Abstract
Obesity has been identified as an independent risk factor for cardiovascular disease. Recent reports have highlighted the significance of stimulator of interferon genes (STING)-NOD-like receptor protein 3 (NLRP3) signaling pathway mediated pyroptosis, and inflammation in cardiovascular disease. Previous studies have demonstrated that exercise training effectively prevents cardiac pyroptosis and inflammation in high-fat diet (HFD)-fed mice. However, it is currently unknown whether exercise reduces pyroptosis and inflammation in obese hearts by targeting the STING-NLRP3 signaling pathway. We investigated the impact of an 8-week aerobic exercise regimen on cardiac function, pyroptosis, inflammation, and the STING-NLRP3 signaling pathway in HFD-induced obese mice. Additionally, to explore the underlying mechanism of STING in exercise-mediated cardioprotection, we administered intraperitoneal injections of the STING agonist diABZI to the mice. Furthermore, to investigate the role of the STING-NLRP3 signaling pathway in HFD-induced cardiac dysfunction, we administered adeno-associated virus 9 (AAV9) encoding shRNA targeting STING (shRNA-STING) via tail vein injection to knockdown STING expression specifically in mouse hearts. After one week of AAV9 injection, we intraperitoneally injected nigericin as an NLRP3 agonist. We first found that aerobic exercise effectively suppressed HFD-mediated upregulation of STING and NLRP3 in the hearts. Moreover, we demonstrated that the protective effect of aerobic exercise in HFD-induced cardiac dysfunction, pyroptosis, and inflammation was impaired by stimulating the STING pathway using diABZI. Additionally, activation of the NLRP3 with nigericin abolished the ameliorative effect of STING deficiency in HFD-induced cardiac dysfunction, pyroptosis, and inflammation. Based on these findings, we concluded that 8-week aerobic exercise alleviates HFD-induced cardiac dysfunction, pyroptosis, and inflammation by targeting STING-NLRP3 signaling pathway. Inhibition of STING-NLRP3 signaling pathway may serve as a promising therapeutic strategy against obesity-induced cardiomyopathy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Obesity is known to be an independent risk factor for cardiovascular disease [1]. Consumption of a high-fat diet (HFD) can lead to obesity and result in abnormal cardiac structure and dysfunction, characterized by myocardial hypertrophy, increased fibrosis, impaired contractile function, lipid accumulation, and various forms of cellular death [2,3,4]. Numerous studies have provided evidence supporting aerobic exercise (AE) as an effective intervention for mitigating HFD-induced cardiac dysfunction, underlying mechanisms involving inhibition of inflammation, reduction of oxidative stress, improvement in mitochondrial function, and promotion of cardiac autophagy [5,6,7,8]. Nevertheless, the precise mechanism responsible for exercise’s protective effect against obesity-induced cardiomyopathy remains elusive.
Pyroptosis is a form of programmed cell death accompanied by an inflammatory response. Previous studies have shown that chronic inflammation serves as a crucial role in obesity-related cardiomyopathy and can activate NOD-like receptor protein 3 (NLRP3) inflammasome, thereby initiating pyroptosis [9, 10]. The NLRP3 inflammasome is a complex composed of NLRP3, apoptosis-associated speck-like protein (ASC) and Caspase-1 [11]. Upon activation, the NLRP3 inflammasome triggers Caspase-1 activation, which leads to the production of pro-inflammatory cytokines interleukin (IL)-1β and IL18, initiating an inflammatory response [12]. Furthermore, the activation of NLRP3 inflammasome also induces pyroptosis through the formation of membrane pores mediated by gasdermin D (GSDMD) [13]. Although the crucial role of NLRP3 in obesity-induced cardiac dysfunction has been highlighted in recent study, the underlying molecular mechanisms are still not fully understood.
Stimulator of interferon gene (STING), localized predominantly in the endoplasmic reticulum, plays a pivotal regulatory role in the innate immune response and has been implicated in cardiovascular and metabolic diseases [14]. A previous study has shown that STING activation induces pyroptosis and inflammation in hepatic cells through the activation of NLRP3 [15]. However, it remains unclear whether STING is also involved in modulating NLRP3-mediated pyroptosis and inflammation in obese cardiac tissue. Previous studies have demonstrated that exercise training effectively mitigated HFD-induced cardiac pyroptosis and inflammation [16, 17]. However, it is currently unknown whether exercise reduces pyroptosis and inflammation in obese hearts by targeting the STING-NLRP3 signaling pathway.
Therefore, the objective of this study is to investigate the role of STING-NLRP3 signaling pathway in improving HFD-induced cardiac dysfunction, pyroptosis, and inflammation after 8-week aerobic exercise. Collectively, these findings provide further support for aerobic exercise as a promising therapeutic strategy for improving cardiac dysfunction among individuals with obesity.
