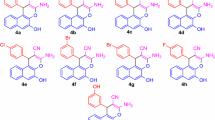
Abstract
A concise, efficient one-pot synthesis of functionalized chromeno[4,3-b]pyridine derivatives via a three-component reaction of 4-oxo-4H-chromene-3-carbaldehydes, malononitrile or cyanoacetates, and aromatic amines under catalyst-free conditions in an environmentally friendly medium (ethanol–water, 3:1 v/v) is described. This synthesis involves a group-assisted purification process, which avoids traditional recrystallization and chromatographic purification methods.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nitrogen-containing heterocyclic compounds are important because of their presence in a broad range of natural products and synthetic organic molecules with various biological activities [1, 2]. The chromenopyridine nucleus has a wide range of pharmacological activities, including antibacterial [3], antiinflammotory [4, 5], antimicrobial [6, 7], anti-HIV [8], and anticancer [9]. Several methods have been developed for the construction of the chromenopyridine skeleton [10,11,12,13,14,15,16,17,18], but these methods involve long multistep processes and afford low yields of the desired products. There is therefore a need to develop concise and efficient methods for the synthesis of these important molecules [19,20,21].
The development of a simple and eco-friendly protocol for the construction of nitrogen-containing heterocyclic compound libraries of medical motifs is an attractive area of research in both academia and the pharmaceutical industry. Multicomponent reactions (MCRs) are promising and powerful tools in organic, combinatorial, and medicinal chemistry because of their atom economy, high complexity and diversity of products, multiple bond formation efficiency, and environmental friendliness [22,23,24,25,26]. These features make MCRs suitable for the easy construction of complex heterocyclic scaffolds from readily available starting materials [27,28,29]. In the past decade, various MCRs have been used for the construction of complex organic heterocyclic molecules [30,31,32,33,34,35].


The development of environmentally friendly synthetic methods is a challenge in modern organic synthesis. The need to reduce the amount of toxic wastes and byproducts arising from chemical processes has resulted in an increasing emphasis on the use of less-toxic and environmentally compatible materials in the design of new synthetic methods. Traditional purification methods such as recrystallization and column chromatography have problems in terms of consumption of organic solvents and energy, waste generation, and pollution. The concept of group-assisted purification (GAP) techniques, which avoid traditional recrystallization and chromatographic purification methods and reduce waste generation from silica and solvents, particularly toxic solvents, was first developed by the Li group in the design of asymmetric synthesis of new imine reagents [36, 37]. To date, GAP chemistry has been used in many asymmetric synthetic reactions [38,39,40,41,42,43] and MCRs [44,45,46,47,48,49]. As part of our current studies on the development of environmentally friendly routes to heterocyclic systems [50,51,52,53,54,55], we now report an efficient and clean synthesis of chromeno[4,3-b]pyridine derivatives under catalyst-free conditions in an environmentally friendly medium (ethanol–water, 3:1 v/v).
Results and discussion
We initially evaluated the three-component reaction of a 1:1:2 mixtures of 4-oxo-4H-chromene- 3-carbaldehyde (1a), ethyl cyanoacetate (2a), and 4-methylaniline (3a) under various conditions. The results are summarized in Table 1. The desired product 4a was obtained in 89% yield when the reaction was carried out in ethanol at 80 \({^{\circ }}\)C for 2 h under catalyst-free conditions (Table 1, entry 1). Various solvents were then evaluated to determine the impact of the solvent on the yield. Of all the solvents tested, i.e., ethanol, toluene, 1,4-dioxane, water, chloroform, and DMF, ethanol gave the best result (Table 1, entries 1–6). Water is a greener solvent; therefore, we evaluated the effect on the yield of mixing ethanol and water in different ratios. Screening experiments showed that a 3:1 (v/v) mixture of ethanol and water was the best solvent for this transformation (Table 1, entries 7–11). The optimum reaction temperature was determined by performing the reaction at ambient temperature, room temperature, and 40, 60, and 80 \({^{\circ }}\)C; the best reaction temperature was 80 \({^{\circ }}\)C (Table 1, entries 11 and 12–14). These experiments showed that the optimum reaction conditions were ethanol–water (3:1 v/v) as the solvent, 80 \({^{\circ }}\)C for 2 h, and catalyst-free conditions.
With the optimum reaction conditions in hand, we then evaluated the substrate scope using various 4-oxo-4H-chromene-3-carbaldehydes, malononitrile or cyanoacetates, and amines. The results are summarized in Table 2. In this reaction, when malononitrile, methyl cyanoacetate, and ethyl cyanoacetate were used, the desired products were obtained in excellent yields. The data in Table 2 show that 4-oxo- 4H-chromene-3-carbaldehydes with no substituents or with electron-withdrawing groups were tolerated under the reaction conditions. However, when 4-oxo-4H-chromene-3-carbaldehydes with electron-donating groups were used, the desired products were not obtained (Table 2, entries 31–33). It can been seen from Table 2 that aromatic amines with either electron-donating or electron-withdrawing groups were also tolerated under these reaction conditions. However, when an aliphatic amine or heterocyclic amine was used in this transformation, the desired products 4 were not obtained (Table 2, entries 34 and 35).
It is important that this synthesis followed a GAP process, which avoids traditional recrystallization or column chromatographic purification methods. Pure products were obtained simply by filtration and washing the solid with a little cold ethanol.
The structures of compounds 4 were identified from their IR, \(^{1}\)H NMR, and \(^{13}\)C NMR spectra, and by HRMS analysis. The structure of compound 4e was further confirmed using single-crystal X-ray diffraction analysis (Fig. 1).
Based on the experimental observations, we propose the following mechanism for this new three-component reaction (Scheme 1). The initial Knoevenagel condensation of a 4-oxo-4H-chromene- 3-carbaldehyde 1 with malononitrile or a cyanoacetate 2 gives intermediate A. Michael addition of an aromatic amine 3 to intermediate A gives intermediate B. Then another aromatic amine 3 adds to intermediate B to give intermediate C, which tautomerizes to give intermediate D. Intermediate D undergoes intramolecular cyclization and dehydration to give the product 4. When the 4-oxo-4H-chromene- 3-carbaldehydes with electron-donating groups (such as methyl and methoxy) at \(\hbox {C}_{6}\) position were used, the desired products were not obtained. The reason was that the electron-donating groups reduced the reaction activity of intermediate A to anilines 3.
In summary, we have developed a highly efficient, catalyst-free, green protocol for the one-pot three-component synthesis of chromeno[4,3-b] pyridine derivatives; these occur as structural units in a number of biologically active compounds. This protocol has the advantages of mild reaction conditions, high yields, convenient operation, and environmental friendliness.
Experimental section
General
All reagents and solvents were commercially available with analytical grade and used as received. All evaporations of organic solvents were carried out with a rotary evaporator in conjunction with a water aspirator. Melting points were measured using an electrothermal XT-5 apparatus and uncorrected. IR spectra were recorded with a Varian F-1000 spectrometer in KBr with absorption in \(\hbox {cm}^{-1}\). \(^{1}\)H NMR and \(^{13}\)C NMR spectra were obtained in \(\hbox {CDCl}_{3}\) or DMSO-\(d_{6}\) solution, using a Bruker 400 MHz spectrometer. J values are reported in hertz. Chemical shifts are expressed in ppm downfield from internal standard TMS. The abbreviations used for NMR signals are: s = singlet, d = doublet, t = triplet, q = quartet, and m = multiplet. HRMS analyses were carried out using a Bruker micrOTOF-Q instrument. X-ray data were collected on a Bruker SMART diffractometer. 4-oxo-4H-chromene-3-carbaldehydes were obtained from Alfa Aesar Company.
General procedure for synthesis of functionalized chromeno[4,3-b]pyridine derivatives 4
A 25-mL flask was charged with a 4-oxo-4H-chromene-3-carbaldehyde 1 (1 mmol), malononitrile or a cyanoacetate 2 (1 mmol), an aromatic amine 3 (2 mmol), ethanol (1.5 mL), and water (0.5 mL). The mixture was stirred at 80 \({^{\circ }}\)C for 2 h. After completion of the reaction (confirmed by TLC), the reaction mixture was cooled to room temperature. The crystalline solids were collected and washed with a small amount of cold ethanol to give the pure products 4a–4d’ for analysis.
