Abstract
Heart disease causing cardiac cell death due to ischemia–reperfusion injury is a major cause of morbidity and mortality in the United States. Coronary heart disease and cardiomyopathies are the major cause for congestive heart failure, and thrombosis of the coronary arteries is the most common cause of myocardial infarction. Cardiac injury is followed by post-injury cardiac remodeling or fibrosis. Cardiac fibrosis is characterized by net accumulation of extracellular matrix proteins in the cardiac interstitium and results in both systolic and diastolic dysfunctions. It has been suggested by both experimental and clinical evidence that fibrotic changes in the heart are reversible. Hence, it is vital to understand the mechanism involved in the initiation, progression, and resolution of cardiac fibrosis to design anti-fibrotic treatment modalities. Animal models are of great importance for cardiovascular research studies. With the developing research field, the choice of selecting an animal model for the proposed research study is crucial for its outcome and translational purpose. Compared to large animal models for cardiac research, the mouse model is preferred by many investigators because of genetic manipulations and easier handling. This critical review is focused to provide insight to young researchers about the various mouse models, advantages and disadvantages, and their use in research pertaining to cardiac fibrosis and hypertrophy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Heart disease is the foremost cause of death in the United States. Cell death induced by ischemia/reperfusion (IR) is a major cause of morbidity and mortality among those suffering from heart disease [1, 2]. Both survival and quality of life in patients suffering from myocardial infarction (MI) can be improved by minimizing events occurring due to IR. The incidence, prevalence, and mortality, as well as the economic cost, for MI and congestive heart failure (CHF) are increasing due to an increase in life expectancy and a decrease in mortality after such events. The prevalence of CHF is 1–2 % in middle-aged people and 2–3 and 5–10 % in person older than 65 and 75 years, respectively [3]. Currently in the United States, the prevalence of heart failure exceeds 5.8 million cases and each year >550,000 new cases are diagnosed [4]. In 2013, in the United States, 1 of every 20 deaths was caused by stroke, 1 of every 7 deaths by coronary artery disease, and 1 of every 9 deaths due to heart failure [5]. Coronary heart disease (CHD) is the leading cause of CHF and accounted for 67 % cases in the 1980s [6], and may contribute up to 73 % cases of heart failure [7]. Other causes of CHF are valvular heart disease (10 %); primary myocardial disease mainly dilated cardiomyopathy (20 %); and arterial hypertension (HTN) (57 %). Overall 5-year survival rates after heart failure (HF) were found to be 25 % in men and 38 % in women in the Framingham study [6]. The causes of death in post CHF patients were sudden death (approximately 40 % of patients), worsening of HF (approximately 40 %) and other factors (20 %) [8]. Duration and severity of the disease, patient gender, and therapeutic strategies decide the survival of patients suffering from HF.
Ischemic injury to the heart leads to cardiac remodeling and fibrosis, and causes HF as an end result. Advanced heart failure syndrome presents a complex picture, including disturbed myocardial function, ventricular remodeling, altered hemodynamics, neuro-humoral activation, cytokine overexpression, and vascular and endothelial dysfunction [8]. Insight into the complex pathophysiology of cardiac remodeling, fibrosis, and HF may help in understanding the disease and lead to development of better therapeutic approaches. Cardiovascular researchers are working in this direction and experimental animal models are of immense help in understanding the ongoing disease process. Animal models are also helpful in understanding the effect of interventions on the disease process, which in turn helps in development of better therapeutic approach. Mouse model can be used to study the transition process occurring in heart failure; the effect of new pharmacological strategies on hemodynamics, neuro-humoral activation, and survival under some preclinical conditions; molecular alterations underlying diseases; the pathophysiology of disease development; molecular techniques used to correct sub-cellular processes in failing heart; and for the study of consequence of gene transfer. This review is focused on the various mouse models used in cardiovascular research.
Normal myocardium of heart
To understand the changes in the myocardium after an injury, the knowledge of the structure of the normal myocardium is important. The structural support of the heart is attributed to its extracellular matrix (ECM) network. The heart’s pumping action is due to the force generated by the cardiomyocyte. The ECM network mainly consist of type I collagen with smaller amounts of type III collagen. This collagen network facilitates the transmission of a systolic force. Tensile strength is provided by thick fibers mainly of type I collagen (85 % of total myocardial collagen). Type III collagen (11 % of the total collagen protein in the heart) forms thin fibers and is responsible for the matrix network’s elasticity [9].
Morphologically, the cardiac matrix network can be subdivided into three constituents: the epimysium, perimysium, and endomysium [10] (Fig. 1). The entire cardiac muscle is enveloped by the epimysium and is located on the endocardial and epicardial surface providing support for endothelial and mesothelial cells. The perimysium arises from the epimysium and surrounds groups of muscle fibers. The individual muscle fibers are enwrapped by endomysium, the final arborization of the perimysium. Endomysial struts bind muscle fibers together to their nutrient microvasculature. They also function as the sites for connections to cardiomyocyte cytoskeletal proteins across the plasma membrane. Increased perimysial deposition of collagen results in interstitial fibrosis, and involvement of the intramural coronary arterial vasculature causes perivascular fibrosis in a fibrotic heart. In addition to collagens, glycoproteins, glycosaminoglycans (hyaluronan), and proteoglycans also form the cardiac ECM. Latent growth factors and proteases in the cardiac ECM may trigger the fibrotic response with their activation following injury [11].
Post infarction myocardial repair
Post-infarction healing and scarring in the heart occurs as a result of activation of the inflammatory response triggered by the hypoxia-induced death of cardiomyocyte [12]. The three overlapping phases in the process of cardiac repair are the inflammatory phase, proliferative phase, and maturation phase (Fig. 2; Table 1). Cardiomyocyte death due to hypoxia triggers the initial inflammation modulated through various steps involving initiation of the complement cascade, free radical generation, and activation of nuclear Factor kappa B (NF-κB), release of endogenous ligands called danger-associated molecular patterns (DAMPs) [13] and toll-like receptor (TLR)-mediated signaling pathways in the inflammatory phase. Neutrophils are the first cells to arrive at the area of injury; they release chemokines that induce the accumulation of monocytes. They digest the damaged tissue and clear the cellular debris by phagocytosis [14].
Cardiac fibrosis following ischemic injury: myocardial injury results in death of cardiomyocyte and apoptosis. Cardiac repair consists of three phases: inflammatory phase, proliferative phase and maturation phase. Inflammation mediated by cytokines, chemokines, and other inflammatory mediators causes wound clearing and apoptosis of dead cells followed by formation of collagen and then cross linkage of collagen fibers leads to formation of scar. ROS reactive oxygen species; ECM extracellular matrix; TGF (transforming growth factor)-beta; IL-(interleukin)-4; TLR toll-like receptor; NF-kB nuclear factor kappa-light-chain-enhancer of activated B cells; MMP matrix metalloproteinase
The proliferative phase starts as soon as the dead cardiomyocytes and matrix are removed. Suppression in expression of inflammatory mediators occurs during the proliferative phase of healing. Timely repression of inflammatory mediator synthesis is important for the transition to the proliferative phase. This mechanism is poorly understood however the probable role of Npr 1 in repressing the expression of cardiac pro-inflammatory mediators, hypertrophic markers, and NF-κB/AP-1 mediated mechanisms, in an Npr1 gene-dose-dependent manner has been discussed [15]. In the proliferation phase, fibroblasts, lymphocytes, mast cells, and endothelial progenitor cells infiltrate the wound. Upregulation of angiogenesis factors, such as CXCL12 and CXL1, in this phase allows endothelial progenitor cells to promote vascularization of the tissue. Activated myofibroblasts form the ECM providing support to the newly formed microvasculature. This is followed by apoptosis of fibroblasts and vascular cells, and loss of most cellular components. The maturation phase consists of formation of a scar containing cross-linked collagen bundles, myofibroblast stem cells, and newly formed vessels (Fig. 2). Cellular and molecular events associated with infarct healing influence the left ventricular remodeling and the prognosis in patients [14, 16]. Understanding cellular and molecular processes after MI may provide therapeutic options to control wound healing and scar formation, and mouse models may play an important role in such studies.
Post-injury cardiac remodeling, fibrosis, and regulation
The changes in size, shape, and function of the left ventricle in response to neuro-hormonal activation, changes in hemodynamic loading conditions and induction of local mediators that alters the structural characteristics of the myocardium are collectively called “ventricular remodeling.” Although mostly seen after MI, ventricular remodeling develops in response to a variety of forms of myocardial stress and injury [17]. Activation of cellular and molecular pathways involving the cardiomyocytes, ECM, and fibroblasts leads to ventricular remodeling.
Role of transforming growth factor-β
Transforming growth factor (TGF)-β is a key mediator in the inflammatory and fibrotic phase of post-infarcted myocardium and, critically, modulates the cellular steps in remodeling (Fig. 3). Markedly induced and activated TGF-β plays a crucial role in modulating fibroblast phenotype and gene expression, which in turn promotes ECM deposition in the infarcted myocardium by up-regulating collagen and fibronectin synthesis and by decreasing matrix degradation through induction of protease inhibitors. TGF-β suppresses inflammatory mediator synthesis from macrophages by repression of inflammatory gene synthesis regulated by TGF-β signaling, while simultaneously enhancing myofibroblast differentiation and matrix deposition through Smad3-dependent pathways [16, 18].
Role of TGF-beta in cardiac fibrosis: TGF-β is activated by MMP-2, MMP-9, TSP-1, ROS and activated TGF-β plays an important role in infarct healing, cardiac fibrosis, ECM formation, angiogenesis, cardiac remodeling and cardiac hypertrophy. MMP matrix metalloproteinase; TSP (thrombospondin)-1; ROS reactive oxygen species; ECM extracellular matrix
TGF-β-induced connective tissue growth factor (CTGF) may promote cardiomyocyte hypertrophy in the remodeling heart [19] and may enhance fibrosis through synergistic interactions with TGF-β. TGF-β 1 also networks with the renin-angiotensin system (RAS) to promote cardiomyocyte hypertrophy, fibroblast proliferation, and expression of ECM proteins in cardiac remodeling [20]. Because of the wide range of actions mediated by TGF-β, TGF-β blockade using a novel orally active TGF-β type I receptor inhibitor (activin receptor-like kinase-5) is a promising therapeutic target for MI and cardiomyopathic conditions associated with fibrosis and hypertrophy [21]. By stimulating growth of cardiomyocyte and inducing interstitial fibrosis, TGF-β plays an important role in the pathogenesis of hypertrophic and dilative ventricular remodeling [14]. Post-infarction ventricular remodeling results in dilation, hypertrophy, and enhanced sphericity of the ventricle and is associated with adverse prognosis [22].
Role of chemokines and cytokines
Following MI, reparative fibrosis and cardiac remodeling is mediated by several members of the chemokine family. The fibrotic process may be regulated by cytokines exerting direct effect on resident fibroblasts, recruitment and activation of mononuclear cell subsets and fibroblast progenitors (fibrocytes), and modulating angiogenesis [23]. Post-ischemic injury, monocytes/macrophages, mast cells, lymphocytes, vascular cells, and cardiomyocytes contribute to the fibrotic response by secreting important fibrogenic mediators. These mediators include inflammatory cytokines (TNF-α, IL-1β, IL-6) and chemokines (CC, CXC, XC, CX3C) regulating the recruitment, chemotaxis and activation of monocytes, lymphocytes and neutrophils. Endothelin-1, matricellular proteins, matrix metalloproteinases (MMP)-12, mast cell-derived proteases, reactive oxygen species, the rennin-angiotensin-aldosterone system, and growth factors such as platelet-derived growth factors (PDGF-AA, -BB, -AB, -CC, -DD) and TGF-β also play an important role in fibrotic response [11]. MCP-1 is a most studied chemokine and plays a role in ischemic cardiomyopathy, ischemic fibrotic cardiomyopathy, and cardiac fibrosis in pressure and volume overloaded heart [23]. CXCL10/IP-10 prevents premature angiogenesis and fibrous tissue deposition until the debridement of infarct and formation of granulation tissue [24]. Interleukin (IL)-1 mediates adverse dilative remodeling in the post-infarction inflammatory reaction. IL-1 regulates post-infarction repair and remodeling through cell-specific actions on leukocytes and fibroblasts. Saxena et al. [25] proposed that IL-1 induces pro-inflammatory leukocyte infiltration and regulates the fibroblast phenotype in the infarcted myocardium. Premature activation of a matrix-synthetic contractile phenotype until the wound is cleared can be prevented by early stimulation of fibroblast IL-1R1 signaling during the inflammatory phase. These studies suggest the potential role of cardiac fibroblast involvement in post-injury myocardial repair.
