Abstract
Moderate exercise positively impacts innate immune functions, bringing about a better resistance against infections and general immunosurveillance. Exercise of high workloads (i.e., high intensity and/or duration) such as elite marathon, on the other hand, may have detrimental effects over immune function, but neither how long nor how intense should be the exercise sessions to be deleterious is known, this being a matter of intense dispute. Exercise is, at the same time, one of the most powerful inducers of the 70 kDa family of heat shock proteins (HSPAs, formerly known as HSP70s), which are protein chaperones characterized by a marked anti-inflammatory potency, when located intracellularly (iHSPA), but may act as pro-inflammatory cytokines if in the extracellular space (eHSPA). The above observations led us to suppose that short-term exercise could impose long-lasting effects on macrophage function that should be related to the eHSPA-to-iHSPA ratio, viz. H-index. Sedentary adult male Wistar rats were then submitted to 20 min swimming sessions with an overload (as a percentage of body weight attached to the tail base) of either 2, 4, 6, or 8 %. Control animals were maintained at rest in shallow water. Monocyte/macrophage functions (phagocytic capacity, nitric oxide [NO], and hydrogen peroxide [H2O2]) were assessed just after and 12 h after exercise and compared with HSPA status and oxidative stress markers. The results showed that exercise increased phagocytosis and H2O2 immediately after the bouts in a workload-dependent way. This was accompanied by increased H-index but no alteration in the redox status. Enhanced phagocytic capacity persisted for up to 12 h, when a marked rise in NO production was also observed, but H-index resumes its control values, suggesting that immune alertness returned to basal levels. Of note was the detection of the cognate form of eHSPA (encoded by hspa8 gene and formerly known as HSP73) in the rat sera. In total, acute exercise may evoke 12 h long workload-dependent effects associated with HSPA status.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute and chronic exercise alter the number and function of circulating cells of the innate immune system (e.g., neutrophils, monocytes, and natural killer [NK] cells) depending on the duration and intensity [1]. It is generally supposed that innate immune responding capacity correlates with exercise workload (i.e., time-power binomium) in an inverted U-shape way, being either immunostimulatory (if the workload is moderate) or immunosuppressive (if strenuous). Unfortunately, however, although exercise may present indisputable benefits to health, there is no consensus on the mechanisms by which exercise is able to modulate innate immune responses [1]. This assumption is particularly valid in relation to short-term exercise effects. We have shown that acute exercise (swimming, 60 min) stimulates macrophage phagocytic capacity, nitric oxide (NO), and hydrogen peroxide production by circulating monocytes/macrophages [2], which is partially dependent on adrenergic stimulation [3]. Nonetheless, whether (and how long) the immunostimulatory effects of short-term exercise bouts still persist during the time is a matter of debate.
Since exercise is a homeostasis-threatening situation, it activates a myriad of physiological responses at both cellular and organism level aimed to reestablish a new homeostatic equilibrium. As exercise changes intracellular Ca2+ contents and pH, elevates body temperature, and accelerates the production of free radicals, protein conformation might also be compromised if protein chaperoning mechanisms have not evolved. These include those based on the heat shock protein (HSP) response. In fact, exercise is one of the principal stimuli for a robust increase in different HSPs in several tissues, which is also fundamental in facilitating cellular remodeling processes related to the training regime [4]. Specifically, HSPs of the 70 kDa family (HSPAs, formerly known as HSP70s) play a crucial role not only in protecting protein structure and function from denaturation, but also in immune system. Accordingly, intracellular HSPA (iHSPA) potently inhibits the activation of nuclear factor kappa B (NF–κB) [5, 6] thus disarming immune responses. On the other hand, under stressful conditions, exercise included, cells of the innate immune system, as well as from other tissues, are able to export HSPA towards the extracellular space [7]. However, extracellularly located HSPA (eHSPA) works exactly in opposite manner as regards its intracellular function. eHSPA may bind to macrophage Toll-like receptors (TLR-2, -4, and -7) eventually triggering pro-inflammatory responses that elevate immunosurveillance [8]. In severe inflammatory illnesses (e.g., sepsis), high levels of eHSPA in an oxidative plasma background are frankly correlated with poor prognosis of septic patients [9]. This is because eHSPA can bind to TLR2 and TLR4 in a variety of cells, including pancreatic β-cells, lymphocytes, endothelial and skeletal muscle cells aside from monocytes/macrophages, leading to the activation of pro-inflammatory pathways via MyD88 and TIRAP that signal downstream to NF-κB via IRAK4, TRAF6, and IKK, and inducing JNK activation via MEKK4/7 [10, 11].
