Abstract
Gloriosa superba L., an endangered medicinal plant with global interest due to presence of colchicine, an important alkaloid used in formulations of Indian and Traditional medicine. The plant has become endangered due to its unscientifically exploitation and high medicinal values. In the Present study 10 randomly amplified polymorphic DNA (RAPD) and 6 ISSR markers were employed to assess genetic divergence among micro propagated, wild and field cultivated plants of Gloriosa superba collected from different parts of India. In RAPD analysis, all the 10 accession with 10 RAPD primers amplified 466 fragments, with 96.43 % polymorphism and with an average of 46.6 bands per primer. The size of amplicons varied from 1656 to 100 bp. While, ISSR primers produced 328 fragments of which 298 were polymorphic with an average of 49.7 bands per primer with 91.83% polymorphism. The size of amplicons ranges from 2395 to 181 bp. RAPD, ISSR markers were also assessed by calculating polymorphic information content (PIC) to discriminate the genotypes, Average PIC value for RAPD, ISSR and combined RAPD + ISSR markers obtained was ≤ 0.50 suggesting the informativeness of markers. Jaccard’s coefficient ranges from 0.18 to 0.75 (RAPD) and 0.17 to 0.61 (ISSR) and 0.21–0.52 for pooled ISSR and RAPD markers. The clustering pattern based on UPGMA analysis of the genotypes in the combined analysis revealed that the majority of the genotypes remained similar to the ISSR dendrogram, while the RAPD-based dendrogram showed some variation in the clustering of genotypes. The result of PCA scattered plot obtained were in agreement with the UPGMA dendrogram, which further confirms the genetic relationships explain by cluster analysis. Results confirmed that the genotype studied had good genetic diversity and can be used for identification, conservation, and future breeding program of Gloriosa species and consequently for the benefit of the pharmaceutical industries.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gloriosa superba Linn. An important medicinal plant belongs to the family Liliaceae, commonly known as glory lily, superb lily, tiger lily or tiger claws, is a native of tropical Asia and Africa. Gloriosa superba is commercially important plant with diverse applications in pharmaceutical, cosmetics, medicines, and alternative natural products.
Recently, this plant gains very high medicinal interest due to its medicinal potential and shows various pharmacological properties like anti-inflammatory [1], Antimicrobial [2], Antithrombotic/Anticoagulent potential [3], Enzyme inhibition activities [4] Anticancer activity [5,6,7], Hapatoprotactive activity [8], Antioxidant activity [9] and Anti-ulcerogenic and Anthelmintic Activity [10, 11] etc. due to presence of a highly active alkaloid [12], Colchicine and other important bioactive compound. Colchicine contents of Gloriosa superba were also studied by many scientists from different parts of India [7, 13, 14].
Various important physicochemical properties have been reported from G. superba including carbohydrates, reducing sugars, proteins, amino acids, steroids, flavonoids, terpenoids, saponins, alkaloids, tannins, and phlobatannins [15]. It is also reported that its seed showed the presence of Carbohydrates, Alkaloids, Glycosides, Flavanoids, Steroids, Terpenoids and Phenolics, its tuber showed the presence of Carbohydrates, Alkaloids and Flavanoids; and leaves of Gloriosa superba showed the presence of Carbohydrates, Alkaloids, Flavanoids, Steroids and Terpenoids [16].
Increasing demand in Indian as well as international market due to presence of highly active alkaloid colchicines has created exploitation pressure on natural forests. It has been over exploited by local peoples, such as harvesting from the natural forest, habitat destruction, uncontrolled cattle’s grazing and frequent forest fires during the last decade, this commercially traded plant is becoming extinct due to which it has been declared as endangered plant by IUCN [17]. In wild in Darjeeling Himalaya it became extinct (EW), while in Tamil Nadu, [18], it has given an endangered ‘B’ status.
To meet the ever -increasing demand for export and from the industries, improvement of the breeding and cultivation of plants, lies the challenges for the development of conservation strategies and understanding its genetic assortment to increase number of germplasm [19]. Therefore, it is very important to conserve this plant by in situ and ex situ multiplication in general and micropropogation. Particularly, seeing its profitability in southern districts of Tamil Nadu, farmers are practicing its mass cultivation which is necessary to cope up with the today’s demand [20].
Nag et al. [21] reported that morphological and biochemical parameters cannot give significant diversity among the accessions, as these characters may not be significantly diverse and requires the plant to fully mature and influenced by the environmental factors to be identified by the farmers. Thus, for multiplication and preservation of promising accessions of such plant species, it is essential to determine their genetic diversity.
Significant development of molecular marker approach, have been proven to be powerful and effective tools as compared to the traditional morphological marker system, for measuring genetic variation and patterns of population/species (intra-/inter-species level), assessment and evaluation of genetic diversity among biota, helps breeders to draw appropriate information and methodology for its future genetic improvement, selective breeding, rapid domestication and/or conservation which will allows to prevent the loss of habitat [22]. For efficient conservation and management, the genetic composition of the species in different geographic locations needs to be assessed using different molecular markers [23]. Due to technical simplicity and speed, RAPD and ISSR marker system has been extensively used to distinguish the genetic variation at a molecular level in several medicinal and aromatic plants [24,25,26,27]. Many researchers have studied genetic diversity, and genetic fidelity of Gloriosa superba from diverse geographical origin as well as Micropropagated plants [28,29,30,31].
The genetic molecular markers will have an important role in protecting proprietary germplasm in the future, securing plant variety rights by virtue of its unique efficiency in distinguishing even closely related germplasm of medicnal and aromatic plants [32]. ISSR markers are highly polymorphic, reproducible as compared to other assay procedure [33] and widely used for genetic diversity, evolutionary biology, phylogeny, gene tagging and genome mapping [34,35,36,37,38]. RAPD markers have been used efficiently to study the genetic diversity among different medicinal plant species, such as Asparagus officinalis [39], Azadirachta indica [40] and Aloe vera [41] Ocimum sanctum [42], Cassia tora [43], Murraya Koenigii [44], Pimpinella anisum L. [45], Lathyrus species [46], Clerodendrum species [47], Pouteria lucuma [48].
