Abstract
Chrysanthemums (Dendranthema morifolium) are one of the most economically important perennial flowering plants, with floricultural (cut flowers), ornamental crop (pot and garden flowers) and, for some cultivars, medicinal uses. Plant architecture is an important agronomic trait for plants with a high ornamental and economic value. In this study, two miniature-related genes, DmCPD and DmGA20ox, were cloned and their tissue-specific expression patterns were analyzed. The results showed that the two genes were both highly expressed in stems, mature leaves, and flowers, and that DmCPD was also highly expressed in pedicels. To generate miniature plants, an RNAi expression vector targeting both DmCPD and DmGA20ox was constructed and transformed into chrysanthemum plants. Smaller plant size and slower growth and development of flowers were observed in dual-silenced chrysanthemums. Brassinosteroid and gibberellin contents in leaves and flower buds of transgenic plants were significantly decreased. Furthermore, the expressions of brassinolide-, gibberellin-, and flowering-related genes were down-regulated by varying degrees in dual-silenced plants. These results suggest that DmCPD and DmGA20ox play important roles in plant architecture, and brassinolide and gibberellin are important hormones in controlling plant architecture. This miniaturization strategy provides an efficient approach for generating new varieties of ornamental plants and crops.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chrysanthemums (Dendranthema morifolium) are one of the most important perennial flowering plants, and can be used as cut flowers, pot and garden flowers, and medicinal materials (Teixeira da Silva 2003). In China, the greenhouse production of chrysanthemums is on the rise. Most chrysanthemum cultivars are short-day that bloom in autumn (Shchennikova et al. 2003), which constrains their year-round production as cut flowers. To meet the year-round market demand, regulation of flowering initiation and development is a highly desired goal in chrysanthemum breeding.
Plant architecture is an important agronomic trait in plants. Dwarfism in cereal crops grown under intensive agriculture has a positive influence on crop agronomy by increasing lodging resistance and decreasing the chances of damage due to wind and rain (Cai et al. 2012). For ornamental plants, plant architecture is more important. Miniature Rosa chinensis is a new variety of the rose family. As its leaves and flowers are small and beautiful, it is admired by many people and has become one of the most popular potted flowers on the international flower market. The tomato cultivar Micro-Tom, produced for ornamental purposes, displays a very dwarf phenotype with small and red ripened fruits (Scott 1989), and has become a popular potted plant. Thus, breeding miniature plant varieties should meet the demands of the intensive agriculture and flower markets. Although conventional breeding has made a great contribution to improve the traits of plants, it is time-consuming. In recent years, genetic engineering has become an important approach in plant breeding.
Plant growth and development are regulated by both environmental and hormonal signals (Depuydt and Hardtke 2011). Based on the phenotype of mutants with disrupted hormone biosynthesis or perception, cytokinin, auxin, gibberellins, and brassinosteroids are considered to be essential for growth (Depuydt and Hardtke 2011). Moreover, brassinosteroids (BRs) and gibberellins (GAs) are inferred to be two major growth-promoting hormones that have similar effects on promoting cell and stem elongation and division (Bai et al. 2012).
Brassinosteroids (BRs) are growth-promoting hormones in plants with structural similarity to animal steroid hormones, regulating senescence, male fertility, pollen and root development, and fruit ripening. In addition, they modulate the plant’s response to numerous environmental signals (Kim and Wang 2010; Clouse 2011). The biochemical definition of the BR biosynthetic pathway and the discovery of BR-deficient and BR-insensitive mutants in Arabidopsis and several crop plants provided convincing evidence that BRs were essential for normal plant development (Fujioka and Yokota 2003). In Arabidopsis, the CPD gene encodes a cytochrome P450 (CYP90) protein, which is required for the C23 hydroxylation of cathasterone to teasterone and plays an essential role in the synthesis of brassinolide (Szekeres et al. 1996; Kauschmann et al. 1996). The hypocotyl elongation defect of the cpd mutant can be rescued by C23-hydroxylated derivatives of cathasterone (Mandava 1988). These previous studies showed that regulating BR biosynthesis or signaling could control the development and height of plants.
Gibberellins (GAs) are involved in many developmental processes, including seed germination, stem elongation, leaf expansion, and flower, seed, and fruit development (Peng et al. 1999b; Sasaki et al. 2002a). Modification of these processes by application of chemicals that alter GA content was a common agronomic practice. For example, GA3 was used to stimulate berry growth in seedless grape production (Christodoulou et al. 1968). Inhibitors of GA biosynthesis were used as growth retardants to control the stature of cereals and ornamental pot plants (Hedden and Hoad 1994). GA is one of the most important determinants of plant height (Sasaki et al. 2002b; Qiao and Zhao 2011). Dwarfism is often caused by mutations in genes controlling the biosynthesis or signaling pathway of GA. In the 1960s, a high-yielding semi-cpd variety of rice, IR8, was achieved by introducing a major dwarfing gene (sd-1) that encoded a defective GA20-oxidase (GA20ox) gene (Spielmeyer et al. 2002b), which led to the rice “green revolution” (Sui et al. 2012; Sasaki et al. 2002b). At the same time, a dominant wheat semi-cpd cultivar Norin 10 carrying Rht genes facilitated a burst in productivity and led to the wheat “green revolution” (Evans 1998). Norin 10 contained the Rht-B1b (formerly called Rht1) and Rht-D1b (formerly called Rht2) alleles that encode a mutant form of DELLA protein, a GA signaling repressor (Peng et al. 1999a; Hedden 2003). These studies demonstrated that controlling GA biosynthesis or signaling was crucial for the production of plants with suitable height for modern production methods.
The flowering time is affected by many variables, including environmental and growth factors such as temperature, photoperiod, light intensity, nutrient status, and phytohormones (Cockshull and Kofranek 1994; Damann and Lyons 1996; Karlsson and Hanscom 1992; Smith and Kamp-Glass 1990). Flowering can thus be induced artificially at specific times by altering environmental factors and externally applying phytohormones, but the process is labor- and energy-intensive. Genetic transformation techniques provide a promising way of introducing new traits by regulating the expression levels of exogenous and endogenous genes in plants (Shimada et al. 2001; Zhang et al. 2013).
In this work, we aimed to obtain miniature chrysanthemums which require a small area and may have better ornamental value. From previous studies, we found that BR/GA biosynthesis or signaling genes could affect the height of a plant. Only silencing one dwarf-related gene may not achieve our desired degree of dwarfness, and silencing the combination of genes that have a similar function may achieve better results. Thus, we constructed an RNAi expression vector targeting both DmCPD and DmGA20ox, which are related to BR and GA biosynthesis, respectively, transformed chrysanthemum plants, and obtained a miniaturized type of chrysanthemum. This study provides a theoretical and technical foundation for future molecular breeding of ornamental plants.