As HSPAs present a dual behavior in inflammation, depending on their location and amounts, the relative abundance of eHSPA to iHSPA dictates the immunoinflammatory balance [10, 11]. Taking into account the above propositions, we aimed to investigate the effects of short bouts of swimming exercise of increasing workloads on circulating rat monocyte/macrophage innate functions, just after and 12 h after exercise sessions. The results were compared with serum eHSPA levels and monocyte/macrophage iHSPA.
Methods
Animals and ethics
Sedentary male Wistar rats (90 days old; weighing approx 250 g; n = 12–16 per experimental group) obtained from The Federal University of Rio Grande do Sul Institute of Basic Health Sciences Animal Care Facility (CREAL) were maintained under controlled temperature (23 ± 1 °C) and humidity (60 %) in a 12/12 h light/dark cycle (lights on at 0700 a.m.), housed in plastic cages (49 × 34 × 16 cm). Throughout the experiments, the animals had free access to water and were fed with standard pelleted laboratory chow (NUVILAB CR-1, Nuvital Nutrients S.A., Curitiba, Brazil) ad libitum. After 1 week of acclimation (five rats per cage), the animals were randomly assigned to the experimental groups described below. The investigation followed all ethical rules established by Arouca’s Act (Federal Law 11794/2008) and the Guide for Care and Use of Experimental Animals published by the National Institutes of Health (NIH publication no. 85-23, revised in 1996). All the procedures were approved by the Federal University of Rio Grande do Sul Committee of Animal Welfare (CEUA-UFRGS, protocol # 23451).
Study design and exercise protocol
The animals were accustomed to the water environment prior to exercise to avoid any stress response related to the new environment and situation, as described elsewhere [3]. The adaptation period consisted in keeping the animals for 8 min in individual swimming pool chambers (20 × 20 × 56 cm) filled with water at 31 ± 1 °C (50 cm depth), for 3 consecutive days, without any overload (avoiding any type of exercise training). Afterward, overnight fed animals (five per group) were randomly assigned for each round of swimming experiments according to exercise burden imposed by a lead overload (2, 4, 6, or 8 % of body weight) attached to the base of the tail and were, then, submitted to an acute swimming exercise bout of 20 min under the same circumstances as during the adaptation, one rat per chamber. Control animals remained at rest in shallow water (3 cm depth; 31 ± 1 °C, 20 min) during each experiment. Then, the following groups were formed: Control (rest), 2, 4, 6, 8 %. Swimming experiments were always carried out between 8:00 and 11:00 a.m. Room temperature during the experiments was kept at 24 °C. All the procedures were in accordance with those prescribed in the American physiological society’s resource book for the design of animal exercise protocols [12] and an experienced researcher was present at all times to avoid drowning. Swimming time (20 min) was chosen because this is the time limit within which an untrained animal really swims before learning how to perform bobbing, which is a survival strategy used to conserve energy without doing exercise.
Euthanasia
Animal killing, by decapitation, always occurred in a separate laboratory environment where there is exhaustion (to remove fear pheromones in the air) in order to prevent unavoidable stress. Between the death of an animal and another, the guillotine and the rest of the material were completely sanitized with water, detergent, and alcohol. Considering the need to obtain peripheral blood for additional cellular and biochemical analyses, animal killing under anesthesia, while desirable, is incompatible with the experimental goals because all anesthetics commonly used in studies with experimental animals lead to an intense hyperglycemia in rodents [13, 14]. Moreover, these anesthetics interfere in the function of cells involved in the production of HSPA, such as leukocytes [15]. Considering also that plasma concentrations of the major molecular object of the study (HSPA) is greatly affected (complete reduction) by high levels of plasma glucose [16], animal killing was performed without anesthesia. The animals were killed either immediately or 12 h after exercise sessions.
