Abstract
Colorectal cancer is the third most common malignancy in the world having a high mortality rate. Flavonoids possess many biological activities including anti-cancer activity. lawsonaringenin (LSG) is a flavonoid isolated from leaves of Lawsonia alba Lam. The objective of this study was to demonstrate the anti-cancer potential of LSG in colorectal cancer for the first time. The HT-29 cells were treated with LSG or 5-fluoruracil, as a positive control, to determine its effect on cell cytotoxicity by a MTT cell proliferation assay, and cell cycle progression and apoptosis using flowcytometry. We also determined the mechanisms underlying LSG-mediated growth inhibition of HT-29 cells by by investigating the expression of key oncogenes and apoptosis genes using q-RT PCR and immunocytochemical analysis. The cell cytotoxicity data showed that the IC50 value of LSG was significantly less than the IC50 value of 5-FU (50 µM). The anti-proliferative effect of LSG was mediated by arresting cells in the S phase of the cell cycle which then led to the induction of apoptosis the q-RT PCR and immunocytochemical analysis showed that LSG reduced the expression of ß-catenin (non-phosphorylated) and its downstream signalling target c-Myc, whereas it increased the phosphorylation of ß-catenin. Furthermore, LSG also downregulated the expression of oncogene K-Ras and anti-apoptotic proteins, Bcl-2, and Bcl-xL. In conclusion, our data demonstrates that LSG exerted its anti-tumor activity by arresting the cell cycle in S phase, and by downregulating the expression of oncogenes including ß-catenin, c-Myc, K-Ras and anti-apoptosis proteins Bcl-2 and Bcl-xL. This study suggests a potential use of natural flavonoid, lawsonaringenin, to attenuate colorectal cancer growth; however, further pre-clinical/clinical studies are required to establish its role as a therapeutic agent.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal cancer is ranked as the third most common cancer and fourth leading cause of mortality around the world and its incidence is rapidly increasing in Asia [1, 2]. The chances for the colorectal cancer to be diagnosed during a lifetime is 5% in men and 4.7% in women, with 30–40% higher prevalence and mortality rates in men than in women [3, 4]. Epidemiological data indicates that environmental factors play an important role in the induction of tumour leading to malignancy. Among cancer-related deaths, 75% of cases are linked to type of dietary intake and sedentary lifestyle [5]. In addition, studies report that majority of the colorectal cases are also linked to both genetic and environmental factors which interact with each other during the process of tumorigenesis of colorectal carcinoma [6]. During the malignant transformation from adenoma to carcinoma, Wnt/beta-catenin pathway plays its effector role by stimulating the transcriptional activity of the c-MYC oncogene [7]. β-Catenin gene is mutated in majority of the colon cancers [8]. Similarly, mutations in RAS and BRAF are found in 50% and 20% of colorectal cancers, respectively, leading to hyperplasia and dysplasia [9, 10].
The deregulation in the process of programmed cell death known as apoptosis is linked to uncontrolled proliferation, and to the development of neoplasm [11]. The disturbance in the ratio of pro-apoptotic and survival proteins can be the determining factor in the process of tumorigenesis. Bcl-2 and Bcl-xL are anti-apoptotic proteins and their constitutive high expression contributes towards the malignancy and cause drug resistance against the cancer chemotherapy [8].
5-Fluorouracil (5-FU), a fluorine derivative of pyrimidine, is used as the clinical treatment for colorectal cancer. Its mechanism of action involves the inhibition of enzyme thymidylate synthase, which is the key enzyme involved in the biosynthesis of pyrimidine nucleotides [12]. Treatment of colorectal cancer with 5-fluorouracil has a survival and response rate from 10 to 15% only [13]. However, this response rate was improved from 40 to 50% when 5-FU is used in combination therapy with other drugs [14]. It is, therefore, important to find novel compounds that can potentially perform better than 5-FU or can be used as an adjunct therapeutic agents in a combination therapy with 5-FU.
Extensive studies report that dietary use of flavonoids containing fruits, vegetables and other medicinal plants may have a preventive effect against colorectal cancer. Several studies suggest that flavonoids might act on the signalling pathways involved in the development and progression of colorectal cancer [15, 16]. The flavonoids manifest anticancer activity by different mechanisms including free-radical scavenging activities [17], arresting cell cycle by altering the expression of cyclins and cyclin dependent kinases [18], and inducing apoptosis by regulating the expression of apoptotic proteins including caspases 3, 8, 9, t-Bid, Bcl-2, Bcl-xL, Bax, and Bak [19, 20].
The current study for the first time investigated the effect of lawsonaringenin (LSG), a natural flavonoid isolated from the leaves of the plant Lawsonia alba Lam [21]. on colorectal cancer and its possible molecular mechanism of action. We studied the effects of LSG on apoptosis, cell cycle progression, and expression of signalling proteins including ß-catenin, c-Myc, K-Ras, Bcl-2 and Bcl-xL using qRT-PCR and immunocytochemistry approaches. The cytotoxic effect of LSG was also studied on non-cancerous 3T3 mouse fibroblast cell line.