Materials and methods
Plant materials and growth conditions
Pink chanetii was used as the test material in this study. The culture conditions were as follow: 16 h per day illumination intensity of 1,000–2,000 lx at 25 ± 2 °C, and 8 h per day of darkness at 18 ± 2 °C, for 2 days. Wild-type and transgenic plants with six leaves were transplanted into plastic pots, moved into the experimental field, and managed routinely.
Isolation of DmCPD and DmGA20ox gene fragments from chrysanthemum and sequence analysis
Total RNA of chrysanthemum was extracted using TRIzol (Invitrogen, USA) according to the manufacturer’s instructions, then 1 µg total RNA was used to synthesize first-strand cDNA through reverse transcription polymerase chain reaction (M-MLV reverse transcriptase, Promega) with Oligo d(T)18 primer. One to two microliters of the cDNA obtained was used to clone the fragments of DmCPD and DmGA20ox genes with primers (DmCPD-F, DmCPD-R) and (DmGA20ox-F, DmGA20ox-R) (Table 1S) through high-fidelity PCR (Prime STARTM HS DNA polymerase, Takara). The amplified products were tailed using DNA A-Tailing kit (Takara) and linked into pGEM-T Easy vector (Promega). Positive clones were selected out via E. coli JM109 transformation and confirmed by sequencing (Invitrogen). Multiple sequence alignments were performed by DNAMAN version 5.2.2. The phylogenic tree was calculated by MEGA (Molecular Evolutionary Genetics Analysis) version 3.1 (Kumar et al. 2004).
Construction of DmCPD and DmGA20ox RNAi vector
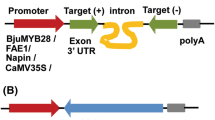
In order to achieve the purpose of dual silencing, we first connected the above two specific gene fragments together: the DmCPD fragment was amplified from the above plasmid CPD-pGEM-T Easy with DmCPD(F + XbaI) and DmCPD(R + XhoI) primers (Table 1S), then double-digested with XbaI and XhoI restriction endonucleases and linked into GA20ox-pGEM-T plasmid at SpeI and SalI restriction sites. After that, the two gene fusion fragments were amplified from plasmid CPD/GA20ox-pGEM-T with DmGA20ox-F + KH and DmCPD-R + X primers (Table 1S), then double-digested with XbaI and HindIII restriction endonucleases and linked into pHANNIBAL plasmid at XbaI and HindIII restriction sites in the sense orientation to obtain GA20ox/CPD-pHANNIBAL plasmid. At the same time, the two gene fusion fragments were amplified from plasmid CPD/GA20ox-pGEM-T with DmGA20ox-F + KH and DmCPD-R + XS primers (Table 1S), then double-digested with KpnI and XhoI restriction endonucleases and linked into GA20ox/CPD-pHANNIBAL plasmid at KpnI and XhoI restriction sites in the antisense orientation. Finally, the double-stranded (ds) RNA expression unit, containing the DmCPD/GA20ox fragment in sense orientation, PDK intron, and DmCPD/GA20ox fragment in antisense orientation was purified and linked into the plant binary vector pBI121 with SacI and XbaI restriction sites.
Plant transformation of DmCPD/GA20ox RNAi vector
The resulted binary vector was transformed into the Agrobacterium LBA4404 strain following the protocols described by Chen et al. (2004). An Agrobacterium-mediated transformation was performed as follows: young leaves of chrysanthemums were cut and washed in tap water for 45–60 min, then washed with 70 % (v/v) ethanol for 2 min, stirring constantly, and rinsed 3 times with sterile water. They were then washed with a 0.1 % (w/w) solution of mercuric chloride for 12 min, rinsed 5–6 times with sterile water, and finally dried with sterile absorbent paper. The above leaves were cut into small pieces having a diameter of 6–8 mm, and were placed on the MS solid agar medium (pH 5.8). The medium was then replaced with a fresh MS medium and the leaves were cultured under the same conditions to obtain chrysanthemum leaves explants. For transformation, the chrysanthemum leaf explants were cut into small pieces having a diameter less than 6 mm, then soaked in an Agrobacterium bacteria solution for 15 min, with excess broth sucked away by sterile absorbent paper, and cultured on a co-culture medium (MS solid medium containing 2 mg/L 6-BA and 0.5 mg/L NAA, pH 5.8), 25 ± 2 °C under dark conditions for 48 h. After being washed with sterile water, the explants were transferred to the bud-inducing medium (MS solid medium containing 4 mg/L 6-BA, 0.1 mg/L NAA, 3 mg/L AgNO3, 400 mg/L Carb and 15–20 mg/L Kan, 0.1 % activated carbon, pH 5.8), and cultured under these conditions: 16 h per day illumination intensity of 1,000–2,000 lx at 25 ± 2 °C and 8 h per day darkness at 18 ± 2 °C. The medium was replaced every 3 weeks until green shoots grew to 2–3 cm.
After that, the shoots were transferred to a rooting medium (1/2 MS solid medium containing 0.1 mg/L NAA, 200 mg/L Carb, and 15 mg/L Kan, pH 5.8) and cultured under the same conditions of illumination and temperature. The transgenic plants were detected with primers NPTII-F (5′-TTGTCACTGAAGCGGGAAGG-3′) and NPTII-R (5′-CGATACCGTAAAGCACGAGGAA-3′).
Quantitative real-time PCR analysis
RNA extraction and cDNA synthesis were performed as described above. The synthesized cDNAs were diluted twice with RNase/DNase-free water. Quantitative real-time PCR analysis was carried out using the CFX96™ Real-Time System (C1000™ Thermal Cycler, BioRad). All reactions were performed using the SYBR® Go Taq II kit (Promega) in a 10 μL total sample volume (5.0 μL 2 × SYBR Premix Go Taq, 0.5 μL primers, 1.0 μL cDNA, and 3.5 μL ddH2O). To remove the effect of genomic DNA and the template from the environment, NTC (no template control) and NRT (no reverse transcription control) were performed. Three replications for each sample were used and standard curves were run simultaneously. Melting curve analysis of qPCR samples revealed that there was only one product for each gene primer reaction. The PCR products were sequenced to confirm the specific amplification. The DmACTIN (Ohmiya et al. 2006) gene was used as the internal standard in tissues. The primers DmCPD(RT)-F/DmCPD(RT)-R and DmGA20ox(RT)-F/DmGA20ox(RT)-R (Table 1S) were used to determine the expression levels of DmCPD and DmGA20ox in wild-type and transgenic lines. Furthermore, the expression levels of GAs, BRs, and flowering-related genes, such as EXP1 (Expansion), XTH5 (Xyloglucan endotransglucosylase/hydrolase), XET (Xyloglucan endotransglucosylase), IAA4 (a member of the auxin-induced Aux/IAA family), CDM36 (a MADS-Box gene), CDM111 (a MADS-Box gene), CmFL (FLORICAULA/LEAFY), CsFTL3 (a chrysanthemum FLOWERING LOCUS T-like gene), and CmNRRa (an orthologous gene of OsNRRa) were detected simultaneously. Primers are shown in Table 1S.
