Abstract
A series of novel pyrazole-4-carboxamides were rationally designed, synthesized, and their structures were characterized by 1H NMR, 13C NMR and HRMS. Preliminary bioassay showed that four compounds 8g, 8j, 8o and 8s exhibited more than 90% and even completed inhibition against Alternaria solani at 100 μg/mL; and 8d displayed 100% inhibition against Fusarium oxysporum at the same concentration. Moreover, 8j exhibited good in vitro fungicidal activity against A. solani with EC50 value of 3.06 μg/mL, and it also displayed completed in vivo protective antifungal activity against A. solani on tomato at 10 mg/L, as boscalid did. The molecular docking results indicated that 8j exhibited the high affinity with SDH protein by H-bond and π–π stacking interactions, which may explain the reasons for its good activities. These data support that compound 8j could be used as a fungicide candidate for further study.
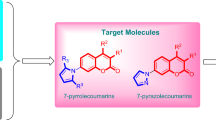
Graphic abstract
A practical method for the synthesis of pyrazole-4-carboxamides were provided and evaluation of their antifungal activities.

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Fungicides play an important role in agricultural production for controlling plant diseases [1, 2]. Succinate dehydrogenase inhibitors as the key class of fungicides can block the energy synthesis of the pathogens by targeting succinate dehydrogenase (SDH) to inhibit mitochondrial electron transfer between succinate and ubiquinone, which system is critical for the oxygen-sensing and has been one of the most significant targets for developing fungicides [3,4,5,6]. To date, over a dozen commercial SDHIs have been launched as fungicides with the characteristics of efficient, broad spectrum activities [7].
SDHIs are structurally very diversity but their essential common feature consists of three parts: the amide bond, the five- or six-membered ring systems attached to carbonyl of the amide bond and the amino group on the side of the amide bond [8, 9]. Among the SDHIs, CF2H group-substituted pyrazole carboxamides scaffold were the newer represented type (Fig. 1) [10], which are the core moiety binding to the active site of SDH, and exhibit excellent antifungal activity. However, the amino substituent group is also important for activity, which can be split into two main components: the one is the liner such as a phenyl group, and the other is the hydrophobic rest that still affects the biological spectrum and potency of the molecules [8]. For example, pyrazole carboxamides containing diphenylamine scaffold and biphenyl ether amine were developed, respectively, and exhibited good antifungal activities [11,12,13,14]. Thus, making the amino group structural diversity based on the important CF2H group-substituted pyrazole moiety may discover novel efficient SDHIs.
Therefore, a series of novel SDHIs were rationally designed and synthesized based on CF2H-substituted pyrazole carboxamide by employing 2-arylbenzylamines as substructures into the target molecules (Fig. 2), and homology model and docking simulation were used to study structure–activity relationship. The target molecules with high antifungal activities were found by the bioassay.
Results and discussion
Chemistry
The synthetic procedures of the target compounds 8a–8u are described in Fig. 3. Firstly, compound 1 was produced from the reaction between the starting materials methyl hydrazine and ethyl 4,4-difluoro-3-oxobutanoate, then which reacted with POCl3 in DMF to give compound 2, and compound 2 was successively oxidized, chlorinated to obtain the acid chloride compound 4. The 2-arylbenzylamines 7 were synthesized from corresponding aldehyde compound 5 by the Suzuki cross-coupling reaction, condensation and reduction. Finally, compound 4 reacted with different 2-arylbenzylamines to give the target compounds. However, the substrate of Ar with substituted Br group could not give the corresponding products 6.
Crystal structure analysis
The crystal structure of 8u was obtained by culturing a mixture of ethyl acetate and dichloromethane (Fig. 4). Single crystal diffraction data were collected using graphite monochromatic oxidation of MoKα radiation (λ = 0.71073 Å) in the range of 1.821° ≤ θ ≤ 27.879° (h, − 14 to 14; k, − 11 to 11; l, − 29 to 29) on the Bruker SMART 1000 CCD diffractometer. A total of 5189 independent diffraction data (Rint = 0.0375) were used for structural analysis, of which 4161 observed reflections with I > 2σ(I).The structure was processed using the direct method of the SHELXS-97 system [15]. The data are improved by the full matrix least squares method. All non-hydrogen atom coordinates were determined by Doron Fourier synthesis. The atomic coordinates and the anisotropy temperature factors were corrected by the full matrix least squares method. The final deviation factors were R = 0.0527 and wR = 0.1385 (w = 1/[σ2((F 2o ) + (0.0824P)2 + 1.1121P], where P = (F 2o + 2F 2c )/3 with (Δ/σ)max = 0.998 and S = 1.045.
Antifungal activity
The in vitro antifungal activities of the target compounds were evaluated at 100 μg/mL, and the results are listed in Table 1 and showed that compound 8d with the olefin at the Ar exhibited 100% growth inhibition activity against F. oxysporum, which is slightly better than that of the positive control boscalid. Both compounds 8j and 8s displayed 100% growth inhibition against A. solani, which were similar to that of boscalid, and 8j exhibited the higher inhibition against F. graminearum and P. sasaki than that of boscalid additionally.
In order to obtain the fungicidal potency of the compounds with inhibition activities more than 90% at 100 μg/mL, in vitro antifungal activity of compound 8d against F. oxysporum, and 8g, 8j, 8o and 8s against A. solani at 50 μg/mL were further determined, respectively (Table 2). The results showed that compound 8j remained 93% growth inhibition against A. solani, which was a similar to that of boscalid, and better than that of fluxapyroxad [16]. Subsequently, the median effective concentration (EC50) of compound 8j was determined (Table 3), and it exhibited good fungicidal activity against A. solani with EC50 value of 3.06 μg/mL, which was a similar to that of boscalid.