Ethyl 2,5-bis(p-tolylamino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4a)
Yellow solid, yield 88%. m.p. 212–214 \({^{\circ }}\)C. IR (KBr): 3450, 2925, 1676, 1615, 1526, 1415, 1230, 1104, 939, 813, 793, 693 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.25 (s, 1H, NH), 8.29 (s, 1H, ArH), 8.13–8.11 (m, 1H, ArH), 7.73 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH), 7.40–7.35 (m, 1H, ArH), 7.24 (d, \(J = 8.0\hbox { Hz}\), 2H, ArH), 7.14 (t, \(J = 6.8 \hbox { Hz}\), 1H, ArH), 6.98 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH), 6.91–6.88 (m, 2H, ArH), 6.83 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH + CH), 6.74 (d, \(J = 8.8\hbox { Hz}\), 1H, NH), 4.37 (q, \(J = 7.2\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 2.32 (s, 3H, \(\hbox {CH}_{3}\)), 2.19 (s, 3H, \(\hbox {CH}_{3}\)), 1.35 (t, \(J = 7.2\hbox { Hz}\), 3H, \(\hbox {CH}_{3}\)). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \)(ppm): 167.2, 156.0, 154.6, 142.0, 138.3, 137.1, 132.2, 129.8, 129.3, 125.6, 122.0, 121.7, 120.8, 118.4, 115.8, 114.6, 106.1, 100.0, 81.9, 61.3, 20.9, 20.5, 14.3. HRMS (ESI) Calcd. for \(\hbox {C}_{29}\hbox {H}_{28}\hbox {N}_{3}\hbox {O}_{3}\) ([M + H]\(^{+})\): 466.2131. Found: 466.2132.
Ethyl 2,5-bis(phenylamino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4b)
Yellow solid, yield 90%. m.p. 230–232 \({^{\circ }}\)C. IR (KBr): 3394, 2970, 1679, 1602, 1519, 1436, 1371, 1293, 1231, 1105, 922, 794, 749 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.35 (s, 1H, NH), 8.33 (s, 1H, ArH), 8.15 (d, \(J = 7.6\hbox { Hz}\), 1H, ArH), 7.86 (d, \(J = 8.0\hbox { Hz}\), 2H, ArH), 7.46–7.37 (m, 3H, ArH), 7.20–7.06 (m, 5H, ArH), 6.92 (d, \(J = 7.6\hbox { Hz}\), 3H, ArH), 6.79 (t, \(J = 8.8\hbox { Hz}\), 1H, CH), 6.73 (t, \(J = 7.2\hbox { Hz}\), 1H, NH), 4.38 (q, \(J = 7.2\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 1.36 (t, \(J = 7.2\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 166.8, 155.1, 154.8, 150.0, 145.5, 139.7, 138.9, 132.5, 129.0, 125.1, 122.7, 121.9, 121.5, 120.2, 118.3, 116.6, 113.8, 106.2, 80.7, 61.5, 14.2. HRMS (ESI) Calcd. for \(\hbox {C}_{27}\hbox {H}_{24}\hbox {N}_{3}\hbox {O}_{3}\) ([M+H]\(^{+}\)): 438.1818. Found: 438.1812.
Ethyl 2,5-bis((4-methoxyphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4c)
Yellow solid, yield 89%. m.p. 180–182 \({^{\circ }}\)C. IR (KBr): 3371, 2972, 2931, 2834, 1677, 1614, 1510, 1421, 1301, 1090, 1029, 944, 821, 764 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \)(ppm): 10.16 (s, 1H, NH), 8.27 (s, 1H, ArH), 8.11–8.09 (m, 1H, ArH), 7.75–7.72 (m, 2H, ArH), 7.39–7.35 (m, 1H, ArH), 7.13 (t, \(J = 8.0\hbox { Hz}\), 1H, ArH), 7.01 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH), 6.88 (t, \(J = 9.2\hbox { Hz}\), 3H, ArH), 6.79 (d, \(J = 9.2\hbox { Hz}\), 2H, ArH), 6.74 (d, \(J = 8.8\hbox { Hz}\), 1H, CH), 6.68 (d, \(J = 8.8\hbox { Hz}\), 1H, NH), 4.36 (q, \(J = 7.2\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 3.78 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 3.68 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 1.35 (t, \(J = 7.2\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 166.8, 155.2, 155.1, 154.9, 152.3, 150.0, 139.3, 138.8, 132.7, 132.4, 125.1, 122.0, 121.7, 121.6, 118.3, 116.3, 115.0, 114.6, 114.1, 105.5, 81.7, 61.4, 55.4, 55.3, 14.2. HRMS (ESI) Calcd. for \(\hbox {C}_{29}\hbox {H}_{28}\hbox {N}_{3}\hbox {O}_{5}\) ([M+H]\(^{+})\): 498.2029. Found: 498.2027.
Ethyl 2,5-bis(o-tolylamino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4d)
Yellow solid, m.p. 190–192 \({^{\circ }}\)C. IR (KBr): 3444, 2985, 1676, 1596, 1520, 1460, 1344, 1234, 1109, 903, 803, 734 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.20 (s, 1H, NH), 8.48 (d, \(J = 7.6\hbox { Hz}\), 1H, ArH), 8.31 (s, 1H, ArH), 8.07–8.05 (m, 1H, ArH), 7.39–7.24 (m, 4H, ArH), 7.16–7.01 (m, 4H, ArH), 6.91 (d, \(J = 8.0\hbox { Hz}\), 1H, ArH), 6.80–6.71 (m, 2H, ArH+CH), 6.16 (d, \(J = 8.8\hbox { Hz}\), 1H, NH), 4.37 (q, \(J = 7.2\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 2.37 (s, 3H, \(\hbox {CH}_{3})\), 2.01 (s, 3H, \(\hbox {CH}_{3})\), 1.35 (t, \(J = 7.2\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.2, 156.2, 154.5, 150.6, 142.6, 138.4, 138.1, 132.3, 130.4, 128.8, 127.2, 126.2, 125.6, 123.2, 122.1, 121.9, 121.5, 119.7, 118.4, 115.9, 112.9, 106.4, 81.6, 61.3, 18.5, 17.5, 14.3. HRMS (ESI) Calcd. for \(\hbox {C}_{29}\hbox {H}_{28}\hbox {N}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 466.2131. Found: 466.2132.
Ethyl 2,5-bis((2,4-dimethylphenyl)amino)-5H-chromen[4,3-b]pyridine-3-carboxylate (4e)
Yellow solid, yield 88%. m.p. 183–185 \({^{\circ }}\)C. IR (KBr): 3402, 2991, 2912, 1686, 1601, 1517, 1440, 1300, 1245, 1225, 1135, 946, 819, 798, 694 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.09 (s, 1H, NH), 8.29 (d, \(J = 9.2\hbox { Hz}\), 2H, ArH), 8.04–8.01 (m, 1H, ArH), 7.38–7.34 (m, 1H, ArH), 7.15–7.01 (m, 4H, ArH), 6.94–6.84 (m, 3H, ArH), 6.71 (d, \(J = 9.2\hbox { Hz}\), 2H, CH), 5.99 (d, \(J = 9.2\hbox { Hz}\), 1H, NH), 4.36 (q, \(J = 6.8 \hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 2.30 (d, \(J = 5.6\hbox { Hz}\), 6H, \(2 \times \hbox {CH}_{3})\), 2.19 (s, 3H, \(\hbox {CH}_{3})\), 1.98 (s, 3H, \(\hbox {CH}_{3})\), 1.34 (t, \(J = 6.8\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.3, 156.2, 154.5, 150.6, 140.3, 138.4, 135.4, 132.7, 132.2, 131.2, 131.0, 129.0, 128.9, 127.5, 126.6, 125.6, 123.3, 122.2, 122.0, 121.6, 118.4, 115.8, 113.2, 106.2, 82.1, 61.2, 20.9, 20.5, 18.4, 17.4, 14.3. HRMS (ESI) Calcd. for \(\hbox {C}_{31}\hbox {H}_{32}\hbox {N}_{3}\hbox {O}_{3}\) ([M+H]\(^{+}\)): 494.2444. Found: 494.2436.
Ethyl 2,5-bis((3,5-dimethylphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4f)
Yellow solid, yield 88%. m.p. 229–230 \({^{\circ }}\)C. IR (KBr): 3442, 2921, 1686, 1602, 1521, 1425, 1230, 1180, 1105, 923, 828, 798, 768, 694 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 10.32 (s, 1H, NH), 8.26–8.24 (m, 1H, CH), 8.17 (s, 1H, NH), 7.55 (s, 2H, ArH), 7.37–7.32 (m, 1H, ArH), 7.12 (t, \(J = 7.2\hbox { Hz}\), 1H, ArH), 7.00 (d, \(J = 8.8\hbox { Hz}\), 1H, ArH), 6.76 (s, 1H, ArH), 6.56 (d, J = 10.4 Hz, 4H, ArH), 4.36 (q, \(J = 6.8\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 2.38 (s, 6H, \(2\times \hbox {CH}_{3})\), 2.29 (s, 6H, \(2\times \hbox {CH}_{3})\), 1.40 (t, \(J = 6.8\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.1, 155.9, 154.6, 150.5, 144.4, 139.6, 139.1, 138.2, 132.2, 125.5, 124.4, 122.0, 121.7, 121.7, 118.5, 118.4, 115.8, 112.1, 106.2, 81.5, 61.3, 21.5, 14.3. HRMS (ESI) Calcd. for \(\hbox {C}_{31}\hbox {H}_{32}\hbox {N}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 494.2444. Found: 494.2434.