Measuring the stem height, leaf size, and flowering time
In order to understand the differences between wild-type and transgenic lines, we measured the stem height, width, and length of mature leaves (4–5 circles from top) from wild-type and transgenic plants. Ten mature leaves of each plant were measured for width and length, and ten plants were measured for stem height. Flowering time and stem growth speed of wild-type and transgenic lines were also counted. For stem growth, stem heights were measured from 40 days after colonization, once every 20 days. Flowering time was calculated from colonization to flowering (Table 1).
Quantification of brassinosteroid and gibberellin content by ELISA
The one-step double-antibody sandwich method, enzyme-linked immunosorbent assay (ELISA), was used to detect the concentration of endogenous BR and GA in mature leaves and flower buds of wild-type and transgenic lines. For BR extraction, a 0.5-g sample was ground in a mortar and homogenized in a 5-mL extraction solution (isopropyl alcohol). Extracts were centrifuged at 10,000 rpm for 20 min at 4 °C, and the supernatant was analyzed by ELISA (Chai et al. 2013). For the extraction of GA, a 0.5-g sample was ground in an ice-cooled mortar in 5 mL 80 % (v/v) methanol extraction medium. The extract was incubated at 4 °C for 4 h and centrifuged at 4,000 rpm for 15 min at the same temperature. The supernatant was also analyzed by ELISA. The BR and GA ELISA experiments were carried out using the same procedure according to the manufacturer’s instructions (http://www.wksubio.com/product/5794959.html). BR and GA contents were measured at 450 nm using a Bio-Rad 680 microplate reader. The results are the means of three replicates. The correlation coefficients (R 2) of BR and GA standard curves were 0.996 and 0.992, respectively. The concentration of each sample was calculated using a linear regression equation.
Measurement of chlorophyll contents
One gram of weighed fresh mature leaves was pounded to pieces in liquid nitrogen, extracted with 10 mL mixed acetone and ethanol (2:1, v/v) solution for 48 h in the dark, and centrifuged at 5,000 rpm for 10 min at 4 °C. The absorbance of the supernatant was measured at 645 and 663 nm in a PerkinElmer Lambda 900 UV/VIS/NIR spectrophotometer using the above-mentioned mixed solution as a blank. Total chlorophyll content was calculated using the formula according to the method of Arnon (1949): Chl (mg/g) = 20.29 × A 645 + 8.02 × A 663. The chlorophyll of each sample was extracted and measured in triplicate.
Results
Isolation and expression of DmCPD and DmGA20ox genes in chrysanthemum
Following the sequences of DmCPD (GenBank: KC522361) and DmGA20ox (GenBank: AB371601) in NCBI (http://www.ncbi.nlm.nih.gov), the two gene fragments were cloned with specific primers (DmCPD-F, DmCPD-R) and (DmGA20ox-F, DmGA20ox-R), respectively (Table 1S), and sequenced. Phylogenetic and amino acid analysis showed that DmCPD had a high identity to EpCPD and AaCPD (Supplemental Fig. 1a), and belonged to a very conservative cytochrome P450 family (Supplemental Fig. 1b), and DmGA20ox shared a high identity with HaGA20ox and LsGA20ox (Supplemental Fig. 2a), and belonged to the 2OG-Fe(II) oxygenase superfamily [this family contains members of the 2-oxoglutarate (2OG) and Fe(II)-dependent oxygenases] (Supplemental Fig. 2b).
Quantitative real-time PCR technology was performed to analyze the expression of DmCPD and DmGA20ox. The results showed that expression patterns of the two genes were similar except in the pedicel (P). They were all highly expressed in rapid growth or development tissues, such as stems (ST), young leaves (YL), mature leaves (ML), flower buds (FB), and flowers (F), especially in stems, mature leaves, and flowers, while in other tissues, such as roots (R) and senescent leaves (SL), a notably low expression was observed (Fig. 1a, b). These results suggest that these two genes play important roles in the growth and development of plant height, leaves, and flowers.
Expression pattern and silencing effects of DmCPD and DmGA20ox genes in wild-type chrysanthemums and transgenic lines. The expression pattern of DmCPD (a) and DmGA20ox (b) genes of chrysanthemum plants (3 months old) by qRT-PCR analysis. R root, ST stem, YL young leaf, ML mature leaf, SL senescent leaf, P pedicel, FB flower bud, F flower. Silencing effects of DmCPD (c) and DmGA20ox (d). WT, wild type; RNAi-1, RNAi-3, RNAi-5, RNAi-9, RNAi-10 and RNAi-11 were different transgenic lines. Relative expression was normalized using a house-keeping gene DmACTIN (AB205087). The data represent the mean from three replicates with three biological repeats. Error bars SE. The expression level of genes in the wild type was set to 1
Generation of transgenic chrysanthemum plants
An RNAi construct targeting the specific fragment of DmCPD and DmGA20ox was created and transformed into wild-type plants Pink chanetii via Agrobacterium-mediated T-DNA transformation. Six independent transgenic lines were confirmed to be positive transgenic lines by PCR analysis using specific primers for the reporter gene NPTII. Quantitative real-time PCR results showed that transcripts of DmCPD and DmGA20ox were clearly reduced in the transgenic lines compared with the wild type, and three transgenic lines RNAi-1, RNAi-3, and RNAi-10 showed better inhibitory effect than the others (Fig. 1c, d).
Transgenic chrysanthemum plants show a miniature phenotype
During plant development, dual-silenced transgenic plants displayed a slower growth rate and smaller size than the wild type in the same time (Fig. 2a–e). To quantify the differences between wild-type and transgenic lines, we measured the width and length of mature leaves (4–5 circles from top), height of stems, and the growth rate of stems, respectively. The results showed that the leaf size of transgenic plants is about 60 % of that of the wild type and plant height is approximately 30 % of that of the wild type (Fig. 3a–c). The speed of stem growth of transgenic lines is approximately 30 % of that of the wild type (Fig. 3d). The results showed that dual-silenced transgenic plants have shorter stems, smaller leaves, and smaller plants. In addition, transgenic chrysanthemums showed dark-green leaves. We extracted the total chlorophyll from mature leaves. The result revealed that the transgenic lines contained 5–20 % more chlorophyll than the wild type (Fig. 3g).