In the present study, authors assess genetic diversity among the micropropagated , field cultivated and wild genotypes collected from different parts of India, using discriminating power of random amplification of polymorphic DNA (RAPD) markers and inter single sequence repeats (ISSR) markers to provide genetic data, and a theoretical basis for protection of the species.
To the best of author’s knowledge, this is first report where wild genotypes of Gloriosa superba from Pachmarhi Biosphere reserve have been used to analyze its genetic diversity.
Materials and methods
Plant material and sample collection
Ten different wild, tissue culture raised and field cultivated samples of Gloriosa suerba were collected from different geographic locations of India were used in this study (Table 1). Tubers of Wild plant were collected from Pachmarhi Biosphere Reserves, Madhya Pradesh (Tamia region, Pipariya and Bagra Tawa region), Tamilnadu (Sathyamangalam region) and Andhra Pradesh (Sri Penusilanarasimha Nellore region), in vitro tissue cultured raised tubers were collected from Plant tissue culture laboratory, MPCST, Bhopal and cultivated tubers were collected from Andhra Pradesh (Srikakulam region), Chattisgarh (Raipur), Tamilnadu (Karur and Salem region) (Fig. 1 and Table 1). Samples were harvested and placed in sealable polythene bag with appropriate label. The collected tubers were used for DNA extraction, while excess tuber materials were stored in – 80 °C for future use.
Molecular characterization
Isolation of genomic DNA
Total genomic DNA was extracted from tubers using a modified CTAB method based on the protocol of Doyle and Doyle [49] with some modifications [43]. The purified DNA was quantified using a Nano Drop UV–Spectrophotometer (ND-1000) at 260 nm. The ratio of absorbance at 260 to that of 280 nm determines the purity of DNA. DNA’s concentration and purity was also determined by running the samples on 0.8% agarose gel based on the intensities of band when compared with the DNA marker (used to determine the concentration) [50]. Before PCR amplification DNA was diluted to a concentration of 50 ng/µl in TE buffer and stored at 4 °C until use.
RAPD and ISSR PCR amplification
Total of 16 primers (10 RAPD and 6 ISSR) were used to study the genetic diversity among the Gloriosa superba accessions are shown in Table 2 and Fig 4. RAPD profiles were generated using 10 single decamer random oligonucleotide primers (Bangalore Genei, India), while 6 ISSR primers of UBC series were selected from the report of Aghaei et al. [51] (supplementary data is attached for reference). Each amplification reaction mixture of 50 µl contained about 1 µl of genomic template DNA (50 ng), 25 µl of 2X red dye master mix, 2 µl of primer, (Bangalore Genei Pvt. Ltd., India) and 22 µl de-ionized water (RNAse, DNAse free). In negative control’s template DNA was replaced with nuclease free water. The reactions were performed in a Gradient Automatic Thermal Cycler (Eppendrof master cycle gradient, Hamburg, Germany) according to [52] in the following cycles: for RAPD, holding at 94 °C for 5 min, followed by 8 cycles of 94 °C for 45 s, 55 °C for 1 min and 72 °C for 1.5 min and 35 cycles of 94 °C for 45 s, 55 °C for 1 min and 72 °C for 1 min and a final additional extension at 72 °C for 10 min and a holding temperature of 4 °C. PCR reaction mix and program profile for ISSR marker amplification, were similar to RAPD markers analysis, only there was variation in annealing temperature depending on primer sequence (adjusted from 35 to 58 °C for 60 s) [53]. The amplified products were stored at − 20 °C until further process.
Agarose Gel electrophoresis
The amplified PCR products were separated on 1.5% agarose gel (Sigma, USA) stained with ethidium bromide in 1xTAE buffer (pH-8.0) with 1kb ladder (Bangalore Genei) as standard molecular size of fragments and visualized under UV radiation and photographed with alfainnotech gel documentation system (Alpha Innotech). Reactions were repeated thrice to check the Reproducibility of the amplification pattern
Data analysis
In RAPD and ISSR analysis, only reproducible, scorable and clear bands (unit character) were considered and are used for construction of binary matrix (1 for present and 0 for absent) and data analysis was performed using dice coefficients, with PAST (Paleontological statistics) software [54].
The Pair wise genetic similarities coefficient was calculated using Jaccard’s coefficient [55], with PAST (Paleontological statistics) software [54].
The Similarity index (SI) were calculated using the Nei’s genetic similarity index [56], based on the basis of the equation,
where Nij = Number of common bands shared between 2 samples i and j, Ni and Nj = Total number of DNA bands for genotypes i and j, respectively.
Dendrogram was constructed using the unweighted pair group method for arithmetic average (UPGMA) based on Jaccard’s similarity coefficients. All analysis was performed using PAST software version 1.62 [54]. UPGMA and PCA analysis were done to evaluate the reliability of generated dendrograms. The Polymorphic information content (PIC), as a marker discrimination power was computed to determine their application in genetic mapping as per Roldán-Ruiz et al. [57] and Soengas et al. [58]
where fi = Frequency of the amplified allele (band present) (1 − fi) = Frequency of the null allele (band absent).
Relationship among the genotypes was analyzed using Principal component analysis (PCA), which was generated using cluster similarity matrix. A software was used to analyze principal component analysis (PCA) and results were graphically expressed in the form of scattered plots. Amplifications were repeated twice to confirm the results.
Results
Diversity and authentication of medicinal plants was mainly characterized based on morphological and biochemical traits. But now a day the morphological and biochemical diversity is restricted due to changes in the characters at different maturing stages of the plants, and also due to influence of the environmental conditions. Therefore, variety of molecular markers has revolutionized and modernized our ability to characterize genetic variation in medicinal plants, as a complementary strategy upon traditional approaches for management of genetic resources [22]. These molecular tools detect the variation at the DNA level and provide valuable data for genetic diversity.
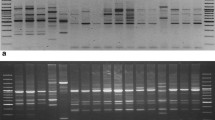
All the 10 accessions of Gloriosa superba used in the present study comprised wild, tissue cultured and field cultivated plant tubers, collected from Madhya Pradesh, Chhattisgarh, Tamilnadu and Andhra Pradesh states of India (Table 1). Molecular profiling of all the 10 accessions were done using RAPD and ISSR markers (Figs. 5, 6).