Role of fibroblast in post-injury myocardial repair
Fibroblasts are mainly involved in the process of fibrosis and regulate the turnover of collagen in tissue, and sometimes changes into myofibroblast by activation and phenotypic transition. Myofibroblasts are the main cells in the fibrotic process. In normally healing wounds, myofibroblasts are involved in tissue repair but activated myofibroblasts act as cellular effectors of the fibrotic process. Contractile proteins, like α-smooth muscle actin expression, characterize the phenotype of the myofibroblast. Endocrine hormones in circulation and autocrine and paracrine factors formed within the myocardium regulate the process of conversion of fibroblasts to myofibroblasts and myofibroblast-mediated collagen turnover. TGF-β1, angiotensin II and endothelin-1 are the main regulators involved in these processes [26].
Proliferation and activation of fibroblast in the myocardium results in origin of the activated myofibroblasts during fibrosis; however, the proliferating fibroblast-like cells in the process of reactive interstitial fibrosis and hypertrophy may be from the neighborhood of blood vessels [27], fibrocytes [28], endothelial cells, pericytes [29], circulating monocytes [30], and bone marrow progenitor cells [31]. TGF-β, platelet-derived growth factor, and fibroblast growth factors mainly stimulate the acquisition of a fibroblast phenotype [32]. Epithelial–mesenchymal transition (EMT) and endothelial–mesenchymal transition (EndMT) are mainly responsible for the production of cardiac fibroblasts from epithelial and endothelial cells during embryonic development [33] (Fig. 4). The role of EndMT in cardiac fibrosis has been suggested by fate mapping studies demonstrating that while in the normal adult heart, there is no significant endothelial contribution to the fibroblast population [34], endothelial origin may contribute up to 30 % of fibroblasts in the damaged myocardium [35]. EndMT causes emergence of fibroblasts from endothelial cells and fibroblast causes excessive deposition of ECM resulting in cardiac fibrosis characterized by decreased extent of microvasculature and disruption of normal myocardial structures. TGF-β1 induces endothelial cells to undergo EndMT, whereas bone morphogenic protein (BMP)-7 preserves the endothelial phenotype by inhibiting EndMT [35]. Further, the monocytic origin of the fibroblast is mediated by sequential Th1 and Th2 induction promoting polarization of the analogous M1 (classically activated) and M2 (alternatively activated) macrophage. The pre-dominant Th2 and M2 responses involving MCP-1 stimuli increase IL-13 playing a role in fibrosis [36].
Origin of myofibroblast and cardiac fibrosis: myofibroblast can originate from circulating fibroblast, fibrocytes, pericytes, endothelial cells, progenitor cells, smooth muscles cells. Origin of myofibroblast results due to the stress, EMT epithelial–mesenchymal transition and EndMT endothelial–mesenchymal transition. TGF-β aids in transformation of promyofibroblast to myofibroblast. Myofibroblast helps in collagen synthesis, ECM formation, and fibrosis and scar formation
Effect of cardiac fibrosis on cardiac function
Cardiac injuries ultimately result in the development of cardiac fibrosis due to the negligible regenerative capacity of the heart. Cardiac fibrosis is associated with increased deposition of matrix proteins in the myocardium. The expansion of the cardiac interstitial space in the absence of significant cardiomyocyte loss is known as “reactive interstitial fibrosis” and the formation of a scar in response to myocardial infarction is called “reparative fibrosis” [37, 38]. Increased deposition of endomysial and perimysial collagen leads to interstitial fibrosis in the fibrotic heart (Fig. 5). Increased collagen deposition in the adventitia of intramural coronary arterioles is caused by perivascular fibrosis. The initial reactive perivascular fibrosis progressing without loss of cardiomyocytes is found in an animal model of left ventricular pressure overload [26].
Pathogenesis of cardiac fibrosis: Ischemic injury to the heart is caused by coronary artery disease due to plaque formation and reduced blood supply. Ischemic injury is followed by a cascade of mechanism consisting of three overlapping phases, to heal the infarcted heart and results in cardiac fibrosis. a Plaque formation, b, c healed scar in a post MI heart with normal myocyte in upper portion of the image, and d collagen deposition
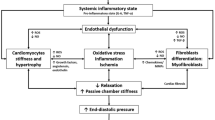
The adaptive response to preserve cardiac output while normalizing wall stress consist of reactive interstitial fibrosis and initial reactive perivascular fibrosis, along with cardiomyocyte hypertrophy. Eventually, however, cardiomyocytes undergoing necrosis and apoptosis cause reparative fibrosis [39]. Interstitial fibrosis of the heart plays an important role in the pathogenesis of ischemic cardiomyopathy, contributing to systolic and diastolic dysfunction. Cardiac fibrosis may lead to heart failure due to loss of contractile function and rhythm disturbances. Atrial fibrillation (AF) is the most common abnormal rhythm for hospital admissions and atrial fibrosis plays an important role in the development of AF [40]. A novel transgenic LKB1 (cardiac-specific liver kinase B1) knockout mouse model is useful in understanding the mechanism of atrial electroanatomical remodeling in the generation of AF [41].
Mouse models in cardiac research
Animal models are used to better understand the pathogenesis of cardiovascular diseases to improve diagnosis, prevention and therapy of ischemic heart disease and to help develop and test new diagnostic, preventive and therapeutic procedures. The advent of sophisticated microsurgical instruments and microscopes favored the shift from rat to mouse model in cardiovascular research. Previously, rat was used due to its larger size. Increased popularity of mouse models to use in cardiac research studies is due to its short gestational period, smaller size, easy to handle and breed large litter size, low maintenance cost, extensive characterization of its genome, and less criticism regarding animal protection, so larger number of animals can be used (Table 2). Also mouse is being used in gene-targeted knockouts and transgenic overexpression experiments [42]. Electrocardiography and high resolution echocardiography allow accurate evaluation of heart function, determination of infarct size and characterization of post-infarct remodeling in mouse [43, 44]. Also the procedures, including examination of electrophysiological aspects of heart rhythm and hemodynamic function, the failing or infarcted rodent myocardium in isolated perfused organ (ex vivo), can be done in Langendorff apparatus [44].
Mouse models for cardiovascular research can be produced surgically, pharmacologically, or electrically. Pharmacological and electrical methods had limitations. The three main surgical procedures being used in research labs for a myocardial infarction mouse model are aortic constriction, pulmonary artery banding, and myocardial infarction with ischemic-reperfusion (I/R) injury. Genetic models have been developed via gene overexpression, deletion, or mutation. These genetic models overcome the need of administering factors or their inhibitors, whose quantification could be problematic and difficult. There is better tolerance to prolonged monoclonal antibody treatments in genetic mouse models. Increased interest to use these mouse models in research studies is due to the availability of a broad variety of genetically altered mice [45]. With various studies, it has now been established that the development of heart and vasculature is being regulated by similar genes and signaling pathways in both mice and human and molecular genetics can play a crucial role in diagnosing and treatment of cardiovascular diseases [46].
While comparing to transgenic model, there can be more biological variation with surgical models, due to the variability in the surgical procedure. So, it is crucial to minimize experimental variability to keep the number of required independent biological replicates and cost of experiment to a minimum. Biological variations are influenced by genetic and/or environmental factors. Limiting the experimental variability will enhance the reproducibility [46, 47]. These mouse models have helped a lot in understanding the mechanism of cardiac fibrosis (Table 3), and in paving a way in the development of newer therapies.
Surgical mouse models of I/R injury
Ischemic-reperfusion mouse models
Occlusion of one of the coronary artery exposes the heart to ischemia and results in acute coronary syndrome. IR of left ventricle (LV) can be created by the temporary occlusion of the LAD coronary artery [57] and produces transient ischemia of the LV. The ischemic condition is reversible if it is for a short duration (<20 min) and is followed by reperfusion, but if the ischemic condition is prolonged, the process of necrosis starts and progresses from sub-endocardium to sub-pericardium. Reperfusion of ischemic tissues is often associated with microvascular dysfunction, which manifests as impaired endothelial-dependent dilatation in arterioles and leukocytes plugging in capillaries [45].
Open-chest ischemia–reperfusion model
With the development of genetically altered mice, there is intense interest in developing murine models to study mechanisms operative in cardiovascular disease. To study the effect of coronary artery occlusion and reperfusion on the myocardium and the methods involved in performing such studies both acutely and chronically, Michael et al. [58] developed the open-chest mouse model using male FVB mice. The LAD coronary artery was occluded permanently or for 30–60 min followed by reperfusion. It was suggested that, as compared with permanent occlusion, there was a significant decrease in infarct size as a percentage of the area at risk in reperfusion. Infiltration of leukocytes into the ischemic region as well as contraction bands was found in the re-perfused myocardium. With the help of this model, the complex pathophysiology, area at risk, infarct size and cardiac function in mouse can be assessed. This model allows the induction of myocardial infarctions of varying degrees and the long-term survival of mouse. It also allows the study of the effect of the absence or overexpression of a gene product in the progression of injury and inflammation in ischemic reperfusion in a transgenic mouse.
Minimal invasive ischemia–reperfusion model or close chest mouse model
Percutaneous transluminal coronary angiography and thrombolysis are widely used in the treatment of acute MI in humans, but they can have the side effects of cardiac dysfunction resulting from IR injury or “myocardial stunning.” Myocardial stunning can also occur in mouse models in cardiac surgery as reperfusion cannot be avoided in operations using extracorporal circulation and cardioplegia techniques. The duration of coronary artery occlusion is crucial for the pathophysiological outcome. Increasing the duration of occlusion increases the risk for infarction of the stunned myocardium. Regional I/R in the anesthetized animal is the standard model to induce ischemia, which can be verified by the sudden regional paleness of the myocardium and changes in ECG. Reperfusion is verified by the appearance of hyperemia in the previously pale region developed during ischemia [59]. In comparison to the permanent occlusion model, the reperfusion MI model leads to a higher infiltration of inflammatory cells, attenuated fibrotic remodeling, and enhanced neo-vascularization in the area of infarction [60].
In order to minimize interfering inflammatory effects induced by surgery, Nossuli et al. [61] developed a “closed chest” ligation model of myocardial I/R injury in the mouse. After thoracotomy, a thin suture is passed under the LAD and the ends of the suture are threaded through a 0.5-mm piece of PE-10 tubing resulting in a loose snare around the LAD. The ends of the suture are exteriorized through each side of the chest wall; the chest is closed and the suture ends are tucked under the skin. The ultimate I/R experiment is performed a few days later by pulling and releasing the free suture ends, when inflammatory cytokines have returned to baseline. It was found that the levels of tumor necrosis factor (TNF)-α and IL-6 mRNA in this sham-operated animal had decreased through day 3 to week 1 after instrumentation. The advantages of developing the in vivo minimally invasive IR model were the implantable device for occlusion of LAD coronary artery and the ability to overcome the problems of high-level background inflammation due to surgical trauma. In addition, this technique enables a more accurate and interpretable response of the involvement of different cytokines, endothelial cells, and other mediators in the I/R injury of the myocardium. Elevated levels of TNF-alpha are found in advanced heart failure not only in the infarcted and peri-infarct zone but also in the myocardium contra-lateral to infarct and play an important role [54].
Different mechanisms involved in IR can be modified by immune responses after the surgical trauma by thoracotomy in open-chest models of coronary ligation. The immune response includes secretion of endogenous ligands of innate immune receptors and cytokine expression and can modulate infarct size. A modified murine closed-chest model using hanging weights used for studying myocardial pre- and post-conditioning and the role of innate immunity in myocardial IR demonstrated that mice can recover from surgical trauma before the onset of myocardial ischemia. The advantage is that volatile anesthetics can be used in a controllable way for instrumentation in a chronic closed-chest model, since their preconditioning effect lasts up to 72 h in contrast to an open-chest model of coronary artery ligation, in which these pre-conditioning protective effects prevent its use. Further, the infarct size assessment and perfusion fixation of the heart for morphometric studies, in addition to histology and immunohistochemistry, can be done in this model [55]. Gao et al. [62] developed a novel and rapid surgical method for inducing MI without requiring ventilation in mice, representing a more efficient and less damaging model of myocardial ischemic injury.