Phenotype of DmCPD and DmGA20ox-silenced lines and wild type. a Phenotype of wild-type and transgenic plants, 90 days after colonization. b Leaf phenotype of wild-type and transgenic plants, 90 days after colonization. c Phenotype of wild-type and transgenic plants, 150 days after colonization. d Top phenotype of wild-type and transgenic plants, 150 days after colonization. e Top phenotype of wild-type and transgenic plants, 150 days after colonization. f Different stages of flower bud development of wild type. WT wild type; RNAi-1, RNAi-3, RNAi-10 were different transgenic lines
Growth and physiological index detection of wild-type and transgenic plants. Length (a) and width (b) of mature leaves (4–5 circles from top) (mm), n = 10. c Height of plant (mm), n = 10. d Stem growth speed of wild-type and transgenic lines, n = 10. BR (e) and GA (f) content in leaves and flower buds of wild-type and transgenic lines. g Total chlorophyll content in mature leaves of wild-type and transgenic lines. WT, wild type; RNAi-1, RNAi-3 and RNAi-10 are different transgenic lines. The data represent the mean from three replicates with three biological repeats. Asterisks indicate P values below 0.05 between the wild type and others by t test. Error bars SE
Transgenic chrysanthemum plants delayed flowering time
Flowering time is an important ornamental trait for chrysanthemum floricultural production. Under the same conditions, wild-type plants start to grow flower buds 142 days after field planting, 12 days later than the transgenic lines start to grow flower buds (Table 1). The time of first flowering of wild-type plants was 154 days after field planting, and that of transgenic lines was about 162–167 days under the same conditions (Fig. 2c–e, Table 1).
Contents of gibberellins and brassinosteroids in transgenic plants were significantly decreased
In order to explore the underlying reasons for the smaller shapes of transgenic plants, endogenous gibberellin and brassinosteroid contents were determined in mature leaves and flower buds using an enzyme-linked immunosorbent assay. The results showed that the contents of the two plant hormone were significantly decreased in transgenic plants: the brassinosteroids of transgenic lines were reduced by 30 % compared to the wild type, and the gibberellins of transgenic mature leaves were about 60 % that of the wild type (Fig. 3e, f).
Expressions of gibberellin-, brassinosteroid- and flowering-related genes were significantly affected in transgenic plants
To further characterize the molecular mechanism of the growth rate and flowering of transgenic plants, a set of GA-, BR-, and flowering-related genes were examined. According to previous research, the plant hormone gibberellin (GA) induces flower formation in a number of species, including chrysanthemums (Lang 1957; Pharis and King 1985; Wilson et al. 1992).
In our study, the transcripts of GA- and BR-related genes that mainly focused on promoting cell elongation, EXP1, XTH5, XET and IAA4, were all significantly down-regulated in the leaves and stems of transgenic lines (Fig. 4a–d). These results showed that the decreased expression of cell wall expansion and cell elongation genes and growth-promoting genes in dual-silenced lines might affect plant cell elongation and development.
The detection results of brassinolide-, gibberellin- and flowering-related genes. Expression analysis of EXP1 (a), XTH5 (b), XET (c) and IAA4 (d) in leaf and stem of wild-type and transgenic lines. Expression analysis of CDM36 (e), CDM111 (f), FL (g), FTL (h) and NRRa (i) in F1 and F2 stage flower buds of wild-type and transgenic lines. WT wild-type; RNAi-1, RNAi-3 are different transgenic lines; F1 and F2 were the initial two development phases of flower buds. The data represent the mean from three replicates with three biological repeats. Asterisks indicate P values below 0.05 between the wild type and others by t test. Error bars SE
Furthermore, a set of flowering-related genes, CDM36, CDM111, FL, FTL, and NRRa, were detected in flower buds of wild-type and transgenic lines. These results showed that, except for NRRa, the other four flowering-related genes were all markedly down-regulated in the initial two stages of flower bud development in dual-silenced lines (Fig. 4e–i). These results suggested that down-regulated flowering-related genes in the transgenic lines might affect the flowering of plants.
Discussion
The chrysanthemum is one of the most popular ornamental plants in the world. The market continously demands new cultivars, and thus chrysanthemum breeding is of great importance. We obtained a smaller type of chrysanthemum by the dual-silencing of DmCPD and DmGA20ox. This study indicates that dual-silencing similar functioning genes involved in plant architecture is a feasible method of achieving miniaturization of the plant, and lays the theoretical and technical foundation for the molecular breeding of miniaturized flowers or ornamental plants in the future.
An optimized Agrobacterium-mediated transformation system of chrysanthemums
The chrysanthemum is an excellent edible, freezing-tolerant landscape plant known for its varied and beautiful leaves. To improve its ornamental traits, transgenic technology has become a more effective means than traditional breeding methods. To date, much research on the tissue cultures of chrysanthemums has been done (Gertsson and Andersson 1985; Hill 1968; Kaul et al. 1990; Malaure et al. 1991; Teixeira da Silva 2003) and the genetic transformation of chrysanthemums has also been studied (van Wordragen et al. 1991; Seiichim et al. 1995; Lemieux et al. 1990; Sherman et al. 1998; Pavingerová et al. 1994; Takatsu et al. 1998). In previous studies, many cultivars of chrysanthemums, such as Dendranthema grandiflorum (Ramat.) Kitamura, Peach Margaret, Polaris, and two potted plants, Heklaand and Iridon, have been used as materials. These results indicate that the genotype of chrysanthemums is an important factor affecting the transformation efficiency. In this study, a new Chinese variety Pink chanetii was selected as the material, and factors affecting the adventitious shoot regeneration in vitro were studied in detail. We found that whatever the culture medium, a small piece of young leaf with wounds on four sides was the best explant. The addition of 3 mg/L AgNO3 and 0.1 % activated carbon in the medium could greatly increase the frequency of shoot regeneration and prevent browning. The optimum rooting medium was 1/2 MS + NAA 0.1 mg/L + Carb 200 mg/L + Kan 10–15 mg/L. The transformation rate was 4.2 %.