Materials and methods
Isolation of lawsonaringenin
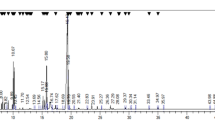
Lawsonaringenin was isolated from the dichloromethane and ethanol extracts of the leaves of L. alba through solvent fractionation and several chromatographic techniques, including vacuum liquid chromatography (VLC), column chromatography (CC) and reversed phase column chromatography as described earlier [21]. The purity was checked by various spectroscopic techniques such as NMR, HR-EIMS, FT-IR and UV–visible. Its molecular formula was deduced as C20H20O5 on the basis of HR-EIMS, which showed the molecular ion at m/z 340.1298.
Cell culture
HT-29 cells (ATCC, USA) were cultured in Dulbecco’s Modified Eagle’s Medium (Complete-DMEM; Gibco, Thermo Fisher Scientific, USA) containing high glucose (500 mg/ml) and L-glutamine (584 mg/ml) with no added sodium pyruvate and supplemented with 10% FBS (foetal bovine serum), and antibiotics (penicillin 100 units/mL and streptomycin 100 mg/mL). Cells were grown in 75 cm2 flasks under sterile conditions and incubated in a humidified incubator with 5% CO2 at 37 °C. Sub-culturing was done every third day based on the confluent state of the cells [22].
Treatment of cells with lawsonaringenin or 5-FU
Lawsonaringenin was dissolved in DMSO as a stock solution at 20 mM and stored at − 20 °C. Cells were treated under sterile conditions with varying concertation of LSG by further diluting (0–50 µM) the stock solution into fresh medium. The final concentration of DMSO was adjusted to < 0.5%. 5-FU was dissolved in DMSO as a stock solution and also stored at − 20 °C. Cells with treated with either 50 or 100 µM concentration of 5-FU under conditions as described above. The control cells were only treated (untreated-control) with an equal amounts of DMSO that was used in LSG-treated cells. The treated- and untreated- (control) cells were incubated in a humidified incubator with 5% CO2 at 37 °C for 24 or 48 h as indicated elsewhere.
MTT assay
Exponentially growing cells (3000 cell/well) were initially seeded in 96-well cell culture-treated plate in complete-DMEM. After 24 h of incubation, media was removed and 200 µL of fresh medium was added containing varying concentrations (0–50 µM) of LSG and cells were incubated in a humidified incubator with 5% CO2 at 37 °C. Cells were also treated with 5-FU (CID 3385, Sigma Aldrich, USA) as a positive control. After 48 h of treatment, MTT dye (0.5 mg/mL) was added to each well, and plates were incubated for 3–4 h. Thereafter, DMSO was added to solubilize formazan crystals, and absorbance was measured at 570 nm using a microplate reader (Spectra Max plus, Molecular Devices, CA, USA). Percentage (%) inhibition and IC50 values were calculated using Soft-Max Pro software (Molecular Devices, USA). as described [23]. After initial experiments, the cells were subsequently treated with IC50 of 5-FU (50 µM) or IC50 of LSG (23.4 µM) or a high dose of LSG (50 µM) that was an equivalent to IC50 of 5-FU.
Analysis of apoptotic profile
Nuclear condensation
Cells (20, 000 cells per well/48 well plate) were analyzed for nuclear fragmentation after the treatment with LSG. Staining of the cells was performed with the fluorescent binding dye DAPI (4, 6-diamidino-2-phenylindole). Cells were grown and treated with test compound for 48 h as described above. After incubation, cells were fixed with 4% paraformaldehyde, washed with PBS, and then stained with DAPI. Cells were then visualized under a fluorescent microscope (Nikon, Japan) [24]. Nuclear area factor which is circularity 4 pi (area/perimeter2) and roundness (perimeter2)/(4*pi*area) was calculated after processing the images by ImageJ software as described [25].
Annexin V/PI staining
After treatment cells (1 million cells per well/6 well plate) were trypsinized, and then washed with PBS. Both floating dead and adherent live cells were collected and finally suspended in annexin binding buffer. Cells were labelled with annexin V-FITC (5 µL) and propidium iodide (1 µL, 0.1 mg/mL), by incubating in the dark for 15 min. Samples were analyzed for apoptosis on flowcytometer (BD FACS Calibur, BD Biosciences, USA) within half an hour [25].
Cell cycle analysis
Cells (1 million cells per well/6 well plate) were washed with PBS twice after treatment with LSG for 48 h, and then were fixed overnight with chilled 70% ethanol. Before flowcytometric analysis, ethanol was removed and the cells were washed with PBS to remove the traces of ethanol. RNAse A (10 mg/mL) was added to the cell pellet, and kept for 3–4 h at room temperature. Staining with propidium iodide prepared in 1.12% sodium citrate was done by incubating the samples for 30 min, and then cell cycle analysis was performed on flow cytometer (BD FACS Calibur™, BD Biosciences) [26].