Ischemia without reperfusion mouse model
This model is similar to the open-chest model; the only difference is that the LAD ligation suture is done with an 8-0 silk and is not removed, as reperfusion is not needed. With any chosen time of ischemia, per the protocol, mice can be sacrificed after transmural MI. Just like the open-chest models, these models are interesting for studying the pathophysiology of heart failure [45].
Ischemia–reperfusion with ischemic pre-conditioning mouse model
Various studies have suggested that myocardial infarct size can be limited by ischemic preconditioning [56, 63]. In this model, LAD ligation is done using an 8-0 silk suture with a U-shaped needle passed under the LAD after dissecting the pericardium. Discoloration of the left ventricle will become evident after LAD ligation. For preconditioning, the knot on top of the 1 mm PE-10 tube is tightened carefully and loosens after the given time, and the mouse undergoes three cycles of 5 min. artery occlusion followed by 5 min. reperfusion, respectively. After 10, 30 min. of coronary artery occlusion followed by 2 h of reperfusion is done. A reduction of the infarct size of about 50 % is demonstrable using the ischemic preconditioning protocol. The mechanism responsible for this reduction is still not fully elucidated. Some of the possible signaling pathways may be reactive oxygen species, isoforms of protein kinase C (PKC) and adenosine [45]. PKC plays an important role in ischemic pre-conditioning by providing some cardiac protection to the myocardium undergoing IR; however, ischemic pre-conditioning can also exaggerate cardiac damage [56].
Langendorff model
In this ex vivo mouse model, animals are given heparin (1000 U/kg, i. p.) 20 min prior to the experimental protocol. The heart is removed from anesthetized animals and placed in a weigh bath with some Ca2+-containing buffer. The heart is lifted from the buffer by holding the aorta with two forceps and is placed on a perfusion cannula with buffer running at a slow rate. The buffer flow rate can be increased after clamping the aorta to the perfusion needle. The heart will begin to beat rhythmically and the beat drops will become clear in a successfully performed procedure [45].
Coronary ligation model of mice
In murine models of MI or ischemic injury, permanent or temporary occlusion of the left main descending coronary artery is performed by coronary ligation [59]. The coronary ligation model requires significant expertise in the techniques and proper anesthesia, thus limiting the performance of large-scale experiments. Additional problem with this model is that the artificial effects from the operation and anesthesia cannot be avoided and it does not have atherosclerotic lesions. The standard murine models for atherosclerosis, apolipoprotein E (apoE) KO mice [64], and LDL receptor knockout [65], exhibit atherosclerotic lesions in the aorta but usually do not develop MI. To overcome these problems, Braun et al. [66] reported a HDL receptor scavenger receptor class B type I (SR-BI)-deficient and apoE-deficient double knockout mice murine model exhibiting coronary lesions, multiple MIs, cardiac dysfunction, and strong atherogenicity. However, all of these mice died early (by 8 weeks of age, 50 % mortality: 6 weeks). A “HypoE mice” SRBI-deficient and hypomorphic apoE (ApoeR61 h/h) mouse model was reported by Zhang et al. [67]; it exhibited diet-induced hypercholesterolemia, coronary atherosclerosis, and MI but with premature death (50 % mortality: 33 ± 4.9 days). Overall, the major problem with these mouse models was premature death, prompting to develop better models, as discussed below.
Using SR-BI KO/ApoeR61 h/h mice (mixed C57BL/66129 background) Nakaoka et al. [68] established a novel murine model called as “modified HypoE mouse” of ischemic cardiomyopathy with multiple diffuse coronary lesions. They shortened the sustained Paigen diet for the HypoE mice causing mice to survive longer. Longer survival of the modified HypoE mouse makes this model suitable for cardiac research involving cardiac remodeling on a highly atherogenic background. They can also be used to investigate pharmacological targets in MI and cardiac remodeling, and to evaluate pharmacological effects. Further to evade the limitations of the coronary ligation model, Weinheimer et al. [69] described a surgical mouse model of pressure overload by a combination of TAC and distal left anterior coronary ligation. The advantage of this model is its progressive deterioration of cardiac structural and functional properties leading to gradual and predictable progression of adverse LV remodeling with HF.
Genetic mouse models
Transgenic mice models of cardiac fibrosis
Transgenic mouse encompasses an additional, artificially introduced genetic material in every cell; this addition may result in either gain of function (production of new protein) or in loss of function (alteration or loss of protein) if integrated DNA interrupts another gene. DNA integration via injection into the pro-nucleus of a fertilized ovum can be done anywhere in the genome with multiple copies integrating in a head-to-tail fashion. It is not necessary for there to be a homology between the injected DNA and the host genome [45]. Transgenic mouse models are serving an important role in understanding the mechanisms involved in the pathogenesis of cardiac fibrosis. The clustered regularly interspaced short palindromic repeats (CRISPR) method has further added the advantage of genotyping and genome editing to answer queries and rapid edition of gene of interest to generate desired mouse models [70, 71]. Manipulating the mouse genome for the desired effect is important and researcher must understand the methodology, limitations, and pitfalls [72]. Transgenic mice with the modulation in the promoter-specific expression of various genes, including fibroblast-specific protein-1; c-fos; activating transcription factor 3; cre expression, have been used to study altered heart function and cardiac fibrosis [33, 73–75].
Knockout mice
Knockout mice are produced by gene targeting in mouse embryonic stem (ES) cells. Gene targeting is replacement of a gene sequence of the mouse’s own genome with a modified related one with a mutation; replacement is done by lining up two similar DNA sequences next to each other to exchange parts (homologous recombination). Knockout mice are enormously useful in studying gene function but there are limitations due to developmental defects, which can cause death in the embryonic period. It is also expensive to produce and take care of knockout mice in special facilities. In these knockout mice, both alleles are knocked out and the gene is entirely absent from all cells [45].
Conditional knockout mice
Conditional knockouts are more advanced types of knockouts with gene deletion in a particular organ, specific cell type, or stage of development. The generation of mice that harbor floxed alleles, containing LoxP recognition sequences flanking a critical exon- essential for the expression or function of the gene of interest, is the most successful approach [76]. Homologous recombination of targeting vectors in ES cells brings the LoxP sites into the germ line and is used to generate conditional knockout mice. Due to the splicing of introns containing LoxP sites during RNA processing, the floxed allele mice express the normal gene product. However, the expression of CRE recombinase, brought into the genetic background of the floxed allele mice via interbreeding, can excise the intervening sequences between the LoxP sites. By controlling the expression of CRE recombinase to a specific tissue, the role of a given gene within the myocardium can be examined.
Mouse models of cardiac fibrosis
Understanding the mechanism of cardiomyocyte hypertrophy is growing area of research because cardiac hypertrophy causes most of the sudden cardiac death, and the signaling pathways involved in the cardiac hypertrophy are still emerging. Cardiac fibrosis is a feature of remodeling heart as well as an outcome of excess ECM deposition and a common histological finding in various cardiomyopathies [77–79]. Mouse models (in vivo or in vitro) could play an important role in understanding these signaling pathways. In vitro models help in understanding the morphology, characterization and phenotypic change in the myocardial fibroblast, ECM synthesis, collagen synthesis, cell proliferation, autocrine and paracrine effects of the cell–cell interactions and the sources of fibroblasts, and are used for drug development. In vivo models help in understanding and testing the effects of genetic changes, drugs, signaling pathways on hypertrophy and fibrosis, and are best to study a specific disease [77].
Mouse models for heart failure and hypertrophy
Animal models of heart failure and hypertrophy have played a major role in understanding the pathophysiology and treatment of CHF. Various animal models (coronary ligation model, aortic banding model, Dahl salt-sensitive rat model, spontaneous hypertensive rat model and spontaneous hypertensive with heart failure) have been used in research labs, discussed in the literature and each one has its unique advantages and disadvantages. Which model and species should be used in cardiac research to create CHF depends on various factors such as reproducibility and accessibility of the model, ethical and economical considerations, and the question being dealt with. This scientific question decides the proximity of the model in mimicking the CHF syndrome in human beings [8]. Various models being used in cardiovascular research for heart failure are rat, dog, pig, rabbit, guinea pig, Syrian hamster, cat, turkey, bovine, and sheep. Animal models for hypertrophy include rat, rabbit, dog, pig, cat, hamster, ferret, sheep, baboon, guinea pig, mouse, and transgenic (gene overexpression, gene mutation, and knock out gene) animals. Other than ethical concern, the use of animal models for cardiovascular research (fibrosis, heart failure, and hypertrophy) depends on following factors: (1) feasibility of the animal (2) similarity to human anatomy and physiology (3) association of disease with pain and discomfort to animal and (4) whether the results from animal studies can be readily transferred to heart failure situations in human being. Replacement, reduction, and refinement must also be taken into consideration [8, 80].
Pressure and volume overload mouse models
Cardiac fibrosis is a feature of remodeling, whereas cardiac hypertrophy is a physiological response of the pressure or volume overload resulting in increased cardiac myocyte size. Minimally invasive transverse aortic banding (MTAB) pressure/volume overload mouse model has been used to study the cardiac hypertrophy [78]. Similarly, transverse aortic constriction (TAC) mouse model of pressure overload has been used to study the fundamental signaling processes playing a role in cardiac hypertrophy [81], Pressure overload can also be induced by ascending as well as abdominal aortic constriction. Ascending aortic constriction results in extreme and more rapid overload, whereas abdominal aortic constriction leaves the possibilities of compensation. Similarly, pulmonary hypertension can cause right ventricular pressure overload and subsequent right ventricular failure as well as the left ventricular systolic and diastolic dysfunction [82, 83]. Pulmonary artery banding (PAB) model for pulmonary hypertension has the advantages of having a constant overload with fixed constriction, and not having any systemic or toxic effects. However, the systemic effects in PAB model depend on tightness of the constriction [84]. PAB causing right- and left heart failure and heart dysfunction may lead to cardiac fibrosis [85, 91, 92] (Table 4).
Transgenic mouse models of cardiac fibrosis
Cardiac fibrosis and hypertrophy often co-exist in cardiomyopathies and understanding their contribution is challenging. However, with the advent of transgenic mice, the pathophysiology of heart failure and fibrosis can be studied in detail. These models are also useful in the identification of various genes and mechanisms responsible for the development of congestive heart failure and fibrosis. ECM and ECM-cardiomyocyte adhesion play important role in transduction of contractile force, structural alignment of cardiomyocytes, and intracellular signaling, cell growth, hypertrophy, and survival respectively. In response to physical and chemical stimuli, cardiac fibroblast remodels the ECM through MMPs and tissue inhibitors of metalloproteinases (TIMPs) by changing its phenotype to cardiac myofibroblast. Further, cardiac fibroblast increases the secretion of TIMPs in response to increased damage-associated molecular patterns after a cardiac injury [93]. Upregulation of TIMP1 expression has been associated with increased cardiac fibrosis due to collagen deposition in human as well as in mouse models [94–96]. TIMPs have an impact on cell growth and hypertrophy, and severe cardiac fibrosis and hypertrophy due to pressure overload has been shown using mice lacking TIMP2 and TIMP3. TIMP2−/− and TIPM3−/− mice are helpful in studying the myocardial hypertrophy and fibrosis independently, as well as the effect on cardiac dysfunction [79].
A new MLP knockout mice model for dilated cardiomyopathy (DCMP) and HF with myocardial hypertrophy, interstitial cell proliferation, and fibrosis has been developed with resemblance for sign and symptoms to human [97]. Similarly, knockout of myogenic factor-5, overexpression of the cardiac stimulatory G protein α-subunit [98], and overexpression of tropomodulin [99] in mice can also lead to development of cardiomyopathy.
