Abstract
Background
One of the main health issues that can affect women's health is reproductive diseases, such as polycystic ovary syndrome (PCOS), endometriosis (EMs), uterine leiomyomas (ULs), and ovarian cancer (OC). Although these diseases are very common, we do not have a complete understanding of their underlying cellular and molecular mechanisms. It is important to mention that the majority of patients are diagnosed with these diseases at later stages because of the absence of early diagnostic techniques and dependable molecular indicators. Hence, it is crucial to discover novel and non-invasive biomarkers that have prognostic, diagnostic and therapeutic capabilities. MiRNAs, also known as microRNAs, are small non-coding RNAs that play a crucial role in regulating gene expression at the post-transcriptional level. They are short in length, typically consisting of around 22 nucleotides, and are highly conserved across species. Numerous studies have shown that miRNAs are expressed differently in various diseases and can act as either oncogenes or tumor suppressors.
Methods
The author conducted a comprehensive review of all the pertinent papers available in web of science, PubMed, Google Scholar, and Scopus databases.
Results
We achieved three goals: providing readers with better information, enhancing search results, and making peer review easier.
Conclusions
This review focuses on the investigation of miRNAs and their involvement in various reproductive disorders in women, including their molecular targets. Additionally, it explores the role of miRNAs in the development and progression of these disorders.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
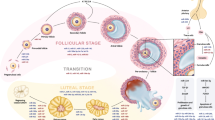
MicroRNAs (miRNAs) are single-stranded non-coding RNAs with an average of 22 nucleotides [1]. These molecules encode about 1% of the human genome sequences [2] and miRNAs regulate the expression of about 30% of mRNA [3]. MiRNAs play a key role in regulating biological processes through the regulation of gene expression at the post-transcriptional level [4,5,6,7]. The expression profile of miRNAs is unique in each tissue, disease, and normal condition [8, 9]. Therefore, the dysregulation of miRNA expression can be critical to the pathogenesis of various diseases, including women’s reproductive disorders [7, 10,11,12], which are the main concern of modern women’s health and may have a negative effect on fertility [13, 14]. The most common women’s reproductive diseases include polycystic ovary syndrome (PCOS) [15], endometriosis (EMs) [16, 17], uterine leiomyomas (ULs) [18], and ovarian cancer (OC) [12, 19]. Since these diseases do not exhibit obvious symptoms at the onset, they are typically diagnosed during the advanced stage [20]. The mechanistic diagram of ovarian/uterine dysfunctions was shown in Fig. 1. In contrast, traditional diagnostic techniques like laparoscopy, ultrasonography, and protein biomarkers are both costly and time-consuming. Therefore, it is essential to discover new biomarkers in order to diagnose and treatment these diseases. Cellular miRNAs can be found in the cellular environment, while extracellular miRNAs (also known as circulating miRNAs) can be easily obtained from various body fluids such as serum, plasma, tears, saliva, urine, breast milk, colostrum, peritoneal fluid, cerebrospinal fluid, bronchial lavage, seminal fluid, and follicular fluid [21]. Additionally, circulating hormones or cytokines are less stable in blood compared to these molecules, making them excellent biomarkers for non-invasive diagnosis of female reproductive diseases. However, the main feature in using miRNAs as diagnostic biomarkers include identifying specific miRNAs applicable to a diverse range of patients and creating cost-effective and straightforward methods for analysing these molecules [11].
Consequently, this study aims to shed light how dysregulation of miRNA affects reproductive diseases in women as well as the signaling pathways that are associated with these conditions.
Polycystic ovary syndrome
Polycystic ovarian syndrome (PCOS) is a common reproductive endocrine syndrome in women of childbearing age, with an incidence of about 5–15% [22]. According to previous research, PCOS is usually accompanied by ovulatory dysfunction, hyperandrogenism, and insulin resistance (IR) [23]. Hence, PCOS is the cause of 40% of female infertility [24]. Some common symptoms of PCOS are a disrupted menstrual cycle, excessive hair growth, acne, being overweight, and infertility [25]. Currently, the etiology of PCOS is still unclear, but environmental and genetic factors, as well as alterations in the expression of miRNAs, may contribute to the pathogenesis of PCOS [15, 26]. Numerous studies have shown that abnormal levels of miRNAs can play a significant role in the pathogenesis and diagnosis of PCOS patients, often targeting key signaling pathways [27, 28]. Long et al. discovered that some miRNAs, namely miR-222, miR-16, miR-19a, miR-24, and miR-186, were upregulated while miR-320 was downregulated in individuals with PCOS. These miRNAs were identified as new biomarkers for diagnosing PCOS [27]. MiR-222, among these miRNAs, has been found to be associated with the development of PCOS, with its levels increasing. When this miRNA is overexpressed, it can lead to a decrease in the expression of p27 Kip1 in human granulosa tumor cells (KGN). P27 Kip1 is a member of the CDKI family and plays a role as a tumor-suppressor gene, promoting apoptosis and regulating the cell cycle. Both miR-222 and its target, p27 Kip1, have been proposed as potential biomarkers for PCOS patients [29, 30]. The miR-16 has been found as a downregulated miRNA in ovarian cortex tissues and serums of PCOS patients. The overexpression of this miRNA promotes ovarian granulosa cell (GC) proliferation and suppresses apoptosis by directly targeting programmed cell death protein 4 (PDCD4) [31]. However, another study conducted by Wang et al. found that overexpressing miR-16 could actually inhibit GC growth and increase cell death in PCOS patients by targeting Apelin13 and inhibiting the PI3K/Akt pathway [32]. In PCOS patients, the miR-19a has been found to target tumor necrosis factor α (TNF-α), leading to the apoptosis of KGN cells. Additionally, a study conducted by Song et al. revealed that miR-186 and miR-135a were elevated in GCs of PCOS patients and had a detrimental impact on the function of GCs by inhibiting the expression of estrogen receptor 2 (ESR2). Therefore, the levels of miR-186 and miR-135a in the GCs are associated with the levels of serum estradiol in individuals with PCOS [33]. A research study discovered that the expression of miR-24 was downregulated in the follicular fluid and serum of women who have PCOS. This particular miRNA plays a crucial role in the process of steroid hormone biosynthesis [34]. MiR-24 targets cytochrome P450 family 11A1 (CYP11A1), which is a critical enzyme involved in the biosynthesis of steroid hormones [35, 36]. A decrease in miR-24 levels could result in an increase in CYP11A1 expression, ultimately leading to an enhanced synthesis of androgens. The study suggests that miR-24 could be utilized as a diagnostic biomarker for PCOS [36].
In a separate study by Kong et al., it was observed that miR-9 was highly expressed in the follicular fluid of PCOS patients, leading to ovarian dysfunction by targeting vitamin D receptors (VDR) [37]. Moreover, Rashad et al. revealed that the levels of miR-320 were reduced in individuals with PCOS and it targeted the endothelin-1 (ET-1) gene [38]. The ET-1 gene is a powerful biomolecule peptide that plays a role in cell division, tumor growth, and mitosis [39, 40]. Similarly, Yuan et al. found that the expression of this particular miRNA was significantly decreased in the ovarian tissue of PCOS patients with insulin resistance (IR) compared to healthy individuals. Interestingly, they also found that this miRNA could regulate IR in PCOS patients by inhibiting the ERK1/2 pathway through targeting insulin receptor substrate 1 (IRS-1) [41]. Additionally, Sang et al. found that there was a significant decrease in miR-132 levels in the follicular fluid of patients with PCOS compared to healthy individuals [42]. However, another study observed an upregulation of miR-132 in GCs of PCOS patients [43]. MiR-132 directly targets forkhead box A1 (Foxa1), resulting in the suppression of KGN cell viability [43]. Foxa1 is a transcription factor that determines genomic regions for binding the other transcription factors, including estrogen, progesterone, and androgen receptors [44, 45]. In PCOS patients, Chen et al. discovered an overexpression of miR-93 in adipocytes. They observed that miR-93 upregulation resulted in a reduction of glucose transporter type 4 (GLUT4) gene expression [46]. GLUT4 is a crucial protein that facilitates glucose transportation to adipocytes via insulin, and its expression is decreased in adipocytes of PCOS patients, which affects glucose homeostasis [47]. One study revealed that miR-145 decreases in human GCs in PCOS patients. This miRNA could suppress human GC cell proliferation by targeting IRS1, leading to blocked MAPK/ERK signaling pathways in PCOS patients. This study also reported that high insulin levels decreased the expression of miR-145 in PCOS [48]. Xia et al. found that miR-155 was highly expressed in the ovarian cortical tissue of individuals with PCOS and could promote proliferation, migration, and invasion in human KGN cells. However, they also observed that this miRNA could decrease PDCD4 levels and trigger the PI3K/AKT and JNK signaling pathways [49]. Other studies have also found that this miRNA is multifunctional and is used as a biomarker for diagnosing PCOS [50, 51]. PDCD4 is a translational repressor that binds to the eIF4A factor and inhibits its RNA helicase activity [52, 53]. This protein is highly expressed in women with PCOS [54]. Geng et al. reported the relationship between miR-99a and PCOS. They found that miR-99a could promote the apoptosis of human GCs by targeting the 3′-UTR of insulin-like growth factor 1 receptor (IGF-1R). IGF-1R is a member of the transmembrane tyrosine kinase receptor family, activated by insulin-like growth factor 1 (IGF-1) or IGF-2. They also reported that the expression of this miRNA decreased in PCOS patients, whereas the protein level of IGF-1R increased [55].