Real time RT-PCR for mRNA expression
Total RNA was isolated from the treated HT-29 cells (1 million cells per well/6 well plate) by the TRIzol reagent method, as per manufacturer’s instructions. The concentration and purity of RNA was analysed by using NanoDrop 2000, Thermo Scientific USA. RNA (1 µg) was used for the cDNA synthesis by using Thermo Scientific RevertAid First Strand cDNA Synthesis Kit (Cat # K1622). Real time PCR of c-DNA samples was performed by using Thermo Scientific Maxima Sybr Green/ROX qPCR Master Mix (2X), (Cat # K0222). GAP-DH was used as the normalizing control. Comparative CT method was used to calculate the fold change in gene expression [23].
Immunofluorescent microscopy for protein expression analysis
HT-29 cells (20,000 cells/well) were seeded in 48-well plate and were allowed to adhere overnight. Cells were treated for 48 h with LSG and then cells were washed with PBS and fixed in 4% paraformaldehyde for 30 min. Afterwards, subsequent washing with PBS was performed and cells were incubated with primary antibodies overnight at 4 °C. Next day, primary antibodies were removed, cells were washed and incubated with secondary antibody for 1 h at room temperature in the dark. DAPI (4, 6-diamidino-2-phenylindole) staining was also performed to observe the nuclei of the cells. Images were taken using a fluorescent microscope (Nikon, Japan) and processed by Adobe Photoshop (Version 7) [22]. The fluorescent intensity was measured by using ImageJ software (NIH, USA). Monoclonal antibodies used ß-catenin (phosphorylated and non-phosphorylated) were from Cell Signaling, USA, c-Myc, Bcl-2, and Bcl-xL from Sigma Aldrich, USA, K-Ras from Abcam, UK. Secondary antibodies: Alexa Fluor® 488 goat anti-mouse IgG and Alexa Fluor® 594 goat anti-rabbit IgG were from Merck Millipore, USA.
Statistical analysis
Statistical analysis was performed using SPSS 20.0 software (IBM, USA). The data were presented as mean ± SEM The differences between groups were calculated by one way ANOVA test with least significant difference (LSD). Data with different symbols represent a statistical difference for at least at P < 0.05, whereas *P < 0.05, **P < 0.01, and ***P < 0.001 are reported as statistically significant.
Results
Determination of LSG effect on HT-29 colorectal carcinoma cells proliferation
The anti-proliferative activity of LSG against HT-29 cells was performed on varying dose range 0–50 µM and the IC50 values were calculated. Table 1 shows the comparison of % inhibition exhibited by 5-FU and LSG at their calculated IC50 values and at a selected high dose. 5-FU inhibited cell proliferation by 50.96% at 50 µM, which was only slightly increased to 53.36% at 100 µM. The IC50 value of LSG was found to be 23.4 µM which is two-fold (***P < 0.001) less than that of the IC50 value of 5-FU (50 µM) (Table 1). LSG at 50 µM concentration inhibited the proliferation of HT-29 cells by 79.71%. However, 5-FU showed 30% less inhibition at the similar concentration. The toxic effect of the LSG was also performed on 3T3 mouse fibroblast cell line, and the test compound was found to be non-toxic (data not shown).
Effect of lawsonaringenin on apoptosis
The effect of LSG was studied on apoptosis by performing DAPI staining. Reduced nuclear area factor is a morphological marker of apoptosis. After 48 h of treatment with LSG colorectal cancer cells underwent nuclear condensation, as shown in the Fig. 1. Nuclear area factor after 5-FU and LSG treatment was significantly decreased (**P < 0.01, ***P < 0.001) by both concentrations.
Analysis of nuclear area factor after LSG induced apoptosis in DAPI stained HT-29 cells. The data are represented as mean ± SEM of three independent experiments. Cells were stained with DAPI and observed under fluorescence microscope at ×20 lens magnification. Scale bar corresponded to 50 µm. Circularity 4 pi (area/perimeter2) and roundness (perimeter2)/(4*pi*area) was calculated by using ImageJ software. Nuclear area factor was calculated as the product of object area (in pixels2)*roundness. Data indicated by dissimilar alphabets represent a significant difference as determined by One-way ANOVA with LSD
Apoptotic analysis after 24 and 48 h of treatment with LSG in HT-29 cells by annexin V/PI assay exhibited that 5-FU induced a significant (**P < 0.01, ***P < 0.001) apoptosis at 50 and 100 µM of concentrations. However, only 50 µM concentration of LSG induced apoptosis in the CRC cells significantly (**P < 0.01), after 24 h of treatment. Whereas, significant induction of apoptosis was not observed by other concentrations of LSG after 24 and 48 h of treatment. Although dead cell population after LSG treatment was increased significantly (***P < 0.001), and subsequent decrease (***P < 0.001) in live cells was observed as compared to the control cells (Fig. 2).
Apoptotic profile of LSG, and 5-FU treated HT-29 cells. The data are represented as mean ± SEM of three independent experiments. a Live cells. b Dead cells. c Apoptotic cells. The graph of apoptotic cell population (Fig. 2c) indicated by dissimilar alphabets represent a significant difference as determined by One-way ANOVA with LSD
Effect of lawsonaringenin on cell cycle progression
Since LSG significantly induced cytotoxic effects in HT-29 cells, further investigation was carried out to determine the effect of LSG on cell cycle progression by performing flow cytometric analysis using PI staining method. G1 population was significantly (**P < 0.01, ***P < 0.001) decreased by both the concentrations of LSG (23.4 and 50 µM). However, cell population in the S-phase of the cell cycle was increased significantly (**P < 0.01) at 50 µM only. The G2/M phase of the cell cycle remained unaffected. These results (Fig. 3) depict that LSG arrests the cells in S phase of the cell cycle and thus inhibiting its progression.