Collagen mutant mouse model
Proliferation of the extracellular matrix and collagen deposition plays a major role in cardiac fibrosis. As mentioned earlier, MMPs and TIMPs regulate the collagen deposition as well as cardiac fibroblast activity thus regulating the fibrosis, and a balance between MMPs and collagen is essential for the integrity of ECM. Studies involving mouse models (TIMP gene knockout, MMP-9 gene knockout, TGFβ-1 knockout, p53 knockout, ACE2 knockout, db/db Smad3 null mice) have shown that disruption of this balance would lead to cardiac remodeling, fibrosis, and dysfunction [16, 18, 74, 100, 101]. Mouse models with Collagen1a1-GFP transgene to recognize the cells producing Collagen-I matrix in wild-type mice exposed to injury, and those mutated at the dystrophin gene locus (mdx) as a model for Duchenne muscular dystrophy (DMD) have been used to elucidate the cellular and molecular mechanisms responsible for fibrosis. Increased fibrosis and attenuated repair phase due to conditional expression of the constitutively active PDGFRα D842 V mutation in Collagen-I + fibroblasts during injury/repair have been reported making PDGFRα pathway as a potential new target for treatment of progressive DMD [102].
Diabetic mutant mouse model
Diabetes increases the risk of myocardial infarction and hypertrophy. Diabetic-cardiomyopathy-related complications are the major cause of morbidity and mortality in diabetes. Diabetes not only affects the heart causing diabetic-cardiomyopathy independent of coronary artery disease, hypertension, and hyperlipidemia but also exacerbate the cardiac injury after myocardial infarction or IR. Association of cardiac fibrosis, hypertrophy, and cardiomyocyte apoptosis has been discussed in literature. Diabetic mouse models have been used in cardiac research to elucidate the underlying cellular and molecular mechanism of fibrosis and hypertrophy [103]. Hyperglycemia of the left ventricular wall in diabetes stimulates cardiac remodeling and disrupts contractile function, thereby impairing the heart function by declining the systolic and diastolic function of the heart [104]. The mutation in diabetic (db/db) mice results in expression of a non-functional truncated long-form of the leptin receptor leading to hypothalamic resistance to leptin. This results in voracious appetite, marked obesity and overt diabetes at a young age in mice. Obesity and diabetes at young age (4–6 months) results in cardiac interstitial fibrosis in db/db mice and has been documented using both histological and biochemical techniques. Activation and expansion of the resident cardiac fibroblast in diabetes may result in cardiac fibrosis [105]. Further the cardiac fibroblast-dependent matrix accumulation in type II diabetes has been reported using the genetically induced mouse model of type II diabetes (db/db murine model). Increased expression of collagen type I, TIMP2 and TGF-β, and phenotypic changes in diabetic db/db fibroblast isolated from diabetic mouse was associated with collagen accumulation, myocardial stiffness and diastolic dysfunction [106].
Mouse models for hypertrophic cardiomyopathy
Cardiomyopathies are a heterogeneous group of diseases affecting the heart muscle and frequently lead to progressive heart failure-related disability or cardiovascular death. Since genetics have been shown to play an increasingly important role in the onset of cardiomyopathies [107], mouse models have emerged as the most commonly used animal model for cardiomyopathy research, based on the availability of genetic information and technologies to manipulate the mouse genome [108, 109]. Hypertrophic cardiomyopathy (HCM) is a disease of the cardiac muscle that occurs due to mutations in genes encoding for the cardiac sarcomere. To date, approximately 1400 mutations have been identified as responsible for HCM pathology. About 70 % of these mutations are in the sarcomere genes encoding cardiac β-myosin heavy chain (MYH7) and cardiac myosin-binding protein C (MYBPC3). Other sarcomeric genes, such as regulatory myosin light chain (MYL2), cardiac troponin T (TNNT2), cardiac troponin I (TNNI3), and actin (ACTC) account for the other cases of HCM [19].
The phenotypic expression of HCM includes massive hypertrophy primarily involving the ventricular septum. The mechanisms of HCM are complex and include dynamic LV outflow tract obstruction due to bulging of the thickened basal anterior septum beneath the aortic valve, mitral regurgitation, diastolic dysfunction, myocardial ischemia, and cardiac arrhythmias that may lead to sudden death [8]. The microscopic hallmarks are myocyte hypertrophy and disarray together with expansion of the interstitial fibrosis. Myocyte disarray is characterized by architectural disorganization of the myocardium, in which adjacent hypertrophied myocytes are aligned perpendicularly or obliquely to each other around central cores of collagen in either a pinwheel configuration or herringbone pattern (Fig. 6).
Pathogenesis of cardiomyopathy: Pressure overload, inflammation or ischemic injury results in congestive heart failure and dilated cardiomyopathy. Genetic abnormality can result in hypertrophic cardiomyopathy (HCM asymmetrical left ventricular hypertrophy). TGF (transforming growth factor)-β plays an important role in both dilated cardiomyopathy as well as hypertrophic cardiomyopathy and can be a target for therapeutic approach. Timing of targeting the TGF- β by pharmacological agents in an infarcted heart is very important
A number of myosin heavy chain transgenic animal models of HCM have been created in an attempt to understand the pathogenesis of HCM phenotypes typically seen in humans. A transgenic mouse model with a point mutation of the Arg403 –Gln in α-myosin heavy chain shows similarities with familial HCM in humans [110]. A similar phenotype has been seen in transgenic mice lacking the light chain-binding domain of β-myosin heavy chain [111]. These models will help in understanding the pathophysiology of HCM. Overexpression of the H-ras gene targeted to the ventricle with MLC2v promoter causes ventricular hypertrophy with myofiber disarray, which causes left ventricular outflow tract obstruction leading to diastolic dysfunction [112]. Cardiac hypertrophy is associated with the overexpression of calmodulin [113], and interleukin (IL)-6 receptor is associated with activation of gp-130 in transgenic mice [114], overexpression of α-1 adrenergic receptor [115] and knocking out; these may act as a novel therapeutic approach.
Knockout of heart/muscle isoform of adenine nucleotide translocator (Ant 1) can also result in a transgenic mouse model for mitochondrial myopathy exhibiting skeletal muscle and cardiac hypertrophy [116]. There is a confounding effect of background genetics and lifestyle or environmental differences between subjects even from the same family or phenotype of HCM. Blakenburg et al. [117] studied the effect of βMHC Val606Met substitution (VM) on a different phenotype of HCM; they proposed that b-myosin heavy chain variant (Met606Val) causes mild hypertrophic cardiomyopathy in mice, but exacerbates HCM phenotypes in mice having a combination with other HCM activators, causing a wide variation in clinical outcome.
Studies indicate that tropomyosin (Tm) phosphorylation status varies in different mouse models of cardiac disease. To study the beneficial effects of decreasing α-Tm phosphorylation in the context of a chronic, intrinsic stressor, Schulz et al. [118] used the familial hypertrophic cardiomyopathy (FHC) α-TmE180G mouse model and showed that α-Tm E180G/S283A double mutant transgenic mice show no signs of cardiac hypertrophy and display improved cardiac function. Increased phosphorylation of phospholamban Ser-16 and Thr-17 as compared with the α-Tm E180G mice was also present in the hearts of these double mutant transgenic mice. This study demonstrated for the first time that decreasing phosphorylation of tropomyosin can prevent a hypertrophic cardiomyopathic phenotype. Recently, Zhao et al. [119] suggested the DBA/2J (D2) mouse model for in-depth genetic analysis of hypertrophic cardiomyopathy vulnerability and modifier screens. Davis et al. [120] recently described a tension-based (calcium-dependent tension generation within the sarcomeres due to the familial mutations in genes-encoding sarcomeric proteins) mouse model that is advantageous in distinguishing HCM from DCP.
Mouse models for dilated cardiomyopathy
Surgical interruption of coronary arteries either through permanent coronary ligation [121, 122] or re-perfused infarction (IR) [58, 123] results in myocardial injury in mice and leads to development of a DCM phenotype progressively. In place of surgery, cryo-injury is often used as an alternative technique to interrupt coronary blood flow; it can give a more reliable area of injury and can be used as an efficient tool in cardiovascular translational research [124, 125]. Drugs can also induce DCM in a dose-dependent manner, leading to HF. Mouse models of DCM with doxorubicin [126, 127] or isoproterenol [128, 129] have been reported in this context. Toxic models are characterized by oxidant stress and myocyte apoptosis [130] and are useful in assessing cardiac responses to stress.
Myocarditis is an inflammatory disease of the cardiac muscle, and is associated with development of dilated cardiomyopathy and heart failure [131, 132]. Knocking out the gene encoding interferon regulatory factor (IRF)-1 [133], knockout of the TGF-β1gene [134], immunization of CAF1/J mice with monoclonal anti-dog sarcoplasmic reticulum Ca2+ ATPase antibody [135], experimental-autoimmune -myocarditis induced by immunization of autoimmune-susceptible-SWXJ-or parental -SJL/J and SWR/J -mice-with whole cardiac alpha-myosin heavy chain [136] have been documented in various studies.
Mouse models of restrictive cardiomyopathy
Restrictive cardiomyopathy (RCM) is a disease of the cardiac muscle that is characterized by restricted ventricular filling due to high ventricular stiffness. While RCM cases are less common than both HCM and DCM and represent about 2–5 % of all pediatric cardiomyopathies, they have the worst prognosis and are associated with cases of sudden death. Primary RCM includes RCM due to idiopathic causes or genetically inherited or sporadically acquired mutations. Primary RCM is confined to the myocardium. Secondary RCM develops from extrinsic factors and includes infiltrative disorders (amyloidosis and sarcoidosis); storage diseases (hemochromatosis, glycogen storage disease, and Fabry disease); inflammatory diseases such as Loeffler cardiomyopathy, endomyocardial fibrosis, and eosinophilic endomyocardial disease and radiations [116, 137–139]; and scleroderma (systemic sclerosis). Fibrillin-1 overexpression as a responsible factor for excessive fibrosis has been demonstrated using a tight skin (Tsk) mouse as a model of scleroderma [140].
Transgenic mouse models with an R192H mutant of cTnI (R193H in the mouse genome) and R145W cTnI mutant with a worst prognosis for RCM has been described. Similarly, transthyretin mice (V30M ATTR and CMV-γ6 transgenic mice) overexpressing amyloidogenic proteins can also mimic systemic amyloidosis causing amyloid deposition in the heart and resulting in diastolic and systolic dysfunction, CHF, and death [141, 142]. Overexpression or mutation of various genes gives different phenotypes and results in cardiac hypertrophy, hyperplasia, and heart failure [8]. Thus, the transgenic mouse model of hypertrophy can be used for identification of genes involved in the process of heart failure and the molecular mechanism involved in development and disease progression.
Dewald et al. [143] reported a murine model of ischemic cardiomyopathy induced by repetitive IR using C57/BL6 mice. They demonstrated that cardiomyopathy induced by brief repetitive myocardial IR in mouse is associated with reversible regional systolic dysfunction and interstitial fibrosis. Repetitive brief myocardial I/R induce the reversible fibrotic remodeling and ventricular dysfunction without MI and necrosis. This model induces cardiomyopathy with features of myocardial hibernation and can be used in investigating mechanisms responsible for the progression and regression of ischemic segmental myocardial dysfunction.
Recent development in therapeutic approaches
Cardiac hypertrophy and fibrosis as a result of myocardial injury are the leading cause of the cardiac mortality. Mouse models are effective in understanding the pathogenesis of hypertrophy and fibrosis and developing the therapeutic approach to delay, prevent, or reverse the process of hypertrophy and fibrosis. Studies have suggested the prevention of apoptosis, the leading cause of heart failure and cardiac mortality, using C57BL/6J wild-type mice and syngenetic lpr mice and gld mice with coronary ligation [144], reversal of hypertrophy and fibrosis using a mouse model of human HCM transgenic cTnT-Q92 mice [145, 146], and prevention of trans-endothelial migration to decrease fibrosis using mice lacking the FcRγ chain protein (FcR−/−) [147].
The cardioprotective effect of decreased iNOS levels in iron dextran-treated mice from I/R injury to the myocardium due to iron deposition or overload suggests that short-term iron overload can protect the heart from I/R injury [148]. Reduction in infarct size and improvement in left ventricular function can also be caused by proteasome inhibition [149], inhaled nitric oxide [150], and FR167653 [151]. Proteasome inhibition can also cause regression of left ventricular hypertrophy [152]. Similarly, IKKbeta inhibition attenuates myocardial injury and dysfunction following IR injury [153]. The important role of type-3 p90 ribosomal S6 kinase (RSK3) in concentric myocyte hypertrophy and cardiomyopathy under in vivo pathological conditions resulting in myocardial interstitial fibrosis and heart failure was described using mice of a mixed C57BL/6: FVB/N background [154]. Improved cardiac function with reduced interstitial fibrosis shown with TM180: RSK3 −/− mice suggests that specific inhibition of RSK3 should be considered as a potential novel therapeutic strategy.