Dendrogram on the basis of ISSR Paired Group Jaccard similarity coefficient matrix data by unweighted pair group method with average (UPGMA) cluster Analysis of G. superba accessions described in Table 1
Dendrogram based on RAPD+ ISSR combined Paired Group Jaccard similarity coefficient matrix data by unweighted pair group method with average (UPGMA) cluster Analysis of 10 G. superba accessions as described in Table 1
RAPD analysis
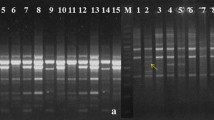
10 RAPD primers (Bangalore Genei) were used to screen 10 G. superba genotypes, of which 9 primers revealed 100% polymorphic banding patterns. A total of 466 amplicons were amplified, out of which 446 were polymorphic, with 96.43 % polymorphism. The number of amplicons varied from 33 (RPI-1) to 64 (RPI-5) with an average of 44.6 amplicons per primer and molecular size of amplicons ranges from 100 bp (RPI-4) to 1656 bp (RPI-5). The extent of polymorphism among all 10 accessions, as revealed by RAPD primers is shown in (Fig. 2a, b). Some primers have also produced accession specific amplicons and monomorphic pattern (Fig. 2).
Jaccard’s co-efficient based on RAPD markers illustrated the mean genetic distance was 0.48. Moreover, the highest genetic distance 0.78 was recorded between GSTIC-05 and GSTIC-09, while the lowest genetic distance 0.18 was obtained between GSTIC-03 and GSTIC-10 (Table 3). Cluster analysis was performed based on similarity co-efficient generated from RAPD profiles, divided the accessions into two major clusters (Fig. 4). Cluster II comprises 8 genotypes, which can be further divided into two sub clusters, namely IIa and IIb, where IIb again divided in two sub clusters IIb1 and IIb2. The group I and IIa and IIb1 comprise a single genotype each namely GSTIC-01, GSTIC-03 and GSTIC-08 respectively. GSTIC-10 is out grouped (Fig. 4). Sub Cluster IIb2 comprises 6 genotypes namely GSTIC-02, GSTIC-04, GSTIC-07, GSTIC-6, GSTIC-5, GSTIC-9 among these GSTIC-2, GSTIC-04 and GSTIC-05, GSTIC-09 respectively are very close with each other. The result of principal component analysis was comparable to the UPGMA cluster analysis with minor deviations (Figs. 4, 7).
The average PIC (Polymorphism Information Content) value for RAPD was 0.344. Highest PIC value obtained was 0.42 for RPI-7 marker and the lowest PIC value was 0.258 for RPI-10 (Table 2). All the RAPD markers showed PIC value ≤ 0.50 suggesting the informativeness of a marker.
ISSR analysis
ISSR primers produced different numbers of amplicons, depending upon their simple sequence repeat motifs. Among 10 ISSR primers screened, 6 primers, with reproducible polymorphic banding patterns were selected for genetic diversity study (Fig. 2c, d and Table 4). Six primers produced a total of 328 amplicons, and out of which 298 amplicons were polymorphic, with an average of 91.83% polymorphism. 100% polymorphism was generated by 4 ISSR primers, while remaining primers exhibited variable percentage of polymorphism. Molecular size of amplicons ranges from 181 (UBC 835) bp to 2395 (UBC 835) bp and total amplicons per primer varied between 40 (UBC 18) to 70 bp (UBC 834), with an average of 54.6 amplicons per primer, while average number of polymorphic amplicons per primer was 49.66 (Table 4). The extent of polymorphism among G. superba genotypes as revealed by ISSR primer is shown in Figs. 2c, d, 3 and Table 4. Genetic distance was obtained with UPGMA analysis using Jaccard’s similarity coefficient ranged from 0.17 between GSTIC-1 and GSTIC-2 to 0.61 between GSTIC-7 and GSTIC-8 (Table 5).
The dendrogram based on ISSR data separated the genotypes into two major clusters. Cluster I comprised of two genotypes GSTIC-1 and GSTIC-2 with similarity coefficient equal to 0.54. Cluster II comprised of two sub clusters IIa and IIb, where IIa comprised of 4 genotypes as GSTIC-6, GSTIC-7, GSTIC-8, GSTIC-9 respectively and IIb comprised of GSTIC-3, GSTIC-4, GSTIC-5 and GSTIC-10 is out grouped (Fig. 5). The result of PCA was comparable to cluster analysis based on UPGMA with minor deviations (Figs. 5, 7). The PIC value ranged from 0.273 (UBC 815) to 0.381 (UBC 835) with mean of 0.341. All the six ISSR primers have shown PIC value ≤ 0.50 suggesting the informativeness of a marker. Molecular markers specific to some accessions were shown in Fig. 3. These markers could be converted to SCAR primers for enhancing the reliability of the markers germplasm identification.
Combined RAPD and ISSR analysis
Combined data of both RAPD and ISSR primers were used to generate an overview of genetic similarities between the genotypes. Data from Both combined RAPD and ISSR primers produced a total of 794 amplicons, of which 744 amplicons were polymorphic and 50 amplicons were monomorphic with 94.12% polymorphism (Table 6). Molecular size of amplicons ranges from 100 (RPI-04) bp to 2395 (UBC-835) bp, with average no. of amplicons 50.66 per primer, while average number of polymorphic amplicons per primer was 47.13 (Table 6).
The Jaccard's similarity coefficient obtained from the pooled data of RAPD and ISSR showed a wide range of correlations among all the genotypes. Maximum similarity coefficient (0.208) was observed between GSTIC-03 and GSTIC-10. However, GSTIC-03 and GSTIC-04 were found distantly related with a similarity coefficient value of 0.521, with an average similarity coefficient 0.36 (Table 7). Pooled RAPD and ISSR data was used for constructing the dendrogram by using Jaccard's similarity coefficient. Dendrogram generated using this combined data separated the accessions into two major clusters, with GSTIC-10 as an out group. Cluster I comprised of two genotypes, namely, GSTIC-01 and GSTIC-02, while cluster II was the largest cluster consisting of seven genotypes, which is further divided into two IIa and IIb sub clusters. Cluster IIb comprised of two genotypes namely GSTIC-03 and GSTIC-04, while Cluster IIa again divided into IIa1 and IIa2, where IIa1 comprised of 3 genotypes GSTIC-07, GSTIC-08 and GSTIC-09 while IIa2 comprised of 2 genotypes namely GSTIC-05 and GSTIC-06 with similarity coefficient of 0.40 (Fig. 6). Result of the PCA based on similarity coefficient of combined RAPD+ISSR markers was comparable with its cluster analysis. The clustering pattern of the genotypes in the combined analysis revealed that the majority of the genotypes remained similar to the ISSR dendrogram, while the RAPD-based dendrogram showed some variation in the clustering of genotypes (Figs. 4, 5, 6, 7). PCA plot is a reflection of broad genetic base of this species. A PCA result further confirms the genetic relationships, based on the dendrogram produced and delineate by cluster analysis.