Methyl 2,5-bis(p-tolylamino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4g)
Yellow solid, yield 88%. m.p. 178–180 \({^{\circ }}\)C. IR (KBr): 3402, 3021, 2947, 2921, 1677, 1612, 1519, 1440, 1296, 1232, 1105, 929, 804, 754 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.21 (s, 1H, NH), 8.25 (s, 1H, ArH), 8.11 (d, \(J = 7.6\hbox { Hz}\), 1H, ArH), 7.73 (d, \(J = 8.0\hbox { Hz}\), 2H, ArH), 7.37–7.32 (m, 1H, ArH), 7.23 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH), 7.14 (t, \(J = 7.2\hbox { Hz}\), 1H, ArH), 6.98 (d, \(J = 8.0\hbox { Hz}\), 2H, ArH), 6.91–6.88 (m, 2H, ArH), 6.82 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH+CH), 6.69 (d, \(J = 8.8\hbox { Hz}\), 1H, NH), 3.89 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 2.32 (s, 3H, \(\hbox {CH}_{3})\), 2.20 (s, 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.5, 155.9, 154.6, 150.8, 142.0, 138.2, 137.0, 132.3, 129.8, 129.3, 129.1, 125.6, 122.0, 121.6, 120.8, 118.4, 115.8, 114.6, 105.7, 81.9, 52.2, 20.9, 20.5. HRMS (ESI) Calcd. for \(\hbox {C}_{28}\hbox {H}_{26}\hbox {N}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 452.1974. Found: 452.1973.
Methyl 2,5-bis((4-bromophenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4h)
Yellow solid, yield 83%. m.p. 182–184 \({^{\circ }}\)C. IR (KBr): 3413, 3033, 2957, 2931, 1670, 1616, 1520, 1455, 1300, 1255, 1137, 939, 811, 768 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.30 (s, 1H, NH), 8.32 (s, 1H, ArH), 8.13 (d, \(J = 7.2\hbox { Hz}\), 1H, ArH), 7.84 (d, \(J = 8.0\hbox { Hz}\), 2H, ArH), 7.60 (d, \(J = 8.0\hbox { Hz}\), 2H, ArH), 7.41–7.14 (m, 5H, ArH), 6.93–6.86 (m, 3H, ArH+CH), 6.75 (d, \(J = 8.0\hbox { Hz}\), 1H, NH), 3.90 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)). \(^{13}\)C NMR (100 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 167.0, 154.7, 154.6, 149.9, 144.8, 138.9, 132.7 131.7, 131.6, 125.2, 122.2, 122.1, 121.3, 118.3, 116.7, 115.9, 114.2, 109.3, 106.4, 80.4, 52.7. HRMS (ESI) Calcd. for \(\hbox {C}_{26}\hbox {H}_{20}\)Br\(_{2}\hbox {N}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 579.9873. Found: 579.9871.
2,5-Bis((4-methoxyphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carbonitrile (4i)
Yellow solid, yield 92%. m.p. 187–188 \({^{\circ }}\)C. IR (KBr): 3330, 2955, 2834, 2218, 1601, 1510, 1446, 1425, 1240, 1034, 823, 753 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 9.11 (s, 1H, NH), 8.10 (s, 1H, ArH), 7.96 (d, \(J = 7.2\hbox { Hz}\), 1H, ArH), 7.60 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH), 7.35 (t, \(J = 7.2\hbox { Hz}\), 1H, ArH), 7.08 (t, \(J = 7.6\hbox { Hz}\), 1H, ArH), 6.98 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH), 6.90–6.73 (m, 6H, ArH+CH), 6.57 (d, \(J = 8.4\hbox { Hz}\), 1H, NH), 3.78 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 3.67 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)). \(^{13}\)C NMR (100 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 155.9, 155.5, 154.7, 152.4, 149.3, 141.7, 139.3, 132.6, 132.5, 125.0, 123.5, 121.8, 121.4, 118.3, 116.9, 116.7, 115.0, 114.6, 113.7, 90.8, 81.5, 55.4, 55.3. HRMS (ESI) Calcd. for \(\hbox {C}_{27}\hbox {H}_{22}\hbox {N}_{4}\hbox {NaO}_{3}\) ([M+Na]\(^{+)}\): 473.1590. Found: 473.1582.
2,5-Bis(phenylamino)-5H-chromeno[4,3-b]pyridine-3-carbonitrile (4j)
Yellow solid, yield 90%. m.p. 215–217 \({^{\circ }}\)C. IR (KBr): 3398, 3313, 2225, 1599, 1521, 1496, 1443, 1300, 1242, 1195, 1099, 929, 749 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 8.17–8.14 (m, 1H, NH), 7.75–7.72 (m, 3H, ArH), 7.46–7.42 (m, 2H, ArH), 7.39–7.34 (m, 1H, ArH), 7.29–7.25 (m, 2H, ArH), 7.20–7.10 (m, 3H, ArH), 6.98–6.89 (m, 4H, ArH), 6.53 (d, J = 9. 6 Hz, 1H, CH), 4.63 (d, \(J = 9.2\hbox { Hz}\), 1H, NH). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 155.6, 154.5, 150.4, 143.9, 139.4, 138.5, 132.9, 129.4, 129.0, 125.6, 123.9, 122.4, 121.0, 120.5, 120.2, 118.5, 116.7, 116.4, 114.7, 91.5, 81.1. HRMS (ESI) Calcd. for \(\hbox {C}_{25}\hbox {H}_{18}\hbox {N}_{4}\)NaO ([M+Na]\(^{+})\): 413.1378. Found: 413.1374.
2,5-Bis(p-tolylamino)-5H-chromeno[4,3-b]pyridine-3-carbonitrile (4k)
Yellow solid, yield 92%. m.p. 187–188 \({^{\circ }}\)C. IR (KBr): 3421, 2921, 2212, 1602, 1520, 1450, 1420, 1245, 1205, 1090, 808, 753 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 9.17 (s, 1H, NH), 8.12 (s, 1H, CH), 7.98 (d, \(J = 7.6\,\hbox {Hz}\), 1H, NH), 7.60 (d, \(J = 7.6\,\hbox {Hz}\), 2H, ArH), 7.36 (t, \(J = 7.2\hbox { Hz}\), 1H, ArH), 7.20 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 7.10 (t, \(J = 7.2\hbox { Hz}\), 1H, ArH), 6.99 (d, \(J = 8.0\hbox { Hz}\), 2H, ArH), 6.90 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 6.81 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 6.62 (d, \(J = 8.4\hbox { Hz}\), 1H, ArH), 2.32 (s, 3H, \(\hbox {CH}_{3})\), 2.19 (s, 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 155.7, 154.7, 149.3, 143.0, 141.7, 137.2, 132.6, 132.1, 129.5, 129.0, 126.9, 125.0, 121.9, 121.5, 121.4, 118.3, 116.9, 116.8, 113.9, 91.4, 80.9, 20.6, 20.3. HRMS (ESI) Calcd. for \(\hbox {C}_{27}\hbox {H}_{22}\hbox {N}_{4}\)NaO ([M+Na]\(^{+})\): 441.1691. Found: 441.1690.
2,5-Bis((3,5-dimethylphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carbonitrile (4l)
Yellow solid, yield 90%. m.p. 190–192 \({^{\circ }}\)C. IR (KBr): 3410, 2916, 2208, 1526, 1450, 1329, 1240, 1178, 1104, 923, 833, 754 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 8.17 (d, \(J = 7.6\hbox { Hz}\), 1H, NH), 7.65 (s, 1H, ArH), 7.37 (t, \(J = 7.6\hbox { Hz}\), 3H, ArH), 7.12 (t, \(J = 7.6\hbox { Hz}\), 1H, ArH), 7.00 (t, \(J = 8.0\hbox { Hz}\), 2H, ArH), 6.83 (s, 1H, ArH), 6.57–6.48 (m, 4H, CH+ArH), 4.57 (d, J = 9.6 Hz, 1H, NH), 2.40 (s, 6H, \(2 \times \hbox {CH}_{3})\), 2.29 (s, 6H, \(2 \times \hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 155.6, 154.6, 150.3, 144.0, 139.3, 139.1, 138.5, 138.3, 132.8, 125.5, 122.2, 122.1, 121.1, 118.5, 118.2, 116.6, 116.4, 112.5, 91.4, 81.2, 21.5, 21.4. HRMS (ESI) Calcd. for \(\hbox {C}_{29}\hbox {H}_{26}\hbox {N}_{4}\)NaO ([M+Na]\(^{+})\): 469.2004. Found: 469.2006.