Methods and materials
Animals and exercise protocol
The procedures were approved by the Institutional Animal Care and Use Committee of Taiyuan University of Technology and Tsinghua University. Five-week-old male C57BL/6 J mice were randomly maintained with the normal diet (ND, 10% kcal from fat) or HFD (60% kcal from fat, Research Diets, D12492) for 24 weeks. The body weight was recorded weekly.
A cohort of HFD-fed mice were subjected to 8-week aerobic exercise since the eighteen weeks. The aerobic exercise protocol was reported in our previous study [18]. In brief, aerobic exercise mice ran on the treadmill at a speed of 14–17.5 m/min for 60 min a day, 5 days a week. During the first week, the mice ran at a speed of 14 m/min, with an incremental increase of 0.5 m/min in the subsequent weeks. Aerobic exercise protocol is detailed in Supplementary Table 1.
Adeno-associated virus 9 (AAV9) and agonist treatment
To explore the potential involvement of STING in exercise-mediated cardioprotection, mice were subjected to intraperitoneal injection of the STING agonist diABZI at a dose of 1.5 mg/kg every three days for eight weeks, beginning at eighteen weeks [19]. The control mice were injected with an equal volume of solvent.
To investigate the role of STING in HFD-induced cardiac dysfunction, mice were administered with AAV9 encoding shRNA targeting STING (shRNA-STING) (at the dose of 1 × 1011 vg/mice) to knockdown STING in mice hearts via the tail vein injection, or with AAV9 control vector as a negative control (shRNA-NC). The AAV9 used above were purchased from Hanheng Biotechnology. After one week of AAV9 injection, mice were intraperitoneally injected with the NLRP3 agonist nigericin at 5 mg/kg every other day for eight weeks [19]. The control mice were injected with an equal volume of solvent.
Echocardiographic assessment
Mice were anesthetized with 1.5% isoflurane, maintaining heart rate ranging from 400 to 500 beats per minute. The mice were placed supine on a heated 37 °C platform to maintain body temperature. Transthoracic echocardiography was conducted using the high-resolution ultrasound imaging system (VINNO 6, Vinno Corporation, Suzhou, China) with a 23 MHz probe. M-mode echocardiograms were obtained at the level of the papillary muscle to indirectly measure the left ventricle internal dimension diastole (LVIDd), left ventricle internal dimension systole (LVIDs), left ventricular anterior wall thickness systolic (LVAWs), left ventricular anterior wall thickness diastolic (LVAWd), left ventricular posterior wall thickness systolic (LVPWs), left ventricular posterior wall thickness diastolic (LVPWd), left ventricular end-diastolic volume (LVEDV) and left ventricular end-systolic volume (LVESV). Ejection fraction (%) was calculated according to the formula: EF (%) = (LVEDV − LVESV)/LVEDV × 100. Fractional shortening (FS%) was calculated according to the formula: FS(%) = (LVIDd − LVIDs)/LVIDd × 100. The echocardiographer was blinded to the experimental treatments. All the echocardiography parameters measured in this study have been listed in Supplementary Table 2.
Histological examination
The cardiac tissue specimens were embedded in optimal cutting temperature (OCT) compound and subsequently cryosectioned into 10-μm slices. Oil Red O staining was employed to evaluate lipid deposition within the cardiac tissue, while 5-μm heart sections were subjected to picrosirius red staining for the detection of cardiac fibrosis. Histopathological images were visualized using an Olympus optical microscope (Olympus, Tokyo, Japan).
Serum total cholesterol and triglycerides assay
Serum total cholesterol and triglycerides were measured using a commercial assay kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). All detecting operations were performed following the kits manufacturer’s instructions.
Real-time qPCR
The total RNA of the frozen heart tissues was isolated using TRIzol reagent, followed by reverse transcription into cDNA using a Transcriptor First Strand cDNA Synthesis Kit. Real-Time qPCR was performed employing SYBR Green PCR Master Mix (Beyotime, Nanjing, China). The expression levels of target genes were analyzed with the 2−ΔΔCt method and normalized against glyceraldehyde-3-phosphate dehydrogenase (Gapdh) as an internal control. The primer sequences employed in our study are provided in Supplementary Table 3.
Western blot
The heart tissue samples were lysed by cold RIPA buffer. The protein concentration was quantified employing a BCA kit. The protein samples were separated by SDS-PAGE and transferred to PVDF membranes. Following blocking with 5% non-fat dry milk for 1 h at room temperature, the membranes were incubated with primary antibodies including STING (1: 1000, 13647, Cell Signaling Technology), NLRP3 (1: 1000, 15101, Cell Signaling Technology), pro-caspase-1 (1: 1000, ab179515, Abcam), cleaved caspase-1 (1: 1000, YC0003, Immunoway), GSDMD-N (1: 1000, ab215203, Abcam), GSDMD (1: 1000, ab219800, Abcam), L-1β (1: 1000, ab254360, Abcam), IL-18 (1: 1000, ab207323, Abcam), TNF-α (1: 1000, 3707, Cell Signaling Technology) and β-actin (1: 5000, GTX109639, Gene Tex) at 4 °C overnight. The membranes were incubated with the horseradish peroxidase-coupled secondary antibodies for 1 h at room temperature. Immunopositive bands were observed with ChemiDoc Touch Imaging System (Bio-Rad, USA).