The PIC value ranged from 0.258 (RPI-10) to 0.420 (RPI-7), with mean PIC value of 0.343. All the sixteen combined RAPD+ ISSR primers have shown PIC value ≤ 0.50 suggesting the informativeness of a marker (Table 6). In this study, it was observed that genetic relation/ variability among the genotypes were found more or less similar to RAPD and ISSR analysis.
Principal component analysis
To overview the genetic variation among 10 genotypes of G. superba, PCA was performed with 10 RAPD and 6 ISSR markers. Cluster analysis result of RAPD markers was comparable with Principal Component Analysis based on its similarity coefficient (Figs. 4, 7). Only a few differences were observed with UPGMA clustering and principal component analysis clustering. The PCA based on RAPD data revealed that genotypes belonging to a particular cluster were grouped together in the PCA plot with minor differences (Fig. 7a). It is fascinatingly to observe that the genotypes belong to same region were clustered together in PCA. Similarly, PCA analysis based on ISSR data revealed that the majority of genotypes were grouped as illustrated by the UPGMA dendrogram (Fig. 7b). RAPD + ISSR marker tools based PCA analysis revealed that genotypes was impartially distributed on PCA plots, which imitate a good genetic relatedness among the G. superba genotypes (Fig. 7c). PCA’s method help in converting a set of variables into few dimensions, by which genotypes can be depicted in two or three-dimensional space. Such a graphical representation help in identifying the individuals that tend to cluster together.
Discussion
To facilitate the efficient conservation, management, and utilization of the species of medicinal plants, it is important to study their genetic diversity, for selection of useful and effective genotypes for the development of cultivars by the farmers and breeders. Genetic markers plays important role in plant improvement programs such as genetic mapping, varietal and parentage identification, and evaluation of polymorphic genetic loci affecting quantitative economic traits. Although many researchers attempted extensive work on Biochemical and morphological characterization of G. superba, from different geographic locations [15, 16], but for effective identification and cultivar development there is an urgent need to study its genetic diversity using different molecular markers. Several non-receptive properties especially, environmental, epistatic and pleiotropic effects, make these genetic markers highly advantageous and useful over traditional Biochemical, morphological and phenotypic markers [59]. It is, therefore, in the present study efforts have been made to characterize 10 different accessions (wild, cultivated and microprogated) of G. superba collected from different parts of India, using random amplified polymorphic DNA (RAPD) and Inter-simple sequence repeats (ISSR Markers). To the best of authors knowledge G. superba accessions from Pachmarhi Biosphere reserve of Madhya Pradesh is first time included in the genetic diversity studies. RAPD and ISSR markers are highly polymorphic and very useful tool for characterization of germplasm, Phylogenetic studies, clonal fidelity testing, genetic relationship, assessment of genetic diversity, varietal identification, hybrid testing, gene tagging and marker-assisted selection, systemic and evolutionary biology, with easy application and results interpretation [60].
In present study RAPD and ISSR markers were used to assess molecular diversity among 10 geographically and technically different genotypes of G. superba, an endangered plant from different parts of India. 10 RAPD primers amplified 466 amplicons, out of which 446 were polymorphic, with 96.43 % polymorphism. Molecular size of amplicons ranges from 100 bp (RPI-4) to 1656 bp (RPI-5) with an average of 44.6 amplicons per primer. Some primers have also produced accession specific amplicons and monomorphic pattern (Fig. 2a, b). The average percentage of polymorphism is closely related with earlier studies as 87.53% in Gloriosa superba from Tamiladu accession [28], 96% in Angelica, genus from China [61], 100% in Cassia tora genotypes from different parts of Madhya Pradesh [43], 73.33% of polymorphism in 14 closely related accessions of Costus speciosus [62], 92.26 % in C. borivilianum [63]. Percent polymorphism is higher than studied in other endangered plants as 49.61% in Oroxylum indicum [64], 27.6% in Paeonia rockii [65], 24.5% Lactoris fernandeziana (Lactoridaceae) [66], Lycium varities [67], which indicates that these wild, cultivated and Micropropagated genotypes of G. superba possess high genetic distance from each other. The average genetic distance for Jaccard’s co-efficients was 0.48 and PIC value was 0.344, which is ≤ 0.50 suggesting the informativeness of a marker.
Similarly, six ISSR primers produced a total of 328 amplicons (Table 4), and out of which 298 amplicons were polymorphic, with an average of 91.83% polymorphism. 100% polymorphism was generated by 4 ISSR primers, while remaining primers exhibited variable percentage of polymorphism. Molecular size of amplicons ranges from 181 (UBC 835) bp to 2395 (UBC 835) bp and total amplicons per primer varied between 40 (UBC 181) and 70 bp (UBC 834), with an average of 54.3 amplicons per primer, while average number of polymorphic amplicons per primer was 49.66 (Table 4). The average percentage of polymorphism is closely related with earlier studies as 93% in gladiolus collection [68], 99% in indigenous and exotic genotypes of Gladiolus hybrids [69], 82.76 in C. borivilianum [63], 65.05% in C. colocynthis [70], Acacia species [71]. Moreover, low-level polymorphism in ISSR markers was also reported in many previous studies, for examples, 24.36% in Bruguiera gymnorrhiza and 12.73% in Heritiera fomes [72]. However, it was noted in the present study that high reproducibility for ISSR markers was achieved due to well—defined PCR conditions.