Cell cycle progression on treatment with LSG. The data showing% cell distribution in G1, S and G2/M phases of the cell cycle are mean ± SEM of three independent experiments. Data indicated by dissimilar alphabets represent a significant difference as determined by One-way ANOVA with LSD. Data obtained in the form of .fcs files from FACS was analysed by the FlowJo software, version 7.0 (Life Sciences, Ashland, Oregon)
Effect of lawsonaringenin on expression of ß-catenin, c-Myc, K-Ras, Bcl-2, and Bcl-xL
The effect of LSG on expression of genes were determined using qRT-PCR analysis (Fig. 4), which showed that both doses (23.4 and 50 µM) of LSG significantly (*P < 0.05, **P < 0.01, and ***P < 0.001) decreased the expression of ß-catenin, K-Ras, Bcl-2, and Bcl-xL, when compared to the control. However, c-Myc expression remained unaffected by the LSG treatment. Figure 4. shows a decrease in fold change of mRNA expression of the selected genes. 5-FU was used as the standard drug and it showed a significant (*P < 0.05, **P < 0.01, and ***P < 0.001) reduction in expression of all the selected oncogenes, as compared to the control HT-29 cells. Fold change was calculated by the comparative CT method (2−∆∆CT method).
Changes in mRNA expression of ß-Catenin, c-Myc, K-Ras, Bcl-2, and Bcl-xL. GAPDH was used as a control gene. CT values were obtained, data was normalized against GAPDH and fold change was calculated by ∆CT method. The data are mean ± SEM of three independent experiments. Data indicated by dissimilar alphabets represent a significant difference as determined by One-way ANOVA with LSD
Effect of lawsonaringenin on expression of ß-catenin, c-Myc, K-Ras, Bcl-2, and Bcl-xL by immunocytochemistry
The effect of LSG on the expression of different genes, as shown in Fig. 4 was further validated by determining the expression of corresponding proteins using a immunohistochemical approach. The expression of ß-actin, used as the control protein, remained the same in treated and untreated HT-29 cells. LSG and 5-FU significantly (*P < 0.05, and ***P < 0.001) decreased the levels of total ß-catenin (non-phosphorylated form) equally. Interestingly only 23.32 µM concentration of LSG (P < 0.001) and 50 µM of 5-FU (*P < 0.05) significantly increased the levels of phosphorylated ß-catenin, as compared to the respective higher concentrations (50 of LSG and 100 µM of 5-FU). c-Myc and Bcl-2 expression was reduced significantly (P < 0.001) by both LSG and 5-FU. LSG significantly decreased the level of K-Ras and Bcl-xL expression also in CRC cells, but no reduction in expression was observed by 5-FU treatment (Figs. 5, 6 and Supplementary Figs. 1, 2, 3, 4, 5, and 6).
Mean fluorescent intensity measured from the protein expression of ß-catenin, c-Myc, K-Ras, Bcl-2, and Bcl-xL, after the treatment with LSG. ß-Actin was used as a control protein. Equal number of cells were counted in different fields and fluorescent intensity was calculated by using Image J software. The data are mean ± SEM of three independent experiments, showing significance *P < 0.05, **P < 0.01, and ***P < 0.001, compared with the respective controls (One-way ANOVA with LSD)
Immunocytochemistry of HT-29 cells demonstrating K-Ras expression. Images were taken at 20X magnification by using microscope TE-2000, Nikon, Japan. K-Ras is shown in Texas-Red channel. The nuclei were stained with DAPI (blue) and images were merged by processing with Adobe Photoshop Version 7.0. Scale bar is 50 µm. (Color figure online)
Discussion
Flavonoids exhibit various biological and pharmacological properties and are mainly found in fruits, vegetables, and other medicinal plants [27]. Flavonoids are reported to have anticancer effects on various cell lines, such as on Caco-2 and HT-29 of colorectal cancer (CRC). These natural flavonoids are also found to be non-toxic on normal cells [28, 29].
Lawsonaringenin is a flavonoid of natural origin and it was isolated from the leaves of the plant Lawsonia alba Lam (Henna) [21]. The anticancer activity of LSG against colorectal cancer cell line HT-29 was evaluated and LSG inhibited the proliferation of HT-29 cells with significantly more efficacy when compared to that of the standard drug 5-Fluorouracil. As stated earlier, 5-FU is used as the main line of clinical treatment for colorectal cancer, therefore it was used as the standard drug [14].
Initially the IC50 value of 5-FU was calculated which was 50 µM. This concentration (50 µM) was selected as the highest dose for screening the % inhibition of the compound LSG. To calculate IC50 value of LSG, % inhibition was measured at different concentrations through MTT assay. In the absence of normal human colon cells availability, LSG was tested also on mouse fibroblast cells (3T3 cells) and found to be nontoxic for the tested doses (data not shown). Normal fibroblasts are generally used as a choice for investigating effects on normal cells [30]. These cells have a finite lifespan in culture, are non-transformed, and non-malignant.