Ethyl 9-fluoro-2,5-bis((4-methoxyphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4m)
Yellow solid, yield 89%. m.p. 190–192 \({^{\circ }}\)C. IR (KBr): 3442, 2996, 2930, 1686, 1616, 1516, 1455, 1213, 1099, 1044, 949, 828, 798, 698 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.13 (s, 1H, NH), 8.29 (s, 1H, ArH), 7.71–7.68 (m, 3H, ArH), 7.25–7.20 (m, 1H, ArH), 7.02 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH), 6.95–6.92 (m, 1H, ArH), 6.87 (d, \(J = 9.2\hbox { Hz}\), 2H, ArH), 6.81–6.75 (m, 3H, ArH+CH), 6.68 (d, \(J = 9.2\hbox { Hz}\), 1H, NH), 4.37 (q, \(J = 6.8\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 3.79 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 3.68 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 1.36 (t, \(J = 7.2\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.0, 155.7, 154.9 (d, J = 234 Hz), 150.4, 149.7, 138.3, 138.1, 132.6, 122.6, 119.6, 118.9 (d, \(J = 25\hbox { Hz}\)), 116.1, 115.7, 114.8, 114.0, 111.4, 111.1, 106.3, 82.6, 61.4, 55.7, 55.5, 14.3. HRMS (ESI) Calcd. for \(\hbox {C}_{29}\hbox {H}_{26}\hbox {FN}_{3}\hbox {NaO}_{5}\) ([M+Na]\(^{+})\): 538.1754. Found: 538.1751.
Ethyl 9-fluoro-2,5-bis(phenylamino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4n)
Yellow solid, yield 90%. m.p. 237–239 \({^{\circ }}\)C. IR (KBr): 3398, 2990, 1681, 1602, 1526, 1445, 1289, 1219, 1109, 933, 877, 798 752, 698 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.31 (s, 1H, NH), 8.36 (s, 1H, ArH), 7.81 (d, \(J = 8.0\hbox { Hz}\), 2H, ArH), 7.76–7.73 (m, 1H, ArH), 7.44 (t, 2H, ArH), 7.24–7.07 (m, 5H, ArH), 6.97–6.90 (m, 3H, ArH), 6.81 (d, \(J = 8.4\hbox { Hz}\), 1H, CH), 6.73 (t, 1H, NH), 4.38 (q, \(J = 7.2\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 1.36 (t, \(J = 7.2\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, DMSO\(-d_{6}\)) \(\delta \) (ppm): 116.6, 157.1 (d, J = 237 Hz), 155.0, 150.9, 148.9, 145.3, 139.4, 139.1, 129.0 (d, J = 5 Hz), 123.0, 120.5, 119.3, 119.1, 118.4, 116.8, 113.8, 110.3 (d, J = 24 Hz), 106.8. HRMS (ESI) Calcd. for \(\hbox {C}_{27}\hbox {H}_{23}\hbox {FN}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 456.1723. Found: 456.1733.
Ethyl 9-fluoro-2,5-bis((3-isopropylphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4o)
Yellow solid, yield 88%. m.p. 187–189 \({^{\circ }}\)C. IR (KBr): 3404, 2960, 2878, 1681, 1611, 1526, 1450, 1225, 1129, 943, 884, 793, 702 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.29 (s, 1H, NH), 8.34 (s, 1H, CH), 7.92 (s, 1H, NH), 7.84–7.81 (m, 1H, ArH), 7.44 (d, \(J = 8.0\hbox { Hz}\), 1H, ArH), 7.33 (t, \(J = 8.0\hbox { Hz}\), 1H, ArH), 7.26–7.21 (m, 1H, ArH), 7.09 (t, \(J = 8.4\hbox { Hz}\), 1H, ArH), 7.02–6.95 (m, 3H, ArH), 6.77 (t, \(J = 7.2\hbox { Hz}\), 3H, ArH), 6.62 (d, \(J = 7.6\hbox { Hz}\), 1H, ArH), 4.40-4.35 (m, 2H, \(\hbox {CH}_{2}\hbox {O}\)), 2.99-2.92 (m, 1H, CH), 2.79-2.74 (m, 1H, CH), 1.36 (t, \(J = 7.2\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\), 1.29 (d, \(J = 6.8\hbox { Hz}\), 6H, (\(\hbox {CH}_{3})_{2}\hbox {C}\)), 1.17 (d, \(J = 6.8\hbox { Hz}\), 6H, (\(\hbox {CH}_{3})_{2}\hbox {C}\)). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.0, 156.0, 150.4, 149.7, 144.3, 139.4, 138.4, 129.4, 128.7, 122.6, 121.5, 119.8, 119.7, 119.1, 119.0, 118.3, 118.2, 115.9, 112.8, 111.7, 111.4 (d, \(J = 25\hbox { Hz}\)), 106.7, 81.7, 61.5, 34.4, 34.2, 24.1, 24.0, 23.9, 14.3. HRMS (ESI) Calcd. for \(\hbox {C}_{33}\hbox {H}_{35}\hbox {FN}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 540.2662. Found: 540.2659.
Methyl 9-fluoro-2,5-bis((3-isopropylphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4p)
Yellow solid, yield 88%. m.p. 184–185 \({^{\circ }}\)C. IR (KBr): 3402, 2958, 2880, 1681, 1611, 1526, 1446, 1295, 1220, 1130, 940, 889, 794, 698 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.24 (s, 1H, NH), 8.32 (s, 1H, ArH), 7.91 (s, 1H, ArH), 7.83–7.79 (m, 1H, ArH), 7.43 (d, \(J = 8.4\hbox { Hz}\), 1H, ArH), 7.32 (t, \(J = 8.0\hbox { Hz}\), 1H, ArH), 7.25–7.20 (m, 1H, ArH), 7.09 (t, \(J = 8.0\hbox { Hz}\), 1H, ArH), 7.02–6.94 (m, 3H, ArH), 6.75 (s, 3H, ArH+CH), 6.62 (d, \(J = 7.6\hbox { Hz}\), 1H, NH), 3.90 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 2.97–2.92 (m, 1H, CH), 2.79–2.75 (m, 1H, CH), 1.30–1.28 (m, 6H, \(2 \times \hbox {CH}_{3})\), 1.17 (d, \(J = 6.8\hbox { Hz}\), 6H, \(2\times \hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.4, 155.9, 150.3, 149.7, 144.3, 139.4, 138.4, 129.3, 128.7, 122.6, 122.5, 121.6, 119.7, 119.6, 119.2, 119.0, 118.4, 118.2, 115.9, 112.8, 111.8, 111.4 (d, J = 24 Hz), 106.4, 81.8, 52.3, 34.4, 34.2, 24.1, 24.0, 23.9. HRMS (ESI) Calcd. for \(\hbox {C}_{32}\hbox {H}_{33}\hbox {FN}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 526.2506. Found: 526.2509.
9-Fluoro-2,5-bis((4-isopropylphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carbonitrile (4q)
Yellow solid, yield 88%. m.p. 161–163 \({^{\circ }}\)C. IR (KBr): 3417, 2958, 2870, 2218, 1611, 1517, 1455, 1260, 1176, 1135, 939, 824 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 7.81–7.78 (m, 1H, NH), 7.74 (s, 1H, ArH), 7.62 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 7.31 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 7.13 (t, \(J = 8.4\hbox { Hz}\), 3H, ArH), 7.08–7.03 (m, 1H, ArH), 6.94–6.91 (m, 1H, ArH), 6.86 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 6.49 (d, \(J = 9.2\hbox { Hz}\), 1H, CH), 4.54 (d, \(J = 9.2\hbox { Hz}\), 1H, NH), 3.00–2.93 (m, 1H, CH), 2.90–2.83 (m, 1H, CH), 1.31 (d, \(J = 6.8\hbox { Hz}\), 6H, (\(\hbox {CH}_{3})_{2}\hbox {C}\)), 1.24 (d, \(J = 6.8\hbox { Hz}\), 6H, (\(\hbox {CH}_{3})_{2}\hbox {C}\)). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3})\) \(\delta \) (ppm): 158.0 (d, J = 239 Hz), 155.7, 150.5, 149.7, 145.0, 141.7, 141.0, 139.5, 135.9, 127.3, 127.0, 120.7, 119.9, 119.8, 119.6, 116.8, 116.3, 114.8, 111.4 (d, \(J = 25\hbox { Hz}\)), 92.0, 81.7, 33.6, 33.3, 24.2, 24.0. HRMS (ESI) Calcd. for \(\hbox {C}_{31}\hbox {H}_{29}\hbox {FN}_{4}\)NaO ([M+Na]\(^{+})\): 515.2223. Found: 515.2224.