In this study, the average similarity index for G. superba genotypes using RAPD ranged from 0.18 to 0.78 and ISSRs ranged from 0.17 to 0.61, While similarity index of the combined data RAPD+ISSR ranged from 0.208 to 0.521 (Tables 3, 5, 7). Low range of similarity index from data analyzed clearly indicated there was high genetic variability between genotypes collected from different geographical regions using different techniques (wild, cultivated and Micropropagated). When similarity index data from RAPD, ISSR and combined RAPD+ISSR were compared with each other showed high polymorphisms and large variability, and distinguishing genotypes clearly. High polymorphism demonstrates that the level of genetic variation in the species is substantial and indicated that genetic base is quite broad. From previous observations it was also found that the RAPD markers are more effective than ISSR markers for differentiation of closely related cultivars in various plant species [73, 74]. This divergence may be due to the proportion of coding and non-coding sequences within the genome and differences in genome composition of species and are selectively neutral.
In the UPGMA-based dendrograms obtained from the combined RAPD and ISSR data, the wild genotypes from Bagra Tawa region of Pachmarhi Biosphere reserve, Madhya Pradesh (GSTIC-10) out grouped and shows broad genetic diversity with other genotypes indicates an earlier introduction of these species to Central India, and subsequently leading to accumulation of variation. Genotypes collected from Pipariya region of Pachmarhi biosphere reserve Madhya Pradesh, wild (GSTIC-01) and tissue culture raised plants (GSTIC-02) with very low genetic diversity coefficient value of 0.510 and grouped into similar clustered, which also signifies our result as the plant tissue culture was from the same region pipariya and originated from Bagra Tawa. Same kind of results was observed from the Genotypes collected from Tamia region of Pachmarhi biosphere reserve Madhya Pradesh, wild (GSTIC-03) and cultivated genotype from Karur region of Tamilnadu (GSTIC-04) and cluster together with the highest genetic similarity coefficient value of 0.521, indicated that the cultivated genotype was travelled from the Central India to south. Wild genotype from Sathyamangalam region of Tamilnadu (GSTIC-07), wild genotype from Sri Penusilanarasimha Nellore region, Andhra Pradesh (GSTIC-08) and cultivated genotype from Srikakulam region of Andhra Pradesh (GSTIC-09) were genetically very close but genetically different from other accessions and were clustered together. Cultivated genotypes from Raipur, Chhattisgarh (GSTIC- 04) and Salem region of Tamilnadu (GSTIC- 05) with genetic similarity coefficient value of 0.491, were clustered together. Similar results were also reported in many plant species such Bacopa monnieri (L.) [75], wormwood capillary [76], Lycium varieties [67], Pimpinella anisum L. [45]. Preethi et al. [77] studied the genetic diversity of Cocoa nucifera L. using EST-SSR markers, which is also a useful and reliable tools for the assessment of genetic diversity and marker-assisted selection which strongly indicates high polymorphism as compared to ISSR and RAPD. The PCA analysis was performed to demonstrate the multiple dimension distribution of the genotypes in a scatter-plot, which show some difference in the separation of individual genotypes at their respective clusters when compared between UPGMA dendrogram and PCA for both the markers. According to Taran et al. [78], this multivariate approach will complement the information obtained from the cluster analysis as it is more informative to study genetic distance among major groups. The results based on cluster analysis were correlated some extent to the geographical distribution, as revealed in the present results. These results are in agreement with some previous study, which concluded that accessions from diverse geographical regions can be genetically similar.
In the Present study we have found that RAPD markers were somewhat significant than those of the ISSR markers, but ISSR was more favorable than the RAPD techniques in terms of presenting the phylogenetic relationships in many germplasms. However, when both the markers were combined, it resulted in significant high genetic diversity among the genotypes of G. superba (wild, cultivated and micropropagated) from different parts of India. Likewise, genetic relationship of many cultivars and plant species was studied such as Dioscorea spp. [79], Cymbopogon flexuosus [80], and sandalwood [74] using these markers. Thus, molecular marker approach used were very important and informative for genetic variation, genetic pool and to develop conservation strategies and breeding among medicinal plants and cultivars.
Conclusion
In the present study the genotypes of G. superba (Wild, Cultivated, and Micropropagated) were collected from different geographic regions of India and were analyzed for marker polymorphisms, genetic diversity using RAPD and ISSR markers. There are many other molecular markers such as inter primer binding site (iPBS) and start codon targeted (SCoT) and Sequence related amplified polymorphisms (SRAP) molecular markers etc which reveals the genetic relationships in molecular level and provides a theoretical basis for genetic improvement and conservation. But RAPD and ISSR molecular markers, proved to be a powerful tool for the generation of potential diagnostic of markers for cultivar analysis. High genetic variation observed among all the 10 genotypes based on dendrogram constructed using UPGMA and scattered plot obtained from PCA analysis. Some RAPD and ISSR markers have also produced species specific amplicons which can be used for the development of co-dominant SCAR markers. Genetic molecular markers constitute useful and reliable tools to understand the genetic pool and to develop conservation strategies and breeding programs.