Reduction of GA and BR content slowed the growth rate and affected the morphological development of DmCPD–DmGA20ox silenced plants
To date, the functions of GA and BR associated with plant growth and development have been intensively investigated. The two plant hormones are involved in many aspects of plant growth and development, which necessarily require precise regulation of GA and BR levels in the plant body. Gibberellin mainly promotes stem extension, germination, seed dormancy, flowering, gender performance, root development, and the aging suppression of leaves and fruit (Tyler et al. 2004; Kende and Lang 1964; Olszewski et al. 2002; Fu and Harberd 2003; Groot et al. 1987). BR plays important regulatory roles in seed dormancy and germination, organ differentiation, vascular tissue development, flowering and senescence, morphogenesis, and other various important growth and development processes (Souter et al. 2002; Schlagnhaufer and Arteca 1985; Diener et al. 2000; Topping et al. 1997).
In this study, DmCPD–DmGA20ox silenced plants showed slower growth speed and smaller size than wild-type plants. To reveal the molecular mechanism of this phenotype, a set of GA- and BR-related genes mainly promoting cell elongation, EXP1, XTH5, XET, and IAA4, were detected in wild-type and transgenic lines. Plant cell wall expansion protein EXP1, regulated by GAs and BRs, unlocks the network of polysaccharides, permitting turgor-driven cell enlargement (Cosgrove 2000, 1998). Xyloglucan is one of the primary structural components of the plant cell wall. Therefore, XTHs function may affect both cell shape and plant morphogenesis. XTH5 and XET encoding xyloglucan endotransglucosylase/hydrolases, regulated by GAs and BRs, affect the recruitment of cell wall-modifying activity in response to environmental stress and growth (Xu et al. 1995; Campbell and Braam 1998; Iliev et al. 2002). IAA4, a BR- and auxin-regulated gene, promotes plant growth and development (Wong et al. 1996; Abel et al. 1995). These genes were all significantly down-regulated in transgenic lines (Fig. 4a–d).
In addition, brassinosteroid and gibberellin content were markedly decreased in transgenic plants. These results indicated that dual-silencing of DmCPD and DmGA20ox in chrysanthemums reduced the GA and BR levels in plants, and down-regulated the expression of cell wall expansion and cell elongation genes, and subsequently affected plant cell elongation to produce smaller plants with a slower growth speed.
DmCPD–DmGA20ox silencing affected the flowering and chlorophyll content of plant
Flower development includes the formation and maintenance of inflorescence and floral meristems, and is controlled by environmental conditions and developmental regulation (Blázquez et al. 1998; Parcy et al. 1998). In Arabidopsis, the flowering time of GA-biosynthetic and GA-signaling mutants is well correlated with the expression level of SOC1. The soc1 null mutant shows reduced sensitivity to GA for flowering, while overexpression of SOC1 rescues the non-flowering phenotype of ga1-3, which shows that the GA pathway provides a positive factor for SOC1 activation (Moon et al. 2003). Arabidopsis mutant dd1 (cpd and delayed-flowering 1) shows dwarfism and late-flowering, and is deficient in gibberellin biosynthesis, and the phenotype is rescued by exogenous GA3 (Magome et al. 2004). In this study, transgenic plants showed a late-flowering phenotype (Fig. 2c–e). To explore the molecular mechanism of this phenotype, a set of flowering related genes mainly promoting flowering, CDM36, CDM111, FL, and FTL, were detected in flower buds of wild-type and transgenic lines. CDM36 is a chrysanthemum MADS-box transcription factor, belonging to the SOC1 protein subfamily (Shchennikova et al. 2004), which plays an essential role in integrating multiple flowering signals to regulate the transition from vegetative to reproductive development (Moon et al. 2003). CDM111, a MADS-box gene, is a member of the APETALA1(AP1) subfamily, and overexpression of CDM111 in Arabidopsis plants results in dramatically reduced time to flowering (Honma and Goto 2001; Mandel and Yanofsky 1995). In addition, CDM111 is able to partially complement the ap1-1 mutant from Arabidopsis, illustrating that CDM111 is the functional equivalent of AP1 (Bowman et al. 1993). GA promotes the expression of FL, which belongs to the FLORICAULA/LEAFY homologous gene from chrysanthemums (Blázquez et al. 1998). FTL3, a chrysanthemum FLOWERING LOCUS T-like gene, is a key regulator of flowering in chrysanthemums. FTL3 has the potential to induce early flowering because of its overexpression in chrysanthemums (Oda et al. 2012). These genes were all significantly down-regulated in transgenic lines (Fig. 4e–h). Judging from these results, silencing of the gibberellin biosynthetic gene GA20ox resulted in reduced GA content in the flower buds, thereby down-regulating the expression levels of CDM36, CDM111, FL, and FTL3, and eventually might lead to delayed flowering of transgenic plants. We also detected NRRa, which was strongly expressed in flower buds and peduncles, and NRRa RNAi transgenic plants flowered 40–64 days earlier, whereas NRRa-overexpressing plants exhibited a delayed flowering phenotype (Zhang et al. 2013). However, the expression levels in wild-type and transgenic lines had no significant difference (Fig. 4i). Why was NRRa not affected by the dual-silencing of DmCPD and DmGA20ox? That needs further study.
Many Arabidopsis BR synthesis and signaling pathway mutants show extension of nutritional growth and delayed flowering time. For instance, under the same growth conditions, BR synthesis mutants det2 (Fujioka et al. 1997), dwf4 (Azpiroz et al. 1998), and cpd (Li and Chory 1997) and BR-insensitive mutant bri1 (Domagalska et al. 2007) all show late-flowering phenotypes. Studies have found that the endogenous BR content of det2 mutant is 10 % of that of the wild type, and cpd, dwf4, and bri1 mutants accumulate different BR precursors in vivo (Szekeres et al. 1996; Azpiroz et al. 1998; Noguchi et al. 1999). These data show that endogenous BR content and change of BR signal transduction are involved in flowering time regulation. Therefore, in our study, reduced brassinolide content in the transgenic flower buds may delay the flowering time.
Moreover, dual-silenced chrysanthemums contained approximately 5–21 % more chlorophyll than wild-type plants (Fig. 3g). A similar phenotype exists in GA-insensitive or GA-deficient mutants of Arabidopsis, which have high levels of chlorophyll (Koorneef et al. 1985).
Simultaneous silencing of DmCPD and DmGA20ox is an effective way of achieving miniaturization of chrysanthemum
Currently, dwarfing genes are concentrated in the GA, BR, auxin biosynthesis, and signal transduction and light signaling pathways (Ross et al. 2001; Yin et al. 2007). Genes of cytokinins and ethylene synthesis and signaling pathways (De Grauwe et al. 2005) may also become important factors in research on ornamental plant miniaturization. Moreover, cell wall synthesis and metabolism, temperature and other environmental factors, DNA methylation (Finnegan et al. 1996), microRNA (Achard et al. 2004), and transcription factors are important regulatory elements for plant growth and development. In this study, BR/GA biosynthesis genes DmCPD and DmGA20ox were selected to construct an RNAi vector. Through transgenic technology, simultaneously silenced chrysanthemum plants were produced and exhibited dwarf and late flower phenotypes. This result indicates that jointly silencing DmCPD and DmGA20ox genes is a feasible method for miniature chrysanthemum breeding.