Statistical analysis
All data are expressed as the mean ± standard deviation (SD). Analyses were performed using GraphPad Prism 9.0. Data were compared using two-way ANOVA followed by a Tukey’s multiple-comparison post hoc test. p < 0.05 was considered statistically significant.
Results
Aerobic exercise alleviates HFD-induced cardiac dysfunction, pyroptosis, and inflammation
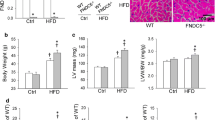
Echocardiography and cardiac morphological properties were performed in ND and HFD mice following 8-week aerobic exercise (Fig. 1A). We observed that HFD feeding resulted in increased body weight, serum total cholesterol and triglycerides, these effects were alleviated by aerobic exercise (Fig. 1B–D). In addition, echocardiography analysis depicted that HFD intake significantly increased LVIDs and LVIDd and decreased EF and FS, but these effects were reversed by aerobic exercise (Fig. 1E–I). Oil red O and Picrosirius Red staining showed that lipid accumulation and cardiac fibrosis were observed in HFD-induced obese hearts, while aerobic exercise alleviated these histological alterations (Fig. 1J–M).
Aerobic exercise improves HFD-induced cardiac dysfunction, pyroptosis, and inflammation. A Schematic diagram of aerobic exercise and HFD-induced obesity model establishment. B–D Statistical analysis of body weight, serum total cholesterol and triglycerides (n = 10). E Representative echocardiographic images. F–I Quantitative data on LVIDs, LVIDd, FS, and EF (n = 10). J–M Representative images and quantitative analysis of Oil red O staining (scale bar: 20 μm) and Picrosirius Red staining (scale bar: 50 μm; n = 3). N–S Western blot images and quantitative densitometry analysis of cleaved caspase-1, pro-caspase-1, GSDMD-N, GSDMD, IL-1β, IL-18, and TNF-α (n = 3). Data are shown as means ± SD. *p < 0.05, **p < 0.01. ns: no significant difference
Subsequently, we investigated whether aerobic exercise attenuated HFD-induced cardiomyocyte pyroptosis and inflammation. Western blot analysis showed that the pyroptosis biomarkers cleaved caspase-1 and GSDMD-N, and the inflammation biomarkers IL-1β, IL-18, and TNF-α were up-regulated by HFD feeding. More importantly, aerobic exercise effectively inversed the above alterations induced by HFD (Fig. 1N–S). Collectively, these results indicated that aerobic exercise alleviates HFD-induced cardiac dysfunction, pyroptosis, and inflammation.
The cardiac STING and NLRP3 expression are increased after HFD and decreased after aerobic exercise
Aberrant activation of the STING-NRLP3 signaling pathway plays an essential role in cardiovascular destruction, pyroptosis, and inflammation [19, 20]. Previous research has shown that STING and NLRP3 were increased in the hearts of diabetic mice [21]. In this study, we found that HFD feeding significantly increased the cardiac mRNA and protein levels of STING and NLRP3, but these effects were reversed by aerobic exercise (Fig. 2A–E). These data suggest that aerobic exercise might protect against HFD-induced cardiac dysfunction, pyroptosis, and inflammation by inhibiting STING-NLRP3 signaling pathway.
Aerobic exercise effectively suppressed HFD-mediated up-regulation of STING and NLRP3 in the hearts. A–B The mRNA levels of Sting and Nlrp3 in cardiac tissues by RT-qPCR (n = 4). C–E Western blot images and quantitative densitometry analysis of STING and NLRP3 (n = 5). Data are shown as means ± SD. *p < 0.05, **p < 0.01. ns: no significant difference
STING activation impairs the protective effect of aerobic exercise in HFD-induced cardiac dysfunction, pyroptosis, and inflammation
To clarify whether a decrease in STING is responsible for the protective effect of aerobic exercise in HFD-induced cardiac dysfunction, a STING agonist (diABZI) was intraperitoneally injected in both the sedentary group and the aerobic exercise group of HFD-feed mice (Fig. 3A). The other 2 parallel mice groups were injected with an equal volume of solvent. We observed intraperitoneal injection of diABZI had no significant effect on body weight in HFD mice (Fig. 3B). As expected, diABZI significantly increased the cardiac expression of STING and exacerbated HFD-induced cardiac dysfunction, as evidenced by the increased LVIDs and LVIDd and the decreased FS and EF (Fig. 3E–I). Histological analyses showed that diABZI also exacerbated HFD-induced lipid accumulation and cardiac fibrosis (Fig. 3J–M). Notably, the beneficial effects of aerobic exercise in HFD-induced cardiac dysfunction appears to have been negated by diABZI, as there was no significant difference in the serum total cholesterol, triglycerides, LVIDs, LVIDd, FS, EF, lipid accumulation, and cardiac fibrosis between the HFD + diABZI + SED and HFD + diABZI + AE groups (Fig. 3C–M), indicating that inhibition of STING is essential for aerobic exercise induced cardiac protection.