According to the results of in vitro antifungal activity assay, in vivo protective activity against A. solani on tomato of compound 8j was also evaluated at 10 μg/mL. It provided 100% protection activity as the positive control boscalid did (Table 4).
Molecular docking
The compound 8j was docked into the active site of Gallus gallus SDH complex to study the structure–activity relationship, and its binding affinity results showed the second highest affinities among the 21 molecules, and its binding model was also performed by the Pymol software and is shown in Fig. 5. The compound 8j was surrounded by residues (e.g., Tyr58, Ile40, Trp173, Pro169, Arg43) of the SDH protein, and H-bond was predicted between 8j and the residue Trp173, which was a similar to that of Carboxin. In addition, 2-chloro-phenyl group in Ar of 8j could form a π–π stacking interaction with Tyr58, which may contribute to its binding potency additionally. These results may explain why 8j showed good antifungal activities.
Conclusions
In summary, a series of pyrazole-4-carboxamides compounds were designed and synthesized as potential SDH inhibitors. All target compounds structures were confirmed by 1H NMR, 13C NMR and HRMS. The fungicidal activities result showed that 8j exhibited the best fungicidal potency against A. solani in vitro with EC50 value of 3.06 μg/mL and displayed 100% protective activity against A. solani in vivo at 10 μg/mL. The molecular docking results also explained the reasons for its good activities. These results support that 8j could be a potential fungicidal candidate and deserved for further studies.
Experimental
Instrumentation and materials
YASARAY 17.6.21 software was used for molecular docking. Melting points of all compounds were determined on an X-4 melting point apparatus and uncorrected. NMR spectrums were obtained in deuteron chloroform (CDCl3) and tetramethyl silane (TMS) as internal standards on a Bruker AV-400 spectrometer operating at 400 MHz for 1H NMR and 101 MHz for 13C NMR. High-resolution mass spectra (HRMS) were determined on a 7.0T FTICR-MS instrument. Crystal structure was recorded by Bruker SMART 1000 CCD diffraction meter. All solvents were of analytical grade. General procedures for the preparation of target compounds 8a–8u.
Synthesis procedure for compound 1
Forty percent methylhydrazine (28.0 mmol) in a solution of water was added dropwise to a stirred solution of ethyl 4,4-difluoro-3-oxobutanoate (24.0 mmol) dissolved in ethanol (20 mL). The mixture was adjusted to pH = 5 by concentrated hydrochloric acid and then was heated to reflux for 4 h. After the reaction was completed, it was cooled to room temperature and filtered. The filtrate was collected and the solvent was evaporated to give crude product, which was used for the next step without further purification.
Synthesis procedure for compound 2
POCl3 (50.0 mmol) was added dropwise to anhydrous N,N-dimethylformamide (DMF) under 0–5 °C, and the mixture was reacted for 30 min at this temperature, and then, a solution of compound 1 (17.0 mmol) in DMF was added dropwise. The mixture was stirred at 90–95 °C for 6 h. After the reaction was completed, it was slowly poured into ice water, stirred for 30 min until the solid precipitated and then filtered to give yellow solid crude compound, which was used for the next step without further purification.
Synthesis procedure for compound 3
Compound 3 can be synthesized according to Ref. [17]. Compound 2 (70.0 mmol), acetone (1.53 mL) and H2O (7.71 mL) were mixed, and the mixture was stirred at 60 °C for 2 min, and a solution of KMnO4 (80.0 mol) in water (15.42 mL) was slowly added and allowed it to stir at 80–85 °C for 4.5 h. When completed, it was filtered, and the filtrate was adjusted to pH = 2 by concentrated hydrochloric acid. The white solid was completely precipitated and filtered to give compound 3, which was used for the next step without further purification.
Synthesis procedure for compound 4
Sulfoxide (14.0 mmol) was added to a solution of compound 3 (1.4 mmol) in acetonitrile under ice bath, and a catalytic amount of DMF (0.1 mmol) was also added, and then, it was refluxed for 2 h. When completed, the solvent was evaporated to give compound 4, which was used for the next step without further purification.
Synthesis procedure for compounds 6a–6u
Compound 5 (4.0 mmol), Ar-boronic acid (6.0 mmol), palladium acetate (0.4 mmol), tricyclohexylphosphine (0.8 mmol), potassium phosphate (11.8 mmol, 2.5 g) and H2O (5 mL) were added to toluene (20 mL). The reaction mixture was stirred at 110 °C overnight under nitrogen. When completed, it was poured into water (50 mL) and extracted with ethyl acetate (20 mL × 3). The organic layer was collected and dried over anhydrous sodium sulfate and filtered. The solvent was evaporated to give compounds 6a–6u, which were used for the next step without further purification.
Synthesis procedure for compounds 7a–7u
Cyclopropylamine (0.78 mol) was added to a solution of compounds 6 (0.65 mol) in methanol (15 mL) and stirred about 1–2 h, and then, sodium borohydride (2.26 mol) was solely added. It was allowed to react at room temperature for 2 h. When completed, the mixture was washed with 30 mL of saturated brine and then was extracted with ethyl acetate. The solvent was evaporated and compounds 7a–7u were obtained. They can be used for the next step without further purification.