References
Abel S, Nguyen MD, Theologis A (1995) The PS-IAA4/5-like family of early auxin-inducible mRNAs in Arabidopsis thaliana. J Mol Biol 251(4):533–549
Achard P, Herr A, Baulcombe DC, Harberd NP (2004) Modulation of floral development by a gibberellin-regulated microRNA. Sci Signal 131(14):3357
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24(1):1
Azpiroz R, Wu Y, LoCascio JC, Feldmann KA (1998) An Arabidopsis brassinosteroid-dependent mutant is blocked in cell elongation. Plant Cell Online 10(2):219–230
Bai M-Y, Shang J-X, Oh E, Fan M, Bai Y, Zentella R, Sun T-P, Wang Z-Y (2012) Brassinosteroid, gibberellin and phytochrome impinge on a common transcription module in Arabidopsis. Nat Cell Biol 14(8):810–817
Blázquez MA, Green R, Nilsson O, Sussman MR, Weigel D (1998) Gibberellins promote flowering of Arabidopsis by activating the LEAFY promoter. Plant Cell Online 10(5):791–800
Bowman JL, Alvarez J, Weigel D, Meyerowitz EM, Smyth DR (1993) Control of flower development in Arabidopsis thaliana by APETALA1 and interacting genes. Development 119(3):721–743
Cai P, Long H, Deng G, Pan Z, Peng Z, Yu M (2012) Molecular cloning, characterization, and expression analysis of genes encoding gibberellin 20-oxidase in Dasypyrum villosum dwarf mutant. Plant Mol Biol Report 30(5):1110–1116
Campbell P, Braam J (1998) Co- and/or post-translational modifications are critical for TCH4 XET activity. Plant J 15(4):553–561
Chai Y-m, Zhang Q, Tian L, Li C-L, Xing Y, Qin L, Shen Y-Y (2013) Brassinosteroid is involved in strawberry fruit ripening. Plant Growth Regul 69(1):63–69
Chen G, Hackett R, Walker D, Taylor A, Lin Z, Grierson D (2004) Identification of a specific isoform of tomato lipoxygenase (TomloxC) involved in the generation of fatty acid-derived flavor compounds. Plant Physiol 136(1):2641–2651
Christodoulou A, Weaver R, Pool R (1968) Relation of gibberellin treatment to fruit-set, berry development, and cluster compactness in Vitis vinifera grapes. Proc Am Soc Hortic Sci 92:301–310
Clouse SD (2011) Brassinosteroid signal transduction: from receptor kinase activation to transcriptional networks regulating plant development. Plant Cell Online 23(4):1219–1230
Cockshull K, Kofranek A (1994) High night temperatures delay flowering, produce abnormal flowers and retard stem growth of cut-flower chrysanthemums. Sci Hortic 56(3):217–234
Cosgrove DJ (1998) Cell wall loosening by expansins. Plant Physiol 118(2):333–339
Cosgrove DJ (2000) Loosening of plant cell walls by expansins. Nature 407(6802):321–326
Damann MP, Lyons RE (1996) Natural chilling and limited inductive photoperiod affect flowering in two Asteraceae genera. J Am Soc Hortic Sci 121(4):694–698
De Grauwe L, Vandenbussche F, Tietz O, Palme K, Van Der Straeten D (2005) Auxin, ethylene and brassinosteroids: tripartite control of growth in the Arabidopsis hypocotyl. Plant Cell Physiol 46(6):827–836
Depuydt S, Hardtke CS (2011) Hormone signalling crosstalk in plant growth regulation. Curr Biol 21(9):R365–R373
Diener AC, Li H, Zhou W-X, Whoriskey WJ, Nes WD, Fink GR (2000) Sterol methyltransferase 1 controls the level of cholesterol in plants. Plant Cell Online 12(6):853–870
Domagalska MA, Schomburg FM, Amasino RM, Vierstra RD, Nagy F, Davis SJ (2007) Attenuation of brassinosteroid signaling enhances FLC expression and delays flowering. Development 134(15):2841–2850
Evans LT (1998) Feeding the ten billion: plants and population growth. Cambridge University Press, Cambridge
Finnegan EJ, Peacock WJ, Dennis ES (1996) Reduced DNA methylation in Arabidopsis thaliana results in abnormal plant development. Proc Natl Acad Sci USA 93(16):8449–8454
Fu X, Harberd NP (2003) Auxin promotes Arabidopsis root growth by modulating gibberellin response. Nature 421(6924):740–743
Fujioka S, Yokota T (2003) Biosynthesis and metabolism of brassinosteroids. Annu Rev Plant Biol 54(1):137–164
Fujioka S, Li J, Choi Y-H, Seto H, Takatsuto S, Noguchi T, Watanabe T, Kuriyama H, Yokota T, Chory J (1997) The Arabidopsis deetiolated2 mutant is blocked early in brassinosteroid biosynthesis. Plant Cell Online 9(11):1951–1962
Gertsson U, Andersson E (1985) Propagation of Chrysanthemum × hortorum and Philodendron scandens by tissue culture. Rapport-Sveriges Lantbruksuniversitet, Institutionen foer Traedgaardsvetenskap
Groot SP, Bruinsma J, Karssen CM (1987) The role of endogenous gibberellin in seed and fruit development of tomato: studies with a gibberellin-deficient mutant. Physiol Plant 71(2):184–190
Hedden P (2003) The genes of the green revolution. Trends Genet 19(1):5–9
Hedden P, Hoad GV (1994) Growth regulators and crop productivity. Marcel Dekker, New York
Hill G (1968) Shoot formation in tissue cultures of chrysanthemum ‘Bronze Pride’. Physiol Plant 21(2):386–389
Honma T, Goto K (2001) Complexes of MADS-box proteins are sufficient to convert leaves into floral organs. Nature 409(6819):525–529
Iliev EA, Xu W, Polisensky DH, Oh M-H, Torisky RS, Clouse SD, Braam J (2002) Transcriptional and posttranscriptional regulation of Arabidopsis TCH4 expression by diverse stimuli. Roles of cis regions and brassinosteroids. Plant Physiol 130(2):770–783
Karlsson MG, Hanscom JT (1992) Influence of low temperature on floral development in chrysanthemum. HortScience 27(6):649
Kaul V, Miller RM, Hutchinson JF, Richards D (1990) Shoot regeneration from stem and leaf explants of Dendranthema grandiflora Tzvelev (syn. Chrysanthemum morifolium Ramat.). Plant Cell Tissue Organ Cult 21(1):21–30