Different studied flavonoids such as genistein, quercetin, silymarin are reported to have anti-proliferative effect against colon cancer cells by arresting the cells in G1/S or G2/M phases of cell division cycle [31, 32].
5-FU is reported to halt the cell cycle progression in S phase in various colorectal cancer cell lines [33]. 5-FU and paclitaxel were used as the standards for protocol optimization. 5-FU arrested the cells in S-phase, whereas G2/M phase was arrested by the treatment with paclitaxel (data not shown).The effect of LSG on cell cycle profile was likewise analysed and it significantly decreased cell population in G1 phase of the cell cycle, whereas subsequent S phase arrest led to increase in the S phase population resulting in slowing down or halting the cell cycle. This data further provides an evidence that antitumor activity for the effect of flavonoids are mediated through their effects on cell cycle progression.
Apoptosis is an important physiological process and the balance between programmed cell death and cell growth is needed for the homeostasis. The deregulation in apoptosis lead to many diseases such as neurodegenerative and autoimmune disorders or cancers [34, 35]. Many studies carried out in the last few decades, have shown that flavonoids induce apoptosis in cancer cells by the modulation of signal transduction pathways associated with apoptosis, proliferation and differentiation [19, 34, 35].
Annexin V and PI staining was performed to determine the apoptotic induction by LSG. 5-FU is reported to have potent pro-apoptotic activity [36], which was shown by the increase in cell population in early and late apoptotic phases after 5-FU treatment (50 and 100 µM). Whereas, only 50 µM concentration of LSG significantly induced apoptosis after 24 h. DAPI stained nuclei in LSG treated cells were condensed in size, which is the morphological marker of apoptosis induction.
Exogenous stress for example: radiation, chemotherapeutic agent or endogenous stress like DNA mutation causes the cells to be arrested first in the G1 or S phase of the cell cycle, to repair the damage. Upon irreparable damage, the cells will commit suicide in order to prevent the passage of damaged DNA to the progeny. This type of death is defined as “stress-induced apoptosis-like cell death (SIaLCD)”. In apoptosis, the dying cells are engulfed by macrophages, whereas, in SIaLCD another type of scavenger cells may be involved. However, as per the available clearance capacity of the scavenger cells present, the dead cells get decomposed to debris through necrotic process, also known as ‘secondary necrosis’ [37]. Therefore, we can suggest that after 48 h, LSG treated cells showed stress induced necrosis-like cell death, as depicted in the increased population of dead cells (Fig. 2b).
Mutation or abnormalities in Wnt/β-catenin signalling is reported in more than 90% of colorectal cancer cases and is considered as an important molecular drug target for chemotherapy. Accumulation of nuclear β-catenin, Tcf/c-Myc signalling is constitutively active in cancer cells [38, 39]. c-Myc targeted genes are involved in cell cycle progression, cell survival, cellular metabolism and proliferation. These genes are deregulated in different cancers [40, 41].
So, by targeting the expression of β-catenin protein and c-Myc, signal transduction, the activated proliferative pathways can be inhibited. Quercetin has been shown to downregulate the expression on different oncogenes involved in signal transduction pathways including c-Myc in Caco-2 CRC cells [42]. 5-FU has also been shown to downregulate the expression of c-Myc in HT-29 cells, showing its effect on Wnt/ß-catenin pathway [43]. In the current study, the effect of LSG was evaluated on the expression of some key mediators in carcinogenesis at mRNA and protein level. As mentioned earlier, the proteins ß-catenin and c-Myc belong to the Wnt/β-catenin signaling pathway and their deregulation play a major role in signaling of growth and proliferation.
The data shows that the expression of c-Myc was significantly decreased by 50 and 100 µM concentrations of 5-FU. However, LSG did not show any change in expression at the gene level whereas a significantly decreased the expression of c-Myc proteins in the cell. Gene expression of β-Catenin was significantly decreased after treatment of LSG and 5-FU in a dose dependent manner. The levels of whole β-catenin were decreased significantly after LSG treatment. However, phosphorylated form of β-catenin was increased only by of LSG at IC50, but at 50 µM concentration no significant increase was observed. Similar results were observed after 5-FU treatment in the phosphorylated ß-catenin levels. This suggests that LSG may be effecting the phosphorylation status of β-catenin protein, which leads to the degradation of whole β-catenin protein in the cytoplasm, inhibiting its translocation inside the nucleus, and through decreased expression of c-Myc, thus inhibiting the cell proliferation. Consistent to our results genestein, a flavonoid has shown to target the upstream mediators of Wnt/ß-catenin signaling by inducing phosphorylated ß-catenin, leading to its ubiquitylation and subsequent degradation [44]. β-catenin is an oncogene. Its activation can result by a direct mutation or by activation of Wnt receptor [45].