Abbreviations
- RAPD:
-
Random amplified polymorphic DNA
- ISSR:
-
Inter simple sequence repeats
- RFLP:
-
Restriction fragment length polymorphism
- AFLP:
-
Amplified fragment length polymorphism
- PIC:
-
Polymorphic information content
- DNA:
-
Deoxyribonucleic acid
- PCR:
-
Polymerase chain reaction
- UPGMA:
-
Unweighted pair group method with the arithmetic averaging algorithm
- PCA:
-
Principal component analysis
References
Jomy J, Jennifer F, Nandgude Tanaji D, Niphade S, Salva A, Deshmukh PT (2009) Analgesic and anti-inflammatory activities of the hydroalcoholic extract from Gloriosa superba Linn. Int J Green Phar. https://doi.org/10.4103/0973-8258.56277
Hemaiswarya S, Raja R, Anbazhagan C, Venkatesan V (2009) Antimicrobial and mutagenic properties of the root tubers of Gloriosa superba Linn (Kalihari). Pakistan J Bot 41(1):293–299
Kee NLA, Mnonopi N, Davids H, Naude RJ, Frost CL (2008) Antithrombotic/anticoagulant and anticancer activities of selected medicinal plants from South Africa. African J Biotechnol 7(3):217–223
Haroon K, Murad AK, Iqbal H (2008) Enzyme inhibition activities of the extracts from rhizomes of Gloriosa superba Linn (Colchicaceae). J Enzyme Inhibit Med Chem 22(6):722–725
Reuter S, Prasad S, Phromnoi K, Ravindran J, Sung B, Yadav RV, Kannappan R (2010) Rhizomes of Gloriosa superba Linn (Colchicaceae). J Enzyme Inhibit Med Chem 22(6):722–725
Rokade SS, Joshi KA, Mahajan K, Patil S, Tomar G, Dubal DS, Parihar VS, Kitture R, Bellare JR, Ghosh S (2018) Gloriosa superba mediated synthesis of platinum and palladium nanoparticles for induction of apoptosis in breast cancer. Bioinorg Chem Appl. https://doi.org/10.1155/2018/4924186
Balkrishna A, Das SK, Pokhrel S, Joshi A, Laxmi VS, Sharma VK, Sharma V, Sharma N, Joshi CS (2019) Colchicine: isolation, LC–MS QTof screening, and anticancer activity study of Gloriosa superba seeds. Molecules. https://doi.org/10.3390/molecules24152772
Mohandass S, Indhumathi T (2011) Hepatoprotective efficacy of Gloriosa superba Linn. against 25. paracetamol treated experimental rats-An in vivo study. Golden Res Thoughts 1(4):1–4
Jothi U, Jebamalar JA, Sivakumar T (2019) Study on estimation and antioxidant activity of gloriosa superba l whole plant extract. Int J Sci Res Biol Sci 6(3):50–55
Pawar BM, Wavhal VP, Pawar ND, Agarwal MR, Shinde PB, Kamble HV (2010) Anthelmintic activity of Gloriosa superba Linn (Liliaceae). Int J PharmTech Res 205:1483–1487
Riaz MNA, Chennamaneni SR, Challa SR (2013) Anti-ulcerogenic evaluation of Gloriosa Superba tuber extracts in-vivo and anthelmintic activity in-vitro: a comparison. J Med Health Sci 24:73–79
Nautiyal OP (2011) Isolation of 3-demethylcolchicine from Gloriosa superba sludge and coupling with α- acetobromoglucose to yield colchicoside and thiocolchicoside. J Nat Prod 4:87–93
Kuo MC, Chang SJ, Hsieh MC (2015) Colchicine Significantly Reduces Incident Cancer in Gout Male Patients A 12-Year Cohort Study. Medicine 94:1–6
Sharma S, Sharma YP, Thakur P (2017) Quantification of colchicine in different parts of Gloriosa superba L. IJCS 53:147–149
Sivakumar T, Gajalakshmi D (2019) Phyto chemical analysis and evaluation of antimicrobial activity in the whole plant extract of Gloriosa superba. Asian J Pharmac Clin Res 126:245–249. https://doi.org/10.22159/ajpcr.2019.v12i6.33059
Muthukrishnan SD, Subramaniyan A (2012) Phytochemical constituents of Gloriosa superba seed, tuber and leaves. Res J Pharmac Biol Chem Sci 33:111–117
Pattanaik C, Reddy CS, Reddy KN (2009) Ethno-medicinal survey of threatened plants in Eastern Ghats. India Our Nature 7:122–128
Mishra SB, Dwivedi S, Shashi A, Prajapati K (2008) Ethnomedicinal uses of some plant species by ethnic and rural peoples of the salem district of tamil nadu with special reference to the conservation of vanishing species. Ethnobot Leaflets 12:873–87
Mahajan R (2015) Gloriosa superba L.: an Endangered Medicinal Plant. Hort Flora Res Spectrum 42:168–171
Ranjith KR, Rohini A (2019) Economic analysis of production and marketing of Gloriosa superba L Tamil Nadu. IJCS 7(3):4275–4278
Nag A, Ahuja PS, Sharma RK (2015) Genetic diversity of high-elevation populations of an endangered medicinal plant. AoB PLANTS. https://doi.org/10.1093/aobpla/plu076
Grover A, Sharma PC (2016) Development and use of molecular markers: past and present. Crit Rev Biotechnol 362:290–302
Petit RJ, Hampe A (2006) Some evolutionary consequences of being a tree. Annu Rev Ecol Evol Syst 37:187–214. https://doi.org/10.1146/annurev.ecolsys.37.091305.110215
Prajapat P, Sasidharan N, Ballani A (2015) Assessment of genetic diversity in four brassica species using randomly amplified polymorphic DNA markers. Int J Agric Environ Biotechnol 84:831–836
Bhattacharyya P, Kumaria S (2015) Molecular characterization of Dendrobium nobile Lindl., an endangered medicinal orchid, based on randomly amplified polymorphic DNA. Plant Syst Evol 3011:201–210
Salazar-Laureles ME, Pérez López DDJ, González- Huerta A, Vázquez-García LM, Valadez-Moctezuma E (2015) Genetic variability analysis of faba bean accessions using Inter-simple sequence repeat (ISSR) markers. Chilean J Agric Res. https://doi.org/10.4067/S0718-58392015000100017
Asfaw BM, Dagne K, Keneni G, Kemal S, Kassahu T (2018) Genetic diversity study of Ethiopian Faba bean (Vicia faba L.) varieties based on phenotypic traits and inter simple sequence repeat (ISSR) markers. African J Biotechnol. https://doi.org/10.5897/AJB2017.16331
Rajagopal C, Kandhasamy R (2013) Assessment of genetic diversity of Gloriosa superba L. accessions detected by random amplified polymorphic DNA analysis. J Med Plants Res 7(28):2122–2127. https://doi.org/10.5897/JMPR12.805