Synthesis procedure for compounds 8a-8u
Triethylamine (1.38 mol) was added to a solution of compounds 7 (1.15 mol) in anhydrous dichloromethane (10 mL), and then, a solution of compound 4 (1.38 mol) in anhydrous dichloromethane was slowly added under 0–5 °C. The reaction was stirred at room temperature for 2 h. When completed, the reaction mixture was washed with saturated brine (30 mL × 3) and dried by anhydrous sodium sulfate. After filtration, the solvent was evaporated. The residue was then purified column chromatography on silica gel to give compounds 8a–8u in 50–73% yield.
The yields, physical properties and their 1H NMR, 13C NMR, HRMS of the target compounds 8a–8u were shown as below:
-
Data for 8a: brown solid; yield 65%; m.p.: 82–83 °C; 1H NMR (CDCl3, 400 MHz) δ: 7.51–7.34 (m, 4H), 7.33–7.24 (m, 3H), 7.19 (d, J = 8.1 Hz, 1H), 6.72 (t, J = 54.9 Hz, 1H), 4.71 (s, 2H), 3.89 (s, 3H), 2.65 (s, 1H), 0.37 (m, 4H);13C NMR (CDCl3, 101 MHz) δ: 164.37 (s), 143.09 (t, J = 27.6 Hz), 139.93 (s), 139.65 (s), 136.48 (s), 133.78 (s), 131.31 (s), 129.24 (s), 128.44 (s), 127.60 (s), 127.49 (s), 127.14 (s), 112.76 (s), 110.41 (s), 108.05 (s), 47.66 (s), 36.89 (s), 30.42 (s), 8.41 (s); HRMS calcd for C22H19Cl2F2N3O (M + H)+: 450.0873, found 450.0947.
-
Data for 8b: yellow solid; yield 68%; m.p.: 114–116 °C; 1H NMR (CDCl3, 400 MHz) δ: 7.45 (s, 1H), 7.29–7.15 (m, 6H), 6.71 (t, J = 54.9 Hz, 1H), 4.70 (s, 2H), 3.88 (s, 3H), 2.65 (s, 1H), 2.40 (s, 3H), 0.36 (m, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.37 (s), 143.21 (t, J = 27.6 Hz), 139.92 (s), 137.35 (s), 136.68 (s), 136.53 (s), 133.56 (s), 131.36 (s), 129.13 (s), 127.36 (s), 127.07 (s), 115.13 (s), 112.77 (s), 110.41 (s), 108.06 (s), 47.67 (s), 36.89 (s), 30.40 (s), 21.21 (s), 8.36 (s); HRMS calcd for C23H21Cl2F2N3O (M + H)+: 464.1030, found: 464.1102.
-
Data for 8c: yellow oil; yield 59%; 1H NMR (CDCl3, 400 MHz) δ: 7.72 (s, 1H), 7.58 (t, J = 8.5 Hz, 2H), 7.48 (s, 1H), 7.32 (s, 2H), 7.26–7.18 (m, 1H), 6.75 (t, J = 54.2 Hz, 1H), 4.80 (d, J = 43.6 Hz, 2H), 3.91 (d, J = 7.5 Hz, 3H), 2.71 (s, 1H), 2.07 (s, 2H), 1.61 (s, 1H), 1.28 (t, J = 6.3 Hz, 3H), 0.39 (m, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.35 (s), 142.77 (t, J = 27.6 Hz), 139.94 (s), 137.87 (s), 136.53 (s), 134.86 (s), 133.54 (s), 131.36 (s), 130.59 (s), 129.17 (s), 127.90 (s), 126.95 (s), 112.76 (s), 110.40 (s), 108.04 (s), 47.67 (s), 36.86 (s), 30.41 (s), 28.56 (s), 15.51 (s), 8.38 (s); HRMS calcd for C24H23Cl2F2N3O (M + H)+: 478.1186, found: 478.1257.
-
Data for 8d: yellow oil; yield 63%; 1H NMR (CDCl3, 400 MHz) δ: 7.48 (d, J = 7.1 Hz, 3H), 7.24 (dt, J = 17.4, 16.6 Hz, 4H), 6.75 (dt, J = 66.8, 40.4 Hz, 1H), 5.81 (d, J = 17.6 Hz, 1H), 5.31 (d, J = 10.9 Hz, 1H), 4.72 (s, 2H), 3.88 (s, 3H), 2.66 (s, 1H), 0.37 (m, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.38 (s), 143.08 (t, J = 27.6 Hz), 139.55 (s), 139.08 (s), 136.91 (s), 136.55 (s), 136.24 (s), 133.85 (s), 131.23 (s), 129.48 (s), 127.49 (s), 127.18 (s), 126.26 (s), 114.49 (s), 112.78 (s), 110.42 (s), 108.06 (s), 47.68 (s), 36.89 (s), 30.45 (s), 8.43 (s); HRMS calcd for C24H21Cl2F2N3O (M + H)+: 476.1030, found: 476.1101.
-
Data for 8e: brown crystal; yield 73%; m.p.: 125–127 °C; 1H NMR (400 MHz, CDCl3) δ: 7.46 (s, 1H), 7.32–7.25 (m, 3H), 7.20 (dd, J = 13.8, 8.0 Hz, 3H), 6.73 (t, J = 54.9 Hz, 1H), 4.72 (s, 2H), 3.89 (s, 5H), 2.97 (dt, J = 13.8, 6.8 Hz, 1H), 2.66 (s, 1H), 1.30 (d, J = 6.9 Hz, 6H), 0.37 (m, 4H); 13C NMR(CDCl3, 101 MHz) δ: 164.35 (s), 148.25 (s), 143.08 (t, J = 27.6 Hz), 139.95 (s), 137.01 (s), 136.56 (s), 133.54 (s), 131.38 (s), 129.16 (s), 127.37 (s), 127.06 (s), 126.48 (s), 112.76 (s), 110.41 (s), 108.05 (s), 47.71 (s), 36.87 (s), 33.85 (s), 30.43 (s), 23.99 (s), 8.40 (s); HRMS calcd for C25H25Cl2F2N3O (M + H)+: 492.1343, found: 492.1414.