Moreover, the effect of LSG was also examined on the levels of K-Ras, Bcl-2 and Bcl-xl. The K-Ras is a proto-oncogene which plays a major role in EGFR stimulated RAS/RAF/MAPK signaling pathway. K-Ras mutation is found in over 40% of CRC patients. These mutations lead to the impairment in the intrinsic GTPase activity of K-Ras, resulting in the constitutive active form of GTP-bound K-Ras. This results in the continuous activation of downstream proliferative signaling pathways leading to tumorigenesis [46]. In a study reported by Tseng et al. suggests that oncogenes involved in tumorigenesis, such as Ras, Bcl-2, Bcl-xL, Raf-1 and PI3K pathway is mostly involved in 5-FU induced apoptosis [35]. Similarly in our study, 5-FU downregulated the expression of K-Ras, Bcl-2, and Bcl-xL. LSG at both the 23.4 and 50 µM concentrations and significantly decreased the gene expression of K-Ras. However, significant decrease in K-Ras protein expression was observed only at high concentration.
The observed difference in the results of c-Myc and K-Ras gene and protein expression shows that the correlation of gene and protein expression levels in biological studies is not perfect. This is due to the complex regulatory mechanisms present inside the biological systems. The observed quantitative differences between gene and protein expression can be due to the post-transcriptional and post translational factors, such as secondary RNA structures, regulatory proteins, regulatory miRNAs, sRNAs and drug-gene, drug-protein interactions [47]. This may also be due to the differences in the sensitivity of these analytical techniques.
The expression of antiapoptotic proteins of Bcl-2 family are found to be upregulated in different cancers including colorectal cancer. High levels of Bcl-2, Bcl-xL, and Mc1-1 are observed in CRC and this is associated with the high metastatic behaviour in different cancer cell lines and attributes towards the high grade tumor and poor prognosis in CRC patients [48].
EGCG, a natural flavonoid and the constituent of green tea has shown its anti-proliferative effect by inhibiting the expression antiapoptotic proteins Bcl-2 and Bcl-xL [49]. In this study qRT-PCR analysis and immunocytochemistry data shows that LSG also significantly reduced the expression of Bcl-2 and Bcl-xL in a concentration dependent manner, which is similar to the effect observed by the treatment of standard drug 5-FU.
Conclusion
In conclusion, present study demonstrated for the first time that LSG treatment of HT-29 cells caused S phase arrest of the cell cycle and induced apoptosis by decreasing the levels of survival proteins Bcl-2 and Bcl-xL. Furthermore, the results also provided evidence that LSG plays its anti-proliferative role against colorectal cancer cells by downregulating the transcriptional activity of Wnt/β-catenin signalling pathway and decreasing the levels of K-Ras. These observations suggest a multi targeted anti-tumour effect of the lawsonaringenin. Furthermore, the IC50 value of LSG appears to be low, however pre-clinical studies are required to determine the effective dose and if this can be achieved by dietary vs pharmacological approaches. Our data suggest that LSG has a potential to be a potent anti-cancer drug for colorectal cancer; however, pre-clinical and clinical studies are required for the development of LSG as a chemotherapeutic agent.
Abbreviations
- ß-Cat-NP:
-
Beta catenin (non-phosphorylated)
- ß-Cat-P:
-
Beta catenin (phosphorylated)
- 5-FU:
-
5-Fluorouracil
- Bcl-2:
-
B-cell lymphoma 2 protein
- Bcl-Xl:
-
B-cell lymphoma extra-large protein
- c-Myc:
-
Avian myelocytomatosis virus oncogene cellular homolog
- EGCG:
-
Epigallocatechin-3gallate
- EGFR:
-
Epidermal growth factor receptor
- G2/M phase:
-
Gap2 mitosis phase
- HT-29:
-
Human colonic adenocarcinoma cell line
- K-Ras:
-
Kirsten rat sarcoma
- PI3K:
-
Phosphatidylinositol 3-kinase
- S phase:
-
Synthesis phase of the cell cycle
- Wnt:
-
Wingless-type MMTV integration site family member
References
Siegel R, Naishadham D, Jemal A (2012) Cancer statistics. CA Cancer J Clin 62:10–29
Pourhoseingholi MA (2012) Increased burden of colorectal cancer in Asia. World J Gastrointest Oncol 4:68
Murphy G, Devesa SS, Cross AJ, Inskip PD, McGlynn KA, Cook MB (2011) Sex disparities in colorectal cancer incidence by anatomic subsite, race and age. Int Journal Cancer 128:1668–1675
Meissner HI, Breen N, Klabunde CN, Vernon SW (2006) Patterns of colorectal cancer screening uptake among men and women in the United States. Cancer Epidemiol Biomark 15:389–394