Yadav K, Aggarwal A, Singh N (2013) Evaluation of genetic fidelity among micropropagated plants of Gloriosa superba L using DNA-based markers— a potential medicinal plant. Fitoterapia 89:265–270. https://doi.org/10.1016/j.fitote.2013.06.009
Paul Jasmine JA, Balakrishnan V (2018) Intra S pecific Analysis of Gloriosa superba (L.) through ISSR finger printing and DNA sequencing of ecotypes collected from different accessions of Tamil Nadu State, India. Res Plant Biol 8:17–21. https://doi.org/10.25081/ripb.2018.v8.3600
Sahana KS, Gnanam R, Rajesh S, Rajamani K (2019) Evaluation of genetic diversity in Gloriosa superba L, an endangered medicinal plant using molecular marker. Int J Curr Microbiol App Sci 86:2125–2134
Patel HK, Fougat RS, Kumar S, Mistry JG, Kumar M (2015) Detection of genetic variation in Ocimum species using RAPD and ISSR markers. 3 Biotech 5(5):697–707. https://doi.org/10.1007/s13205-014-0269-y
Gupta M, Chyi Y-S, Romero-Severson J, Owen JL (1994) Amplification of DNA markers from evolutionarily diverse gen-omes using single primers of simple-sequence repeats. TheorAppl Genet 89:998–1006
Godwin ID, Aitken EA, Smith LW (1997) Application of inter simple sequence repeat ISSR markers to plant genetics. Electrophoresis 189:1524–1528
Virk PS, Zhu J, John Newbury H, Bryan G, Jackson MT, Ford-Lloyd BV (2012) Effectiveness of different classes of molecular marker for classifying and revealing variation in rice (Oryza sativa) germplasm. Euphytica 112(3):275–284. https://doi.org/10.1023/A:1003952720758
Tiwari JK, Chandel P, Gupta S, Gopal J, Singh BP, Bhardwaj V (2013) Analysis of genetic stability of in vitro propagated potato microtubers using DNA markers. Physiol Mol Biol Plants: Int J Funct Plant Biol 194:587–595. https://doi.org/10.1007/s12298-013-0190-6
Badar Z et al (2017) Analysis of genetic fidelity of wild type and in vitro regenerated aloe vera plants through RAPD and ISSR molecular markers. Int J Biotech Bioeng 3(8):259–267
Khatik N, Joshi R (2020) Assessment of genetic fidelity in microclones of curry leaf plants [Murraya koenigii L. Spreng.] using ISSR markers. Indian J Exp Biol 584:286–291
Shasany AK, Darokar MP, Sakia D, Rajkumar S, Sundaresan V, Khanuja SPS (2003) Genetic diversity and species relationship in Asparagus spp. using RAPD analysis. J Med Arom Plant Sci 25:698–704
Singh R and Randhawa G (2008) Comparative assessment of genetic diversity in Indian and exotic neem (Azadirachta indica) using AFLP markers Indian J Agricul Sci 78(10)
Tripathi N, Saini N, Tiwari S (2011) Assessment of genetic diversity among Aloe vera accessions using amplified fragment length polymorphism. Int J Med Arom Plants 1:115–121
Tilwari A, Tamrakar K, Sharma R (2013) Use of random amplified polymorphic DNA (RAPD) for assessing genetic diversity of Ocimum sanctum (Krishna Tulsi) from different environments of Central India. J Med Plant Res. https://doi.org/10.5897/JMPR12.429
Tilwari A, Chauhan D, Sharma R, Singh RK (2016) Assessment of genetic variations among medicinal plant cassia tora from different geographic regions of central india using RAPD markers medicinal and aromatic plants. Med Aromat Plants (Los Angel) 5:6. https://doi.org/10.4172/2167-0412.1000276
Sharma D, Khatik N, Joshi R (2019) Evaluation of genetic stability of Micropropagated plnts of Murraya Koenigii L. Spreng. Using RAPD Markers. Asian J Microbiol Biotechnol Environ Sci 211:146–151
Akçali Giachino RR (2020) Investigation of the genetic variation of anise Pimpinella anisum L. using RAPD and ISSR markers. Genet Resour Crop Evol 67:763–780. https://doi.org/10.1007/s10722-019-00861-y
Samira A, Osman, Honda BM, Ali (2020) Genetic diversity of five lathyrus species using rapd, issr and scot markers. Asian J Plant Sci 19:152–165
Gogoi B, Wann SB, Saikia SP (2020) Comparative assessment of ISSR, RAPD, and SCoT markers for genetic diversity in Clerodendrum species of North East India. Mol Biol Rep 47:7365–7377. https://doi.org/10.1007/s11033-020-05792-x
Arlos HQJ, Zulita APL (2020) Genetic variability in Pouteria lucuma using ISSR markers. Manglar 17(1):7–12. https://doi.org/10.17268/manglar.2020.002
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus. 12:13–15
Sambrook, Russell DW (2001) Molecular cloning: a Laboratory manual. Cold spring Harbour Laboratoey Press, Cold spring harbor, NY, USA
Aghaei M, Darvishzadeh R, Hassani A (2012) Molecular characterization and similarity relationships among Iranian basil (O basilicum) accessions using inter simple sequence repeat markers. Artig Cient 43(2):312–320
Sambrook J, Fritschi EF, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, New York
Mohamad A, Alhasnawi AN, Kadhimi AA, Isahak A et al (2017) DNA Isolation and Optimization of ISSR-PCR Reaction System in Oryza sativa L. Int J. Adv Sci Eng Inf Tech 7(6) ISSN: 2088-5334
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontologia Electronica 4(1):9
Jaccard P (1908) Nouvelles recherches sur la distribution florale. Bull. Soc. Vaud. Sci. Nat. 44:223–270
Nei M, Li WH (1979) Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc Natl Acad Sci 76(10):5269–5273. https://doi.org/10.1073/pnas.76.10.5269
Ruiz R, Gilliland TJ, Calsyn E, Gillilad TJ, Coll R, Van Eijk MJT, De Loose M (2000) Estimating genetic conformity between related ryegrass (Lolium) varieties II. AFLP characterization. Mol Breed 6:593–602
Soengas P, Velasco P, Padilla G, Ordas A, Cartea ME (2006) Genetic relationships among brassica napus crops based on SSR marker. Am Soc Horticul Sci 41(5):1195–1199. https://doi.org/10.21273/HORTSCI.41.5.1195
Mondini L, Noorani A, Pagnotta MA (2009) Assessing plant genetic diversity by molecular tools. Diversity. 1:19–35. https://doi.org/10.3390/d1010019