STING agonist diABZI impairs the protective effect of aerobic exercise in HFD-induced cardiac dysfunction, lipid accumulation, and fibrosis. A Schematic diagram of aerobic exercise, diABZI injection and HFD-induced obesity model establishment. B–D Statistical analysis of body weight, serum total cholesterol and triglycerides (n = 8). E Representative echocardiographic images. F–I Quantitative data on LVIDs, LVIDd, FS, and EF (n = 8). J–M Representative images and quantitative analysis of Oil red O staining (scale bar: 20 μm) and Picrosirius Red staining (scale bar: 50 μm; n = 3). Data are shown as means ± SD. *p < 0.05, **p < 0.01. ns: no significant difference
Given the role of STING in pyroptosis and inflammation, we next investigated whether diABZI could abolish the anti-pyroptotic and anti-inflammation effect of aerobic exercise. As expected, STING agonist exacerbated HFD-induced cardiomyocyte pyroptosis and inflammation, as evidenced by the increased NLRP3, cleaved caspase-1, GSDMD-N, IL-1β, IL-18, and TNF-α. Notably, there was no significant difference in pyroptosis biomarkers and inflammation biomarkers levels between the HFD + diABZI + SED and HFD + diABZI + AE groups (Fig. 4A–H). These results demonstrated that aerobic exercise attenuated HFD-induced cardiac dysfunction, pyroptosis, and inflammation by inhibiting STING.
STING agonist diABZI abolishes the anti-pyroptotic and anti-inflammation effect of aerobic exercise. A–H Representative western blot images and quantitative densitometry analysis of pyroptosis biomarkers and inflammation biomarkers by detection of STING, NLRP3, cleaved caspase-1, pro-caspase-1, GSDMD-N, GSDMD, IL-1β, IL-18, and TNF-α expression levels (n = 3). Data are shown as means ± SD. *p < 0.05, **p < 0.01. ns: no significant difference
NLRP3 activation abolishes the ameliorative effect of STING deficiency in HFD-induced cardiac dysfunction, pyroptosis, and inflammation
NLRP3, as a downstream effector of STING, is essential for the activation of pyroptosis and inflammation [22]. Therefore, we explored whether STING deficiency exerts an anti-pyroptotic and anti-inflammation effect on HFD-induced cardiac dysfunction by inhibiting NLRP3. We used AAV9 encoding shRNA targeting STING (shRNA-STING) to knockdown STING in the hearts of HFD mice via the tail vein in vivo (Fig. 5A). Cardiac STING downregulation was verified by western blotting upon shRNA-STING (Fig. 6A–B). We first observed that AAV9 encoding shRNA-STING had no significant effect on body weight, serum total cholesterol and triglycerides in HFD-fed mice (Fig. 5B–D). We found that STING knockdown improved HFD-induced cardiac dysfunction as demonstrated by the decreased LVIDs and LVIDd and the increased FS and EF (Fig. 5E–I). In addition, STING deficiency also reduced lipid accumulation and cardiac fibrosis in HFD-induced obese hearts (Fig. 5J–M).
NLRP3 agonist nigericin abolishes the ameliorative effect of STING deficiency in HFD-induced cardiac dysfunction, lipid accumulation, and fibrosis. A Schematic diagram of AAV9 injection, diABZI injection, and HFD-induced obesity model establishment. B–D Statistical analysis of body weight, serum total cholesterol and triglycerides (n = 8). E Representative echocardiographic images. F–I Quantitative data on LVIDs, LVIDd, FS and EF (n = 8). J–M Representative images and quantitative analysis of Oil red O staining (scale bar: 20 μm) and Picrosirius Red staining (scale bar: 50 μm; n = 3). Data are shown as means ± SD. *p < 0.05, **p < 0.01. ns: no significant difference
NLRP3 agonist nigericin abolishes the ameliorative effect of STING deficiency in HFD-induced cardiomyocyte pyroptosis, and inflammation. A–H Representative western blot images and quantitative densitometry analysis of STING, NLRP3, cleaved caspase-1, pro-caspase-1, GSDMD-N, GSDMD, IL-1β, IL-18, and TNF-α (n = 3). Data are shown as means ± SD. *p < 0.05, **p < 0.01. ns: no significant difference
One week after AAV9 injection, a NLRP3 agonist (nigericin) was intraperitoneally injected on alternate days in both the HFD + shRNA-NC group and the HFD + shRNA-STING group of HFD-fed mice. The other 2 parallel mice groups were injected with an equal volume of solvent. As expected, nigericin significantly increased the cardiac expression of NLRP3 and exacerbated HFD-induced cardiac dysfunction, as evidenced by the increased LVIDs and LVIDd and the decreased FS and EF (Fig. 5E–I). Nigericin also exacerbated HFD-induced lipid accumulation and cardiac fibrosis (Fig. 5J–M). Importantly, the ameliorative effects of STING knockdown in HFD-induced cardiac dysfunction appears to have been blocked by nigericin, as there was no significant difference in the serum total cholesterol, triglycerides, LVIDs, LVIDd, FS, EF, lipid accumulation, and cardiac fibrosis between the HFD + Nigericin + shRNA-NC and HFD + Nigericin + shRNA-STING groups (Fig. 5C–M), suggesting that STING knockdown attenuated HFD-induced cardiac dysfunction by inhibiting NLRP3.