-
Data for 8f: yellow solid; yield 74%; m.p.: 106–108 °C; 1H NMR (CDCl3, 400 MHz)δ: 7.44 (d, J = 8.1 Hz, 3H), 7.29–7.13 (m, 4H), 6.71 (t, J = 54.8 Hz, 1H), 4.72 (s, 2H), 3.88 (s, 3H), 2.66 (s, 1H), 1.36 (s, 9H), 0.36 (m, 4H); 13C NMR(CDCl3, 101 MHz) δ: 164.36 (s), 143.08 (t, J = 27.6 Hz), 139.88 (s), 136.56 (s), 133.53 (s), 131.40 (s), 128.91 (s), 127.33 (s), 125.33 (s), 112.76 (s), 112.08 (s), 110.41 (s), 109.73 (s), 108.05 (s), 107.37 (s), 47.73 (s), 36.89 (s), 34.62 (s), 31.37 (s), 30.45 (s), 8.42 (s); HRMS calcd for C26H27Cl2F2N3O (M + H)+: 506.1499, found: 506.1568.
-
Data for 8g: brown solid; yield 43%; m.p.: 53–54 °C; 1H NMR (CDCl3, 400 MHz)δ: 7.49 (s, 1H), 7.38 (d, J = 5.4 Hz, 1H), 7.30 (dd, J = 8.1, 1.7 Hz, 1H), 7.27–7.08 (m, 4H), 6.71 (t, J = 54.7 Hz, 1H), 4.61 (s, 2H), 3.87 (s, 3H), 2.66 (s, 1H), 0.38 (m, 4H); 13C NMR(CDCl3, 101 MHz)δ: 164.34 (s), 160.68 (s), 158.24 (s), 143.07 (t, J = 27.5 Hz), 137.67 (s), 134.54 (s), 133.32 (s), 131.68 (s), 130.03 (s), 129.95 (s), 127.56 (s), 127.26 (s), 124.35 (s), 115.65 (s), 112.75 (s), 110.40 (s), 108.04 (s), 47.57 (s), 36.87 (s), 30.42 (s), 8.40 (s); HRMS calcd for C22H18Cl2F3N3O (M + H)+: 468.0799, found: 468.0854.
-
Data for 8h: brown oil; yield 58%; 1H NMR (CDCl3, 400 MHz) δ: 7.48 (s, 1H), 7.45–7.36 (m, 1H), 7.30 (d, J = 8.2 Hz, 1H), 7.18 (d, J = 8.0 Hz, 1H), 7.09 (dd, J = 8.0, 6.1 Hz, 2H), 7.02 (d, J = 7.6 Hz, 1H), 6.73 (t, J = 55.2 Hz, 1H), 4.69 (s, 2H), 3.90 (s, 3H), 2.68 (s, 1H), 0.40 (m, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.36 (s), 163.82 (s), 161.36 (s), 142.77 (t, J = 77.2 Hz), 138.56 (s), 136.50 (s), 134.28 (s), 131.13 (s), 130.08 (s), 129.99 (s), 127.61 (s), 127.27 (s), 125.10 (s), 114.68 (s), 112.77 (s), 110.42 (s), 108.06 (s), 47.66 (s), 36.89 (s), 30.48 (s), 8.48 (s); HRMS calcd for C22H18Cl2F3N3O (M + H)+: 468.0799, found: 468.0851.
-
Data for 8i: brown solid; yield 63%; m.p.: 100–102 °C; 1H NMR (CDCl3, 400 MHz)δ: 7.46 (s, 1H), 7.27 (dd, J = 6.9, 5.1 Hz, 3H), 7.14 (dd, J = 16.7, 8.3 Hz, 3H), 6.72 (t, J = 54.6 Hz, 1H), 4.67 (s, 2H), 3.89 (s, 3H), 2.68 (s, 1H), 0.39 (m, 4H); 13C NMR(CDCl3, 101 MHz) δ: 164.34 (s), 163.55 (s), 161.09 (s), 143.07 (t, J = 27.7 Hz), 138.80 (s), 136.61 (s), 135.58 (s), 133.98 (s), 131.37 (s), 130.90 (s), 130.82 (s), 127.50 (s), 127.20 (s), 115.53 (s), 115.32 (s), 112.79 (s), 110.43 (s), 47.70 (s), 36.89 (s), 30.48 (s), 8.43 (s); HRMS calcd for C22H18Cl2F3N3O (M + H)+: 468.0799, found: 468.0853.