Endometriosis
Endometriosis (EMs) is a multifactorial estrogen-dependent gynecologic condition. This disease affects approximately 10% of all reproductive-aged women worldwide [56]. The clinical symptoms of EMs typically include dysmenorrhea, chronic pelvic pain, dyspareunia, and infertility [57]. Several studies have indicated that this disease is also related to people’s genetics [58,59,60]. The risk of EMs is seven times higher in women with a family history compared to other women [61]. EMs, despite being highly prevalent, is often difficult to detect due to its variable symptoms and complications in pathogenesis, resulting in delays in diagnosis [62]. There are various methods available for detecting this disease, such as imaging techniques, protein biomarkers, and laparoscopy [56]. Currently, laparoscopic surgery is considered the most reliable method for diagnosing EMs [63], but it is expensive and invasive. As a result, scientists have been exploring alternative diagnostic tests for EMs [56]. Numerous research studies have demonstrated the involvement of multiple miRNAs in the development of EMs, making them a desirable option for non-invasive detection of these diseases [64,65,66,67]. Moreover, miRNA levels have been observed to alter in EMs according to researchers [68].
In their ground-breaking study, Wang et al. discovered that the levels of certain miRNAs in the bloodstream can serve as sensitive markers for detecting endometriosis. They specifically found that miR-199a is associated with the typical symptoms of this condition, indicating its importance in hormone-related pathways. Through experimental analysis, they identified several target genes of miR-199a, including discoidin domain receptor 1 (DDR1), CD44, early growth response 1 (EGR1), and serine/threonine-protein kinase (SGK3B) in the Wnt signaling pathway, as well as SGK3B in the Ras-GTPase pathway. Additionally, the researchers observed that the combination of miR-199a and miR-122, by targeting SRY-related HMG box 4 (SOX4), showed an increase in serum levels and could potentially be used to differentiate the severity of endometriosis [69]. Furthermore, it was observed that the levels of miR-202 were reduced in the endometrial stromal cells (ESCs) of patients with EMs. Additionally, by targeting K-Ras in the Raf-1/MEK/ERK signaling pathway, the overexpression of miR-202 was shown to hinder the invasion and migration of primary ESCs from patients with EMs [70]. The plasma levels of let-7b-5p are significantly downregulated in EMs [71]. The toll-like receptor 4 (TLR-4) gene is targeted by this miRNA, potentially contributing to the inflammation involved in the pathophysiology of EMs [72]. Furthermore, the let-7 family has been shown to target the KRAS gene, which is part of the KRAS pathway that is targeted for therapeutic treatment of EMs [73]. Hawkins et al. discovered that certain miRNAs exhibited altered expression in EMs as compared to the endometrium. One of these miRNAs, miR-29c, targeted the 3′-UTR of several extracellular matrix genes, including laminin subunit gamma 2 (LAMC2), transcription factor AP-2 gamma (TFAP2C), uroplakin 1B (UPK1B), and fraser syndrome 1 (FRAS1). To confirm these targets, a luciferase reporter was used, revealing that miR-29c targets the 3′-UTRs of collagen type III alpha 1 chain (COL3A), collagen type VII alpha 1 chain (COL7A1), collagen type XXI alpha 1 Chain (COL21A1), and TFAP2C mRNAs [74]. A different research demonstrated that the levels of miR-29c were reduced in individuals with EMs. This specific miRNA has the ability to suppress the progression of EMs by impacting the invasion of the endometrium, the growth of cells, and programmed cell death, all by suppressing the expression of c-Jun. Consequently, targeting miR-29c could potentially be a novel approach for the treatment of EMs [75]. Takebayashi et al. discovered that there was an increase in the expression of miR-100-5p in stromal cells of human endometriotic cysts (ECSCs). They found that miR-100-5p directly targeted two genes, namely SWItch/sucrose non-fermentable-related matrix-associated actin-dependent regulator of chromatin subfamily D member 1 (SMARCD1) and matrix metallopeptidase 1 (MMP1) [76]. Furthermore, numerous research studies have discovered that the levels of miR-34a expression were significantly decreased in EM tissues. This decrease in miR-34a expression has been found to have an impact on the p53 protein by inhibiting sirtuin 1 (SIRT1) [77, 78]. It is worth noting that SIRT1plays a crucial role in regulating the homeostasis of vascular endothelial cells [79, 80]. Mounting evidence indicates that the miR-200 family, including miR-141-3p, miR-200a, miR-200b, and miR-200c, changes in the plasma levels of patients with EMs. This family can serve as potential non-invasive biomarkers for diagnosing EMs [81]. Kruppel-like factor 12 (KLF-12) is a transcription factor and plays a regulatory role in some cell processes such as cell differentiation, physiologic function, and phenotypic modulation [82]. It has been reported that miR-141-3p significantly decreases in ectopic ESCs. This miRNA targets KLF-12 mRNA, which leads to the suppression of cell proliferation and migration [83]. In a study by Rekker et al., it was found that the levels of miR-200a and miR-141 were lower in the plasma of women with EMs compared to those who were healthy. The study indicated that these two microRNAs could potentially serve as novel non-invasive biomarkers for diagnosing EMs [81]. while the search results suggest that miR-200a may play a role in endometriosis, the findings are not entirely consistent and further research is needed to fully understand its mechanisms of action in the disease. A different research showed that the levels of miR-200b were reduced in individuals with EMs and this had an impact on the growth and spread of endometriotic cells. This was achieved by targeting three proteins: krüppel-like factor 4 (KLF4), zinc finger E-box binding homeobox 1 (ZEB1), and zinc finger E-box binding homeobox 2 (ZEB2) [84]. Moreover, miR-200c is strikingly downregulated in ectopic endometrial compared with normal endometrial tissues. This miRNA represses EMs by targeting metastasis-associated lung adenocarcinoma transcript 1 (MALAT1) [85]. In another study, Rekker et al. showed that the expression of miR-139-5p and miR-375 increased and decreased, respectively, in endometriotic lesions compared to eutopic cells. Overexpression of miR-139-5p resulted in the suppression of homeobox A9 (HOXA9) and homeobox A10 (HOXA10), while miR-375 was responsible for regulating the endothelin 1 (EDN1) gene [86]. Overall, the exact mechanisms by which miR-139-5p and miR-375 contribute to endometriosis development are not fully understood and require further investigation. However, the search results suggest that these miRNAs may be involved in multiple processes that are dysregulated in endometriosis, including cell proliferation, apoptosis, inflammation, metabolism, and angiogenesis [86]. Multiple pieces of evidence have indicated that miR-145 is a molecular marker that can be used non-invasively to diagnose EMs in patients at an early stage. This miRNA supresses the growth and invasion of endometriotic cells by targeting various factors related to pluripotency (SOX2) and cytoskeletal components (JAM-A and transgelin) [87, 88]. Several research studies have shown that the relationship between miR-126 and CRK proto-oncogene (Crk) can impact the growth and advancement of EMs. Crk has the ability to bind to various signaling molecules using its SH2/SH3 domain. This protein is essential for important cellular processes such as cell proliferation, migration, morphogenesis, invasion, phagocytosis, and survival, and is therefore involved in transmitting cellular signals. As a result, it plays a significant role in EMs [89, 90].
