plaza-Nieto JF, Jiménez-Jiménez FJ (2022) Association between LAG3/CD4 genes variants and risk for multiple sclerosis. Int J Mol Sci 23(23):15244
Kadowaki A, Miyake S, Saga R, Chiba A, Mochizuki H, Yamamura T (2016) Gut environment-induced intraepithelial autoreactive CD4(+) T cells suppress central nervous system autoimmunity via LAG-3. Nat Commun 7(11639). https://doi.org/10.1038/ncomms11639
Do J, Kim D, Kim S, Valentin-Torres A, Dvorina N, Jang E, Nagarajavel V, DeSilva TM, Li X, Ting AH, Vignali DAA, Stohlman SA, Baldwin WM, Min B (2017) Treg-specific IL-27Rα deletion uncovers a key role for IL-27 in Treg function to control autoimmunity. Proceedings of the National Academy of Sciences 114(38): 10190–10195: https://DOI: https://doi.org/10.1073/pnas.1703100114
Maruhashi T, Sugiura D, Okazaki IM, Okazaki T (2020) LAG-3: from molecular functions to clinical applications. J Immunother Cancer 8(2). https://doi.org/10.1136/jitc-2020-001014
Jones A, Opejin A, Henderson JG, Gross C, Jain R, Epstein JA, Flavell RA, Hawiger D (2015) Peripherally Induced Tolerance Depends on Peripheral Regulatory T Cells that require Hopx to inhibit intrinsic IL-2 expression. J Immunol 195(4):1489. https://doi.org/10.4049/jimmunol.1500174
Kitz A, Dominguez-Villar M (2017) Molecular mechanisms underlying Th1-like Treg generation and function. Cellular. Mol Life Sci 74(22):4059–4075. https://doi.org/10.1007/s00018-017-2569-y
Maeda TK, Sugiura D, Okazaki IM, Maruhashi T, Okazaki T (2019) Atypical motifs in the cytoplasmic region of the inhibitory immune co-receptor LAG-3 inhibit T cell activation. J Biol Chem 294(15):6017–6026. https://doi.org/10.1074/jbc.RA119.007455
Kleinewietfeld M, Hafler DA (2013) The plasticity of human Treg and Th17 cells and its role in autoimmunity. Semin Immunol 25(4) 305 – 12: https://DOI:. https://doi.org/10.1016/j.smim.2013.10.009
Dabrowska S, Andrzejewska A, Janowski M, Lukomska B (2021) Immunomodulatory and Regenerative effects of mesenchymal stem cells and extracellular vesicles: Therapeutic Outlook for Inflammatory and degenerative diseases. Front Immunol 11. https://doi.org/10.3389/fimmu.2020.591065
Negi N, Griffin MD (2020) Effects of mesenchymal stromal cells on regulatory T cells: current understanding and clinical relevance. Stem Cells 38(5):596–605. https://doi.org/10.1002/stem.3151. https://DOI
Gomzikova MO, James V, Rizvanov AA (2019) Therapeutic application of mesenchymal stem cells derived Extracellular vesicles for Immunomodulation. Front Immunol 10(2663). https://doi.org/10.3389/fimmu.2019.02663
Willis GR, Mitsialis SA, Kourembanas S (2018) Good things come in small packages: application of exosome-based therapeutics in neonatal lung injury. Pediatr Res 83(1–2):298–307. https://doi.org/10.1038/pr.2017.256
Di Trapani M, Bassi G, Midolo M, Gatti A, Takam Kamga P, Cassaro A, Carusone R, Adamo A, Krampera M (2016) Differential and transferable modulatory effects of mesenchymal stromal cell-derived extracellular vesicles on T, B and NK cell functions. Sci Rep 6(1):24120. https://doi.org/10.1038/srep24120
Zhang B, Yeo RWY, Lai RC, Sim EWK, Chin KC, Lim SK (2018) Mesenchymal stromal cell exosome–enhanced regulatory T-cell production through an antigen-presenting cell–mediated pathway. Cytotherapy 20(5):687–696. https://doi.org/10.1016/j.jcyt.2018.02.372. https://DOI
Varderidou-Minasian S, Lorenowicz MJ (2020) Mesenchymal stromal/stem cell-derived extracellular vesicles in tissue repair: challenges and opportunities. Theranostics 10(13):5979–5997. https://doi.org/10.7150/thno.40122
Acknowledgements
We would like to express our gratitude to Dr. Behzad Baradaran and Elham Baghbani, Tabriz University of Medical Sciences, for their assistance in performing flow cytometry.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Mohammadzadeh Adel, designed and performed many of the experiments and conceptual interpretations. Masoud Lahouti, Hamed, Arash and Sahand were involved in performing the practical examination.Jafar Rezaei helped in exosome isolation and interpretation. Shahria Alipour and Vahid designed and Helped in RT-PCR. Dr Hassan Ansari revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper.
Ethical approval
This study was granted by the Ethics Committee of the Urmia University of Medical Science Animal Ethics Committee (IR.UMSU.REC.1399.069).
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohammadzadeh, A., Lahouty, M., charkhian, H. et al. Human umbilical cord mesenchymal stem cell-derived exosomes alleviate the severity of experimental autoimmune encephalomyelitis and enhance lag-3 expression on foxp3 + CD4 + T cells. Mol Biol Rep 51, 522 (2024). https://doi.org/10.1007/s11033-024-09433-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11033-024-09433-5