The role of transgenic (TG) expression of either Ca2+/calmodulin-dependent protein kinase IV (CaMKIV), CaMKIIdeltaB, and CaMKIIdeltaC in the pathogenesis of dilated cardiomyopathy and heart failure was demonstrated using adult C57BL/6 male mice. The potential role of the multifunctional Ca2+ calmodulin (CaM)-dependent protein kinase II (CaMKII) in treatment of myocardial dysfunction and arrhythmias in the setting of structural heart disease has been reported [155, 156]. The role of nucleotide-binding oligomerization domain-2 (NOD2) deficiency in promoting cardiac hypertrophy and fibrosis was demonstrated using C57/BL6 mice and NOD2-knockout mice (C57BL/6 background). NOD2 deficiency was also associated with the enhanced activation of TLR4 and the MAPKs, NF-κB and TGF-β/Smad pathways in NOD2-knockout (KO) mice. These results suggest the attenuating role of NOD2 in hypertrophy and fibrosis [157].
Along with cardioprotective approaches, mouse models are also useful in studying approaches for cardiac regeneration through stem cell therapy. Endogenous regeneration in an infarcted myocardium by the stimulation of cardiomyocyte proliferation can be initiated by adenoviral delivery of cyclin A into the infarct border zone; this leads to improved cardiac function [158]. There are promising results of transplantation of exogenous cells, such as embryonic cells [159] and adult bone marrow-derived cells, in cardiac regeneration. The metabolism of bone marrow-derived stem cells by the cytokine granulocyte-colony stimulating factor in mouse models [160] also has demonstrated the prevention of apoptosis of myocytes, electrophysiological coupling stabilization between the infarcted area, and improved vascularization in the infarcted area [160, 161]. Transplantation of mesenchymal stem cells, umbilical cord blood cells, skeletal myoblasts, endothelial progenitor cell, cardiac stem cells, embryonic stem cells, and induced pluripotent cells and their roles in cardioprotection, cardiac regeneration, and re-endothelialization have been described in mouse and porcine models [162–164].
The role of the coronary adventitial cells and TGFβ1 in the formation of perivascular fibrosis and TGFβR1 signaling inhibition in collagen synthesis using mouse model of DMD suggests that coronary adventitia can be a promising target for anti-fibrotic therapy development [165]. Further, because DMD leads to DCM, a female mdx-XistΔhs mice model has been used to study the required levels of dystrophin in the heart to prevent or delay cardiomyopathy in mice. Mosaic expression of 4–15 % dystrophin in the heart is sufficient to delay onset and ameliorate cardiomyopathy in mice [166].
Pathological hypertrophy is mal-remodeling of the mammalian heart in response to mechanical and pathological stress. Pathological hypertrophy is associated with upregulation of fetal genes, resulting in increased cardiac fibrosis and reduction of cardiac dysfunction. Using miR-155 mutant mice and calcineurin transgenic mice (Tg(Myh6-Ppp3ca)37Eno), Hu et al. [167] suggest that miR-155 is an inducer of pathological cardiomyocyte hypertrophy. Inhibition of endogenous miR-155 might have a clinical perspective to suppress cardiac hypertrophy and heart failure. Similarly, severe DCM progression with downregulation of miR-669a has been shown in Sgcb-null dystrophic mice. Treatment with miR-669a causes a decrease in hypertrophic remodeling, fibrosis, and cardiomyocyte apoptosis and an increase sarcomere organization. It also has been shown to reduce ventricular atrial natriuretic peptide levels and ameliorate gene/miRNA profile of DCM markers. It also leads to significantly reduced adverse remodeling and enhanced systolic fractional shortening of the left ventricle, suggesting that adeno-associated viral (AAV)-mediated miRNA therapy can be beneficial in a transgenic model of severe, chronic MD-associated DCM [168]. Similarly, the inhibition of miR-154 is cardioprotective in a mouse model of pressure overload, and may be a novel target for treating the pathologies associated with cardiac fibrosis, hypertrophy, and dysfunction [169]. These studies suggest the role of epi-genetics in developing novel therapeutic approaches for cardiovascular disease; combining epi-genetics with mouse models may play an important role [170].
Conclusion
Ischemic heart disease is the most common cause of cardiac disease resulting in death in the United States, and there are increasing trends for heart disease. Cardiac disease or CHF results in increased morbidity and decreased quality of life, so there is a need to understand the etiology, pathogenesis, progression, and healing of the ischemic heart, as well as post-infarction remodeling and cardiac hypertrophy. Animal models are of immense importance for better understanding and insight into the genetic basis of cardiac disease in human beings. Various animal models have been used in the past; however, at present, researchers have a preference for the mouse model due its genetic homology with humans. In mouse models, the closed-chest model is more commonly used due to its advantages. The choice of the animal model to be used depends on the investigator; it should be made carefully and ensure that the selected model will serve the purpose of study. Mouse models can help researchers understand the cellular and molecular events occurring during fibrosis and remodeling; as an outcome, various therapeutic approaches can be developed. Targeting Treg cells [171] for cell therapy; using TGF-β blockers; blocking transcriptional factors and involved pathways; blocking the regulators of fibrosis; using gene therapy, stem cell therapy, and bone marrow-derived stem cell transfer; or transplant of exogenous cells provide insight into some of the therapeutic approaches that can be developed based on studies done on mouse models.
References
Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Borden WB, Bravata DM, Dai S, Ford ES, Fox CS et al (2013) Heart disease and stroke statistics–2013 update: a report from the American Heart Association. Circulation 127(1):e6–e245
Heron M (2012) Deaths: leading causes for 2008. Natl Vital Stat Rep 60(6):1–94
Yamani M, Massie BM (1993) Congestive heart failure: insights from epidemiology, implications for treatment. Mayo Clin Proc 68(12):1214–1218
Roger VL (2013) Epidemiology of heart failure. Circ Res 113:646–659
Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M (2016) Heart disease and stroke statistics-2016 update: a report from the American Heart association. Circulation 133(4):e38–e360
Ho KK, Anderson KM, Kannel WB, Grossman W, Levy D (1993) Survival after the onset of congestive heart failure in Framingham Heart Study subjects. Circulation 88(1):107–115
Velagaleti RS, Vasan RS (2007) Heart failure in the twenty-first century: is it a coronary artery disease or hypertension problem? Cardiol Clin 25:487–495
Hasenfuss G (1998) Animal models of human cardiovascular disease, heart failure and hypertrophy. Cardiovasc Res 39(1):60–76
Kwak HB (2013) Aging, exercise, and extracellular matrix in the heart. J Exerc Rehabil 9(3):338–347
Berk BC, Fujiwara K, Lehoux S (2007) ECM remodeling in hypertensive heart disease. J Clin Invest 117(3):568–575
Kong P, Christia P, Frangogiannis NG (2014) The pathogenesis of cardiac fibrosis. Cell Mol Life Sci 71(4):549–574
Frangogiannis NG (2006) Targeting the inflammatory response in healing myocardial infarcts. Curr Med Chem 13(16):1877–1893
Arslan F, de Kleijn DP, Pasterkamp G (2011) Innate immune signaling in cardiac ischemia. Nat Rev Cardiol 8(5):292–300
Bujak M, Frangogiannis NG (2007) The role of TGF-beta signaling in myocardial infarction and cardiac remodeling. Cardiovasc Res 74(2):184–195
Vellaichamy E, Das S, Subramanian U, Maeda N, Pandey KN (2014) Genetically altered mutant mouse models of guanylyl cyclase/natriuretic peptide receptor-A exhibit the cardiac expression of proinflammatory mediators in a gene-dose-dependent manner. Endocrinology 155(3):1045–1056
Bujak M, Ren G, Kweon HJ, Dobaczewski M, Reddy A, Taffet G, Wang XF, Frangogiannis NG (2007) Essential role of Smad3 in infarct healing and in the pathogenesis of cardiac remodeling. Circulation 116(19):2127–2138
Konstam MA, Kramer DG, Patel AR, Maron MS, Udelson JE (2011) Left ventricular remodeling in heart failure: current concepts in clinical significance and assessment. JACC Cardiovasc Imaging 4(1):98–108
Biernacka A, Cavalera M, Wang J, Russo I, Shinde A, Kong P, Gonzalez-Quesada C, Rai V, Dobaczewski M, Lee DW, Wang XF, Frangogiannis NG (2015) Smad3 signaling promotes fibrosis while preserving cardiac and aortic geometry in obese diabetic mice. Circ Heart Fail 8(4):788–798
Panek AN, Posch MG, Alenina N, Ghadge SK, Erdmann B, Popova E, Perrot A, Geier C, Dietz R, Morano I, Bader M, Ozcelik C (2009) Connective tissue growth factor overexpression in cardiomyocytes promotes cardiac hypertrophy and protection against pressure overload. PLoS ONE 4(8):e6743
Schneider MD (2002) Serial killer: angiotensin drives cardiac hypertrophy via TGF-Î21. J Clin Investig 109(6):715–716
Hermida N, López B, González A, Dotor J, Lasarte JJ, Sarobe P, Borrás-Cuesta F, Díez J (2009) A synthetic peptide from transforming growth factor-β1 type III receptor prevents myocardial fibrosis in spontaneously hypertensive rats. Cardiovasc Res 81(3):601–609
Yousef ZR, Redwood SR, Marber MS (2000) Postinfarction left ventricular remodelling: where are the theories and trials leading us? Heart 83(1):76–80
Dobaczewski M, Frangogiannis NG (2009) Chemokines and cardiac fibrosis. Front Biosci (Schol Ed). 1:391–405
Frangogiannis NG (2004) Chemokines in the ischemic myocardium: from inflammation to fibrosis. Inflamm Res 53(11):585–595
Saxena A, Chen W, Su Y, Rai V, Uche OU, Li N, Frangogiannis NG (2013) IL-1 induces proinflammatory leukocyte infiltration and regulates fibroblast phenotype in the infarcted myocardium. J Immunol 191(9):4838–4848
Biernacka A, Frangogiannis NG (2011) Aging and cardiac fibrosis. Aging Dis 2(2):158–173
Ljungqvist A, Unge G (1973) The proliferative activity of the myocardial tissue in various forms of experimental cardiac hypertrophy. Acta Pathol Microbiol Scand A 81(3):233–240
Abe R, Donnelly SC, Peng T, Bucala R, Metz CN (2001) Peripheral blood fibrocytes: differentiation pathway and migration to wound sites. J Immunol 166(12):7556–7562
Humphreys BD, Lin SL, Kobayashi A, Hudson TE, Nowlin BT, Bonventre JV, Valerius MT, McMahon AP, Duffield JS (2010) Fate tracing reveals the pericyte and not epithelial origin of myofibroblasts in kidney fibrosis. Am J Pathol 176(1):85–97