In a separate investigation, Jia et al. discovered that, miR-20a was notably decreased in EMs patients when compared to healthy individuals [91]. Interestingly, another study reported an increase in miR-20a in females with EMs in contrast to controls (women with benign ovarian tumors). However, the upregulation of this miRNA was only observed in stages III and IV (advanced stage) and not in stages I and II (mild stage). Additionally, miR-20a has an impact on netrin-4 (NTN4) in EMs [92]. NTN4 is a secreted protein that plays a crucial role in various biological processes such as angiogenesis, tumorigenesis, cell migration, and invasion [93, 94]. Additionally, miR-424-5p is underexpressed in the ectopic endometrium and controls proliferation and apoptosis in EM cells via targeting fibroblast growth factor receptor (FGFR1) in the STAT3 signaling pathway [95]. FGFR1 is a growth factor receptor tyrosine kinase (RTK) involved in cellular processes such as cell proliferation and migration, angiogenesis, and the suppression of apoptosis [96]. In their study, Ma et al. reported that the levels of miR-142-3p decreased in ectopic endometrial tissues, while the expression of krüppel-like factor 9 (KLF9) increased, indicating that KLF9 is targeted by miR-142-3p [97]. KLF9 is known to have a significant impact on abnormal estrogen levels and acts as a progesterone receptor in endometriosis [98, 99]. Zhou et al. revealed that miR-196a was significantly upregulated in patients with EMs (mild stage). This miRNA decreases the expression of progesterone receptor (PGR) isoforms via the activation of the MEK/ERK signaling pathway and repress the progesterone receptor and decidualization in eutopic endometrium from women with EMs. This suggests that miR-196a may be involved in the resistance to progestin therapy that is often observed in EMs [100].
Uterine leiomyomas
Uterine leiomyomas (ULs), known as fibroids or myomas, are benign cancers of uterine smooth muscles (myometrium) and the most common pelvic tumors [18]. ULs mainly affect females during their childbearing years [101]. Most women with ULs are asymptomatic, but about 30% of them show different symptoms, including pelvic and back pain, abnormal uterine bleeding (AUB), anemia, constipation, urinary frequency, recurrent pregnancy loss, and infertility, which can eventually lead to a hysterectomy [102]. This disease was diagnosed in more than 70% of white women and more than 80% of African women [103, 104]. Despite the high prevalence of ULs, their cellular and molecular bases are not entirely understood. However, some alternations in genetics, epigenetics, the expression of miRNAs, steroids (estrogen and progesterone), the disorganized extracellular matrix (ECM), chemokines, cytokines, and growth factors play an important role in the formation and development of ULs [105].
Numerous pieces of evidence have shown that many miRNAs are expressed differently in leiomyoma compared to normal myometrium tissue [106,107,108]. Therefore, it can be argued that miRNAs regulate gene expression in this cancer [105]. The expression of let-7 family miRNAs is significantly upregulated in fibroids relative to the myometrium and is associated with fibroid size. Indeed, small fibroids (≤ 3 cm) have a higher expression of let-7 family miRNAs compared to large fibroids (≥ 10 cm) [106]. Klemke et al. and Peng et al. showed that let-7 family miRNAs suppressed the expression of high motility group A2 (HMGA2) genes in vitro and in vivo. Thus, they contribute to fibroid growth and development [109, 110]. The miR-21 has been reported as a highly upregulated miRNA in fibroids [106]. Furthermore, this miRNA is overexpressed as an oncogenic miRNA (oncomiR) in the (hTERT)-immortalized leiomyoma cell line (UtLM) and leiomyoma tissue. Therefore, it can cause apoptosis and translation by binding to the mRNA of molecules involved in these processes [111]. On the other hand, PDCD-4 is a potential target for miR-21. Unexpectedly, the overexpression of miR-21 increases the expression of PDCD-4 at the protein level in ULs, while the expression of this molecule decreases in most cancers and acts as a tumor suppressor [111]. Interestingly, miR-21, both directly and indirectly, affects the TGF/β pathway by suppressing SMAD7 and the TGF/β receptor, respectively, and increases ECM formation in fibroids [112]. Cardozo et al. showed that the overexpression of miR-21a-5p increased the expression of TGF/β3 protein in fibroids and altered the expression of several genes, including matrix metalloproteinase-2 (MMP-2), matrix metalloproteinase-9 (MMP-9), fibronectin, and collagen. According to these findings, the overexpression of miR-21 can increase proliferation in UL cells [113]. The miR-29 family (miR-29a, miR-29b, and miR-29c), especially miR-29b and miR-29c, is significantly downregulated in leiomyoma relative to the myometrium [107]. This family generally targets more than 15 ECM-related genes, including collagen subtypes and elastin in fibroids [114]. The miRNAs from this family directly bind to the 3`-UTR of collagen type I and inhibit the translation of this protein [114]. Therefore, the downregulation of the miR-29 members increases the collagen fibers in leiomyoma [115, 116]. Xu et al. found that this family could directly or indirectly regulate the expression of several genes in the protease kinase B (PKB or AKT) pathway, increasing growth and survival in response to extracellular signals [117]. Another study revealed that miR-29c could target CDK (a key regulator of the G1/S phase). Thus, increasing the expression of miR-29c in fibroids inhibits the expression of CDK, and the knockdown of this miRNA increases the expression of CDK [118]. The two miR-93 and miR-106 families have overlapping functions in fibroids [119]. Melo et al. showed that miR-93 and miR-106b had lower expression levels in fibroids than in the myometrium and were predicted to be regulators of the HMGA2 gene [120]. Furthermore, miR-93 and miR-106b can directly or indirectly regulate the expression of several factors, including plasminogen activator inhibitor-1 (PAI-1), tissue factor 3 (TF3), connective tissue growth factor (CTGF), and interleukin-8 (IL-8), which are associated with inflammation and tissue turnover. Accordingly, these two miRNAs can play an important role in fibroid biology [121]. Furthermore, it has been shown that miR-93 targets E2F transcription factor 1 (E2F1) and cyclin D1 (CCND1) in primary uterine fibroid cells [118]. The miR-197 is a miRNA tumor suppressor, the expression of which is reduced in human UFs. This miRNA induces apoptosis and inhibits cell proliferation and migration in vitro [122, 123]. Ling et al. found that the downregulation of miR-197 could increase cell growth and stop the cell cycle in the G0/G1 phase in human UL cells. It has also been shown that miR-197 inhibits cell proliferation by directly targeting insulin-like growth factor binding proteins 5 (IGFBP5) [124]. Numerous pieces of evidence have shown that miR-200a, miR-200b, and miR-200c (especially miR-200c) are significantly downregulated in ULs compared to the healthy surrounding uterine tissue [119]. The miR-200 family may act as tumor suppressors by inhibiting epithelial-mesenchymal transition (EMT) [125]. Furthermore, these miRNAs regulate the activation of the NF-kB pathway via the phosphorylation of its inhibitor [121]. Like the miR-29 family, the miR-200 family regulates the expression of several genes in the AKT pathway [117]. Additionally, miR-200c, like miR-29c, targets CDK2 mRNA in fibroids [118].
Ovarian cancer
Ovarian cancer (OC) is one of the most common gynecologic malignancies, ranking third after cervical and uterine cancers [126, 127]. This cancer has a high mortality rate, and it is estimated that deaths from OC will increase significantly by 2040 [127]. The therapeutic options for OC have been remarkably improved by identifying invasive surgical methods and new drug treatments [128]. Recently, several clinical trials have shown that poly (ADP-ribose) polymerase (PARP) inhibitors (namely olaparib, niraparib, talazoparib, and rucaparib) are effective in patients with platinum-resistant or sensitive OC with BReast CAncer gene 1 (BRCA1) or BReast CAncer gene 1 (BRCA2) mutations [129, 130]. Hence, these inhibitors can be a turning point in treating OC in the early stages and in tumor recurrence [131]. Besides these novel therapeutic methods, multidisciplinary approaches with different specialists are needed to treat OC to determine the treatment protocol for each patient based on the patient’s characteristics and the tumor state [132, 133]. On the other hand, the symptoms of OC are often absent or vague at the onset of the disease, and due to the lack of early diagnostic methods and reliable molecular markers, most patients are diagnosed at an advanced stage, and only 20% of them are diagnosed at stage I of the disease [20].