Kauschmann A, Jessop A, Koncz C, Szekeres M, Willmitzer L, Altmann T (1996) Genetic evidence for an essential role of brassinosteroids in plant development. Plant J 9(5):701–713
Kende H, Lang A (1964) Gibberellins and light inhibition of stem growth in peas. Plant Physiol 39(3):435
Kim T-W, Wang Z-Y (2010) Brassinosteroid signal transduction from receptor kinases to transcription factors. Annu Rev Plant Biol 61:681–704
Koorneef M, Elgersma A, Hanhart C, Loenen-Martinet E-v, Rijn L v, Zeevaart J (1985) A gibberellin insensitive mutant of Arabidopsis thaliana. Physiol Plant 65(1):33–39
Kumar S, Tamura K, Nei M (2004) MEGA3: integrated software for molecular evolutionary genetics analysis and sequence alignment. Brief Bioinform 5(2):150–163
Lang A (1957) The effect of gibberellin upon flower formation. Proc Natl Acad Sci USA 43(8):709
Lemieux C, Firoozabady E, Robinson K, Jong JD (1990) Agrobacterium-mediated transformation of chrysanthemum. In: Integration of in vitro techniques in ornamental plant breeding. Proceedings, symposium, 10–14 November 1990. EUCARPIA, pp 150–155
Li J, Chory J (1997) A putative leucine-rich repeat receptor kinase involved in brassinosteroid signal transduction. Cell 90(5):929–938
Magome H, Yamaguchi S, Hanada A, Kamiya Y, Oda K (2004) dwarf and delayed-flowering 1, a novel Arabidopsis mutant deficient in gibberellin biosynthesis because of overexpression of a putative AP2 transcription factor. Plant J 37(5):720–729
Malaure R, Barclay G, Power J, Davey M (1991) The production of novel plants from florets of Chrysanthemum morifolium using tissue culture. 1. Shoot regeneration from ray florets and somaclonal variation exhibited by the regenerated plants. J Plant Physiol 139(1):8–13
Mandava NB (1988) Plant growth-promoting brassinosteroids. Annu Rev Plant Physiol Plant Mol Biol 39(1):23–52
Mandel MA, Yanofsky MF (1995) A gene triggering flower formation in Arabidopsis. Nature 377(6549):522–524
Moon J, Suh SS, Lee H, Choi KR, Hong CB, Paek NC, Kim SG, Lee I (2003) The SOC1 MADS-box gene integrates vernalization and gibberellin signals for flowering in Arabidopsis. Plant J 35(5):613–623
Noguchi T, Fujioka S, Choe S, Takatsuto S, Yoshida S, Yuan H, Feldmann KA, Tax FE (1999) Brassinosteroid-insensitive dwarf mutants of Arabidopsis accumulate brassinosteroids. Plant Physiol 121(3):743–752
Oda A, Narumi T, Li T, Kando T, Higuchi Y, Sumitomo K, Fukai S, Hisamatsu T (2012) CsFTL3, a chrysanthemum FLOWERING LOCUS T-like gene, is a key regulator of photoperiodic flowering in chrysanthemums. J Exp Bot 63(3):1461–1477
Ohmiya A, Kishimoto S, Aida R, Yoshioka S, Sumitomo K (2006) Carotenoid cleavage dioxygenase (CmCCD4a) contributes to white color formation in chrysanthemum petals. Plant Physiol 142(3):1193–1201
Olszewski N, T-p Sun, Gubler F (2002) Gibberellin signaling biosynthesis, catabolism, and response pathways. Plant Cell Online 14(suppl 1):S61–S80
Parcy F, Nilsson O, Busch MA, Lee I, Weigel D (1998) A genetic framework for floral patterning. Nature 395(6702):561–566
Pavingerová D, Dostál J, Bísková R, Benetka V (1994) Somatic embryogenesis and Agrobacterium-mediated transformation of chrysanthemum. Plant Sci 97(1):95–101
Peng J, Richards DE, Hartley NM, Murphy GP, Devos KM, Flintham JE, Beales J, Fish LJ, Worland AJ, Pelica F (1999a) ‘Green revolution’ genes encode mutant gibberellin response modulators. Nature 400(6741):256–260
Peng J, Richards DE, Hartley NM, Murphy GP, Devos KM, Flintham JE, Beales J, Fish LJ, Worland AJ, Pelica F (1999b) ‘Green revolution’ genes encode mutant gibberellin response modulators. Nature 400(6741):256–261
Pharis RP, King RW (1985) Gibberellins and reproductive development in seed plants. Annu Rev Plant Physiol 36(1):517–568
Qiao F, Zhao K-J (2011) The influence of RNAi targeting of OsGA20ox2 gene on plant height in rice. Plant Mol Biol Report 29(4):952–960
Ross JJ, O’Neill DP, Wolbang CM, Symons GM, Reid JB (2001) Auxin-gibberellin interactions and their role in plant growth. J Plant Growth Regul 20(4):336–353
Sasaki A, Ashikari M, Ueguchi-Tanaka M, Itoh H, Nishimura A, Swapan D, Ishiyama K, Saito T, Kobayashi M, Khush G (2002) Green revolution: a mutant gibberellin-synthesis gene in rice. Nature 416(6882):701–702
Sasaki A, Ashikari M, Ueguchi-Tanaka M, Itoh H, Nishimura A, Swapan D, Ishiyama K, Saito T, Kobayashi M, Khush G (2002b) A mutant gibberellin-synthesis gene in rice. Nature 416(6882):701
Schlagnhaufer CD, Arteca RN (1985) Brassinosteroid-induced epinasty in tomato plants. Plant Physiol 78(2):300–303
Scott JW (1989) Micro-Tom—a miniature dwarf tomato. Florida Agric Exp Stn Circ 370:1–6
Seiichim F, de Jongzl J, Rademakerz W (1995) Efficient genetic transformation of chrysanthemum (Dendranthema grandiflorum (Ramat.) Kitamura) using stem segments. Breed Sci 45:179–184
Shchennikova A, Shulga O, Angenent G, Skryabin K (2003) Genetic regulation of inflorescence development in Chrysanthemum. Dokl Biol Sci 391:368–370
Shchennikova AV, Shulga OA, Immink R, Skryabin KG, Angenent GC (2004) Identification and characterization of four chrysanthemum MADS-box genes, belonging to the APETALA1/FRUITFULL and SEPALLATA3 subfamilies. Plant Physiol 134(4):1632–1641
Sherman JM, Moyer JW, Daub ME (1998) A regeneration and Agrobacterium-mediated transformation system for genetically diverse Chrysanthemum cultivars. J Am Soc Hortic Sci 123(2):189–194