-
Data for 8j: brown oil; yield 66%; 1H NMR (CDCl3, 400 MHz) δ: 7.48 (s, 2H), 7.39–7.28 (m, 3H), 7.27 (s, 1H), 7.13 (d, J = 7.5 Hz, 1H), 6.72 (t, J = 54.4 Hz, 1H), 4.53 (s, 2H), 3.90 (s, 3H), 2.73 (s, 1H), 0.62 (s, 1H), 0.42 (m, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.42 (s), 142.96 (t, J = 27.8 Hz), 138.17 (s), 137.33 (s), 136.81 (s), 134.45 (s), 133.87 (s), 133.45 (s), 131.37 (s), 131.21 (s), 129.67 (s), 129.43 (s), 127.20 (s), 127.13 (s), 112.76 (s), 110.41 (s), 108.05 (s), 47.69 (s), 36.90 (s), 30.51 (s), 8.65 (s); HRMS calcd for C22H18Cl3F2N3O (M + H)+: 484.0484, found: 484.0559.
-
Data for 8k: brown oil; yield 77%; 1H NMR (CDCl3, 400 MHz) δ: 7.48 (s, 1H), 7.37 (d, J = 4.1 Hz, 2H), 7.32–7.27 (m, 2H), 7.17 (t, J = 9.0 Hz, 2H), 6.72 (t, J = 54.8 Hz, 1H), 4.68 (s, 2H), 3.89 (s, 3H), 2.67 (s, 1H), 0.40 (m, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.35 (s), 143.11 (t, J = 27.7 Hz), 141.43 (s), 138.40 (s), 136.51 (s), 134.34 (s), 131.16 (s), 129.74 (s), 129.27 (s), 127.79 (s), 127.68 (s), 127.49 (s), 127.30 (s), 114.98 (s), 112.77 (s), 110.41 (s), 108.06 (s), 47.69 (s), 36.91 (s), 30.47 (s), 8.52 (s); HRMS calcd for C22H18Cl3F2N3O (M + H)+: 484.0484, found: 484.0556.
-
Data for 8l: brown solid; yield 67%; m.p.: 113–114 °C; 1H NMR (CDCl3, 400 MHz) δ: 7.51–7.37 (m, 3H), 7.29 (dd, J = 8.2, 1.9 Hz, 2H), 7.25–7.20 (m, 1H), 7.15 (d, J = 8.1 Hz, 1H), 6.72 (t, J = 54.6 Hz, 1H), 4.67 (s, 2H), 3.89 (s, 3H), 2.68 (s, 1H), 0.40 (m, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.34 (s), 143.04 (t, J = 27.7 Hz), 138.55 (s), 138.04 (s), 136.51 (s), 134.16 (s), 133.75 (s), 131.19 (s), 130.55 (s), 128.66 (s), 127.51 (s), 127.25 (s), 126.31 (s), 114.95 (s), 112.77 (s), 110.41 (s), 108.06 (s), 47.70 (s), 36.89 (s), 30.49 (s), 8.45 (s); HRMS calcd for C22H18Cl3F2N3O (M + H)+: 484.0484, found: 484.0555.
-
Data for 8m: yellow oil; yield 55%; 1H NMR (CDCl3, 400 MHz) δ: 7.49 (d, J = 22.4 Hz, 2H), 7.32 (t, J = 7.9 Hz, 2H), 7.21 (d, J = 7.9 Hz, 1H), 7.09 (d, J = 8.0 Hz, 1H), 6.72 (t, J = 54.7 Hz, 1H), 4.51 (s, 2H), 3.90 (s, 3H), 2.75 (s, 1H), 0.44 (m, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.36 (s), 143.06 (t, J = 27.7 Hz), 137.45 (s), 136.74 (s), 135.60 (s), 134.81 (s), 134.65 (s), 134.29 (s), 133.84 (s), 132.11 (s), 131.14 (s), 129.50 (s), 128.90 (s), 127.29 (s), 112.79 (s), 110.43 (s), 108.08 (s), 47.74 (s), 36.89 (s), 30.59 (s), 8.59 (s); HRMS calcd for C22H17Cl4F2N3O (M + H)+: 518.0094, found: 518.0163.
-
Data for 8n: yellow oil; yield 71%; 1H NMR (CDCl3, 400 MHz) δ: 7.88 (s, 1H), 7.82 (d, J = 7.5 Hz, 1H), 7.71 (d, J = 11.3 Hz, 1H), 7.68 (s, 1H), 7.66 (s, 1H), 7.62 (s, 1H), 7.36 (d, J = 7.9 Hz, 1H), 6.71 (t, J = 54.6 Hz, 1H), 4.78 (s, 2H), 3.88 (s, 3H), 2.72 (s, 1H), 0.41 (m, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.51 (s), 143.07 (t, J = 27.8 Hz), 141.17 (s), 139.63 (s), 135.47 (s), 132.73 (s), 131.21 (q, J = 25.1 Hz), 130.76 (s), 130.53 (s), 129.32 (s), 128.99 (s), 126.29 (s), 125.95 (s), 124.35 (s), 123.94 (s), 122.83 (s), 112.90 (s), 110.55 (s), 47.97 (s), 36.87 (s), 30.66 (s), 8.43 (s); HRMS calcd for C23H18Cl2F5N3O (M + H)+: 518.0747, found: 518.0828.
-
Data for 8o: brown solid; yield 95%; m.p.: 129–131 °C; 1H NMR (CDCl3, 400 MHz) δ: 7.46 (s, 1H), 7.42–7.35 (m, 1H), 7.28 (dd, J = 8.2, 1.9 Hz, 1H), 7.14 (d, J = 7.8 Hz, 2H), 7.03 (t, J = 7.3 Hz, 1H), 6.97 (d, J = 7.7 Hz, 1H), 6.72 (t, J = 54.7 Hz, 1H), 4.88 (s, 1H), 4.23 (s, 1H), 3.89 (s, 3H), 3.79 (s, 3H), 2.63 (s, 1H), 0.36 (d, J = 23.6 Hz, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.39 (s), 156.51 (s), 143.21 (t, J = 27.5 Hz), 137.86 (s), 136.32 (s), 133.69 (s), 131.57 (s), 131.07 (s), 129.50 (s), 128.39 (s), 127.06 (s), 120.74 (s), 112.76 (s), 110.64 (s), 110.41 (s), 108.05 (s), 99.99 (s), 55.41 (s), 47.46 (s), 36.88 (s), 30.31 (s), 8.68 (s); HRMS calcd for C23H21Cl2F2N3O2 (M + H)+: 480.0979, found: 480.1050.