Recently, several studies have shown that changes in the expression profiling of miRNAs have been detected in OC. Hence, miRNAs can serve as new biomarkers with prognostic, diagnostic, and therapeutic approaches in this cancer [26, 154, 155]. Here, the most important miRNAs involved in the pathogenesis of OC and their molecular targets are discussed. The dysregulation of the miR-200 family and their role in the onset and progression of OC are well established [134]. Members of the miR-200 family suppress the expression of ZEB1 and ZEB2, which leads to the suppression of EMT [135]. Besides, this family affects the β-tubulin III protein in OC [136]. β-tubulin III is a microtubule protein normally expressed in cells of neuronal origin [137]. The overexpression of this protein has been reported in OC and is associated with poor survival and poor response to treatment [138]. The let-7 family is another family whose expression is significantly reduced in OC [139]. This family inhibits several oncogenes, such as KRAS, HRAS, c-MYC, and HMGA-2, and several cell cycle regulators, such as cyclin A, cyclin D1, cyclin D2, cyclin D3, CDC25, and CDK6, in OC [140,141,142,143]. Intriguingly, miR-34 family members (miR-34a, miR-34b, and miR-34c) are underexpressed in OC [144]. This family also targets several genes, including c-MYC, NOTCH1, CDK6, E2F3, MET, Bcl2, and cyclin D1 in OC [144, 145]. In addition, the expression of miR-100 decreases in most of the OC cell lines and tissues [5, 146]. This miRNA inhibits the mTOR signaling pathway and proto-oncogene of polo-like kinase-1 (PLK1) in OC [146, 147]. The miR-9 is a downregulated miRNA in human OC, and its overexpression inhibits OC cell growth [148]. This miRNA could directly bind to the 3′-UTR of NF-κB1 mRNA and suppress the NF-κB1 expression at both the mRNA and protein levels. Therefore, NF-κB1 is an important target of miR-9 in OC cells [148]. In addition, miR-140-3p is downregulated in OC and is a potential regulator of the RAD51-associated protein 1 gene (RAD51AP1) [149, 150]. The RAD51AP1 gene is involved in double-stranded DNA repair and is overexpressed in OC [149]. Furthermore, the dysregulation of miR-214 has been reported in OC. This miRNA induces cisplatin resistance and cell survival via targeting the 3’-UTR of PTEN mRNA, which leads to the suppression of PTEN protein and the activation of the PI3K/AKT/mTOR signaling pathway [151].
Infertility
Infertility is a global reproductive health problem that affects 10–15% of women worldwide, and women’s reproductive disorders may have adverse effects on fertility [13, 14]. Implantation occurs as a result of complex communication between the blastocyst and endometrium. Some studies have demonstrated that the interaction between various genes and miRNAs could lead to the creation of signaling during implantation, thereby modulating the interaction between the embryo and endometrium [152]. Since miRNAs are secreted from both the blastocyst and endometrium, we mention several examples of different expressions of miRNAs in implanted compared with non-implanted blastocysts and in the endometrium of women with recurrent implantation failure. Cuman et al. discovered that miR-661 is more highly expressed in blastocysts that do not successfully implant compared to those that do. It was found that when primary human endometrial epithelial cells (HEECs) take in miR-661, it reduces the attachment of trophoblast cell line spheroids to HEECs through poliovirus receptor-related 1 (PVRL1) [153]. This suggests that human blastocysts, through changes in miRNA secretion, can cause abnormalities that lead to failed implantation. In addition, certain research studies have indicated that miRNAs exhibit varying levels of expression during the menstrual cycle and that the irregularity of specific miRNAs can impact a woman's fertility [154,155,156]. One such example is the increase in endometrial miR-31 during the reproductive cycle, which targets the regulatory protein forkhead box P3 (FOXP3) that plays a role in immune system responses [157].
MiRNA-based therapeutics in PCOS, EMS, ULS, and OC
MiRNA-based therapeutics have shown promising results in the diagnosis and treatment of various reproductive women diseases. Recent research has shown that miRNAs and their target mRNAs are differentially expressed in EMs, OC, ULS and PCOS. Additionally, miRNAs have been found to regulate a wide range of normal and abnormal cellular functions, suggesting they may have significant roles in the development of these disorders. For example, a study revealed that the downregulation of miR-17-5P and the upregulation of autophagy-related protein 7 (ATG7) in PCOS patients could serve as therapeutic targets in these patients [158]. Moreover, several studies have demonstrated that the let-7 family targets the KRAS gene and that the KRAS pathway is one of the therapeutic targets for treating EMs [73]. Indeed, the local treatment of EMs with let-7b can be considered a promising treatment for EMs because this miRNA can simultaneously affect several signaling pathways, including estrogen, KRAS, and inflammatory signaling [72]. On the other hand, miR-21 is the most common miRNA that is differentially expressed in ULs and is regulated by estradiol. Recently, this miRNA has been a target for treating ULs [101]. The miR-199a-5p is another miRNA that could serve as an evolutionarily conserved anti-oncogene and a therapeutic target for ULs [159]. According to the study by Ahn et al., the expression level of miR-139-5p decreases in myeloma cells, and the modulation of miR-139-5p could be effective in the pathogenesis of ULs by regulating collagen type 1 and the phosphorylation of the p38 MAPK pathway. Therefore, this miRNA can be used as a new therapeutic target in ULs [160]. In patients with OC, reducing miR-150 and miR-214 expression can prevent the proliferation of cancer cells and the tolerance of chemotherapy drugs. Hence, these two miRNAs may be used as an adjuvant treatment in OC [161]. Lan et al. revealed that the downregulation of miR-140-5P can lead to a decrease in the sensitivity of OC cells to the drug cisplatin [162]. The upregulation of this miRNA can inhibit the proliferation and migration of cancer cells and increase the sensitivity to cisplatin; hence, it may be considered a therapeutic target in OC [163]. In addition, the upregulation of miR-21 is correlated with cisplatin resistance in OC, and targeting miR-21 as a therapeutic target can decrease the tumorigenic characteristics in cisplatin-resistant cells [164].
On the other hand, miRNAs could be used as biomarkers to monitor therapeutic efficacy. Metformin is an insulin-sensitizing drug that has been used for decades in PCOS patients. This drug improves IR and impaired glucose tolerance (IGT) and reduces androgen levels in PCOS patients through the possible reduction of the miR-222 expression level [163]. Moreover, treatments based on antiandrogenic (AR blockers), including ethinyl estradiol and cyproterone-acetate, deal with the excess amount of androgens in PCOS patients and increase the levels of miR-27 b and miR-155 [51]. Additionally, it has been reported that miR-125b expression levels change in ovarian endometriomas after treatment with leuprolide acetate. Therefore, this miRNA can be used as a potential biomarker in monitoring response to treatment [165]. Kiba et al. found that five miRNAs, including miR-15b, miR-125b-1, miR-136, miR-142-3p, and miR-146a, were upregulated in ovarian endometrioma after leuprolide acetate treatment [165]. Furthermore, let-7a can be served as a biomarker in monitoring the response to chemotherapy in OC patients. Indeed, let-7a upregulation was associated with increased survival rates in OC patients receiving platinum alone, but with lower survival in patients receiving combination therapy [166].
In the future, personalized medicine could benefit from the use of miRNA-based therapies. Nevertheless, there are still obstacles to overcome, such as standardization and a better comprehension of how miRNAs can be applied in personalized medicine. Despite these challenges, scientists are investigating the potential of miRNA-based therapies as a hopeful new method for treating a variety of illnesses.
Conclusions
Infertility and the risk of fetal and maternal mortality are largely attributed to common reproductive diseases in women, including PCOS, EMs, ULs, and OC. Early detection is crucial for effective treatment, but current diagnostic methods are often time-consuming and expensive. However, miRNAs have emerged as significant players in various processes that impact the functioning of the female reproductive system. On the other hand, an ideal biomarker should meet certain criteria, such as enabling early detection, being easily accessible through non-invasive methods, being specific to each condition or tissue, exhibiting sensitivity, having a long half-life in samples, and allowing for accurate and reasonably priced detection. According to this review, miRNA signatures are different in healthy women compared to women with reproductive disorders (Fig. 2). MiRNAs play a significant role in regulating gene expression by targeting specific molecules, some of which are involved in signaling pathways. These pathways are crucial in the development of reproductive diseases in women, such as MAPK/ERK, PI3K/AKT, ERK1/2, KRAS, WNT, e Ras-GTPase, Adherens junction, Hippo, ECM-receptor interaction, TGF/β, P38 MAPK, NF-κB, NOTCH1, P53, STAT3, JNK, and mTOR. In conclusion, using miRNAs presents a fresh method for identifying reproductive diseases. Consequently, miRNAs have been identified as particular diagnostic biomarkers implicated in the development of reproductive diseases. Our final goal in this study was to assess the impact of miRNAs and their targets on the pathogenesis and diagnosis of reproductive diseases in women (Table 1). Nonetheless, research on miRNAs in women's reproductive diseases is still in its early stages. However, there is a growing body of evidence that suggests that miRNAs have the potential to be used as biomarkers for diagnosis and as therapeutic targets. Moreover, there are several challenges in investigating the role of miRNAs in reproductive women diseases. These challenges include a limited understanding of miRNA function, difficulty in identifying the targets of miRNAs, variability in miRNA expression, lack of standardization in sample collection, RNA isolation, and data analysis, as well as high costs. However, despite these challenges, researchers are actively exploring the potential of miRNA-based therapies as a promising approach for treating various reproductive women diseases. As research progresses, new insights are expected to emerge that will help overcome these challenges and enhance our understanding of how miRNAs can be utilized in personalized medicine.