Shimada Y, Fujioka S, Miyauchi N, Kushiro M, Takatsuto S, Nomura T, Yokota T, Kamiya Y, Bishop GJ, Yoshida S (2001) Brassinosteroid-6-oxidases from Arabidopsis and tomato catalyze multiple C-6 oxidations in brassinosteroid biosynthesis. Plant Physiol 126(2):770–779
Smith J, Kamp-Glass M (1990) Gibberelin promotes flower stem elongation on the chrysanthemums. HortScience 25(9):1118
Souter M, Topping J, Pullen M, Friml J, Palme K, Hackett R, Grierson D, Lindsey K (2002) Hydra mutants of Arabidopsis are defective in sterol profiles and auxin and ethylene signaling. Plant Cell Online 14(5):1017–1031
Spielmeyer W, Ellis MH, Chandler PM (2002) Semidwarf (sd-1), “green revolution” rice, contains a defective gibberellin 20-oxidase gene. Proc Natl Acad Sci USA 99(13):9043–9048
Sui J-M, Guo B-T, Wang J-S, Qiao L-X, Zhou Y, Zhang H-G, Gu M-H, Liang G-H (2012) A new GA-insensitive semidwarf mutant of rice (Oryza sativa L.) with a missense mutation in the SDG gene. Plant Mol Biol Report 30(1):187–194
Szekeres M, Németh K, Koncz-Kálmán Z, Mathur J, Kauschmann A, Altmann T, Rédei GP, Nagy F, Schell J, Koncz C (1996) Brassinosteroids rescue the deficiency of CYP90, a cytochrome P450, controlling cell elongation and de-etiolation in Arabidopsis. Cell 85(2):171–182
Takatsu Y, Tomotsune H, Kasumi M, Sakuma F (1998) Differences in adventitious shoot regeneration capacity among Japanese chrysanthemum (Dendranthema grandiflorum (Ramat.) Kitamura) cultivars and the improved protocol for Agrobacterium-mediated genetic transformation. J Jpn Soc Hortic Sci 67(6):958–964
Teixeira da Silva JA (2003) Chrysanthemum: advances in tissue culture, cryopreservation, postharvest technology, genetics and transgenic biotechnology. Biotechnol Adv 21(8):715–766
Topping JF, May VJ, Muskett PR, Lindsey K (1997) Mutations in the HYDRA1 gene of Arabidopsis perturb cell shape and disrupt embryonic and seedling morphogenesis. Development 124(21):4415–4424
Tyler L, Thomas SG, Hu J, Dill A, Alonso JM, Ecker JR, Sun T-p (2004) DELLA proteins and gibberellin-regulated seed germination and floral development in Arabidopsis. Plant Physiol 135(2):1008–1019
van Wordragen MF, de Jong J, Huitema HB, Dons HJ (1991) Genetic transformation of chrysanthemum using wild type Agrobacterium strains; strain and cultivar specificity. Plant Cell Rep 9(9):505–508
Wilson RN, Heckman JW, Somerville CR (1992) Gibberellin is required for flowering in Arabidopsis thaliana under short days. Plant Physiol 100(1):403–408
Wong LM, Abel S, Shen N, Foata M, Mall Y, Theologis A (1996) Differential activation of the primary auxin response genes, PS-IAA4/5 and PS-IAA6, during early plant development. Plant J 9(5):587–599
Xu W, Purugganan MM, Polisensky DH, Antosiewicz DM, Fry SC, Braam J (1995) Arabidopsis TCH4, regulated by hormones and the environment, encodes a xyloglucan endotransglycosylase. The Plant Cell Online 7(10):1555–1567
Yin C, Gan L, Ng D, Zhou X, Xia K (2007) Decreased panicle-derived indole-3-acetic acid reduces gibberellin A1 level in the uppermost internode, causing panicle enclosure in male sterile rice Zhenshan 97A. J Exp Bot 58(10):2441–2449
Zhang Y, Lian L, Liu Q, Xiao N, Fang R, Liu Q, Chen X (2013) Altered expression of CmNRRa changes flowering time of Chrysanthemum morifolium. Plant Biotechnol J 11(3):373–379
Acknowledgments
This work was supported by National Natural Science Foundation of China (Nos. 31171968 and 31100089), and the Fundamental Research Funds for the Central Universities (CDJXS10232209).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
11032_2015_239_MOESM1_ESM.tif
Supplemental Fig. 1 Phylogenetic analysis and multiple sequence alignment of DmCPD and other cytochrome P450 family proteins. a. Phylogenetic analysis of DmCPD and other proteins was conducted by the neighbor-joining method, and bootstrap analysis of 1,000 replicates. DmCPD is marked with an asterisk. b. Multiple sequence alignment of DmCPD and other cytochrome P450 family proteins. Identical amino acids are shaded in black, and similar amino acids are shaded in gray. DmCPD is marked with an asterisk. Accession numbers and corresponding references for the proteins listed are as follows: DmCPD (BAE16978), EpCPD (AGV40782), AaCPD (ABC94481), SlCPD (XP_004240946), GhCPD (ACR20477), CjCPD (AAZ39038), NtCPD (CAD27417), PhCPD (AGJ98214), PeCPD(ADK66927), CsCPD(AAZ05071). (TIFF 1181 kb)
11032_2015_239_MOESM2_ESM.tif
Supplemental Fig. 2 Phylogenetic analysis and multiple sequence alignment of DmGA20ox and other GA20ox proteins. a. Phylogenetic analysis of DmGA20ox and other GA20ox proteins was conducted by the neighbor-joining method, and bootstrap analysis of 1,000 replicates. DmGA20ox is marked with an asterisk. b. Multiple sequence alignment of DmGA20ox and other GA20ox proteins. DmGA20ox is marked with an asterisk. Identical amino acids are shaded in black, and similar amino acids are shaded in gray. Accession numbers and corresponding references for the proteins listed are as follows: DmGA20ox (BAG48319), CaGA20ox (ADZ96940), GhGA20ox (ACM68923), RcGA20ox (XP_002510873), HaGA20ox (CAQ43616), NtGA20ox (BAC76428), LsGA20ox (BAA37128). (TIFF 1465 kb)
Rights and permissions
About this article
Cite this article
Xie, Q., Chen, G., Liu, Q. et al. Dual silencing of DmCPD and DmGA20ox genes generates a novel miniature and delayed-flowering Dendranthema morifolium variety. Mol Breeding 35, 67 (2015). https://doi.org/10.1007/s11032-015-0239-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11032-015-0239-z