Assessment of exercise effort
Exercise loads were chosen to examine a broad range of intensity effects of swimming exercise over the parameters investigated. In accordance to that, low-intensity (2 %), moderate-intensity (4 %), high-intensity (6 %), and strenuous (8 %) exercise bouts were examined. It was assumed that, at 4 % overload, oxygen consumption under the present conditions was ca. 65–70 % of the maximum \((\dot{\text{{V}}}{\text{O}}_{{2\max}})\), which is estimated to be 85–100 mL min−1 kg−1 [12], so that the anaerobic threshold is reached at 6 % overload in which maximal lactate steady state is around 5.5 mM [17, 18]. Therefore, exercise effort and intensity were evaluated by measuring blood lactate (Accutrend® Lactate, Roche, caudal vein, approx 25 μL) just after the exercise session. The effectiveness of exercise was inferred from the increasing concentrations of blood lactate (Table 1).
Blood processing
After blood sampling for lactate analysis, the animals were either decapitated immediately or placed in their cages for additional 12 h until killing. In both cases, whole blood was collected after killing in heparinized tubes for cellular analyses and used immediately. A one-milliliter blood sample from each animal was also saved for the preparation of serum (without any anticoagulant) that was used in oxidative stress studies and for the opsonization of zymosan particles (see below, please). After obtainment, serum samples were frozen in working aliquots at −86 °C until use.
Monocyte/macrophage preparations
Immediately after animal killing, heparinized whole blood was mixed 1:1 (v/v) with phosphate-buffered saline (PBS, consisting of 136.8 mM NaCl, 2.7 mM KCl, 0.9 mM KH2PO4, and 6.4 mM Na2HPO4, pH 7.4). After blood harvesting, mononuclear cells were separated by layering PBS-diluted blood onto a home-made discontinuous gradient of 6 % (w/v) Ficoll (Sigma, F4375)/10.24 % (w/v) sodium diatrizoate hydrate (Sigma, S4506), ρ = 1.084 g/mL, 311 m Osm/kg [19], using equal amounts of blood and gradient solution, as previously described [3]. The samples were then immediately centrifuged (400×g, at room temperature for 10–20 min in a swinging bucket rotor). Centrifugation times varied depending on exercise load, being shorter for higher intensities and vice versa. Exercise strongly influences rheological properties of whole blood and plasma, including a rise in viscosity and hematocrit which is mainly due to hemoconcentration as a result of fluid transfer from the blood to the interstitial spaces [20, 21]. Since hemoconcentration results in increased osmolality and plasma tonicity, mononuclear cells tend also to lose fluid toward the extracellular space thus affecting cellular density. After centrifugation, the turbid interface containing mononuclear leukocytes was collected, resuspended in 10 volumes of PBS, and centrifuged (400×g for 15 min, at room temperature). Mononuclear cell pellets were then resuspended in either Hanks-balanced salt solution (HBSS, consisting of 137 mM NaCl, 3 mM KCl, 0.3 mM Na2HPO4, 0.5 mM KH2PO4, 0.8 mM Na2SO4, 1.3 mM CaCl2, 0.8 mM MgCl2, 25 mM HEPES, 5.5 mM glucose, pH 7.4) or RPMI 1640 culture medium [Sigma, pH 7.4 containing 11 mM glucose, 2 mM l–glutamine, 25 mM HEPES and 24 mM NaHCO3, and supplemented with 10 % (v/v) heat-inactivated fetal bovine serum (FBS, Cripion, Brazil) and antibiotics (100 U/mL penicillin and 100 μg/mL streptomycin (Gibco))] to be incubated; for phagocytic assays, FBS was replaced with autologous rat serum. These procedures were shown to give 90 % pure monocyte preparations which were immediately incubated under appropriate conditions (see below) to allow cell attachment prior functional evaluations. After preparations, cell viability was higher than 95 %, as inferred from the Trypan blue dye exclusion technique.