Data availability
Not applicable.
Abbreviations
- MiRNAs:
-
MicroRNAs
- PCOS:
-
Polycystic ovarian syndrome
- IR:
-
Insulin resistance
- KGN:
-
Human granulosa tumor cells
- CDKI:
-
Cyclin-dependent kinase inhibitor
- GC:
-
Granulosa cell
- PDCD4:
-
Programmed cell death protein 4
- TNF-α:
-
Tumor necrosis factor α
- ESR2:
-
Estrogen receptor 2
- CYP11A1:
-
Cytochrome P450 family 11A1
- OD:
-
Oocyte donor
- VDR:
-
Vitamin D receptors
- ET-1:
-
Endothelin-1
- Foxa1:
-
Forkhead box A1
- GLUT4:
-
Glucose transporter type 4
- IRS1:
-
Insulin receptor substrate 1
- IGF-1:
-
Insulin-like growth factor 1
- EMs:
-
Endometriosis
- ESCs:
-
Endometrial stromal cells
- TLR-4:
-
Toll-like receptor 4
- LAMC2:
-
Laminin-2
- TFAP2C:
-
Transcription factor AP-2
- UPK1B:
-
Uroplakin1B
- FRAS1:
-
Fraser syndrome 1
- COL3A:
-
Collagen type III A
- ECSCs:
-
Endometriotic cyst stromal cells
- SMARCD1:
-
Chromatin subfamily D member 1
- MMP1:
-
Matrix metallopeptidase 1
- MMP-2:
-
Matrix metalloproteinase-2
- MMP-9:
-
Matrix metalloproteinase-9
- SIRT1:
-
Sirtuin 1
- KLF-12:
-
Kruppel-like factor 12
- KLF4:
-
Krüppel-like factor 4
- ZEB1:
-
Zinc finger E-box binding homeobox 1
- ZEB2:
-
Zinc finger E-box binding homeobox 2
- MALAT1:
-
Metastasis-associated lung adenocarcinoma transcript 1
- HOXA9:
-
Homeobox A9
- HOXA10:
-
Homeobox A10
- NTN4:
-
Netrin-4
- FGFR1:
-
Fibroblast growth factor receptor
- RTK:
-
Receptor tyrosine kinase
- PGR:
-
Progesterone receptor
- KLF9:
-
Krüppel-like factor 9
- ULs:
-
Uterine leiomyomas
- AUB:
-
Abnormal uterine bleeding
- ECM:
-
Extracellular matrix
- HMGA2:
-
High motility group A2
- OncomiR:
-
Oncogenic miRNA
- PKB:
-
Protease kinase B
- PAI-1:
-
Plasminogen activator inhibitor-1
- TF3:
-
Tissue factor 3
- CTGF:
-
Connective tissue growth factor
- IL-8:
-
Interleukin-8
- E2F1:
-
E2F transcription factor 1
- CCND1:
-
Cyclin D1
- IGFBP5:
-
Insulin-like growth factor binding proteins 5
- EMT:
-
Epithelial-mesenchymal transition
- LPCs:
-
Leiomyoma progenitor cells
- OC:
-
Ovarian cancer
- PARP:
-
Poly ADP-ribose polymerase
- PLK1:
-
Polo-like kinase-1
- RAD51AP1:
-
RAD51-associated protein 1 gene
- EAOC:
-
Endometriosis-associated ovarian cancer
- SOC:
-
Serous ovarian cancer
- OCCC:
-
Ovarian clear cell cancer
- ATG7:
-
Autophagy-related protein 7
- IGT:
-
Impaired glucose tolerance
- AR:
-
Antiandrogenic
- HEECs:
-
Human endometrial epithelial cells
- FOXP3:
-
Forkhead box P3
References
Wang J, Chen J, Sen S (2016) MicroRNA as biomarkers and diagnostics. J Cell Physiol 231(1):25–30
Friedman RC et al (2009) Most mammalian mRNAs are conserved targets of microRNAs. Genome Res 19(1):92–105
Berezikov E et al (2005) Phylogenetic shadowing and computational identification of human microRNA genes. Cell 120(1):21–24
O’Brien J et al (2018) Overview of microRNA biogenesis, mechanisms of actions, and circulation. Front Endocrinol 9:402
Nagaraja AK et al (2010) A link between mir-100 and FRAP1/mTOR in clear cell ovarian cancer. Mol Endocrinol 24(2):447–463
Azimi T et al (2021) Pap Smear miR-92a-5p and miR-155-5p as potential diagnostic biomarkers of squamous intraepithelial cervical cancer. Asian Pac J Cancer Prev APJCP 22(4):1271
Mansouri S et al (2020) Alteration in expression of miR-32 and FBXW7 tumor suppressor in plasma samples of patients with T-cell acute lymphoblastic leukemia. Cancer Manag Res 12:1253–1259
Tafti A et al (2023) A systems biology approach and in vitro experiment indicated Rapamycin targets key cancer and cell cycle-related genes and miRNAs in triple-negative breast cancer cells. Mol Carcinog. https://doi.org/10.1002/mc.23628
Bagheri M, Sarabi PZ, Mondanizadeh M (2022) The role of miRNAs as a big master regulator of signaling pathways involved in lymphoblastic leukemia. J Cell Physiol 237(4):2128–2139
Mouillet JF et al (2015) MicroRNAs in placental health and disease. Am J Obstet Gynecol 213(4):S163–S172
Condrat CE et al (2020) miRNAs as biomarkers in disease: latest findings regarding their role in diagnosis and prognosis. Cells 9(2):276
Pan Q, Chegini N (2008) MicroRNA signature and regulatory functions in the endometrium during normal and disease states. Semin Reprod Med. https://doi.org/10.1055/s-0028-1096128
Vannuccini S et al (2016) Infertility and reproductive disorders: impact of hormonal and inflammatory mechanisms on pregnancy outcome. Hum Reprod Update 22(1):104–115
Gebremedhn S et al (2021) MicroRNA-mediated gene regulatory mechanisms in mammalian female reproductive health. Int J Mol Sci 22(2):938
Teng Y et al (2019) Effect of microRNA-409 on the pathogenesis of polycystic ovary syndrome. Eur Rev Med Pharmacol Sci 23(5):1874–1881
Ohlsson Teague EMC, Print CG, Hull ML (2010) The role of microRNAs in endometriosis and associated reproductive conditions. Human Reprod Update 16(2):142–165
Filigheddu N et al (2010) Differential expression of microRNAs between eutopic and ectopic endometrium in ovarian endometriosis. J Biomed Biotechnol 2010:1–29
McWilliams MM, Chennathukuzhi VM (2017) Recent advances in uterine fibroid etiology. Semin Reprod Med 35:181
Zhang L et al (2008) Genomic and epigenetic alterations deregulate microRNA expression in human epithelial ovarian cancer. Proc Natl Acad Sci 105(19):7004–7009
Zaman MS et al (2012) Current status and implications of microRNAs in ovarian cancer diagnosis and therapy. J Ovarian Res 5(1):1–11
Hunter MP et al (2008) Detection of microRNA expression in human peripheral blood microvesicles. PLoS ONE 3(11):e3694
Azziz R (2016) Introduction: determinants of polycystic ovary syndrome. Fertil Steril 106(1):4–5
Liu AL et al (2016) New insights into mTOR signal pathways in ovarian-related diseases: polycystic ovary syndrome and ovarian cancer. Asian Pac J Cancer Prev APJCP 17(12):5087
Khan MJ, Ullah A, Basit S (2019) Genetic basis of polycystic ovary syndrome (PCOS): current perspectives. Appl Clin Genet 12:249
Yildiz BO (2015) Approach to the patient: contraception in women with polycystic ovary syndrome. J Clin Endocrinol Metab 100(3):794–802
Butler AE et al (2020) microRNA expression in women with and without polycystic ovarian syndrome matched for body mass index. Front Endocrinol 11:206
Long W et al (2014) Characterization of serum microRNAs profile of PCOS and identification of novel non-invasive biomarkers. Cell Physiol Biochem 33(5):1304–1315
Shen H et al (2017) Pathway and network-based analysis of genome-wide association studies and RT-PCR validation in polycystic ovary syndrome. Int J Mol Med 40(5):1385–1396
Huang X et al (2019) MiR-222 promotes the progression of polycystic ovary syndrome by targeting p27 Kip1. Pathol Res Pract 215(5):918–923
Lloyd RV et al (1999) p27kip1: a multifunctional cyclin-dependent kinase inhibitor with prognostic significance in human cancers. Am J Pathol 154(2):313–323
Fu X et al (2018) MicroRNA-16 promotes ovarian granulosa cell proliferation and suppresses apoptosis through targeting PDCD4 in polycystic ovarian syndrome. Cell Physiol Biochem 48(2):670–682
Wang W et al (2023) MicroRNA-16 represses granulosa cell proliferation in polycystic ovarian syndrome through inhibition of the PI3K/Akt pathway by downregulation of Apelin13. Hum Fertil 26(3):611–621
Song Y et al (2019) Altered miR-186 and miR-135a contribute to granulosa cell dysfunction by targeting ESR2: a possible role in polycystic ovary syndrome. Mol Cell Endocrinol 494:110478
Chan MC et al (2010) Molecular basis for antagonism between PDGF and the TGFβ family of signalling pathways by control of miR-24 expression. EMBO J 29(3):559–573