-
Data for 8p: brown solid; yield 60%; m.p.: 94–95 °C; 1H NMR (CDCl3, 400 MHz)δ: 7.46 (s, 1H), 7.35 (t, J = 7.8 Hz, 1H), 7.31–7.27 (m, 1H), 7.19 (d, J = 8.2 Hz, 1H), 6.96–6.86 (m, 2H), 6.86–6.57 (m, 2H), 4.71 (s, 2H), 3.90 (s, 2H), 3.84 (s, 3H), 2.67 (s, 1H), 0.39 (m, 4H); 13C NMR(CDCl3, 101 MHz) δ: 164.45 (s), 159.50 (s), 143.21 (t, J = 27.5 Hz), 141.02 (s), 139.75 (s), 136.48 (s), 133.83 (s), 131.16 (s), 129.48 (s), 127.40 (s), 127.09 (s), 126.37 (s), 121.65 (s), 115.04 (s), 112.93 (s), 110.41 (s), 108.05 (s), 55.32 (s), 47.71 (s), 36.89 (s), 30.48 (s), 8.42 (s); HRMS calcd for C23H21Cl2F2N3O2 (M + H)+: 480.0979, found: 480.1051.
-
Data for 8q: brown solid; yield 68%; m.p.: 127–128 °C; 1H NMR (CDCl3, 400 MHz) δ: 7.45 (s, 1H), 7.28 (d, J = 2.1 Hz, 1H), 7.20 (dd, J = 19.9, 8.0 Hz, 3H), 6.97 (d, J = 8.3 Hz, 2H), 6.72 (t, J = 55.0 Hz, 1H), 4.71 (s, 2H), 3.89 (s, 3H), 3.86 (s, 3H), 2.66 (s, 1H), 0.37 (m, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.35 (s), 159.11 (s), 143.07 (t, J = 27.8 Hz), 139.60 (s), 136.61 (s), 133.46 (s), 131.92 (s), 131.45 (s), 130.36 (s), 127.38 (s), 127.07 (s), 113.85 (s), 112.77 (s), 110.41 (s), 108.06 (s), 55.35 (s), 47.66 (s), 36.88 (s), 30.41 (s), 8.38 (s); HRMS calcd for C23H21Cl2F2N3O2 (M + H)+: 480.0979, found: 480.1046.
-
Data for 8r: brown solid; yield 71%; m.p.: 69–70 °C; 1H NMR (CDCl3, 400 MHz) δ: 7.55 (d, J = 10.1 Hz, 2H), 7.40 (s, 1H), 7.28 (dd, J = 8.5, 2.0 Hz, 1H), 6.75 (t, J = 54.4 Hz, 1H), 6.51 (s, 2H), 4.96 (s, 2H), 3.91 (s, 3H), 2.77 (s, 1H), 0.53 (m, 4H); 13C NMR(CDCl3, 101 MHz) δ: 164.54 (s), 142.60 (t, J = 27.8 Hz), 138.77 (s), 135.60 (s), 134.04 (s), 129.45 (s), 128.08 (s), 127.67 (s), 127.39 (s), 126.45 (s), 114.91 (s), 112.76 (s), 111.57 (s), 110.40 (s), 109.36 (s), 48.69 (s), 36.89 (s), 30.79 (s), 8.51 (s); HRMS calcd for C20H17Cl2F2N3O2 (M + H)+: 440.0666, found: 440.0738.
-
Data for 8s: yellow oil; yield 73%; 1H NMR (CDCl3, 400 MHz) δ: 7.46 (s, 1H), 7.39 (d, J = 4.3 Hz, 1H), 7.33 (d, J = 8.1 Hz, 1H), 7.28 (d, J = 1.9 Hz, 1H), 7.15–7.09 (m, 1H), 7.03 (s, 1H), 6.74 (t, J = 55.0 Hz, 1H), 4.86 (s, 2H), 3.90 (s, 3H), 2.71 (s, 1H), 0.44 (m, 4H); 13C NMR (CDCl3, 101 MHz) δ: 165.12 (s), 146.34 (t, J = 27.8 Hz), 140.20 (s), 137.84 (s), 134.42 (s), 132.51 (s), 132.07 (s), 131.42 (s), 127.94 (s), 127.51 (s), 127.35 (s), 126.03 (s), 112.49 (s), 110.14 (s), 107.79 (s), 48.65 (s), 36.91 (s), 31.14 (s), 10.11 (s); HRMS calcd for C20H17Cl2F2N3OS (M + H)+: 456.0437, found: 456.0509.