We further investigated whether NLRP3 activation could abolish the anti-pyroptotic and anti-inflammation effect of STING knockdown. As expected, STING deficiency reduced HFD-induced cardiomyocyte pyroptosis and inflammation, as demonstrated by the decreased NLRP3, cleaved caspase-1, GSDMD-N, IL-1β, IL-18, and TNF-α. In addition, nigericin exacerbated cardiomyocyte pyroptosis and inflammation in HFD-induced obese hearts. Notably, the levels of HFD-induced cardiac pyroptosis biomarkers and inflammation biomarkers were comparable between shRNA-NC and shRNA-STING treatment in Nigericin injected HFD-fed mice (Fig. 6A–H). Taken together, these results suggested that STING knockdown improved HFD-induced cardiac dysfunction, pyroptosis, and inflammation by inhibiting NLRP3.
Discussion
Obesity is recognized as an independent risk factor for cardiovascular disease [1]. Previous studies have demonstrated that cardiomyocyte pyroptosis and inflammation are the primary pathological mechanisms underlying obesity-related cardiomyopathy [9, 23]. Moderate-intensity aerobic exercise has been widely acknowledged to confer beneficial effects on cardiovascular diseases [24, 25]. In this study, we present evidence that 8-week aerobic exercise alleviates HFD-induced cardiac dysfunction, pyroptosis, and inflammation by targeting the STING-NLRP3 signaling pathway.
To date, no approved weight-loss drug has shown efficacy in reducing the risk of cardiovascular events in obese individuals while also promoting weight loss [26]. Aerobic exercise has been proven as an effective approach for fat and weight reduction, as well as a therapeutic intervention to enhance cardiac function [27, 28]. A previous study showed that 12-week exercise training reduced cardiac lipid content and improved left ventricular EF in male overweight/obese subjects [29]. Another study reported significant improvements in FS and EF, along with reductions in cardiac hypertrophy, fibrosis, and lipid accumulation following 8 weeks of swimming exercise in HFD-induced obese hearts [30]. Similarly, our study employed echocardiography along with Oil red O and Picrosirius Red staining to demonstrate that significant amelioration of HFD-induced cardiac dysfunction, fibrosis, and lipid accumulation after 8-week aerobic exercise. However, the precise molecular mechanism underlying exercise-induced protection against obesity-related cardiomyopathy remains unclear.
HFD-induced cardiac dysfunction involves multiple molecular mechanisms, including oxidative stress, inflammation, lipid accumulation, mitochondrial dysfunction, interstitial fibrosis and cardiomyocyte hypertrophy [31, 32]. Recent studies have highlighted the crucial roles played by the NLRP3 inflammasome and pyroptosis in the pathological processes underlying HFD-induced heart injury [9, 33]. Pyroptosis represents a prototypical form of programmed cell death that is closely associated with inflammatory activation [13]. Notably, a study demonstrated that treadmill exercise effectively mitigated cardiac pyroptosis and inflammation in HFD-feeding mice [16]. Consistent with these findings, our study aligns with previous research indicating that aerobic exercise ameliorates HFD-induced cardiac pyroptosis and inflammation. Collectively, these findings suggested that aerobic exercise exerts beneficial effects on HFD-induced cardiac dysfunction through suppression of cardiomyocyte pyroptosis and inflammation.