Sørensen AE et al (2016) MicroRNA species in follicular fluid associating with polycystic ovary syndrome and related intermediary phenotypes. J Clin Endocrinol Metab 101(4):1579–1589
Nanda D et al (2020) Evaluation of serum miRNA-24, miRNA-29a and miRNA-502-3p expression in PCOS subjects: correlation with biochemical parameters related to PCOS and insulin resistance. Indian J Clin Biochem 35(2):169–178
Kong F, Du C, Wang Y (2019) MicroRNA-9 affects isolated ovarian granulosa cells proliferation and apoptosis via targeting vitamin D receptor. Mol Cell Endocrinol 486:18–24
Rashad NM et al (2019) Association of miRNA−320 expression level and its target gene endothelin-1 with the susceptibility and clinical features of polycystic ovary syndrome. J Ovarian Res 12(1):1–10
Flammer J, Konieczka K (2015) Retinal venous pressure: the role of endothelin. EPMA J 6(1):1–12
Li C, Sun Y, Liu B (2003) The relationship between endothelin and tumor. Foreign Med Oncol Branch 30:360–362
Yuan W-N, Tan L (2017) MicroRNA-320 inhibits insulin resistance in patients with PCOS through regulating ERK1/2 signaling pathway. Biomed Res 28(11):4946–4949
Sang Q et al (2013) Identification of microRNAs in human follicular fluid: characterization of microRNAs that govern steroidogenesis in vitro and are associated with polycystic ovary syndrome in vivo. J Clin Endocrinol Metab 98(7):3068–3079
Cui X et al (2020) miR-132 is upregulated in polycystic ovarian syndrome and inhibits granulosa cells viability by targeting Foxa1. Mol Med Rep 22(6):5155–5162
Fu X et al (2019) FOXA1 upregulation promotes enhancer and transcriptional reprogramming in endocrine-resistant breast cancer. Proc Natl Acad Sci 116(52):26823–26834
Gou L, Zou H, Li B (2019) Long noncoding RNA MALAT1 knockdown inhibits progression of anaplastic thyroid carcinoma by regulating miR-200a-3p/FOXA1. Cancer Biol Ther 20(11):1355–1365
Chen YH et al (2013) miRNA-93 inhibits GLUT4 and is overexpressed in adipose tissue of polycystic ovary syndrome patients and women with insulin resistance. Diabetes 62(7):2278–2286
Shepherd PR, Kahn BB (1999) Glucose transporters and insulin action—implications for insulin resistance and diabetes mellitus. N Engl J Med 341(4):248–257
Cai G et al (2017) MicroRNA-145 negatively regulates cell proliferation through targeting IRS1 in isolated ovarian granulosa cells from patients with polycystic ovary syndrome. Reprod Sci 24(6):902–910
Xia H, Zhao Y (2020) miR-155 is high-expressed in polycystic ovarian syndrome and promotes cell proliferation and migration through targeting PDCD4 in KGN cells. Artif Cells Nanomed Biotechnol 48(1):197–205
Tam W (2001) Identification and characterization of human BIC, a gene on chromosome 21 that encodes a noncoding RNA. Gene 274(1–2):157–167
Arancio W et al (2018) Serum miRNAs in women affected by hyperandrogenic polycystic ovary syndrome: the potential role of miR-155 as a biomarker for monitoring the estroprogestinic treatment. Gynecol Endocrinol 34(8):704–708
Yang H-S et al (2004) A novel function of the MA-3 domains in transformation and translation suppressor Pdcd4 is essential for its binding to eukaryotic translation initiation factor 4A. Mol Cell Biol 24(9):3894–3906
Yang H-S et al (2003) The transformation suppressor Pdcd4 is a novel eukaryotic translation initiation factor 4A binding protein that inhibits translation. Mol Cell Biol 23(1):26–37
Ding L et al (2016) Higher PDCD4 expression is associated with obesity, insulin resistance, lipid metabolism disorders, and granulosa cell apoptosis in polycystic ovary syndrome. Fertil Steril 105(5):1330-1337.e3
Geng Y et al (2019) MiRNA-99a can regulate proliferation and apoptosis of human granulosa cells via targeting IGF-1R in polycystic ovary syndrome. J Assist Reprod Genet 36(2):211–221
Moustafa S et al (2020) Accurate diagnosis of endometriosis using serum microRNAs. Am J Obstetrics Gynecol 223(4):557.e1-557.e11
Zhou CF et al (2019) miR-205-5p inhibits human endometriosis progression by targeting ANGPT2 in endometrial stromal cells. Stem Cell Res Ther 10(1):1–13
Matalliotakis M et al (2017) The familial risk of endometriosis among the female relatives of patients with endometriosis in Greece. J Endometriosis Pelvic Pain Disorders 9(3):184–187
Nouri K et al (2010) Family incidence of endometriosis in first-, second-, and third-degree relatives: case-control study. Reprod Biol Endocrinol 8(1):1–7
Kennedy S (1999) The genetics of endometriosis. Eur J Obstet Gynecol Reprod Biol 82(2):129–133
Moen MH, Magnus P (1993) The familial risk of endometriosis. Acta Obstet Gynecol Scand 72(7):560–564
Husby GK, Haugen RS, Moen MH (2003) Diagnostic delay in women with pain and endometriosis. Acta Obstet Gynecol Scand 82(7):649–653
Giudice LC, Kao LC (2004) Endometriosis. Lancet 364(9447):1789–1799
Ohlsson Teague EMC et al (2009) MicroRNA-regulated pathways associated with endometriosis. Mol Endocrinol 23(2):265–275
Zhang J et al (2019) Endometriosis pathoetiology: the role of microRNAs in the dysregulation of endometrial functions. Reprod Develop Med 3(04):247–251
Cho S et al (2015) Circulating microRNAs as potential biomarkers for endometriosis. Fertil Steril 103(5):1252-1260.e1
Cosar E et al (2016) Serum microRNAs as diagnostic markers of endometriosis: a comprehensive array-based analysis. Fertil Steril 106(2):402–409
Bjorkman S, Taylor HS (2019) MicroRNAs in endometriosis: biological function and emerging biomarker candidates. Biol Reprod 101(6):1167–1178
Wang W-T et al (2013) Circulating microRNAs identified in a genome-wide serum microRNA expression analysis as noninvasive biomarkers for endometriosis. J Clin Endocrinol Metab 98(1):281–289
Zhang D et al (2020) MicroRNA-202 inhibits endometrial stromal cell migration and invasion by suppressing the K-Ras/Raf1/MEK/ERK signaling pathway. Int J Mol Med 46(6):2078–2088
Nematian SE et al (2018) Systemic inflammation induced by microRNAs: endometriosis-derived alterations in circulating microRNA 125b–5p and Let-7b-5p regulate macrophage cytokine production. J Clin Endocrinol Metab 103(1):64–74
Sahin C et al (2018) micro RNA Let-7b: a novel treatment for endometriosis. J Cell Mol Med 22(11):5346–5353
Grechukhina O et al (2012) A polymorphism in a let-7 microRNA binding site of KRAS in women with endometriosis. EMBO Mol Med 4(3):206–217
Hawkins SM et al (2011) Functional microRNA involved in endometriosis. Mol Endocrinol 25(5):821–832
Long M et al (2015) miR-29c is downregulated in the ectopic endometrium and exerts its effects on endometrial cell proliferation, apoptosis and invasion by targeting c-Jun. Int J Mol Med 35(4):1119–1125
Takebayashi K et al (2020) hsa-miR-100-5p, an overexpressed miRNA in human ovarian endometriotic stromal cells, promotes invasion through attenuation of SMARCD1 expression. Reprod Biol Endocrinol 18(1):1–10
Yamakuchi M, Ferlito M, Lowenstein CJ (2008) miR-34a repression of SIRT1 regulates apoptosis. Proc Natl Acad Sci 105(36):13421–13426
Rezk NA, Lashin MB, Sabbah NA (2021) MiRNA 34-a regulate SIRT-1 and Foxo-1 expression in endometriosis. Non-coding RNA Res 6(1):35–41
Potente M et al (2007) SIRT1 controls endothelial angiogenic functions during vascular growth. Genes Dev 21(20):2644–2658
Potente M, Dimmeler S (2008) Emerging roles of SIRT1 in vascular endothelial homeostasis. Cell Cycle 7(14):2117–2122