Haudek SB, Xia Y, Huebener P, Lee JM, Carlson S, Crawford JR, Pilling D, Gomer RH, Trial J, Frangogiannis NG, Entman ML (2006) Bone marrow-derived fibroblast precursors mediate ischemic cardiomyopathy in mice. Proc Natl Acad Sci USA 103(48):18284–18289
Kania G, Blyszczuk P, Stein S, Valaperti A, Germano D, Dirnhofer S, Hunziker L, Matter CM, Eriksson U (2009) Heart-infiltrating prominin-1+/CD133+ progenitor cells represent the cellular source of transforming growth factor beta-mediated cardiac fibrosis in experimental autoimmune myocarditis. Circ Res 105(5):462–470
Olivey HE, Mundell NA, Austin AF, Barnett JV (2006) Transforming growth factor-beta stimulates epithelial–mesenchymal transformation in the proepicardium. Dev Dyn 235(1):50–59
Zeisberg EM, Kalluri R (2010) Origins of cardiac fibroblasts. Circ Res 107(11):1304–1312
Lugus JJ, Park C, Ma YD, Choi K (2009) Both primitive and definitive blood cells are derived from Flk-1+ mesoderm. Blood 113(3):563–566
Zeisberg EM, Tarnavski O, Zeisberg M, Dorfman AL, McMullen JR, Gustafsson E, Chandraker A, Yuan X, Pu WT, Roberts AB, Neilson EG, Sayegh MH, Izumo S, Kalluri R (2007) Endothelial-to-mesenchymal transition contributes to cardiac fibrosis. Nat Med 13(8):952–961
Trial J, Cieslik KA, Haudek SB, Duerrschmid C, Entman ML (2013) Th1/M1 conversion to th2/m2 responses in models of inflammation lacking cell death stimulates maturation of monocyte precursors to fibroblasts. Front Immunol 4:287
Weber KT (1989) Cardiac interstitium in health and disease: the fibrillar collagen network. J Am Coll Cardiol 13(7):1637–1652
Anderson KR, Sutton MG, Lie JT (1979) Histopathological types of cardiac fibrosis in myocardial disease. J Pathol 128(2):79–85
Isoyama S, Nitta-Komatsubara Y (2002) Acute and chronic adaptation to hemodynamic overload and ischemia in the aged heart. Heart Fail Rev 7(1):63–69
Verheule S, Sato T, Tt Everett, Engle SK, Otten D, Rubart-von der Lohe M, Nakajima HO, Nakajima H, Field LJ, Olgin JE (2004) Increased vulnerability to atrial fibrillation in transgenic mice with selective atrial fibrosis caused by overexpression of TGF-beta1. Circ Res 94(11):1458–1465
Ozcan C, Battaglia E, Young R, Suzuki G (2015) LKB1 knockout mouse develops spontaneous atrial fibrillation and provides mechanistic insights into human disease process. J Am Heart Assoc 4(3):e001733
Lin MC, Rockman HA, Chien KR (1995) Heart and lung disease in engineered mice. Nat Med 1:749–751
Gehrmann J, Frantz S, Maguire CT, Vargas M, Ducharme A, Wakimoto H, Lee RT, Berul CI (2001) Electrophysiological characterization of murine myocardial ischemia and infarction. Basic Res Cardiol 96(3):237–250
Kuhlmann MT, Kirchhof P, Klocke R, Hasib L, Stypmann J, Fabritz L, Stelljes M, Tian W, Zwiener M, Mueller M, Kienast J, Breithardt G, Nikol S (2006) G-CSF/SCF reduces inducible arrhythmias in the infarcted heart potentially via increased connexin43 expression and arteriogenesis. J Exp Med 203(1):87–97
Conci E, Pachinger O, Metzler B (2006) Mouse models for myocardial ischaemia/reperfusion. J Kardiol-Austrian J Cardiol 13(7–8):239–244
Tarnavski O, McMullen JR, Schinke M, Nie Q, Kong S, Izumo S (2004) Mouse cardiac surgery: comprehensive techniques for the generation of mouse models of human diseases and their application for genomic studies. Physiol Genomics 16(3):349–360
Zaragoza C, Gomez-Guerrero C, Martin-Ventura JL, Blanco-Colio L, Lavin B, Mallavia B, Tarin C, Mas S, Ortiz A, Egido J (2011) Animal models of cardiovascular diseases. J Biomed Biotechnol 2011:497841. doi:10.1155/2011/497841
Wei H, Campbell W, Vander Heide RS (2006) Heat shock-induced cardioprotection activates cytoskeletal-based cell survival pathways. Am J Physiol Heart Circ Physiol 291(2):H638–H647
Perricone AJ, Bivona BJ, Jackson FR, Vander Heide RS (2013) Conditional knockout of myocyte focal adhesion kinase abrogates ischemic preconditioning in adult murine hearts. J Am Heart Assoc 2(5):e000457
Tsukamoto K, Mani DR, Shi J, Zhang S, Haagensen DE, Otsuka F, Guan J, Smith JD, Weng W, Liao R, Kolodgie FD, Virmani R, Krieger M (2013) Identification of apolipoprotein D as a cardioprotective gene using a mouse model of lethal atherosclerotic coronary artery disease. Proc Nat Acad Sci USA 110(42):17023–17028
Vanhoutte D, Schellings MW, Gotte M, Swinnen M, Herias V, Wild MK, Vestweber D, Chorianopoulos E, Cortés V, Rigotti A, Stepp MA, Van de Werf F, Carmeliet P, Pinto YM, Heymans S (2007) Increased expression of syndecan-1 protects against cardiac dilatation and dysfunction after myocardial infarction. Circulation 115(4):475–482
Xia Y, Lee K, Li N, Corbett D, Mendoza L, Frangogiannis NG (2009) Characterization of the inflammatory and fibrotic response in a mouse model of cardiac pressure overload. Histochem Cell Biol 131(4):471–481
Dewald O, Frangogiannis NG, Zoerlein M, Duerr GD, Klemm C, Knuefermann P, Taffet G, Michael LH, Crapo JD, Welz A, Entman ML (2003) Development of murine ischemic cardiomyopathy is associated with a transient inflammatory reaction and depends on reactive oxygen species. Proc Natl Acad Sci USA 100(5):2700–2705
Irwin MW, Mak S, Mann DL, Qu R, Penninger JM, Yan A, Dawood F, Wen WH, Shou Z, Liu P (1999) Tissue expression and immunolocalization of tumor necrosis factor-alpha in postinfarction dysfunctional myocardium. Circulation 99(11):1492–1498
Kim SC, Boehm O, Meyer R, Hoeft A, Knufermann P, Baumgarten G (2012) A murine closed-chest model of myocardial ischemia and reperfusion. J Vis Exp 65:e3896
Mayr M, Metzler B, Chung YL, McGregor E, Mayr U, Troy H, Hu Y, Leitges M, Pachinger O, Griffiths JR, Dunn MJ, Xu Q (2004) Ischemic preconditioning exaggerates cardiac damage in PKC-delta null mice. Am J Physiol Heart Circ Physiol 287(2):H946–H956
Xu Z, Alloush J, Beck E, Weisleder N (2014) A murine model of myocardial ischemia–reperfusion injury through ligation of the left anterior descending artery. J Vis Exp. doi:10.3791/51329
Michael LH, Entman ML, Hartley CJ, Youker KA, Zhu J, Hall SR, Hawkins HK, Berens K, Ballantyne CM (1995) Myocardial ischemia and reperfusion: a murine model. Am J Physiol 269(6 Pt 2):H2147–H2154
Klocke R, Tian W, Kuhlmann MT, Nikol S (2007) Surgical animal models of heart failure related to coronary heart disease. Cardiovasc Res 74(1):29–38
Vandervelde S, van Amerongen MJ, Tio RA, Petersen AH, van Luyn MJ, Harmsen MC (2006) Increased inflammatory response and neovascularization in reperfused versus non-reperfused murine myocardial infarction. Cardiovasc Pathol 15(2):83–90
Nossuli TO, Lakshminarayanan V, Baumgarten G, Taffet GE, Ballantyne CM, Michael LH, Entman ML (2000) A chronic mouse model of myocardial ischemia–reperfusion: essential in cytokine studies. Am J Physiol Heart Circ Physiol 278(4):H1049–H1055
Gao E, Koch WJ (2013) A novel and efficient model of coronary artery ligation in the mouse. Methods Mol Biol 1037:299–311
Miller DL, Van Winkle DM (1999) Ischemic preconditioning limits infarct size following regional ischemia–reperfusion in in situ mouse hearts. Cardiovasc Res 42(3):680–684
Zhang SH, Reddick RL, Burkey B, Maeda N (1994) Diet-induced atherosclerosis in mice heterozygous and homozygous for apolipoprotein E gene disruption. J Clin Invest 94:937–945
Ishibashi S, Goldstein JL, Brown MS, Herz J, Burns DK (1994) Massive xanthomatosis and atherosclerosis in cholesterol-fed low density lipoprotein receptor-negative mice. J Clin Invest 93(5):1885–1893
Braun A, Trigatti BL, Post MJ, Sato K, Simons M, Edelberg JM, Rosenberg RD, Schrenzel M, Krieger M (2002) Loss of SR-BI expression leads to the early onset of occlusive atherosclerotic coronary artery disease, spontaneous myocardial infarctions, severe cardiac dysfunction, and premature death in apolipoprotein E-deficient mice. Circ Res 90(3):270–276
Zhang S, Picard MH, Vasile E, Zhu Y, Raffai RL, Weisgraber KH, Krieger M (2005) Diet-induced occlusive coronary atherosclerosis, myocardial infarction, cardiac dysfunction, and premature death in scavenger receptor class B type I-deficient, hypomorphic apolipoprotein ER61 mice. Circulation 111(25):3457–3464
Nakaoka H, Nakagawa-Toyama Y, Nishida M, Okada T, Kawase R, Yamashita T, Yuasa-Kawase M, Nakatani K, Masuda D, Ohama T, Sonobe T, Shirai M, Komuro I, Yamashita S (2013) Establishment of a novel murine model of ischemic cardiomyopathy with multiple diffuse coronary lesions. PLoS ONE 8(8):e70755
Weinheimer CJ, Lai L, Kelly DP, Kovacs A (2015) Novel mouse model of left ventricular pressure overload and infarction causing predictable ventricular remodelling and progression to heart failure. Clin Exp Pharmacol Physiol 42(1):33–40
Miano JM, Zhu QM, Lowenstein CJ (2016) A CRISPR path to engineering new genetic mouse models for cardiovascular research. Arterioscler Thromb Vasc Biol 36(3):1058–1075
Carroll KJ, Makarewich CA, McAnally J, Anderson DM, Zentilin L, Liu N, Giacca M, Bassel-Duby R, Olson EN (2016) A mouse model for adult cardiac-specific gene deletion with CRISPR/Cas9. Proc Natl Acad Sci USA 113(2):338–343
Davis J, Maillet M, Miano JM, Molkentin JD (2012) Lost in transgenesis: a user’s guide for genetically manipulating the mouse in cardiac research. Circ Res 111(6):761–777
Kong P, Christia P, Saxena A, Su Y, Frangogiannis NG (2013) Lack of specificity of fibroblast-specific protein 1 in cardiac remodeling and fibrosis. Am J Physiol Heart Circ Physiol 305:H1363–H1372
Manabe I, Shindo T, Nagai R (2002) Gene expression in fibroblasts and fibrosis: involvement in cardiac hypertrophy. Circ Res 91:1103–1113
Okamoto Y, Chaves A, Chen J, Kelley R, Jones K et al (2001) Transgenic mice with cardiac-specific expression of activating transcription factor 3, a stress-inducible gene, have conduction abnormalities and contractile dysfunction. Am J Pathol 159:639–650
Rajewsky K, Gu H, Kuhn R, Betz UA, Muller W, Roes J, Schwenk F (1996) Conditional gene targeting. J Clin Invest 98(3):600–603
Rajamannan NM (2006) Models of cardiac fibrosis. Drug Discov Today 3:291–295
Martin TP, Hortigon-Vinagre MP, Findlay JE, Elliott C, Currie S et al (2014) Targeted disruption of the heat shock protein 20-phosphodiesterase 4D (PDE4D) interaction protects against pathological cardiac remodelling in a mouse model of hypertrophy. FEBS Open Bio 4:923–927
Fan D, Takawale A, Basu R, Patel V, Lee J et al (2014) Differential role of TIMP2 and TIMP3 in cardiac hypertrophy, fibrosis, and diastolic dysfunction. Cardiovasc Res 103:268–280