Anand P, Kunnumakara AB, Sundaram C, Harikumar KB, Tharakan ST, Lai OS, Sung B, Aggarwal BB (2008) Cancer is a preventable disease that requires major lifestyle changes. Pharm Res 25:2097–2116
Jasperson KW, Tuohy TM, Neklason DW, Burt RW (2010) Hereditary and familial colon cancer. Gastroenterology 138:2044–2058
Kriegl L, Vieth M, Kirchner T, Menssen A (2012) Up-regulation of c-MYC and SIRT1 expression correlates with malignant transformation in the serrated route to colorectal cancer. Oncotarget 3:1182
Kang MH, Reynolds CP (2009) Bcl-2 inhibitors: targeting mitochondrial apoptotic pathways in cancer therapy. Clin Cancer Res 15:1126–1132
Andreyev HJN, Norman A, Cunningham D, Oates J, Dix B, Iacopetta B, Young J, Walsh T, Ward R, Hawkins N (2001) Kirsten ras mutations in patients with colorectal cancer: the ‘RASCAL II’study. Br J Cancer 85:692
Rajagopalan H, Bardelli A, Lengauer C, Kinzler KW, Vogelstein B, Velculescu VE (2002) Tumorigenesis: RAF/RAS oncogenes and mismatch-repair status. Nature 418:934–934
Kerr JF, Wyllie AH, Currie AR (1972) Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 26:239
Sobrero A, Guglielmi A, Grossi F, Puglisi F, Aschele C (2000) Mechanism of action of fluoropyrimidines: relevance to the new developments in colorectal cancer chemotherapy. In: Paper presented at the seminars in oncology, vol 27, issue 5. Elsevier, New York, pp 72–77
Giacchetti S, Perpoint B, Zidani R, Le Bail N, Faggiuolo R, Focan C, Chollet P, Llory J, Letourneau Y, Coudert B (2000) Phase III multicenter randomized trial of oxaliplatin added to chronomodulated fluorouracil–leucovorin as first-line treatment of metastatic colorectal cancer. J Clin Oncol 18:136–136
Douillard J, Cunningham D, Roth A, Navarro M, James R, Karasek P, Jandik P, Iveson T, Carmichael J, Alakl M (2000) Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet 355:1041–1047
Lin J, Zhang SM, Wu K, Willett WC, Fuchs CS, Giovannucci E (2006) Flavonoid intake and colorectal cancer risk in men and women. Am J Epidemiol 164:644–651
Ogasawara M, Matsunaga T, Suzuki H (2007) Differential effects of antioxidants on the in vitro invasion, growth and lung metastasis of murine colon cancer cells. Biol Pharm Bullet 30:200–204
Duthie SJ, Dobson V (1999) Dietary flavonoids protect human colonocyte DNA from oxidative attack in vitro. Eur J Nutr 38:28–34
Yin F, Giuliano AE, Law RE, Van Herle AJ (2001) Apigenin inhibits growth and induces G2/M arrest by modulating cyclin-CDK regulators and ERK MAP kinase activation in breast carcinoma cells. Anticancer Res 21:413–420
Ramos S (2007) Effects of dietary flavonoids on apoptotic pathways related to cancer chemoprevention. J Nutr Biochem 18:427–442
Ren W, Qiao Z, Wang H, Zhu L, Zhang L (2003) Flavonoids: promising anticancer agents. Med Res Rev 23:519–534
Uddin N, Siddiqui BS, Begum S, Bhatti HA, Khan A, Parveen S, Choudhary MI (2011) Bioactive flavonoids from the leaves of Lawsonia alba (Henna). Phytochem Lett 4:454–458
Akhdar H, Loyer P, Rauch C, Corlu A, Guillouzo A, Morel F (2009) Involvement of Nrf2 activation in resistance to 5-fluorouracil in human colon cancer HT-29 cells. Eur J Cancer 45:2219–2227
Luo J, Li Y-N, Wang F, Zhang W-M, Geng X (2010) S-adenosylmethionine inhibits the growth of cancer cells by reversing the hypomethylation status of c-myc and H-ras in human gastric cancer and colon cancer. Int J Biol Sci 6:784
DeCoster MA (2007) The nuclear area factor (NAF): a measure for cell apoptosis using microscopy and image analysis. Mod Res Educ Top Microsc 378–384
Violette S, Poulain L, Dussaulx E, Pepin D, Faussat AM, Chambaz J, Lacorte JM, Staedel C, Lesuffleur T (2002) Resistance of colon cancer cells to long-term 5-fluorouracil exposure is correlated to the relative level of Bcl-2 and Bcl-XL in addition to Bax and p53 status. Int J Cancer 98:498–504
Lim DY, Jeong Y, Tyner AL, Park JH (2007) Induction of cell cycle arrest and apoptosis in HT-29 human colon cancer cells by the dietary compound luteolin. Am J Physiol Gastrointest Liver Physiol 292:G66–G75
Middleton E, Kandaswami C, Theoharides TC (2000) The effects of plant flavonoids on mammalian cells: implications for inflammation, heart disease, and cancer. Pharm Rev 52:673–751
Gupta S, Hussain T, Mukhtar H (2003) Molecular pathway for (–)-epigallocatechin-3-gallate-induced cell cycle arrest and apoptosis of human prostate carcinoma cells. Arch Biochem Biophys 410:177–185