STING, a key adaptor protein in the innate immune response, is predominantly localized in the endoplasmic reticulum [34]. Growing evidence suggests that activation of STING leads to the release of various pro-inflammatory factors and plays a crucial role in initiating pyroptosis [35]. Recent reports have highlighted the significance of STING-mediated pyroptosis and inflammation in cardiovascular disease[14, 36]. Knockout of STING has been shown to significantly ameliorate lipopolysaccharide (LPS)-induced cardiac injury in mice by inhibiting myocardial pyroptosis, inflammation, and apoptosis [20]. In addition, STING inhibitor C-176 or heart specific knockdown STING has demonstrated promising results in reducing myocardial pyroptosis and inflammatory response while improving cardiac function in diabetic mice[21, 37]. Similarly, our study revealed that up-regulation of STING expression in HFD-induced obese hearts; however, STING knockdown effectively improved cardiac dysfunction, pyroptosis, and inflammation. Interestingly, we found that aerobic exercise effectively reversed the HFD-induced up-regulation of STING expression, suggesting that aerobic exercise may play the anti-pyroptosis and anti-inflammatory role by down-regulating STING expression. Notably, a recent study reported that overexpression of STING compromised the neuroprotective effect exerted by treadmill exercise following traumatic brain injury [38]. In line with these findings, our study showed that intraperitoneal injection of the STING agonist diABZI largely abrogated the protective effect conferred by aerobic exercise against HFD-induced cardiac dysfunction as well as attenuated pyroptosis and inflammation. These findings confirmed that aerobic exercise attenuated HFD-induced cardiac dysfunction, pyroptosis and inflammation by inhibiting STING.
NLRP3 serves as a crucial mediator in initiating immune response and inflammasome formation, thereby promoting the maturation and secretion of pro-inflammatory cytokines such as Caspase1, IL-1β, and IL-18, and initiate pyrodeath [39]. NLRP3 is believed to play a pivotal role in sensing obesity-related metabolic stress and mediating associated inflammatory responses and pyroptosis [40, 41]. A previous study has shown that NLRP3 knockout reduces systemic inflammation and improves cardiac remodeling in HFD-fed mice [9]. Additionally, silencing the NLRP3 gene has been shown to ameliorate cardiac inflammation, pyroptosis, fibrosis, and cardiac function in diabetic rats [42]. In our study, we observed an up-regulation of NLRP3 expression in HFD-induced obese hearts. Moreover, intraperitoneal injection of the NLRP3 agonist Nigericin further exacerbated HFD-induced cardiac dysfunction, pyroptosis, and inflammation. These findings underscore the critical role of NLRP3 activation in obesity-related myocardial injury. A recent study reported that STING activation induces pyroptosis and inflammation in hepatic cells through the activation of NLRP3 [15]. Interestingly, in the present study, we found that aerobic exercise downregulates the expression of NLRP3 in HFD-induced obese hearts. Therefore, we hypothesize that NLRP3 is a downstream target of STING down-regulation to exert anti-pyroptosis and anti-inflammatory effects. Our present study revealed that NLRP3 agonist Nigericin significantly reversed the ameliorative effect of STING deficiency in HFD-induced cardiac dysfunction, pyroptosis and inflammation, suggesting that STING knockdown improves HFD-induced cardiac dysfunction, pyroptosis and inflammation by inhibiting NLRP3.
To the best of our knowledge, this study represents the first evidence demonstrating that aerobic exercise mitigates HFD-induced cardiac dysfunction, pyroptosis, and inflammation by inhibiting the STING-NLRP3 signaling pathway. These findings further support the potential therapeutic efficacy of aerobic exercise in ameliorating heart dysfunction in obese patients and highlight the pivotal role played by the STING-NLRP3 signaling pathway. However, it should be noted that this study did not employ transgenic mice with cardiac-specific knockout or overexpression of STING to confirm the underlying mechanism. Furthermore, a comprehensive investigation into the upstream molecular mechanisms through which aerobic exercise downregulates cardiac STING expression is still warranted.
Conclusion
Overall, this study has demonstrated 8-week aerobic exercise alleviates HFD-induced cardiac dysfunction, pyroptosis, and inflammation by targeting STING-NLRP3 signaling pathway. Therefore, inhibition of STING-NLRP3 signaling pathway may serve as a promising therapeutic strategy against obesity-induced cardiomyopathy.
Data availability
The datasets used and analyzed during the current study are available upon request by contacting with the corresponding author.