Leong XF, Ng CY, Jaarin K (2015) Animal models in cardiovascular research: hypertension and atherosclerosis. Biomed Res Int 2015:528757. doi:10.1155/2015/528757
deAlmeida AC, van Oort RJ, Wehrens XH (2010) Transverse aortic constriction in mice. J Vis Exp 38:e1729–e1729
Kiefer TL, Bashore TM (2011) Pulmonary hypertension related to left-sided cardiac pathology. Pulm Med 2011:381787
Knight DS, Steeden JA, Moledina S, Jones A, Coghlan JG et al (2015) Left ventricular diastolic dysfunction in pulmonary hypertension predicts functional capacity and clinical worsening: a tissue phase mapping study. J Cardiovasc Magn Reson 17:116
Borgdorff MA, Dickinson MG, Berger RM, Bartelds B (2015) Right ventricular failure due to chronic pressure load: what have we learned in animal models since the NIH working group statement? Heart Fail Rev 20:475–491
Hirata M, Ousaka D, Arai S, Okuyama M, Tarui S et al (2015) Novel model of pulmonary artery banding leading to right heart failure in rats. Biomed Res Int 2015:753210
Furihata T, Kinugawa S, Takada S, Fukushima A, Takahashi M et al (2016) The experimental model of transition from compensated cardiac hypertrophy to failure created by transverse aortic constriction in mice. IJC Heart Vasc 11:24–28
Hutchinson KR, Saripalli C, Chung CS, Granzier H (2015) Increased myocardial stiffness due to cardiac titin isoform switching in a mouse model of volume overload limits eccentric remodeling. J Mol Cell Cardiol 79:104–114
Mohamed BA, Schnelle M, Khadjeh S, Lbik D, Herwig M et al (2016) Molecular and structural transition mechanisms in long-term volume overload. Eur J Heart Fail 18:362–371
Abassi Z, Goltsman I, Karram T, Winaver J, Hoffman A (2011) Aortocaval fistula in rat: a unique model of volume-overload congestive heart failure and cardiac hypertrophy. J Biomed Biotechnol 2011:729497
Houser SR, Margulies KB, Murphy AM, Spinale FG, Francis GS et al (2012) Animal models of heart failure: a scientific statement from the American Heart Association. Circ Res 111:131–150
Patten RD, Hall-Porter MR (2009) Small animal models of heart failure: development of novel therapies, past and present. Circ Heart Fail 2:138–144
Egemnazarov B, Schmidt A, Crnkovic S, Sydykov A, Nagy BM et al (2015) Pressure overload creates right ventricular diastolic dysfunction in a mouse model: assessment by echocardiography. J Am Soc Echocardiogr 28:828–843
Turner NA (2016) Inflammatory and fibrotic responses of cardiac fibroblasts to myocardial damage associated molecular patterns (DAMPs). J Mol Cell Cardiol 94:189–200
Fontes MS, Raaijmakers AJ, van Doorn T, Kok B, Nieuwenhuis S et al (2014) Changes in Cx43 and NaV1.5 expression precede the occurrence of substantial fibrosis in calcineurin-induced murine cardiac hypertrophy. PLoS ONE 9:e87226
Chen TH, Chen MR, Chen TY, Wu TC, Liu SW et al (2016) Cardiac fibrosis in mouse expressing DsRed tetramers involves chronic autophagy and proteasome degradation insufficiency. Oncotarget. doi:10.18632/oncotarget.11026
Heymans S, Schroen B, Vermeersch P, Milting H, Gao F et al (2005) Increased cardiac expression of tissue inhibitor of metalloproteinase-1 and tissue inhibitor of metalloproteinase-2 is related to cardiac fibrosis and dysfunction in the chronic pressure-overloaded human heart. Circulation 112:1136–1144
Arber S, Hunter JJ, Ross J Jr, Hongo M, Sansig G, Borg J, Perriard JC, Chien KR, Caroni P (1997) MLP-deficient mice exhibit a disruption of cardiac cytoarchitectural organization, dilated cardiomyopathy, and heart failure. Cell 88(3):393–403
Iwase M, Uechi M, Vatner DE, Asai K, Shannon RP, Kudej RK, Wagner TE, Wight DC, Patrick TA, Ishikawa Y, Homcy CJ, Vatner SF (1997) Cardiomyopathy induced by cardiac Gs alpha overexpression. Am J Physiol 272(1 Pt 2):H585–H589
Sussman MA, Welch S, Cambon N, Klevitsky R, Hewett TE, Price R, Witt SA, Kimball TR (1998) Myofibril degeneration caused by tropomodulin overexpression leads to dilated cardiomyopathy in juvenile mice. J Clin Invest 101(1):51–61
Zhu F, Li Y, Zhang J, Piao C, Liu T et al (2013) Senescent cardiac fibroblast is critical for cardiac fibrosis after myocardial infarction. PLoS ONE 8:e74535
Moritani T, Iwai M, Kanno H, Nakaoka H, Iwanami J et al (2013) ACE2 deficiency induced perivascular fibrosis and cardiac hypertrophy during postnatal development in mice. J Am Soc Hypertens 7:259–266
Ieronimakis N, Hays A, Prasad A, Janebodin K, Duffield JS et al (2016) PDGFRalpha signalling promotes fibrogenic responses in collagen producing cells in Duchenne muscular dystrophy. J Pathol. doi:10.1002/path.4801
Li Y, Ma J, Zhu H, Singh M, Hill D et al (2011) Targeted inhibition of calpain reduces myocardial hypertrophy and fibrosis in mouse models of type 1 diabetes. Diabetes 60:2985–2994
Rajesh M, Mukhopadhyay P, Batkai S, Patel V, Saito K et al (2010) Cannabidiol attenuates cardiac dysfunction, oxidative stress, fibrosis, and inflammatory and cell death signaling pathways in diabetic cardiomyopathy. J Am Coll Cardiol 56:2115–2125
Cavalera M, Wang J, Frangogiannis NG (2014) Obesity, metabolic dysfunction, and cardiac fibrosis: pathophysiological pathways, molecular mechanisms, and therapeutic opportunities. Transl Res 164:323–335
Hutchinson KR, Lord CK, West TA, Stewart JA Jr (2013) Cardiac fibroblast-dependent extracellular matrix accumulation is associated with diastolic stiffness in type 2 diabetes. PLoS ONE 8:e72080
Jacoby D, McKenna WJ (2012) Genetics of inherited cardiomyopathy. Eur Heart J 33(3):296–304
Eppig JT, Blake JA, Bult CJ, Kadin JA, Richardson JE (2015) The mouse genome database (MGD): facilitating mouse as a model for human biology and disease. Nucleic Acids Res 43:D726–D736
Maron BJ, Maron MS (2013) Hypertrophic cardiomyopathy. Lancet 381(9862):242–255
Geisterfer-Lowrance AA, Christe M, Conner DA, Ingwall JS, Schoen FJ, Seidman CE, Seidman JG (1996) A mouse model of familial hypertrophic cardiomyopathy. Science 272(5262):731–734
Welikson RE, Buck SH, Patel JR, Moss RL, Vikstrom KL, Factor SM, Miyata S, Weinberger HD, Leinwand LA (1999) Cardiac myosin heavy chains lacking the light chain binding domain cause hypertrophic cardiomyopathy in mice. Am J Physiol 276(6 Pt 2):H2148–H2158
Gottshall KR, Hunter JJ, Tanaka N, Dalton N, Becker KD, Ross J Jr, Chien KR (1997) Ras-dependent pathways induce obstructive hypertrophy in echo-selected transgenic mice. Proc Natl Acad Sci USA 94(9):4710–4715
Gruver CL, DeMayo F, Goldstein MA, Means AR (1993) Targeted developmental overexpression of calmodulin induces proliferative and hypertrophic growth of cardiomyocytes in transgenic mice. Endocrinology 133(1):376–388
Hirota H, Yoshida K, Kishimoto T, Taga T (1995) Continuous activation of gp130, a signal-transducing receptor component for interleukin 6-related cytokines, causes myocardial hypertrophy in mice. Proc Natl Acad Sci USA 92(11):4862–4866
Milano CA, Dolber PC, Rockman HA, Bond RA, Venable ME, Allen LF, Lefkowitz RJ (1994) Myocardial expression of a constitutively active alpha 1B-adrenergic receptor in transgenic mice induces cardiac hypertrophy. Proc Natl Acad Sci USA 91(21):10109–10113
Graham BH, Waymire KG, Cottrell B, Trounce IA, MacGregor GR, Wallace DC (1997) A mouse model for mitochondrial myopathy and cardiomyopathy resulting from a deficiency in the heart/muscle isoform of the adenine nucleotide translocator. Nat Genet 16(3):226–234
Blankenburg R, Hackert K, Wurster S, Deenen R, Seidman JG, Lohse MJ, Schmitt JP (2014) β-Myosin heavy chain variant Met606Val causes very mild hypertrophic cardiomyopathy in mice, but exacerbates HCM phenotypes in mice carrying other HCM mutations. Circ Res 115(2):227–237
Schulz EM, Wilder T, Chowdhury SA, Sheikh HN, Wolska BM, Solaro RJ, Wieczorek DF (2013) Decreasing tropomyosin phosphorylation rescues tropomyosin-induced familial hypertrophic cardiomyopathy. J Biol Chem 288(40):28925–28935
Zhao W, Zhao T, Chen Y, Zhao F, Gu Q, Williams RW, Bhattacharya SK, Lu L, Sun Y (2015) A murine hypertrophic cardiomyopathy model: the DBA/2J strain. PLoS ONE 10(8):e0133132
Davis J, Davis LC, Correll RN, Makarewich CA, Schwanekamp JA, Moussavi-Harami F, Wang D, York AJ, Wu H, Houser SR, Seidman CE, Seidman JG, Regnier M, Metzger JM, Wu JC, Molkentin JD (2016) A tension-based model distinguishes hypertrophic versus dilated cardiomyopathy. Cell 165(5):1147–1159
Dai S, Yuan F, Mu J, Li C, Chen N, Guo S, Kingery J, Prabhu SD, Bolli R, Rokosh G (2010) Chronic AMD3100 antagonism of SDF-1alpha-CXCR4 exacerbates cardiac dysfunction and remodeling after myocardial infarction. J Mol Cell Cardiol 49(4):587–597
Wang G, Hamid T, Keith RJ, Zhou G, Partridge CR, Xiang X, Kingery JR, Lewis RK, Li Q, Rokosh DG, Ford R, Spinale FG, Riggs DW, Srivastava S, Bhatnagar A, Bolli R, Prabhu SD (2010) Cardioprotective and antiapoptotic effects of heme oxygenase-1 in the failing heart. Circulation 121(17):1912–1925
Liu X, Simpson JA, Brunt KR, Ward CA, Hall SR, Kinobe RT, Barrette V, Tse MY, Pang SC, Pachori AS, Dzau VJ, Ogunyankin KO, Melo LG (2007) Preemptive heme oxygenase-1 gene delivery reveals reduced mortality and preservation of left ventricular function 1 year after acute myocardial infarction. Am J Physiol Heart Circ Physiol 293(1):H48–H59
van den Bos EJ, Mees BM, de Waard MC, de Crom R, Duncker DJ (2005) A novel model of cryoinjury-induced myocardial infarction in the mouse: a comparison with coronary artery ligation. Am J Physiol Heart Circ Physiol 289(3):H1291–H1300
Polizzotti BD, Ganapathy B, Haubner BJ, Penninger JM, Kuhn B (2016) A cryoinjury model in neonatal mice for cardiac translational and regeneration research. Nat Protoc 11(3):542–552
Weinstein DM, Mihm MJ, Bauer JA (2000) Cardiac peroxynitrite formation and left ventricular dysfunction following doxorubicin treatment in mice. J Pharmacol Exp Ther 294(1):396–401
Robert J (2007) Long-term and short-term models for studying anthracycline cardiotoxicity and protectors. Cardiovasc Toxicol 7(2):135–139
Oudit GY, Crackower MA, Eriksson U, Sarao R, Kozieradzki I, Sasaki T, Irie-Sasaki J, Gidrewicz D, Rybin VO, Wada T, Steinberg SF, Backx PH, Penninger JM (2003) Phosphoinositide 3-kinase gamma-deficient mice are protected from isoproterenol-induced heart failure. Circulation 108(17):2147–2152
Shan J, Kushnir A, Betzenhauser MJ, Reiken S, Li J, Lehnart SE, Lindegger N, Mongillo M, Mohler PJ, Marks AR (2010) Phosphorylation of the ryanodine receptor mediates the cardiac fight or flight response in mice. J Clin Invest 120(12):4388–4398