Lee H-J, Wang C-J, Kuo H-C, Chou F-P, Jean L-F, Tseng T-H (2005) Induction apoptosis of luteolin in human hepatoma HepG2 cells involving mitochondria translocation of Bax/Bak and activation of JNK. Toxicol Appl Pharm 203:124–131
Tzvetkova-Chevolleau T, Stéphanou A, Fuard D, Ohayon J, Schiavone P, Tracqui P (2008) The motility of normal and cancer cells in response to the combined influence of the substrate rigidity and anisotropic microstructure. Biomaterials 2008 29:1541–1551
Casagrande F, Darbon J-M (2001) Effects of structurally related flavonoids on cell cycle progression of human melanoma cells: regulation of cyclin-dependent kinases CDK2 and CDK1. Biochem Pharm 61:1205–1215
Zi X, Feyes DK, Agarwal R (1998) Anticarcinogenic effect of a flavonoid antioxidant, silymarin, in human breast cancer cells MDA-MB 468: induction of G1 arrest through an increase in Cip1/p21 concomitant with a decrease in kinase activity of cyclin-dependent kinases and associated cyclins. Clin Cancer 4:1055–1064
Hammond WA, Swaika A, Mody K (2016) Pharmacologic resistance in colorectal cancer: a review. Ther Adv Med Oncol 8:57–84
Watson WH, Cai J, Jones DP (2000) Diet and apoptosis. Annu Rev Nutr 20:485–505
MacFarlane M, Williams AC (2004) Apoptosis and disease: a life or death decision. EMBO Rep 5:674–678
Zhang N, Yin Y, Xu S-J, Chen W-S (2008) 5-Fluorouracil: mechanisms of resistance and reversal strategies. Molecules 13:1551–1569
Zhang J, Lou X, Jin L, Zhou R, Liu S, Xu N, Liao DJ (2014) Necrosis, and then stress induced necrosis-like cell death, but not apoptosis, should be the preferred cell death mode for chemotherapy: clearance of a few misconceptions. Oncoscience 1:407
Polakis P (2000) Wnt signaling and cancer. Gene Dev 14:1837–1851
Zhurinsky J, Shtutman M, Ben-Ze’ev A (2000) Plakoglobin and beta-catenin: protein interactions, regulation and biological roles. J Cell Sci 113:3127–3139
Nesbit CE, Tersak JM, Prochownik EV (1999) MYC oncogenes and human neoplastic disease. Oncogene 18
Dang CV, O’Donnell KA, Zeller KI, Nguyen T, Osthus RC, Li F (2006) The c-Myc target gene network. In: Paper presented at the seminars in cancer biology
van Erk MJ, Roepman P, van der Lende TR, Stierum RH, Aarts J, van Bladeren PJ, van Ommen B (2005) Integrated assessment by multiple gene expression analysis of quercetin bioactivity on anticancer–related mechanisms in colon cancer cells in vitro. Eur J Nutr 44:143–156
Wiebke EA, Grieshop NA, Loehrer PJ, Eckert GJ, Sidner RA (2003) Antitumor effects of 5-fluorouracil on human colon cancer cell lines: antagonism by levamisole. J Surg Res 111:63–69
Amado NG, Fonseca BF, Cerqueira DM, Neto VM, Abreu JG (2011) Flavonoids: potential Wnt/beta-catenin signaling modulators in cancer. Life Sci 89:545–554
Polakis P (1999) The oncogenic activation of β-catenin. Cur Opin Gene Dev 9:15–21
Bokemeyer C, Van Cutsem E, Rougier P, Ciardiello F, Heeger S, Schlichting M, Celik I, Köhne C-H (2012) Addition of cetuximab to chemotherapy as first-line treatment for KRAS wild-type metastatic colorectal cancer: pooled analysis of the CRYSTAL and OPUS randomised clinical trials. Eur J Cancer 48:1466–1475
Maier T, Güell M, Serrano L (2009) Correlation of mRNA and protein in complex biological samples. FEBS Lett 583:3966–3973
Koehler BC, Scherr A-L, Lorenz S, Urbanik T, Kautz N, Elssner C, Welte S, Bermejo JL, Jäger D, Schulze-Bergkamen H (2013) Beyond cell death–antiapoptotic Bcl-2 proteins regulate migration and invasion of colorectal cancer cells in vitro. PloS ONE 8:e76446
Leone M, Zhai D, Sareth S, Kitada S, Reed JC, Pellecchia M (2003) Cancer prevention by tea polyphenols is linked to their direct inhibition of antiapoptotic Bcl-2-family proteins. Cancer Res 63:8118–8121
Acknowledgements
M.I.C. acknowledges the enabling role of the Higher Education Commission, Islamabad, Pakistan, through a financial support under, “Studies on the chemoprevention of mammary carcinogenesis by dietary agents” (Project No. 20-3790). A.A. thanks Ms. Narjis Fatima and Dr. Waqar Hameed for the kind gift of Bcl-2 primer.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Authors declare that there are no known conflicts of interest associated with this publication.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Anwar, A., Uddin, N., Siddiqui, B.S. et al. A natural flavonoid lawsonaringenin induces cell cycle arrest and apoptosis in HT-29 colorectal cancer cells by targeting multiple signalling pathways. Mol Biol Rep 45, 1339–1348 (2018). https://doi.org/10.1007/s11033-018-4294-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-018-4294-5