Abbreviations
- HFD:
-
High-fat diet
- STING:
-
Stimulator of interferon genes
- NLRP3:
-
NOD-like receptor protein 3
- AE:
-
Aerobic exercise
- ASC:
-
Apoptosis-associated speck-like protein
- GSDMD:
-
Gasdermin D
- IL-1β:
-
Interleukin-1β
- ND:
-
Normal diet
- AAV9:
-
Adeno-associated virus 9
- LVIDd:
-
Left ventricle internal dimension diastole
- LVIDs:
-
Left ventricle internal dimension systole
- EF:
-
Ejection fraction
- FS:
-
Fractional shortening
References
Powell-Wiley TM, Poirier P, Burke LE et al (2021) Obesity and cardiovascular disease: a scientific statement from the american heart association. Circulation 143(21):E984–E1010
Xu ZJ, Qin Y, Lv BB et al (2022) Intermittent fasting improves high-fat diet-induced obesity cardiomyopathy via alleviating lipid deposition and apoptosis and decreasing m6a methylation in the heart. Nutrients 14(2):251
Zhu MY, Peng LL, Huo SQ et al (2023) STAT3 signaling promotes cardiac injury by upregulating NCOA4-mediated ferritinophagy and ferroptosis in high-fat-diet fed mice. Free Radical Biol Med 201:111–125
Hu QX, Zhang HL, Cortés NG et al (2020) Increased Drp1 acetylation by lipid overload induces cardiomyocyte death and heart dysfunction. Circ Res 126(4):456–470
Chen XD, Li HY, Wang KW et al (2019) Aerobic exercise ameliorates myocardial inflammation, fibrosis and apoptosis in high-fat-diet rats by inhibiting P2X7 purinergic receptors. Front Physiol 10:1286
Kim K, Ahn N, Jung S (2018) Comparison of endoplasmic reticulum stress and mitochondrial biogenesis responses after 12 weeks of treadmill running and ladder climbing exercises in the cardiac muscle of middle-aged obese rats. Braz J Med Biol Res 51(10):e7508
Ko JB, Jang YC, Quindry J et al (2023) Exercise-induced antisenescence and autophagy restoration mitigate metabolic disorder-induced cardiac disruption in mice. Med Sci Sports Exerc 55(3):376–388
Alexandre-Santos B, Alves R, Matsuura C et al (2020) Modulation of cardiac renin-angiotensin system, redox status and inflammatory profile by different volumes of aerobic exercise training in obese rats. Free Radical Biol Med 156:125–136
Sokolova M, Sjaastad I, Louwe MC et al (2019) NLRP3 inflammasome promotes myocardial remodeling during diet-induced obesity. Front Immunol 10:1621
Yue QX, Liu Y, Ji J et al (2023) Down-regulation of OIP5-AS1 inhibits obesity-induced myocardial pyroptosis and miR-22/NLRP3 inflammasome axis. Immun Inflamm Dis 11(10):e1066
Toldo S, Mezzaroma E, Buckley LF et al (2022) Targeting the NLRP3 inflammasome in cardiovascular diseases. Pharmacol Ther 236:108053
Mangan MSJ, Olhava EJ, Roush WR et al (2018) Targeting the NLRP3 inflammasome in inflammatory diseases. Nat Rev Drug Discovery 17(9):688
Toldo S, Abbate A (2023) The role of the NLRP3 inflammasome and pyroptosis in cardiovascular diseases. Nat Rev Cardiol. https://doi.org/10.1038/s41569-023-00946-3
Oduro PK, Zheng XX, Wei JN et al (2022) The cGAS-STING signaling in cardiovascular and metabolic diseases: Future novel target option for pharmacotherapy. Acta Pharmaceutica Sinica B 12(1):50–75
Xiao Y, Zhao C, Tai Y et al (2023) STING mediates hepatocyte pyroptosis in liver fibrosis by epigenetically activating the NLRP3 inflammasome. Redox Biol 62:102691
Kar S, Shahshahan HR, Hackfort BT et al (2019) Exercise training promotes cardiac hydrogen sulfide biosynthesis and mitigates pyroptosis to prevent high-fat diet-induced diabetic cardiomyopathy. Antioxidants 8(12):638
Kesherwani V, Chavali V, Hackfort BT et al (2015) Exercise ameliorates high fat diet induced cardiac dysfunction by increasing interleukin 10. Front Physiol 6:124
Xu ZJ, Qin Y, Lv BB et al (2022) Effects of moderate-intensity continuous training and high-intensity interval training on testicular oxidative stress, apoptosis and m6A Methylation in obese male mice. Antioxidants 11(10):1874
Fang GY, Li XC, Yang FY et al (2023) Amentoflavone mitigates doxorubicin-induced cardiotoxicity by suppressing cardiomyocyte pyroptosis and inflammation through inhibition of the STING/NLRP3 signalling pathway. Phytomedicine 117:154922
Li N, Zhou H, Wu HM et al (2019) STING-IRF3 contributes to lipopolysaccharide-induced cardiac dysfunction, inflammation, apoptosis and pyroptosis by activating NLRP3. Redox Biol 24:101215
Yan ML, Li Y, Luo QM et al (2022) Mitochondrial damage and activation of the cytosolic DNA sensor cGAS-STING pathway lead to cardiac pyroptosis and hypertrophy in diabetic cardiomyopathy mice. Cell Death Discov 8(1):258
Gaidt MM, Ebert TS, Chauhan D et al (2017) The DNA inflammasome in human myeloid cells is initiated by a STING-cell death program upstream of NLRP3. Cell 171(5):1110–1124
Gastaldi S, Rocca C, Gianquinto E et al (2023) Discovery of a novel 1,3,4-oxadiazol-2-one-based NLRP3 inhibitor as a pharmacological agent to mitigate cardiac and metabolic complications in an experimental model of diet-induced metaflammation. Eur J Med Chem 257:115542