Wang QD, Bohlooly YM, Sjoquist PO (2004) Murine models for the study of congestive heart failure: implications for understanding molecular mechanisms and for drug discovery. J Pharmacol Toxicol Methods 50(3):163–174
Martino TA, Liu P, Sole MJ (1994) Viral infection and the pathogenesis of dilated cardiomyopathy. Circ Res 74(4):182–188
Matsumori A, Kawai C (1982) An experimental model for congestive heart failure after encephalomyocarditis virus myocarditis in mice. Circulation 65(6):1230–1235
Liu P, Penninger J, Aitken K, Sole M, Mak T (1995) The role of transgenic knockout models in defining the pathogenesis of viral heart disease. Eur Heart J 16(Suppl O):25–27
Kulkarni AB, Huh CG, Becker D, Geiser A, Lyght M, Flanders KC, Roberts AB, Sporn MB, Ward JM, Karlsson S (1993) Transforming growth factor beta 1 null mutation in mice causes excessive inflammatory response and early death. Proc Natl Acad Sci USA 90(2):770–774
Sharaf AR, Narula J, Nicol PD, Southern JF, Khaw BA (1994) Cardiac sarcoplasmic reticulum calcium ATPase, an autoimmune antigen in experimental cardiomyopathy. Circulation 89(3):1217–1228
Jane-wit D, Yu M, Edling AE, Kataoka S, Johnson JM, Stull LB, Moravec CS, Tuohy VK (2002) A novel class II-binding motif selects peptides that mediate organ-specific autoimmune disease in SWXJ, SJL/J, and SWR/J mice. J Immunol 169(11):6507–6514
Hirasawa M, Ito Y, Shibata MA, Otsuki Y (2007) Mechanism of inflammation in murine eosinophilic myocarditis produced by adoptive transfer with ovalbumin challenge. Int Arch Allergy Immunol 142(1):28–39
Jean-Charles PY, Li YJ, Nan CL, Huang XP (2011) Insights into restrictive cardiomyopathy from clinical and animal studies. J Geriatr Cardiol 8(3):168–183
Boerma M, Hauer-Jensen M (2010) Preclinical research into basic mechanisms of radiation-induced heart disease. Cardiol Res Pract. doi:10.4061/2011/858262
Lemaire R, Farina G, Kissin E, Shipley JM, Bona C, Korn JH, Lafyatis R (2004) Mutant fibrillin 1 from tight skin mice increases extracellular matrix incorporation of microfibril-associated glycoprotein 2 and type I collagen. Arthritis Rheum 50(3):915–926
Teng MH, Yin JY, Vidal R, Ghiso J, Kumar A, Rabenou R, Shah A, Jacobson DR, Tagoe C, Gallo G, Buxbaum J (2001) Amyloid and nonfibrillar deposits in mice transgenic for wild-type human transthyretin: a possible model for senile systemic amyloidosis. Lab Invest 81(3):385–396
Ruberg FL, Berk JL (2012) Transthyretin (TTR) cardiac amyloidosis. Circulation 126(10):1286–1300
Dewald O, Frangogiannis NG, Zoerlein MP, Duerr GD, Taffet G, Michael LH, Welz A, Entman ML (2004) A murine model of ischemic cardiomyopathy induced by repetitive ischemia and reperfusion. Thorac Cardiovasc Surg 52(5):305–311
Li Y, Takemura G, Kosai K, Takahashi T, Okada H, Miyata S, Yuge K, Nagano S, Esaki M, Khai NC, Goto K, Mikami A, Maruyama R, Minatoguchi S, Fujiwara T, Fujiwara H (2004) Critical roles for the Fas/Fas ligand system in postinfarction ventricular remodeling and heart failure. Circ Res 95(6):627–636
Lim DS, Lutucuta S, Bachireddy P, Youker K, Evans A, Entman M, Roberts R, Marian AJ (2001) Angiotensin II blockade reverses myocardial fibrosis in a transgenic mouse model of human hypertrophic cardiomyopathy. Circulation 103(6):789–791
Teekakirikul P, Eminaga S, Toka O, Alcalai R, Wang L, Wakimoto H, Nayor M, Konno T, Gorham JM, Wolf CM, Kim JB, Schmitt JP, Molkentin JD, Norris RA, Tager AM, Hoffman SR, Markwald RR, Seidman CE, Seidman JG (2010) Cardiac fibrosis in mice with hypertrophic cardiomyopathy is mediated by non-myocyte proliferation and requires Tgf-β. J Clin Invest 120(10):3520–3529
Haudek SB, Trial J, Xia Y, Gupta D, Pilling D, Entman ML (2008) Fc receptor engagement mediates differentiation of cardiac fibroblast precursor cells. Proc Natl Acad Sci USA 105(29):10179–10184
Metzler B, Jehle J, Theurl I, Ludwiczek S, Obrist P, Pachinger O, Weiss G (2007) Short term protective effects of iron in a murine model of ischemia/reperfusion. Biometals 20(2):205–215
Stansfield WE, Moss NC, Willis MS, Tang R, Selzman CH (2007) Proteasome inhibition attenuates infarct size and preserves cardiac function in a murine model of myocardial ischemia–reperfusion injury. Ann Thorac Surg 84(1):120–125
Hataishi R, Rodrigues AC, Neilan TG, Morgan JG, Buys E, Shiva S, Tambouret R, Jassal DS, Raher MJ, Furutani E, Ichinose F, Gladwin MT, Rosenzweig A, Zapol WM, Picard MH, Bloch KD, Scherrer-Crosbie M (2006) Inhaled nitric oxide decreases infarction size and improves left ventricular function in a murine model of myocardial ischemia–reperfusion injury. Am J Physiol Heart Circ Physiol 291(1):H379–H384
Yada M, Shimamoto A, Hampton CR, Chong AJ, Takayama H, Rothnie CL, Spring DJ, Shimpo H, Yada I, Pohlman TH, Verrier ED (2004) FR167653 diminishes infarct size in a murine model of myocardial ischemia–reperfusion injury. J Thorac Cardiovasc Surg 128(4):588–594
Stansfield WE, Tang RH, Moss NC, Baldwin AS, Willis MS, Selzman CH (2008) Proteasome inhibition promotes regression of left ventricular hypertrophy. Am J Physiol Heart Circ Physiol 294(2):H645–H650
Moss NC, Stansfield WE, Willis MS, Tang RH, Selzman CH (2007) IKKbeta inhibition attenuates myocardial injury and dysfunction following acute ischemia–reperfusion injury. Am J Physiol Heart Circ Physiol 293(4):H2248–H2253
Passariello CL, Gayanilo M, Kritzer MD, Thakur H, Cozacov Z, Rusconi F, Wieczorek D, Sanders M, Li J, Kapiloff MS (2013) p90 ribosomal S6 kinase 3 contributes to cardiac insufficiency in alpha-tropomyosin Glu180Gly transgenic mice. Am J Physiol Heart Circ Physiol 305(7):H1010–H1019
Anderson ME (2005) Calmodulin kinase signaling in heart: an intriguing candidate target for therapy of myocardial dysfunction and arrhythmias. Pharmacol Ther 106(1):39–55
Zhang T, Maier LS, Dalton ND, Miyamoto S, Ross J Jr, Bers DM, Brown JH (2003) The deltaC isoform of CaMKII is activated in cardiac hypertrophy and induces dilated cardiomyopathy and heart failure. Circ Res 92(8):912–919
Zong J, Salim M, Zhou H, Bian ZY, Dai J, Yuan Y, Deng W, Zhang JY, Zhang R, Wu QQ, Tang QZ (2013) NOD2 deletion promotes cardiac hypertrophy and fibrosis induced by pressure overload. Lab Invest 93(10):1128–1136
Woo YJ, Panlilio CM, Cheng RK, Liao GP, Atluri P, Hsu VM, Cohen JE, Chaudhry HW (2006) Therapeutic delivery of cyclin A2 induces myocardial regeneration and enhances cardiac function in ischemic heart failure. Circulation 114(1 Suppl):I206–I213
Kolossov E, Bostani T, Roell W, Breitbach M, Pillekamp F, Nygren JM, Sasse P, Rubenchik O, Fries JW, Wenzel D, Geisen C, Xia Y, Lu Z, Duan Y, Kettenhofen R, Jovinge S, Bloch W, Bohlen H, Welz A, Hescheler J, Jacobsen SE, Fleischmann BK (2006) Engraftment of engineered ES cell-derived cardiomyocytes but not BM cells restores contractile function to the infarcted myocardium. J Exp Med 203(10):2315–2327
Orlic D, Kajstura J, Chimenti S, Limana F, Jakoniuk I, Quaini F, Nadal-Ginard B, Bodine DM, Leri A, Anversa P (2001) Mobilized bone marrow cells repair the infarcted heart, improving function and survival. Proc Natl Acad Sci USA 98(18):10344–10349
Harada M, Qin Y, Takano H, Minamino T, Zou Y, Toko H, Ohtsuka M, Matsuura K, Sano M, Nishi J, Iwanaga K, Akazawa H, Kunieda T, Zhu W, Hasegawa H, Kunisada K, Nagai T, Nakaya H, Yamauchi-Takihara K, Komuro I (2005) G-CSF prevents cardiac remodeling after myocardial infarction by activating the Jak-Stat pathway in cardiomyocytes. Nat Med 11(3):305–311
Pankajakshan D, Kansal V, Agrawal DK (2013) In vitro differentiation of bone marrow derived porcine mesenchymal stem cells to endothelial cells. J Tissue Eng Regen Med. 7(11):911–920
Ou L, Li W, Liu Y, Zhang Y, Jie S, Kong D, Steinhoff G, Ma N (2010) Animal models of cardiac disease and stem cell therapy. Open Cardiovasc Med J 4:231–239
Aminzadeh MA, Tseliou E, Sun B, Cheng K, Malliaras K, Makkar RR, Marbán E (2015) Therapeutic efficacy of cardiosphere-derived cells in a transgenic mouse model of non-ischaemic dilated cardiomyopathy. Eur Heart J 36(12):751–762
Ieronimakis N, Hays AL, Janebodin K, Mahoney WM Jr, Duffield JS, Majesky MW, Reyes M (2013) Coronary adventitial cells are linked to perivascular cardiac fibrosis via TGFbeta1 signaling in the mdx mouse model of Duchenne muscular dystrophy. J Mol Cell Cardiol 63:122–134
van Putten M, van der Pijl EM, Hulsker M, Verhaart IE, Nadarajah VD, van der Weerd L, Aartsma-Rus A (2014) Low dystrophin levels in heart can delay heart failure in mdx mice. J Mol Cell Cardiol 69:17–23
Seok HY, Chen J, Kataoka M, Huang ZP, Ding J, Yan J, Hu X, Wang DZ (2014) Loss of MicroRNA-155 protects the heart from pathological cardiac hypertrophy. Circ Res 114(10):1585–1595
Quattrocelli M, Crippa S, Montecchiani C, Camps J, Cornaglia AI, Boldrin L, Morgan J, Calligaro A, Casasco A, Orlacchio A, Gijsbers R, D’Hooge J, Toelen J, Janssens S, Sampaolesi M (2013) Long-term miR-669a therapy alleviates chronic dilated cardiomyopathy in dystrophic mice. J Am Heart Assoc 2(4):e000284
Bernardo BC, Nguyen SS, Gao XM, Tham YK, Ooi JY, Patterson NL, Kiriazis H, Su Y, Thomas CJ, Lin RC, Du XJ, McMullen JR (2016) Inhibition of miR-154 protects against cardiac dysfunction and fibrosis in a mouse model of pressure overload. Sci Rep 6:22442
Napoli C, Grimaldi V, De Pascale MR, Sommese L, Infante T, Soricelli A (2016) Novel epigenetic-based therapies useful in cardiovascular medicine. World J Cardiol 8(2):211–219
Saxena A, Dobaczewski M, Rai V, Haque Z, Chen W, Li N, Frangogiannis NG (2014) Regulatory T cells are recruited in the infarcted mouse myocardium and may modulate fibroblast phenotype and function. Am J Physiol Heart Circ Physiol 307(8):H1233–H1242
Acknowledgments
This work was supported by research Grants R01 HL112597, R01 HL116042, and R01 HL120659 to DK Agrawal from the National Heart, Lung and Blood Institute, National Institutes of Health, USA. The content of this review article is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Financial and competing interests’ disclosure
The authors have no other relevant affiliations or financial involvement with any organization or entity with financial interest or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed. No writing assistance was utilized in the production of this manuscript.
Rights and permissions
About this article
Cite this article
Rai, V., Sharma, P., Agrawal, S. et al. Relevance of mouse models of cardiac fibrosis and hypertrophy in cardiac research. Mol Cell Biochem 424, 123–145 (2017). https://doi.org/10.1007/s11010-016-2849-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-016-2849-0