Ethyl 8-fluoro-2,5-bis((4-methoxyphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4r)
Yellow solid, yield 89%. m.p. 218–220 \({^{\circ }}\)C. IR (KBr): 3442, 3041, 2966, 1681, 1621, 1510, 1420, 1365, 1240, 1034, 929, 834, 743 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.15 (s, 1H, NH), 8.24 (s, 1H, CH), 8.09 (t, \(J = 8.0\hbox { Hz}\), 1H, NH), 7.71 (t, \(J = 8.8\hbox { Hz}\), 2H, ArH), 7.01–6.70 (m, 10H, ArH), 4.35 (q, \(J = 7.2\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 3.78 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 3.68 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 1.35 (t, \(J = 7.2\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 166.7, 164.7 (d, J = 247 Hz), 156.6, 156.5, 155.2, 152.5, 149.4, 139.1, 138.7, 132.6, 127.1, 122.1, 118.3, 115.4, 115.1, 114.6, 114.1, 109.2 (d, J = 23 Hz), 105.5, 105.1, 82.6, 61.4, 55.4, 55.3, 14.2. HRMS (ESI) Calcd. for \(\hbox {C}_{29}\hbox {H}_{27}\hbox {FN}_{3}\hbox {O}_{5}\) ([M+H]\(^{+})\): 516.1935. Found: 516.1925.
Ethyl 8-fluoro-2,5-bis(p-tolylamino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4s)
Yellow solid, yield 90%. m.p. 231–233 \({^{\circ }}\)C. IR (KBr): 3402, 2976, 2921, 1677, 1616, 1516, 1415, 1290, 1209, 1105, 969, 893, 808, 708 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.24 (s, 1H, NH), 8.26 (s, 1H, ArH), 8.12 (t, \(J = 7.6\hbox { Hz}\), 1H, ArH), 7.70 (d, \(J = 8.0\hbox { Hz}\), 2H, ArH), 7.22 (d, \(J = 7.6\hbox { Hz}\), 2H, ArH), 6.98 (t, \(J = 6.8\hbox { Hz}\), 4H, ArH), 6.83–6.76 (m, 4H, ArH+CH+NH), 4.35 (q, \(J = 6.8\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 2.32 (s, 3H, \(\hbox {CH}_{3})\), 2.20 (s, 3H, \(\hbox {CH}_{3})\), 1.34 (t, \(J = 6.8\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.1, 156.1, 141.8, 138.3, 137.0, 132.5, 129.9, 129.4, 129.3, 127.3, 127.2, 120.9, 118.1, 114.8, 114.5, 109.7 (d, J = 22 Hz), 105.9, 105.8, 105.5, 82.5, 61.3, 20.9, 20.5, 14.3. HRMS (ESI) Calcd. for \(\hbox {C}_{29}\hbox {H}_{27}\hbox {FN}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 484.2036. Found: 484.2046.
Ethyl 8-fluoro-2,5-bis((3-isopropylphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4t)
Yellow solid, yield 88%. m.p. 231–233 \({^{\circ }}\)C.; IR (KBr): 3452, 2956, 2930, 1686, 1608, 1526, 1445, 1209, 1104, 973, 788, 698 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.31 (s, 1H, NH), 8.31 (s, 1H, ArH), 8.18 (t, \(J = 7.2\hbox { Hz}\), 1H, ArH), 7.75 (s, 1H, ArH), 7.61 (d, \(J = 8.0\hbox { Hz}\), 1H, ArH), 7.33 (t, \(J = 8.0\hbox { Hz}\), 1H, ArH), 7.12–7.06 (m, 2H, ArH), 6.99 (t, \(J = 6.8\hbox { Hz}\), 2H, ArH), 6.86–6.76 (m, 4H, ArH+CH), 6.63 (d, \(J = 7.6\hbox { Hz}\), 1H, NH), 4.35 (q, \(J = 6.8\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 2.98–2.91 (m, 1H, CH), 2.81–2.74 (m, 1H, CH), 1.35 (t, \(J = 6.8\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\), 1.28 (d, \(J = 6.8\hbox { Hz}\), 6H, (\(\hbox {CH}_{3})_{2}\hbox {C}\)), 1.18 (d, \(J = 6.8\hbox { Hz}\), 6H, (\(\hbox {CH}_{3})_{2}\hbox {C}\)). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.1, 156.0, 150.4, 150.0, 149.6, 144.1, 139.5, 138.4, 129.4, 128.6, 127.4, 127.3, 121.4, 118.9, 118.3, 118.1, 114.9, 112.8, 111.7, 109.6 (d, J = 22 Hz), 106.1, 105.9, 105.6, 82.3, 61.4, 34.3, 34.2, 24.1, 24.0, 23.9, 14.3. HRMS (ESI) Calcd. for \(\hbox {C}_{33}\hbox {H}_{35}\hbox {FN}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 540.2662. Found: 540.2653.
Methyl 8-fluoro-2,5-bis((4-methoxyphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4u)
Yellow solid, yield 89%. m.p. 208–210 \({^{\circ }}\)C. IR (KBr): 3408, 1691, 1626, 1512, 1420, 1310, 1209, 1104, 1034, 979, 823, 793 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.09 (s, 1H, NH), 8.24 (s, 1H, ArH), 8.10–8.07 (m, 1H, ArH), 7.70 (d, \(J = 9.2\hbox { Hz}\), 2H, ArH), 7.01–6.94 (m, 3H, ArH), 6.87–6.69 (m, 7H, ArH+CH+NH), 3.89 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 3.78 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 3.68 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.5, 155.6, 154.9 (d, J = 223 Hz), 150.2, 138.3, 137.9, 132.7, 127.3, 127.2, 122.6, 116.1, 114.8, 113.9, 109.6 (d, J = 22 Hz), 105.4, 105.3, 83.3, 55.7, 55.5, 52.2. HRMS (ESI) Calcd. for \(\hbox {C}_{28}\hbox {H}_{24}\hbox {FN}_{3}\hbox {NaO}_{5}\) (M+Na]\(^{+})\): 524.1598. Found: 524.1592.
Methyl 8-fluoro-2,5-bis(p-tolylamino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4v)
Yellow solid, yield 88%. m.p. 231–233 \({^{\circ }}\)C. IR (KBr): 3402, 3025, 2955, 1686, 1616, 1526, 1431, 1295, 1209, 1105, 974, 848, 793 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.19 (s, 1H, NH), 8.27 (s, 1H, CH), 8.13–8.10 (m, 1H, NH), 7.70 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 7.23 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 7.02–6.95 (m, 4H, ArH), 6.83–6.77 (m, 4H, ArH), 3.89 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 2.32 (s, 3H, \(\hbox {CH}_{3})\), 2.19 (s, 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.5, 156.0, 150.2, 141.7, 138.3, 136.9, 132.5, 129.9, 129.4, 129.3, 127.4, 127.3, 120.9, 118.1, 114.9, 114.6, 109.6 (d, J = 23 Hz), 105.7, 105.6, 82.6, 52.2, 20.9, 20.5. HRMS (ESI) Calcd. for \(\hbox {C}_{28}\hbox {H}_{25}\hbox {FN}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 470.1877. Found: 470.1871.
Methyl 8-fluoro-2,5-bis((3-isopropylphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4w)
Yellow solid, yield 88%. m.p. 183–184 \({^{\circ }}\)C. IR (KBr): 3442, 2966, 1692, 1608, 1526, 1436, 1385, 1210, 1110, 979, 789, 702 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 10.26 (s, 1H, NH), 8.30 (s, 1H, ArH), 8.19–8.15 (m, 1H, ArH), 7.74 (s, 1H, ArH), 7.61 (d, \(J = 8.0\hbox { Hz}\), 1H, ArH), 7.33 (t, \(J = 8.0\hbox { Hz}\), 1H, ArH), 7.12–7.06 (m, 2H, ArH), 7.00–6.95 (m, 2H, ArH), 6.84–6.77 (m, 4H, ArH+CH), 6.63 (d, \(J = 7.6\hbox { Hz}\), 1H, NH), 3.90 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 2.98–2.91 (m, 1H, CH), 2.81–2.74 (m, 1H, CH), 1.28 (d, \(J = 6.8\hbox { Hz}\), 6H, (\(\hbox {CH}_{3})_{2}\hbox {C}\)), 1.17 (d, \(J = 6.8\hbox { Hz}\), 6H, (\(\hbox {CH}_{3})_{2}\hbox {C}\)). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.1, 164.7 (d, J = 248 Hz), 156.7, 156.5, 155.0, 149.3, 149.2, 145.2, 139.5, 138.9, 129.0, 128.9, 127.0, 126.8, 121.1, 118.3, 118.2, 117.9, 116.7, 115.8, 112.2, 111.2, 109.1 (d, J = 22 Hz), 105.8, 105.5, 105.3, 81.8, 52.5, 33.7, 24.0. HRMS (ESI) Calcd. for \(\hbox {C}_{32}\hbox {H}_{33}\hbox {FN}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 526.2506. Found: 526.2529.