Jiang JZ, Ni LY, Zhang XX et al (2023) Moderate-intensity exercise maintains redox homeostasis for cardiovascular health. Adv Biol 7(4):e2200204
Chen HH, Chen C, Spanos M et al (2022) Exercise training maintains cardiovascular health: signaling pathways involved and potential therapeutics. Signal Transduct Target Ther 7(1):306
Wong ND, Karthikeyan H, Fan WJ (2023) US population eligibility and estimated impact of semaglutide treatment on obesity prevalence and cardiovascular disease events. Cardiovasc Drugs Ther. https://doi.org/10.1007/s10557-023-07488-3
Swift DL, Johannsen NM, Lavie CJ et al (2014) The role of exercise and physical activity in weight loss and maintenance. Prog Cardiovasc Dis 56(4):441–447
Vega RB, Konhilas JP, Kelly DP et al (2017) Molecular mechanisms underlying cardiac adaptation to exercise. Cell Metab 25(5):1012–1026
Schrauwen-Hinderling VB, Hesselink MKC, Meex R et al (2010) Improved ejection fraction after exercise training in obesity is accompanied by reduced cardiac lipid content. J Clin Endocrinol Metab 95(4):1932–1938
Li SC, Qian XL, Gong JH et al (2021) Exercise training reverses lipotoxicity-induced cardiomyopathy by inhibiting HMGCS2. Med Sci Sports Exerc 53(1):47–57
Gutiérrez-Cuevas J, Sandoval-Rodriguez A, Meza-Rios A et al (2021) Molecular mechanisms of obesity-linked cardiac dysfunction: an up-date on current knowledge. Cells 10(3):629
Ren J, Wu NN, Wang SY et al (2021) Obesity cardiomyopathy: evidence, mechanisms, and therapeutic implications. Physiol Rev 101(4):1745–1807
Scott L, Fender AC, Saljic A et al (2021) NLRP3 inflammasome is a key driver of obesity-induced atrial arrhythmias. Cardiovasc Res 117(7):1746–1759
Hopfner KP, Hornung V (2020) Molecular mechanisms and cellular functions of cGAS-STING signalling. Nat Rev Mol Cell Biol 21(9):501–521
Decout A, Katz JD, Venkatraman S et al (2021) The cGAS-STING pathway as a therapeutic target in inflammatory diseases. Nat Rev Immunol 21(9):548–569
Luo W, Zou XY, Wang YD et al (2023) Critical role of the cGAS-STING pathway in doxorubicin-induced cardiotoxicity. Circ Res 132(11):E223–E242
Ma XM, Geng K, Law BYK et al (2023) Lipotoxicity-induced mtDNA release promotes diabetic cardiomyopathy by activating the cGAS-STING pathway in obesity-related diabetes. Cell Biol Toxicol 39(1):277–299
Chen J, Zhu T, Yu DY et al (2023) Moderate intensity of treadmill exercise rescues TBI-induced ferroptosis, neurodegeneration, and cognitive impairments via suppressing STING pathway. Mol Neurobiol 60(9):4872–4896
Fu JN, Wu H (2023) Structural mechanisms of NLRP3 inflammasome assembly and activation. Annu Rev Immunol 41:301–316
Pan M, Tai RQ, Liu YR et al (2022) Pyroptosis: a possible link between obesity-related inflammation and inflammatory diseases. J Cell Physiol 237(2):1245–1265
Vandanmagsar B, Youm YH, Ravussin A et al (2011) The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat Med 17(2):179–188
Luo BB, Li B, Wang WK et al (2014) NLRP3 gene silencing ameliorates diabetic cardiomyopathy in a type 2 diabetes rat model. PLoS ONE 9(8):e104771
Acknowledgements
This work is particularly supported by Taiyuan University of Technology high-level talent scientific research funding.
Funding
This study was supported by Science and Technology Service Project of Winter Sports Management Center of General Administration of Sport of China (No. 20212001045) and Taiyuan University of Technology high-level talent scientific research funding.
Author information
Authors and Affiliations
Contributions
ZX and BZ researched and designed the experiment. ZX and ZM performed the experiment and analyzed the data. ZX, XZ, and BZ contributed reagents and materials. ZX wrote and edited the manuscript. All authors have read and critically revised the manuscript and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
All animal experiments were approved by the Institutional Animal Care and Use Committee of Taiyuan University of Technology and Tsinghua University.
Consent for publication
No human studies or human tissues were used in this research study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11010_2024_4950_MOESM1_ESM.docx
Supplementary file1 (DOCX 27 KB)—Table S1 Aerobic exercise protocol. Table S2 Biometric and echocardiographic measurements in experimental mice. Table S3 Primer sequences used for Real-Time qPCR.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, Z., Ma, Z., Zhao, X. et al. Aerobic exercise mitigates high-fat diet-induced cardiac dysfunction, pyroptosis, and inflammation by inhibiting STING-NLRP3 signaling pathway. Mol Cell Biochem (2024). https://doi.org/10.1007/s11010-024-04950-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11010-024-04950-0