Nagaraju J, Reddy K, Nagaraja G et al (2001) Comparison of multilocus RFLPs and PCR-based marker systems for genetic analysis of the silkworm, Bombyx mori. Heredity 86:588–597. https://doi.org/10.1046/j.1365-2540.2001.00861.x
Zhiqiang M, Zhang C, Khan MA et al (2015) Efficiency of improved RAPD and ISSR markers in assessing genetic diversity and relationships in Angelica sinensis Oliv. Diels varieties of China. Electron J Biotechnol 18(2):96–102. https://doi.org/10.1016/j.ejbt.2014.12.006
Mandal AB, Thomas VA, Elanchezhian R (2007) RAPD pattern of Costus speciosus Koen ex. Retz., an important medicinal plant from the Andaman and Nicobar Islands. Curr Sci 933:369–373
Samantaray S, Ngangkham U (2017) Evaluation of genetic diversity in Chlorophytum borivilianum (Santp. and Fernan.) using molecular markers; an endangered medicinal plant. Active ingredients from Aromat Med Plants 50-70
Jayaram K, Prasad MNV (2008) Genetic diversity in Oroxylum indicum L. Vent. Bignoniaceae, a vulnerable medicinal plant by random amplified polymorphic DNA marker. Afr J Biotech 7:254–262
Pei YL, Zou YP, Yin Z, Wang XQ, Zhang ZX, Hong DY (1995) Preliminary report of RAPD analysis in Paeonia suffruticosa subsp. Spontanea and P. rockii. Acta Phys Sin 33:350–356
Brauner S, Crawford DJ, Stuessy TF (1992) Ribosomal DNA and RAPD variation in the rare plant family Lactoridaceae. Am J Bot 79:1436–1439
Xiaoyan Liu DuJ, Khan MA, Cheng J, Wei C et al (2020) Analysis of genetic diversity and similarities between different Lycium varieties based on ISSR analysis and RAMP-PCR markers. World Acad Sci J. https://doi.org/10.3892/wasj.2020.39
Jingang W, Ying G, Daidi C, Shenkui L, Chuanpin Y (2008) ISSR analysis of 26 general species of Gladiolus hybridus Hort. J Northeast Agric Univ 8(154):6–10
Chaudhary V, Kumar M, Sharma S et al (2018) (2018) Assessment of genetic diversity and population structure in gladiolus (Gladiolus hybridus Hort) by ISSR markers. Physiol Mol Biol Plants 24:493–501. https://doi.org/10.1007/s12298-018-0519-2
Verma KS, Haq SU, Kachhwaha S, Kothari SL (2017) RAPD and ISSR marker assessment of genetic diversity in Citrullus colocynthis L. Schrad: a unique source of germplasm highly adapted to drought and high temperature stress. 3 Biotech 75:288. https://doi.org/10.1007/s13205-017-0918-z
Nader RA, Hayssam MA (2020) Mohamed ZMS and Hosam EEW (Quantitative and Qualitative Genetic Studies of Some Acacia Species Grown in Egypt. Plants 9(2):243. https://doi.org/10.3390/plants9020243
Dasgupta N, Nandy P, Sengupta C, Das S (2015) RAPD and ISSR marker mediated genetic polymorphism of two mangroves Bruguiera gymnorrhiza and Heritiera fomes from Indian Sundarbans in relation to their sustainability. Physiol Mol Biol Plants 21(3):375–384. https://doi.org/10.1007/s12298-015-0308-0
Farajpour M, Ebrahimi M, Amiri R et al (2011) Study of genetic variation in yarrow using inter-simple sequence repeat (ISSR) and random amplified polymorphic DNA (RAPD) markers. Afr J Biotechnol 10(54):11137–11141
Patel DM, Fougat RS, Sakure AA, Kumar S, Kumar M, Mistry JG (2016) Detection of genetic variation in sandalwood using various DNA markers. 3 Biotech 61:55.
Tripathi N, Chouhan DS, Saini N, Tiwari S (2012) Assessment of genetic variations among highly endangered medicinal plant Bacopa monnieri L. from Central India using RAPD and ISSR analysis. 3 Biotech 24:327–336. https://doi.org/10.1007/s13205-012-0059-3
Shafie MS, Hasan SM, Zain AM, Shah RM (2011) RAPD and ISSR markers for comparative analysis of genetic diversity in wormwood capillary Artemisia capillaris from Negeri Sembilan, Malaysia. J Med Plants Res 518:4426–4451
Preethi P, Rahman S, Naganeeswaran S et al (2020) Development of EST-SSR markers for genetic diversity analysis in coconut (Cocos nucifera L.). Mol Biol Rep. https://doi.org/10.1007/s11033-020-05981-8
Taran B, Zhang C, Warkentin T, Tullu A, Vanderberg A (2005) Genetic diversity among varieties and wild species accessions of pea Pisum sativum L. based on molecular markers, and morphological and physiological characters. Genome 48:257–272. https://doi.org/10.1139/g04-114
Velasco-Ramirez AP, Torres-Moran MI, Molina-Moret S et al (2014) Efficiency of RAPD, ISSR, AFLP and ISTR markers for the detection of polymorphisms and genetic relationships in camote de cerro Dioscorea spp. Electron J Biotechnol 172:65–71. https://doi.org/10.1016/j.ejbt.2014.01.002
Debajit S, Sukriti D, Sneha G, Mohan L et al (2015) RAPD and ISSR based Intra-specific molecular genetic diversity analysis of Cymbopogon flexuosus L. Stapf with a distinct correlation of morpho-chemical observations. Res J Biotechnol 107:105–113
Acknowledgements
The authors are sincerely thankful to the Director General, Madhya Pradesh Council of Science and Technology, Vigyan Bhawan, Nehru Nagar, for providing all the necessary facilities to conduct the research. We also thank Dr SS Asthana for editing the Manuscript. We also thank our reviewers for improving the earlier draft by their constructive comments to finalize the manuscript.
Author information
Authors and Affiliations
Contributions
Laboratory experiment work was performed by AT with the help of laboratory staff. AT has analyzed the data, written and critically reviewed the manuscript. Collection of samples was done by AT with the help of AK from Different parts of India. Facilities in the lab are provided by RS.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tilwari, A., Sharma, R. Random amplified polymorphic DNA and inter simple sequence repeat markers reveals genetic diversity between micro propagated, wild and field cultivated genotypes of Gloriosa superba: an endangered medicinal plant. Mol Biol Rep 48, 2437–2452 (2021). https://doi.org/10.1007/s11033-021-06278-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06278-0