8-Fluoro-2,5-bis((4-methylphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carbonitrile (4x)
Yellow solid, yield 89%. m.p. 209–210 \({^{\circ }}\)C. IR (KBr): 3330, 3025, 2920, 2225, 1616, 1516, 1461, 1345, 1245, 1010, 969, 813 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, DMSO-\(d_{6}\)) \(\delta \) (ppm): 9.19 (s, 1H, NH), 8.11 (s, 1H, ArH), 8.00–7.96 (m, 1H, ArH), 7.58 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 7.19 (d, \(J = 8.0\hbox { Hz}\), 2H, ArH), 7.00–6.94 (m, 4H, ArH), 6.82–6.76 (m, 3H, ArH+CH), 6.67 (d, \(J = 8.4\hbox { Hz}\), 1H, NH), 2.31 (s, 3H, \(\hbox {CH}_{3})\), 2.19 (s, 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, DMSO-d\(_{6}\)) \(\delta \) (ppm): 166.0, 163.5, 156.4, 156.3, 155.7, 148.6, 142.8, 141.8, 137.1, 132.2, 129.5, 129.0, 127.2, 127.1, 127.0, 121.6, 118.2, 116.8, 116.2, 114.0, 109.4 (d, J = 23 Hz), 105.5, 105.3, 91.4, 81.8, 20.6, 20.3. HRMS (ESI) Calcd. for \(\hbox {C}_{27}\hbox {H}_{22}\hbox {FN}_{4}\hbox {O}\) ([M+H]\(^{+})\): 437.1778. Found: 437.1788.
Ethyl 9-chloro-2,5-bis((4-methoxyphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4y)
Yellow solid, yield 89%. m.p. 209–211 \({^{\circ }}\)C. IR (KBr): 3368, 1682, 1621, 1516, 1440, 1230, 1090, 1039, 949, 828, 694 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 10.20 (s, 1H, NH), 8.18 (s, 1H, ArH), 8.09 (s, 1H, ArH), 7.69 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH), 7.24 (d, \(J = 8.8\hbox { Hz}\), 1H, ArH), 6.96 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH), 6.89–6.83 (m, 5H, ArH), 6.45 (d, \(J = 9.2\hbox { Hz}\), 1H, CH), 6.44 (d, J = 9.6 Hz, 1H, NH), 4.37 (q, \(J = 7.2\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 3.86 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 3.78 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 1.41 (t, \(J = 6.8\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.0, 156.0, 155.7, 153.8, 152.9, 149.4, 138.4, 138.0 132.6, 131.8, 127.1, 125.1, 122.9, 122.5, 119.8, 116.1, 115.5, 114.8, 114.0, 106.4, 82.7, 61.4, 55.7, 55.5, 14.3. HRMS (ESI) Calcd. for \(\hbox {C}_{29}\hbox {H}_{27}\hbox {ClN}_{3}\hbox {O}_{5}\) ([M+H]\(^{+})\): 532.1639. Found: 532.1637.
Ethyl 9-chloro-2,5-bis(p-tolylamino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (4z)
Yellow solid, yield 88%. m.p. 242–244 \({^{\circ }}\)C. IR (KBr): 3370, 2986, 2920, 1681, 1616, 1520, 1440, 1280, 1230, 1099, 954, 814, 693 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 10.31 (s, 1H, NH), 8.20 (s, 1H, ArH), 8.14 (s, 1H, ArH), 7.70 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 7.27–7.22 (m, 3H, ArH), 7.08 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 6.90–6.83 (m, 3H, ArH), 6.53 (d, \(J = 9.2\hbox { Hz}\), 1H, CH), 4.52 (d, \(J = 9.2\hbox { Hz}\), 1H, NH), 4.38 (q, \(J = 7.2\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 2.38 (s, 3H, \(\hbox {CH}_{3})\), 2.29 (s, 3H, \(\hbox {CH}_{3})\), 1.41 (t, \(J = 7.2\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.0 156.0, 153.0, 149.4, 141.8, 138.4, 136.8, 132.6, 131.9, 129.9, 129.3, 127.2, 125.2, 122.9, 120.8, 119.9, 115.6, 114.6, 106.6, 82.1, 61.4, 30.9, 20.9, 20.5, 14.3. HRMS (ESI) Calcd. for \(\hbox {C}_{29}\hbox {H}_{27}\hbox {ClN}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 500.1741. Found: 500.1749.
Ethyl 2,5-bis((4-bromophenyl)amino)-9-chloro-5H-chrome[4,3-b]pyridine-3-carboxylate (\(\mathbf{4a}^\prime \))
Yellow solid, yield 85%. m.p. 242–244 \({^{\circ }}\)C. IR (KBr): 3364, 2986, 2930, 1677, 1612, 1521, 1451, 1296, 1230, 1070, 955, 819, 699 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 10.45 (s, 1H, NH), 8.21 (s, 1H, ArH), 8.10 (s, 1H, ArH), 7.72 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 7.52 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH), 7.36 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH), 7.31–7.28 (m, 1H, ArH), 6.90 (d, \(J = 8.8\hbox { Hz}\), 1H, ArH), 6.81 (d, \(J = 8.8\hbox { Hz}\), 2H, ArH), 6.50 (d, \(J = 8.8\hbox { Hz}\), 1H, CH), 4.62 (d, \(J = 9.2\hbox { Hz}\), 1H, NH), 4.45 (q, \(J = 7.2\hbox { Hz}\), 2H, \(\hbox {CH}_{2}\hbox {O}\)), 1.42 (t, \(J = 7.2\hbox { Hz}\), 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 155.5, 154.7, 149.2, 145.3, 141.7, 139.7, 138.0, 137.4, 132.6, 124.9, 124.5, 121.8, 121.4, 120.3, 118.8, 118.4, 117.1, 116.7, 111.8, 91.8, 80.7, 21.4, 21.3. HRMS (ESI) Calcd. for \(\hbox {C}_{27}\hbox {H}_{20}\)Br\(_{2}\hbox {ClN}_{3}\hbox {NaO}_{3}\) ([M+Na]\(^{+})\): 649.9458. Found: 649.9459.
Methyl 9-chloro-2,5-bis(p-tolylamino)-5H-chromeno[4,3-b]pyridine-3-carboxylate (\(\mathbf{4b}^\prime \))
Yellow solid, yield 88%. m.p. 239–241 \({^{\circ }}\)C. IR (KBr): 3398, 2956, 2921, 1677, 1611, 1521, 1436, 1296, 1235, 1105, 929, 798 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 10.28 (s, 1H, NH), 8.22 (s, 1H, ArH), 8.15 (s, 1H, ArH), 7.71 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 7.29–7.24 (m, 3H, ArH), 7.09 (d, \(J = 8.0\hbox { Hz}\), 2H, ArH), 6.91–6.84 (m, 3H, ArH), 6.53 (d, \(J = 9.2\hbox { Hz}\), 1H, CH), 4.53 (d, \(J = 9.2\hbox { Hz}\), 1H, NH), 3.94 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 2.40 (s, 3H, \(\hbox {CH}_{3})\), 2.19 (s, 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 167.4, 155.9, 153.1, 149.6, 149.2, 141.8, 138.5, 136.8, 132.7, 131.9, 129.9, 129.3, 127.2, 125.2, 122.8, 120.9, 119.9, 115.7, 114.6, 106.3, 82.1, 52.3, 20.9, 20.5. HRMS (ESI) Calcd. for \(\hbox {C}_{28}\hbox {H}_{25}\hbox {ClN}_{3}\hbox {O}_{3}\) ([M+H]\(^{+})\): 486.1584. Found: 486.1581.
9-Chloro-2,5-bis((4-methoxyphenyl)amino)-5H-chromeno[4,3-b]pyridine-3-carbonitrile (\(\mathbf{4c}^\prime \))
Yellow solid, yield 90%. m.p. 212–213 \({^{\circ }}\)C. IR (KBr): 3387, 3336, 2995, 2952, 2840, 2218, 1601, 1516, 1450, 1346, 1247, 1034, 888, 813 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 8.07 (s, 1H, NH), 7.82 (s, 1H, ArH), 7.62–7.60 (m, 2H, ArH), 7.34–7.31 (m, 1H, ArH), 7.08–7.03 (m, 3H, ArH), 6.95–6.88 (m, 5H, ArH), 6.49 (d, J = 9.6 Hz, 1H, CH), 4.45 (d, J = 10.0 Hz, 1H, NH), 3.92 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)), 3.83 (s, 3H, \(\hbox {CH}_{3}\hbox {O}\)). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 156.0, 154.1, 153.0, 149.3, 139.7, 137.5, 132.5, 131.1, 127.4, 125.2, 123.1, 122.3, 119.9, 116.6, 114.8, 114.2, 91.7, 82.5, 55.6. HRMS (ESI) Calcd. for \(\hbox {C}_{27}\hbox {H}_{21}\hbox {ClN}_{4}\hbox {NaO}_{3}\) ([M+Na]\(^{+})\): 507.1200. Found: 507.1204.