-
Data for 8t: brown oil; yield 68%; 1H NMR (CDCl3, 400 MHz) δ: 7.91 (dd, J = 7.8, 4.6 Hz, 2H), 7.62–7.36 (m, 7H), 7.25 (t, J = 6.3 Hz, 1H), 6.71 (t, J = 54.8 Hz, 1H), 4.44 (d, J = 34.6 Hz, 2H), 3.87 (s, 3H), 2.65 (s, 1H), 0.28 (m, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.39 (s), 143.06 (t, J = 27.5 Hz), 137.97 (s), 137.76 (s), 137.10 (s), 134.17 (s), 133.55 (s), 131.96 (s), 128.44 (s), 128.33 (s), 127.14 (s), 126.58 (s), 125.62 (s), 125.33 (s), 112.78 (s), 110.42 (s), 108.06 (s), 47.85 (s), 36.88 (s), 30.42 (s), 8.40 (s); HRMS calcd for C26H21Cl2F2N3O (M + H)+: 500.1030, found: 500.1099.
-
Data for 8u: yellow crystal; yield 70%; m.p.: 157–158 °C; 1H NMR (CDCl3, 400 MHz) δ: 7.71 (d, J = 8.0 Hz, 1H), 7.63 (d, J = 7.5 Hz, 1H), 7.54 (d, J = 7.9 Hz, 1H), 7.48 (s, 1H), 7.34 (dd, J = 13.7, 5.1 Hz, 2H), 7.28 (d, J = 7.7 Hz, 1H), 6.96–6.58 (m, 2H), 5.07 (s, 2H), 3.90 (s, 3H), 2.79 (d, J = 4.3 Hz, 1H), 0.51 (d, J = 12.5 Hz, 4H); 13C NMR (CDCl3, 101 MHz) δ: 164.49 (s), 154.77 (s), 153.94 (s), 143.19 (t, J = 27.5 Hz), 136.88 (s), 135.23 (s), 130.37 (s), 128.71 (s), 127.79 (s), 127.53 (s), 124.81 (s), 123.20 (s), 121.18 (s), 115.02 (s), 112.85 (s), 111.28 (s), 110.50 (s), 108.14 (s), 106.06 (s), 48.76 (s), 36.91 (s), 30.84 (s), 8.54 (s); HRMS calcd for C24H19Cl2F2N3O2 (M + H)+: 490.0822, found: 490.0893.
Determination of antifungal activity
In vitro antifungal activity
In vitro antifungal activities against Fusarium oxysporum, Pythium aphanidermatum, Setosphaeria turcica, Alternaria solani, Fusarium graminearum, Pellicularia sasakii, Physalospora piricola, Sclerotinia sclerotiorum and Botrytis cinereal of all compounds were determined according to an established method [18]. The inhibition activities of any target compounds showed that more than 90% at 100 μg/mL were further evaluated at 50 μg/mL, and the median effective concentration (EC50) of 8j was determined by a reported method [19].
In vivo fungicidal activity
The in vivo fungicidal activity of 8j against A. solani on tomato leaves was evaluated according to the following procedures [20]. The target compound solution (10 μg/mL) was sprayed onto the tomato plants. The control plants were sprayed with water. After 24 h, the tomato plants were infected with spore suspension of the A. solani (concentration: 1 × 105 spore/mL) and then cultured in the greenhouse. The disease percentage of diseased control plants was compared with the healthy control plants by considering the disease control is set as 100 and no disease as 0. Boscalid was used as a positive control.
Molecular docking
The molecular structure 8j was drawn by ChemDraw Ultra 14.0 and then saved as mol2 file. The crystal structure of SDH of Gallus gallus (PDB: 2FBW) was obtained from the PDB database (https://www.rcsb.org/structure/2fbw) [21], which was applied as the target for molecular docking by YASARA software. The docking models were analyzed by the AutoDock Vina program. Pymol software was used to show the docking models.
References
Lamberth C (2018) Agrochemical lead optimization by scaffold hopping. Pest Manage Sci 74(2):282–292. https://doi.org/10.1002/ps.4755
Chen L, Wu Q, Fan Z, Li H, Li J, Hu W, Liu X, Belskaya NP, Glukhareva T, Zhao B (2018) Design, synthesis and biological evaluation of isothiazole based 1,2,4-trizaole derivatives. Chin J Chem 36(8):731–736. https://doi.org/10.1002/cjoc.201700765
Bardas GA, Veloukas T, Koutita O, Karaoglanidis GS (2010) Multiple resistance of Botrytis cinerea from kiwi fruit to SDHIs, QoIs and fungicides of other chemical groups. Pest Manag Sci 66(9):967–973. https://doi.org/10.1002/ps.1968
He L, Cui K, Song Y, Mu W, Liu F (2018) High-efficiency control of gray mold by the novel SDHI fungicide benzovindiflupyr combined with a reasonable application approach of dipping flower. J Agric Food Chem 66(26):6692–6698. https://doi.org/10.1021/acs.jafc.8b01936
Ishii H, Zhen F, Hu M, Li X, Schnabel G (2016) Efficacy of SDHI fungicides, including benzovindiflupyr, against Colletotrichum species. Pest Manage Sci 72(10):1844–1853. https://doi.org/10.1002/ps.4216
Xiong L, Shen Y-Q, Jiang L-N, Zhu X-L, Yang W-C, Huang W, Yang G-F (2015) Succinate dehydrogenase: an ideal target for fungicide discovery. ACS Symp Ser 1204:175–194. https://doi.org/10.1021/bk-2015-1204.ch013
Gutierrez-Alonso O, Hawkins NJ, Cools HJ, Shaw MW, Fraaije BA (2017) Dose-dependent selection drives lineage replacement during the experimental evolution of SDHI fungicide resistance in Zymoseptoria tritici. Evol Appl 10(10):1055–1066. https://doi.org/10.1111/eva.12511