Rekker K et al (2015) Circulating miR-200–family micro-RNAs have altered plasma levels in patients with endometriosis and vary with blood collection time. Fertil Steril 104(4):938-946.e2
Suske G, Bruford E, Philipsen S (2005) Mammalian SP/KLF transcription factors: bring in the family. Genomics 85(5):551–556
Zhang Y, Yan J, Pan X (2019) miR-141-3p affects apoptosis and migration of endometrial stromal cells by targeting KLF-12. Eur J Physiol 471(8):1055–1063
Eggers JC et al (2016) microRNA miR-200b affects proliferation, invasiveness and stemness of endometriotic cells by targeting ZEB1, ZEB2 and KLF4. Reprod Biomed Online 32(4):434–445
Liang Z et al (2017) miR-200c suppresses endometriosis by targeting MALAT1 in vitro and in vivo. Stem Cell Res Ther 8(1):1–11
Rekker K et al (2018) Differentially-expressed miRNAs in ectopic stromal cells contribute to endometriosis development: the plausible role of miR-139-5p and miR-375. Int J Mol Sci 19(12):3789
Bashti O et al (2018) miR-31 and miR-145 as potential non-invasive regulatory biomarkers in patients with endometriosis. Cell J (Yakhteh) 20(1):84
Adammek M et al (2013) MicroRNA miR-145 inhibits proliferation, invasiveness, and stem cell phenotype of an in vitro endometriosis model by targeting multiple cytoskeletal elements and pluripotency factors. Fertil Steril 99(5):1346-1355.e5
Liu S et al (2012) Expression of miR-126 and Crk in endometriosis: miR-126 may affect the progression of endometriosis by regulating Crk expression. Arch Gynecol Obstet 285(4):1065–1072
Feng R et al (2010) miR-126 functions as a tumour suppressor in human gastric cancer. Cancer Lett 298(1):50–63
Jia SZ et al (2013) Plasma miR-17-5p, miR-20a and miR-22 are down-regulated in women with endometriosis. Hum Reprod 28(2):322–330
Zhao M et al (2014) miR-20a contributes to endometriosis by regulating NTN4 expression. Mol Biol Rep 41(9):5793–5797
Liu Y et al (2004) Novel role for Netrins in regulating epithelial behavior during lung branching morphogenesis. Curr Biol 14(10):897–905
Hoang S et al (2009) Netrin-4 enhances angiogenesis and neurologic outcome after cerebral ischemia. J Cereb Blood Flow Metab 29(2):385–397
Yang W et al (2017) MiR-424-5p regulates proliferation and apoptosis by targeting FGFR1 in endometriosis cells. Int J Clin Exp Med 10(1):666–674
Turner N, Grose R (2010) Fibroblast growth factor signalling: from development to cancer. Nat Rev Cancer 10(2):116–129
Ma L et al (2019) MicroRNA-142-3p suppresses endometriosis by regulating KLF9-mediated autophagy in vitro and in vivo. RNA Biol 16(12):1733–1748
Pabona JM et al (2012) Krüppel-like factor 9 and progesterone receptor coregulation of decidualizing endometrial stromal cells: implications for the pathogenesis of endometriosis. J Clin Endocrinol Metab 97(3):E376–E392
Simmen RC et al (2004) Subfertility, uterine hypoplasia, and partial progesterone resistance in mice lacking the Krüppel-like factor 9/basic transcription element-binding protein-1 (Bteb1) gene. J Biol Chem 279(28):29286–29294
Zhou M et al (2016) miR-196a overexpression activates the MEK/ERK signal and represses the progesterone receptor and decidualization in eutopic endometrium from women with endometriosis. Hum Reprod 31(11):2598–2608
Karmon AE et al (2014) MicroRNAs in the development and pathobiology of uterine leiomyomata: does evidence support future strategies for clinical intervention? Hum Reprod Update 20(5):670–687
Giuliani E, As-Sanie S, Marsh EE (2020) Epidemiology and management of uterine fibroids. Int J Gynecol Obstet 149(1):3–9
Stewart EA et al (2017) Epidemiology of uterine fibroids: a systematic review. BJOG Int J Obstet Gynaecol 124(10):1501–1512
Wise LA, Laughlin-Tommaso SK (2016) Epidemiology of uterine fibroids–from menarche to menopause. Clin Obstet Gynecol 59(1):2
Ciavattini A et al (2013) Uterine fibroids: pathogenesis and interactions with endometrium and endomyometrial junction. Obstet Gynecol Int 2013:1–11
Wang T et al (2007) A micro-RNA signature associated with race, tumor size, and target gene activity in human uterine leiomyomas. Genes Chromosom Cancer 46(4):336–347
Marsh EE et al (2008) Differential expression of microRNA species in human uterine leiomyoma versus normal myometrium. Fertil Steril 89(6):1771–1776
Zota AR et al (2020) Phthalate exposures and MicroRNA expression in uterine fibroids: the FORGE study. Epigen Insights 13:2516865720904057
Peng Y et al (2008) Antiproliferative effects by Let-7 repression of high-mobility group A2 in uterine leiomyoma. Mol Cancer Res 6(4):663–673
Klemke M et al (2010) Loss of let-7 binding sites resulting from truncations of the 3′ untranslated region of HMGA2 mRNA in uterine leiomyomas. Cancer Genet Cytogenet 196(2):119–123
Fitzgerald JB et al (2012) Role of microRNA-21 and programmed cell death 4 in the pathogenesis of human uterine leiomyomas. Fertil Steril 98(3):726-734.e2
Noetel A et al (2012) microRNA are central players in anti-and profibrotic gene regulation during liver fibrosis. Front Physiol 3:49
Cardozo ER et al (2018) MicroRNA 21a–5p overexpression impacts mediators of extracellular matrix formation in uterine leiomyoma. Reprod Biol Endocrinol 16(1):1–11
Kriegel AJ et al (2012) The miR-29 family: genomics, cell biology, and relevance to renal and cardiovascular injury. Physiol Genomics 44(4):237–244
Marsh EE et al (2016) Decreased expression of microRNA-29 family in leiomyoma contributes to increased major fibrillar collagen production. Fertil Steril 106(3):766–772
Qiang W et al (2014) Down-regulation of miR-29b is essential for pathogenesis of uterine leiomyoma. Endocrinology 155(3):663–669
Xu X et al (2018) Oxidative stress-induced miRNAs modulate AKT signaling and promote cellular senescence in uterine leiomyoma. J Mol Med 96(10):1095–1106
Chuang T-D, Khorram O (2019) Regulation of cell cycle regulatory proteins by microRNAs in uterine leiomyoma. Reprod Sci 26(2):250–258
Ciebiera M et al (2020) The role of miRNA and related pathways in pathophysiology of uterine fibroids—from bench to bedside. Int J Mol Sci 21(8):3016
Mello J et al (2018) MicroRNAs involved in the HMGA2 deregulation and its co-occurrence with MED12 mutation in uterine leiomyoma. MHR Basic Sci Reprod Med 24(11):556–563
Panda H et al (2012) Endometrial miR-200c is altered during transformation into cancerous states and targets the expression of ZEBs, VEGFA, FLT1, IKKβ, KLF9, and FBLN5. Reprod Sci 19(8):786–796
Kim YJ et al (2018) Variation in microRNA expression profile of uterine leiomyoma with endometrial cavity distortion and endometrial cavity non-distortion. Int J Mol Sci 19(9):2524
Wu X et al (2015) Effects of miRNA-197 overexpression on proliferation, apoptosis and migration in levonorgestrel treated uterine leiomyoma cells. Biomed Pharmacother 71:1–6
Ling J et al (2015) Upregulation of miR-197 inhibits cell proliferation by directly targeting IGFBP5 in human uterine leiomyoma cells. In Vitro Cell Dev Biol Animal 51(8):835–842
Chuang T-D, Khorram O (2017) Tranilast inhibits genes functionally involved in cell proliferation, fibrosis, and epigenetic regulation and epigenetically induces miR-29c expression in leiomyoma cells. Reprod Sci 24(9):1253–1263
Momenimovahed Z et al (2019) Ovarian cancer in the world: epidemiology and risk factors. Int J Women’s Health 11:287
Bray F et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424
Falzone L et al (2021) A multidisciplinary approach remains the best strategy to improve and strengthen the management of ovarian cancer. Int J Oncol 59(1):1–14