9-Chloro-2,5-bis(p-tolylamino)-5H-chromeno[4,3-b]pyridine-3-carbonitrile\( ~(\mathbf{4d}^\prime )\)
Yellow solid, yield 90%. m.p. 229–231 \({^{\circ }}\)C. IR (KBr): 3416, 3031, 2918, 2212, 1597, 1521, 1451, 1351, 1261, 1200, 1141, 899, 808 \(\hbox {cm}^{-1}\). \(^{1}\)H NMR (400 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 8.08 (s, 1H, NH), 7.77 (s, 1H, ArH), 7.59 (d, \(J = 8.4\hbox { Hz}\), 2H, ArH), 7.30 (t, J = 10.0 Hz, 3H, ArH), 7.09 (d, \(J = 7.6\hbox { Hz}\), 3H, ArH), 6.92–6.83 (m, 3H, ArH), 6.52 (d, J = 10.0 Hz, 1H, CH), 4.52 (d, J = 9.6 Hz, 1H, NH), 2.42 (s, 3H, \(\hbox {CH}_{3})\), 2.31 (s, 3H, \(\hbox {CH}_{3})\). \(^{13}\)C NMR (100 MHz, \(\hbox {CDCl}_{3}\)) \(\delta \) (ppm): 155.7, 153.0, 149.3, 141.3, 139.6, 135.6, 134.0, 132.5, 129.9, 129.6, 127.4, 125.2, 122.2, 120.9, 120.0, 116.6, 114.9, 92.0, 81.8, 20.9, 20.5. HRMS (ESI) Calcd. for \(\hbox {C}_{27}\hbox {H}_{22}\hbox {ClN}_{4}\hbox {O}\) ([M+H]\(^{+})\): 453.1482. Found: 453.1487.
References
Clardy J, Walsh C (2004) Lessons from natural and molecules. Nature 432:829–837. doi:10.1038/nature03194
Boger DL, Boyce CW, Labroli MA, Sehon CA, Jim Q (1999) Total synthesis of Ningalin A, Lamellarin O, Lukinol A, and Permethyl Storniamide A utilizing heterocyclic azadiene Diels–Alder reactions. J Am Chem Soc 121:54–62. doi:10.1021/ja982078+
Frolova LV, Malik I, Uglinskii PY, Rogelj S, Kornienko A, Magedov IV (2011) Multicomponent synthesis of 2,3-dihydrochromeno[4,3-d]pyrazolo[3,4-b]pyridine-1,6-diones: a novel heterocyclic scaffold with antibacterial activity. Tetrahedron Lett 52:6643–6645. doi:10.1016/j.tetlet.2011.10.012
Khan IA, Kulkarni MV, Gopal M, Shahabuddin MS, Sun CM (2005) Synthesis and biological evaluation of novel angularly fused polycyclic coumarins. Bioorg Med Chem Lett 15:3584–3587. doi:10.1016/j.bmcl.2005.05.063
Gebhardt P, Dornberger K, Gollmick FA, Gräfe U, Härtl A, Görls H, Schlegel B, Hertweck C (2007) Quercinol, and anti-inflammatory chromene from the wood-rotting fungus Daedalea quercina (Oak Mazegill). Bioorg Med Chem Lett 17:2558–2560. doi:10.1016/j.bmcl.2007.02.008
El-Essaway FA, El-Etraway AS (2014) Synthesis of novel chromeno[4,3-b]pyrazolo[4,3-e]pyridine derivatives with antimicrobial evaluation. J Heterocycl Chem 51:191–195. doi:10.1002/jhet.1687
Patel AA, Lad HB, Pandya KR, Patel CV, Brahmbhatt DI (2013) Synthesis of a new series of 2-(2-oxo-2H-chromen-3-yl)-5H-chromeno[4,3-b]pyridine-5-ones by two facile methods and evaluation of their antimicrobial activity. Med Chem Res 22:4745–4754. doi:10.1007/s00044-013-0489-4
Ishikawa T, Oku Y, Tanaka T, Kumamoto T (1999) An approach to anti-HIV-1 active Calophyllum coumarin synthesis: an enantioselective construction of 2,3-dimethyl-4-chromanone ring by quinine-assisted intramolecular Michael-type addition. Tetrahedron Lett 40:3777–3780. doi:10.1016/S0040-4039(99)00607-3
Thapa U, Thapa P, Karki R, Yun M, Choi JH, Jahng Y, Lee E, Jeon KH, Na Y, Ha EM, Cho WJ, Kwon Y, Lee ES (2011) Synthesis of 2,4-dioxyl chromenopyridines and evaluation of their topoisomerase I and II inhibitory activity, cytotoxicity, and structure-activity realationship. Eur J Med Chem 46:3201–3209. doi:10.1016/j.ejmech.2011.04.029
Korotaev VY, Barkov AY, Sosnovskikh VY (2013) Synthesis of 5-(trifluoromethyl)-5H-chromeno [3,4-b]pyridines from 3-nitro-2-(trifluoromethyl)-2H-chromenes and aminoenones derived from acetylacetone and cyclic anilines. Tetrahedron Lett 54:3091–3093. doi:10.1016/j.tetlet.2013.03.137
Hegab MI, Abdel-Fattah ASM, Yousef NM, Nour HF, Nostafa AM, Ellithey M (2007) Synthesis, X-ray structure, and pharmacological activity of some 6,6-disubstituted chromeno[4,3-b]- and chromeno-[3,4-c]-quinolines. Arch Pharm Chem Life Sci 340:396–403. doi:10.1002/ardp.200700089
Bachi MD, Denenark D (1989) Cyclizations of ene radicals. Imidoyl radicals as intermediates in the synthesis of heterocyclic compounds. J Am Chem Soc 111:1886–1888. doi:10.1021/ja00187a057
Palacios F, Alonso C, Amezua P, Rubiales G (2002) Synthesis of aza polycyclic compounds derived from pyrrolidine, indolizidine, and indole via intramolecular Diels–Alder cycloadditions of neutral 2-azadienes. J Org Chem 67:1941–1946. doi:10.1021/jo016325v
Demuner AJ, Barbosa LCA, Miranda ACM, Geraldo GC, Da Silva CM, Giberti S, Bertazzini M, Forlani G (2013) The furgal phytotoxin alternariol 9-methyl ether and some of its synthetic analogues inhibit the photosynthetic electron transport chain. J Nat Prod 76:2234–2245. doi:10.1021/np4005882
Kand D, Kalle AM, Talukdar P (2013) Chromenoquinoline-based thiol probes: a study on the quencher position for controlling fluorescent off-on characteristics. Org Biomol Chem 11:1691–1701. doi:10.1039/C2OB27192C
Kudale AA, Miller DO, Dawe LN, Bodwell GJ (2011) Intramolecular Povarov reactions involving 3-aminocoumarins. Org Biomol Chem 9:7196–7206. doi:10.1039/C1OB05867C
Keskin S, Balci M (2015) Intramolecular heterocyclization of O-propargylated aromatic hydroxyaldehydes as an expedient route to substituted chromenopyridines under metal-free conditions. Org Lett 17:964–967. doi:10.1021/acs.orglett.5b00067
Goh KKK, Kim S, Zard SZ (2013) Free-radical variant for the synthesis of functionalized 1,5-diketones. Org Lett 15:4818–4821. doi:10.1021/ol402213k
Dolatkhah Z, Nasiri-Aghdam M, Bazgir A (2013) A three-component synthesis of benzochromeno- diazocines and chromenopyridines. Tetrahedron Lett 45:1960–1962. doi:10.1016/j.tetlet.2013.01.122
Safaei-Ghomi J, Shahbazi-Alavi H, Heidari-Baghbahadorani H (2014) SnO nanoparticles as an efficient catalyst for one-pot synthesis of chromeno[2,3-b]pyridines and 2-amino-3,5-dicyano-6-sulfanyl pyridines. RSC Adv 4:50668–50677. doi:10.1039/C4RA04769A
Elinson MN, Gorbunov SV, Vereshchagin AN, Nasybullin RF, Goloveshkin AS, Bushmarinov IS, Egorov MP (2014) Chemical and electrocatalytic cascade cyclization of sailcylaldehyde with three molecules of malononitrile: ‘one-pot’ simple and efficient way to the chromeno[2,3-b]pyridine scaffold. Tetrahedron 70:8559–8563. doi:10.1016/j.tet.2014.09.066
Dömling A (2006) Recent develpoments in isocyanide based mnlticomponent reactions in applied chemistry. Chem Rev 106:17–89. doi:10.1021/cr0505728
Tietze LF (1996) Domino reactions in organic synthesis. Chem Rev 96:115–136. doi:10.1021/cr950027e
Dömling A, Ugi I (2000) Multicomponent reactions with isocyanides. Angew Chem Int Ed 39:3168–3210. doi:10.1002/1521-3773(10000915)39:18<3168::AID-ANIE3168>3.0.CO;2-U
Tietz LF, Kinzel T, Brazel CC (2009) The domino multicomponent allylation reaction for the stereoselective synthesis of homoallylic alcohols. Acc Chem Res 42:367–378. doi:10.1021/ar800170y
Zhu J, Bienayme H (2005) Multicomponent reactions. Wiley, Weinheim
Balme G, Bossharth E, Monteiro N (2003) Pd-assisted multicomponent synthesis of heterocycles. Eur J Org Chem. doi:10.1002/ejoc.200300378