Sierotzki H, Scalliet G (2013) A review of current knowledge of resistance aspects for the next-generation succinate dehydrogenase inhibitor fungicides. Phytopathology 103(9):880–887. https://doi.org/10.1094/phyto-01-13-0009-rvw
Zhang L, Li W, Xiao T, Song Z, Csuk R, Li S (2018) Design and discovery of novel chiral antifungal amides with 2-(2-oxazolinyl)aniline as a promising pharmacophore. J Agric Food Chem 66(34):8957–8965. https://doi.org/10.1021/acs.jafc.8b02778
Yang D, Zhao B, Fan Z, Yu B, Zhang N, Li Z, Zhu Y, Zhou J, Kalinina TA, Glukhareva TV (2019) Synthesis and biological activity of novel succinate dehydrogenase inhibitor derivatives as potent fungicide candidates. J Agric Food Chem 67(47):13185–13194. https://doi.org/10.1021/acs.jafc.9b05751
Xiong L, Li H, Jiang L-N, Ge J-M, Yang W-C, Zhu XL, Yang G-F (2017) Structure-Based Discovery of Potential Fungicides as Succinate Ubiquinone Oxidoreductase Inhibitors. J Agric Food Chem 65(5):1021–1029. https://doi.org/10.1021/acs.jafc.6b05134
Zhang A, Yue Y, Yang J, Shi J, Tao K, Jin H, Hou T (2019) Design, synthesis, and antifungal activities of novel aromatic carboxamides containing a diphenylamine scaffold. J Agric Food Chem 67(17):5008–5016. https://doi.org/10.1021/acs.jafc.9b00151
Zhang A, Yue Y, Yang Y, Yang J, Tao K, Jin H, Hou T (2019) Discovery of N-(4-fluoro-2-(phenylamino)phenyl)-pyrazole-4-carboxamides as potential succinate dehydrogenase inhibitors. Pestic Biochem Physiol 158:175–184. https://doi.org/10.1016/j.pestbp.2019.05.007
Lv X-H, Ren Z-L, Liu P, Li B-X, Li Q-S, Chu M-J, Cao H-Q (2017) Design, synthesis and biological evaluation of novel nicotinamide derivatives bearing a substituted pyrazole moiety as potential SDH inhibitors. Pest Manage Sci 73(8):1585–1592. https://doi.org/10.1002/ps.4488
Altomare A, Burla MC, Camalli M, Cascarano GL, Giacovazzo C, Guagliardi A, Moliterni AGG, Polidori G, Spagna R (1999) SIR97: a new tool for crystal structure determination and refinement. J Appl Crystallogr 32(1):115–119. https://doi.org/10.1107/S0021889898007717
Guo X, Zhao B, Fan Z, Yang D, Zhang N, Wu Q, Yu B, Zhou S, Kalinina TA, Belskaya NP (2019) Discovery of novel thiazole carboxamides as antifungal succinate dehydrogenase inhibitors. J Agric Food Chem 67(6):1647–1655. https://doi.org/10.1021/acs.jafc.8b06935
Wen H-l, Kang J-j, Dai B, Deng R-h, Hu H-w (2015) Syntheses, crystal structures and antibacterial activities of 5-chloro-3-methyl-1-phenyl-1H-pyrazole-4-carboxylic acid and its copper (II) complex. Chin J Struct Chem 34(1):33–40. https://doi.org/10.14102/j.cnki.0254-5861.2011-0447
Chen L, Zhao B, Fan Z, Liu X, Wu Q, Li H, Wang H (2018) Synthesis of novel 3,4-chloroisothiazole-based imidazoles as fungicides and evaluation of their mode of action. J Agric Food Chem 66(28):7319–7327. https://doi.org/10.1021/acs.jafc.8b02332
Fan Z, Yang Z, Zhang H, Mi N, Wang H, Cai F, Zuo X, Zheng Q, Song H (2010) Synthesis, crystal structure, and biological activity of 4-methyl-1,2,3-thiadiazole-containing 1,2,4-triazolo[3,4-b][1,3,4]thiadiazoles. J Agric Food Chem 58(5):2630–2636
Chen L, Zhao B, Fan Z, Hu M, Li Q, Hu W, Li J, Zhang J (2019) Discovery of novel isothiazole, 1,2,3-thiadiazole, and thiazole-based cinnamamides as fungicidal candidates. J Agric Food Chem 67(45):12357–12365. https://doi.org/10.1021/acs.jafc.9b03891
Huang L-s, Sun G, Cobessi D, Wang AC, Shen JT, Tung EY, Anderson VE, Berry EA (2006) 3-Nitropropionic acid is a suicide inhibitor of mitochondrial respiration that, upon oxidation by complex II, forms a covalent adduct with a catalytic base arginine in the active site of the enzyme. J Biol Chem 281(9):5965–5972. https://doi.org/10.1074/jbc.M511270200
Acknowledgements
This work was supported in part by the National Natural Science Foundation of China (No. 31871981); Youth Natural Science Foundation of Hebei Province (No. B2019204030); and the starting Scientific Research Foundation for the introduced talents of Hebei Agricultural University (No. 201842); Modern Agriculture Industry Technology System Innovation Team of Phase II of Hebei Province (No. HB2018020205).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dong, C., Gao, W., Li, X. et al. Synthesis of pyrazole-4-carboxamides as potential fungicide candidates. Mol Divers 25, 2379–2388 (2021). https://doi.org/10.1007/s11030-020-10127-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-020-10127-w