Dockery LE et al (2017) Tolerance and toxicity of the PARP inhibitor olaparib in older women with epithelial ovarian cancer. Gynecol Oncol 147(3):509–513
Foo T, George A, Banerjee S (2021) PARP inhibitors in ovarian cancer: an overview of the practice-changing trials. Genes Chromosom Cancer 60(5):385–397
Jiang X et al (2019) Current status and future prospects of PARP inhibitor clinical trials in ovarian cancer. Cancer Manag Res 11:4371
Aletti GD et al (2017) Multidisciplinary approach in the management of advanced ovarian cancer patients: a personalized approach. Results from a specialized ovarian cancer unit. Gynecol Oncol 144(3):468–473
Bengrine L et al (2022) Multi-disciplinary care planning of ovarian cancer in older patients: general statement—a position paper from SOFOG-GINECO-FRANCOGYN-SFPO. Cancers 14(5):1295
Koutsaki M, Spandidos DA, Zaravinos A (2014) Epithelial–mesenchymal transition-associated miRNAs in ovarian carcinoma, with highlight on the miR-200 family: Prognostic value and prospective role in ovarian cancer therapeutics. Cancer Lett 351(2):173–181
Korpal M et al (2008) The miR-200 family inhibits epithelial-mesenchymal transition and cancer cell migration by direct targeting of E-cadherin transcriptional repressors ZEB1 and ZEB2. J Biol Chem 283(22):14910–14914
Leskelä S et al (2011) The miR-200 family controls β-tubulin III expression and is associated with paclitaxel-based treatment response and progression-free survival in ovarian cancer patients. Endocr Relat Cancer 18(1):85–95
Person F et al (2017) Prevalence of βIII-tubulin (TUBB3) expression in human normal tissues and cancers. Tumor Biol 39(10):1010428317712166
Ferrandina G et al (2006) Class III β-tubulin overexpression is a marker of poor clinical outcome in advanced ovarian cancer patients. Clin Cancer Res 12(9):2774–2779
Yang N et al (2008) MicroRNA microarray identifies Let-7i as a novel biomarker and therapeutic target in human epithelial ovarian cancer. Can Res 68(24):10307–10314
Johnson SM et al (2005) RAS is regulated by the let-7 microRNA family. Cell 120(5):635–647
Büssing I, Slack FJ, Großhans H (2008) Let-7 microRNAs in development, stem cells and cancer. Trends Mol Med 14(9):400–409
Johnson CD et al (2007) The let-7 microRNA represses cell proliferation pathways in human cells. Can Res 67(16):7713–7722
Schultz J et al (2008) MicroRNA let-7b targets important cell cycle molecules in malignant melanoma cells and interferes with anchorage-independent growth. Cell Res 18(5):549–557
Corney DC et al (2010) Frequent downregulation of miR-34 family in human ovarian cancers. Clin Cancer Res 16(4):1119–1128
Suzuki HI et al (2009) Modulation of microRNA processing by p53. Nature 460(7254):529–533
Peng D-X et al (2012) Prognostic implications of microRNA-100 and its functional roles in human epithelial ovarian cancer. Oncol Rep 27(4):1238–1244
Mabuchi S et al (2009) mTOR is a promising therapeutic target both in cisplatin-sensitive and cisplatin-resistant clear cell carcinoma of the ovary. Clin Cancer Res 15(17):5404–5413
Guo LM et al (2009) MicroRNA-9 inhibits ovarian cancer cell growth through regulation of NF-κB1. FEBS J 276(19):5537–5546
Miles GD et al (2012) Identifying microRNA/mRNA dysregulations in ovarian cancer. BMC Res Notes 5(1):1–10
Wilson CA et al (2005) HER-2 overexpression differentially alters transforming growth factor-β responses in luminal versus mesenchymal human breast cancer cells. Breast Cancer Res 7(6):1–22
Yang H et al (2008) MicroRNA expression profiling in human ovarian cancer: miR-214 induces cell survival and cisplatin resistance by targeting PTEN. Can Res 68(2):425–433
Paul A, Sadek ST, Mahesan AM (2019) The role of microRNAs in human embryo implantation: a review. J Assist Reprod Genet 36(2):179–187
Cuman C et al (2015) Human blastocyst secreted microRNA regulate endometrial epithelial cell adhesion. EBioMedicine 2(10):1528–1535
Kuokkanen S et al (2010) Genomic profiling of microRNAs and messenger RNAs reveals hormonal regulation in microRNA expression in human endometrium. Biol Reprod 82(4):791–801
Kresowik JD et al (2014) MicroRNA-31 is significantly elevated in both human endometrium and serum during the window of implantation: a potential biomarker for optimum receptivity. Biol Reprod 91(1):1–6
Tochigi H et al (2017) Loss of miR-542-3p enhances IGFBP-1 expression in decidualizing human endometrial stromal cells. Sci Rep 7(1):1–10
Kresowik J et al (2012) microRNA 31 is upregulated in serum during the window of implantation. Fertil Steril 98(3):S187
Fang L et al (2022) MiR-17–5P regulates autophagy of ovarian granulosa cells in patients with polycystic ovary syndrome via targeting ATG7.
Zhao W et al (2022) Effects of miRNA-199a-5p on cell proliferation and apoptosis of uterine leiomyoma by targeting MED12. Open Med 17(1):151–159
Ahn SH et al (2021) MicroRNA-139-5p regulates fibrotic potentials via modulation of collagen type 1 and phosphorylated p38 MAPK in uterine leiomyoma. Yonsei Med J 62(8):726
Wuerkenbieke D et al (2015) miRNA-150 downregulation promotes pertuzumab resistance in ovarian cancer cells via AKT activation. Arch Gynecol Obstet 292(5):1109–1116
Lan H et al (2015) miR-140-5p inhibits ovarian cancer growth partially by repression of PDGFRA. Biomed Pharmacother 75:117–122
Chen Z et al (2019) Role of microRNA in the pathogenesis of polycystic ovary syndrome. DNA Cell Biol 38(8):754–762
Echevarría-Vargas IM, Valiyeva F, Vivas-Mejía PE (2014) Upregulation of miR-21 in cisplatin resistant ovarian cancer via JNK-1/c-Jun pathway. PLoS ONE 9(5):e97094
Kiba A et al (2016) Differential micro ribonucleic acid expression profiling in ovarian endometrioma with leuprolide acetate treatment. J Obstet Gynaecol Res 42(12):1734–1743
Lu L et al (2011) MicroRNA let-7a: a potential marker for selection of paclitaxel in ovarian cancer management. Gynecol Oncol 122(2):366–371
Li G et al (2022) Long non-coding RNA placenta-specific protein 2 regulates micorRNA-19a/tumor necrosis factor α to participate in polycystic ovary syndrome. Bioengineered 13(1):856–862
Roth LW et al (2014) Altered microRNA and gene expression in the follicular fluid of women with polycystic ovary syndrome. J Assist Reprod Genet 31:355–362
Pei T et al (2018) miR-194-3p represses the progesterone receptor and decidualization in eutopic endometrium from women with endometriosis. Endocrinology 159(7):2554–2562
Acknowledgements
The authors acknowledge the Deputy for research and technology, Arak University of Medical Sciences, Iran for providing spiritual support to accomplish the present study. This study was approved by the Ethics Committee of Arak University of Medical Sciences with the No: IR.ARAKMU.REC.1402.007.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualization: BK, SA, and MM; Investigation: MB, BK, SA, and MM; Resources: MM, MB, and MA; Writing—original draft preparation: MM and MB; Writing review and editing: MA, MB and MM. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that there are no conflicts of interest.
Ethical approval
No applicable.
Informed consent
Not applicable.
Consent for publications
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bagheri, M., Khansarinejad, B., Mondanizadeh, M. et al. MiRNAs related in signaling pathways of women’s reproductive diseases: an overview. Mol Biol Rep 51, 414 (2024). https://doi.org/10.1007/s11033-024-09357-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11033-024-09357-0