Bertozzi F, Gustafsson M, Olsson R (2002) A novel metal iodide promoted three-component synthesis of substituted pyrrolidines. Org Lett 4:3147–3150. doi:10.1021/ol0264814
Nair V, Vinod AU, Rajesh C (2001) A novel synthesis of 2-aminopyrroles using a three-component reaction. J Org Chem 66:4427–4429. doi:10.1021/jo001714v
Li S, Wu J (2011) Synthesis of H-pyrazolo[5,1-a]isoquinolines via copper (II)-catalyzed oxidation of an aliphatic C–H bond of tertiary amine in air. Org Lett 13:712–715. doi:10.1021/ol102939r
Wen LR, Shi YJ, Liu GY, Li M (2012) Modulating the reactivity of functionalized N,S-ketene acetal in MCR: selective synthesis of tetrahydropyridines and thiochromeno[2,3-b]pyridines via DABCO-catalyzed tandem annulation. J Org Chem 77:4252–4260. doi:10.1021/jo202665q
Saha A, Payra S, Banerjee S (2015) One-pot multicomponent synthesis of highly functionalized bio-active pyrano[2,3-c]pyrazole and benzylpyrazolyl coumarin using \(\text{ ZrO }_{2}\) nanoparticles as a reusable catalyst. Green Chem 17:2859–2866. doi:10.1039/C4GC02420F
Poomathi N, Mayakrishnan S, Muralidharan D, Srinivasan R, Perumal PT (2015) Reaction of isatins with 6-amino uracils and isoxazoles: isatin ring-operning vs. annulations and regioselective synthesis of isoxazole fused quinolones scaffolds in water. Green Chem 17:3362–3372. doi:10.1039/C5GC00006H
Vivekanand T, Vinoth P, Agieshkumar B, Sampath N, Sudalai A, Menéndez JC, Sridharan V (2015) Highly efficient regioselective synthesis of pyrroles via a tandem enamine formation-Michael addition-cyclization sequence under catalyst- and solvent-free conditions. Green Chem 17:3415–3423. doi:10.1039/C5GC00365B
Bora PP, Bihani M, Bez G (2015) Beyond enzymatic promiscuity: asymmetric induction by L-proline on lipase catalyzed synthesis of polyfunctionalized 4H-pyrans. RSC Adv 5:50597–50603. doi:10.1039/C5RA08785F
Kattuboina A, Kaur P, Nguyen T, Li G (2008) Chiral N-phosphonyl imine chemistry: asymmetric 1,2-additions of allylmagnesium bromides. Tetrahedron Lett 49:3722–3724. doi:10.1016/j.tetlet.2008.04.038
An G, Seifert C, Li G (2015) N-Phosphonyl/phosphinyl imines and group-assisted purification (GAP) chemistry/technology. Org Biomol Chem 13:1600–1617. doi:10.1039/C4OB02254H
Kaur P, Shakya G, Sun H, Pan Y, Li G (2010) Chiral N-phosphonyl imine chemistry: an efficient asymmetric synthesis of chiral N-phosphonyl propargylamines. Org Biomol Chem 8:1091–1096. doi:10.1039/B929314F
Kattamuri PV, Ai T, Pindi S, Sun Y, Gu P, Shi M, Li G (2011) Asymmetric synthesis of \(\alpha \)-amino-1,3-dithianes via chiral N-phosphonyl imine-based umpolung reaction without using chromatography and recrystallization. J Org Chem 76:2792–2797. doi:10.1021/jo200070d
Kattamuri PV, Xiong Y, Pan Y, Li G (2013) N,N-Diisopropyl-N-phosphonyl imines lead to efficient asymmetric synthesis of aziridine-2-carboxylic esters. Org Biomol Chem 11:3400–3408. doi:10.1039/C3OB40251G
Kaur P, Nguyen T, Li G (2009) Chiral N-phosphonylimine chemistry: Asymmetric synthesis of N-phosphonyl \(\beta \)-amino weinreb amides. Eur J Org Chem. doi:10.1002/ejoc.200801061
Kaur P, Wever W, Pindi S, Milles R, Gu P, Shi M, Li G (2011) The GAP chemistry for chiral N-phosphonyl imine-based Strecker reaction. Green Chem 13:1288–1292. doi:10.1039/C1GC15029D
Pindi S, Wu J, Li G (2013) Design, synthesis, and applications of chiral N-2-phenyl-2-propyl sulfinyl imines for group-assisted purification (GAP) asymmetric synthesis. J Org Chem 78:4006–4012. doi:10.1021/jo400354r
Cheng C, Jiang B, Tu SJ, Li G (2011) [4+2+1] Domino cyclization in water for chemo- and regioselective synthesis if spiro-substituted benzo[b]furo[3,4-e][1,4]diazepine derivatives. Green Chem 13:2107–2215. doi:10.1039/C1GC15183E
Alizadeh A, Rezvanian A, Zhu LG (2012) Synthesis of heterocyclic [3.3.3]propellanes via a sequential four-componentreaction. J Org Chem 77:4385–4390. doi:10.1021/jo300457m
Ahadi S, Kamranifard T, Armaghan M, Khavasia HR, Bazgir A (2014) Dimino Knoevenagel condensation-Michael addition-cyclization for diastereoselective synthesis of dihydrofuropyrido[2.3-d] pyrimidines via pyridinium ylides in water. RSC Adv 4:7296–7300. doi:10.1039/C3RA45795H
Akbarzadeh R, Amanpour T, Khavasi HR, Bazgir A (2014) Atom-economical isocyanide-based multicomponent synthesis of 2,5-dioxopyrrolidines, spirobenzothiazinechromanes and 1,5-benzothiazepines. Tetrahedron 70:169–175. doi:10.1016/j.tet.2013.12.011
Chennapuram M, Emmadi NR, Bingi C, Nanubolu JB, Atmakur K (2014) Group-assisted purification (GAP) chemistry for dihydrofurans: water as a medium for catalyst free synthesis in a one pot four component reaction. Green Chem 16:3237–3246. doi:10.1039/C4GC00388H
Liu J, Zhang HR, Lin XR, Yan SJ, Lin J (2014) Catalyst-free cascade reaction of heterocyclic ketene aminals with N-substituted maleimide to synthesis bicyclic pyrrolidinone derivatives. RSC Adv 4:27582–27590. doi:10.1039/C4RA03863K
Wang H, Shi D (2013) Efficient synthesis of functionalized dihydro-1H-indol-4(5H)-ones via one-pot three-component reaction under catalyst-free conditions. ACS Comb Sci 15:261–266. doi:10.1021/co4000198
Wang H, Liu X, Feng X, Huang Z, Shi D (2013) GAP chemistry for pyrrolyl coumarin derivatives: a highly efficient one-pot synthesis under catalyst-free conditions. Green Chem 15:3307–3311. doi:10.1039/C3GC41799A
Zhang J, Feng X, Liu X, Huang Z, Shi D (2014) An efficient three-component synthesis of highly functionalized tetrahydroacenaphtho[1,2-b]indolone derivatives catalyzed by L-proline. Mol Divers 18:727–736. doi:10.1007/s11030-014-9544-4
Feng X, Wang J, Lin W, Zhang J, Huang Z, Shi D (2014) Catalyst-free reaction in water: synthesis of functionalized tetrahydroindole derivatives via three-component domino reaction. Chin J Chem 32:889–896. doi:10.1002/cjoc.201400312
Wang J, Feng X, Xun Z, Shi D, Huang Z (2015) Multicomponent strategy to pyrazolo[3,4-e]indolizine derivatives under microwave irradiation. J Org Chem 80:8435–8442. doi:10.1021/acs.joc5b01314
Feng X, Wang J, Xun Z, Zhang J, Huang Z, Shi D (2015) Highly selective synthesis of functionalized polyhydroisoquinoline derivatives via a three-component domino reaction. Chem Commun 51:1528–1531. doi:10.1039/C4CC08900f
Acknowledgements
We are grateful for the Major Basic Research Project of the Natural Science Foundation of Jiangsu Higher Education Institutions (15KJA150006), the financial support from the Natural Science Foundation of Jiangsu Province (BK20131160) and a Project Funded by the Priority Academic Project Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zheng, YX., Xun, Z., Zhang, JJ. et al. An efficient one-pot synthesis of functionalized chromeno[4,3-b]pyridine derivatives under catalyst-free conditions. Mol Divers 21, 293–304 (2017). https://doi.org/10.1007/s11030-016-9723-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-016-9723-6