Preparation of zymosan particles for phagocytic assays
Fifty milligrams of Saccharomyces cerevisiae ghosts (Zymosan A, from Sigma, Z4250), consisting of 3–6 μm particles (4.1 ± 0.6 × 109 ghosts), were suspended in 100 mL of PBS, boiled for 30 min and washed three times with PBS in working aliquots in microfuge tubes (15,000×g for 10 s at room temperature). The resulting precipitates were resuspended in PBS at a final concentration of 20 mg/mL, as described elsewhere [22]. Zymosan particles were then labeled with neutral red (3-amino-7-dimethylamino-2-methylphenazine hydrochloride, Sigma, N4638), as previously described [23], by mixing 300 μL of a 200 mg/mL neutral red solution (in dimethyl sulfoxide [DMSO], Sigma) with 1 mL of 20 mg/mL zymosan suspension and further incubating the mixture for 10 min at 37 °C under agitation. Subsequently, the preparation was resuspended and washed in 20 volumes of PBS (1000×g for 10 min, at room temperature) and the working aliquots were passed through a G27-needle insulin syringe for homogenization. At this point, zymosan particles were either used immediately or kept at −86 °C in working aliquots to be further used. Neutral red-labeled zymosan particles are orangish-brown at pH 7.4 and reddish at pH values below 6.8 thus being easily detected under optical microscopy after phagocytosis because lysosomal pH is lower than 6.8. Moreover, within lysosomes, labeled zymosan particles strongly fluoresce at 488 nm (emission above 580 nm) being straightforwardly quantitated by epifluorescence microscope using FITC interference filters/cubes or by laser-assisted flow cytometry. Typical micrographs utilized for the assessment of phagocytosis are given in Fig. 1.
Epifluorescence and differential interference contrast (DIC) microscopy analysis of macrophage phagocytosis. a Serum-opsonized neutral red-labeled zymosan particles under DIC microscopy just prior addition to macrophage preparations. b A coverslip-attached macrophage with three phagocytosed zymosan particles (arrows) under fluorescence microscopy (FITC filter; exc.: 488 nm/emiss.: >580 nm); note that a fourth zymosan particle (asterisk-marked arrow) does not fluoresce because it was not yet taken up, although being clearly visualized at the macrophage membrane surface (c) when DIC image is merged with fluorescence picture. c Merged image (50 % DIC/50 % FITC fluorescence) showing also that another non-phagocytosed zymosan particle does not fluoresce at all (arrowhead). d A typical preparation of coverslip-attached macrophages in the presence of serum-opsonized neutral red-labeled zymosan particles under DIC microscopy. e The same preparation as in part (d) but observed with FITC fluorescence filter. f Merged image from (d) and (e), 50 % each depicting that only phagocytosed particles fluoresce inside macrophages. Original magnification: a, b, and c = X960; d, e, and f = X640. Scale bars in b and c = 4 μm
At the moment of phagocytic assays, equal parts (usually, 50 μL) of 20 mg/mL neutral red-labeled zymosan suspensions were mixed with homologous serum (obtained from each rat in study) and incubated for 30 min at 37 °C under moderate agitation. After incubation, the samples were washed 3 times with 500 μL of PBS, at room temperature for 10 s each at 15,000×g. Then, opsonized zymosan particles were resuspended in 650 μL of culture media (above) resulting in a final suspension of ca 1.2 × 108 particles/mL.
Phagocytic assay
Monocyte/macrophage phagocytic capacity was estimated in peripheral blood leukocyte preparations from the rats after monocyte conversion into adhered macrophages. Mononuclear cell suspensions containing 5 × 105 monocytes/mL [as assessed by the crystal violet (2 mg/mL)-acetic acid (10 %, v/v) technique for nuclear visualization] were plated onto 6-well dishes containing sterile 20 × 20 mm glass coverslips and incubated for 30 min at 37 °C in 500 μL of RPMI 1640 culture medium supplemented with 10 % (v/v) autologous serum in a humidified 5 % (v/v) CO2 atmosphere in air. After this period, non-adherent cells were removed by washing the wells 3 times with pre-warmed PBS and treated with 450 μL of culture medium (as above) at 37 °C. Then, 50 μL of a pre-warmed neutral red-labeled opsonized zymosan particle suspension (approx 5 × 107 particles/well) were added and left to incubate with macrophages for an additional 30 min at 37 °C under the same above conditions. Phagocytosis was stopped by placing the dishes on ice for 10 min, after which the cells were washed 3 times with ice-cold PBS to remove non-phagocytosed particles. Phagocytic activity was assessed by counting the total amount of engulfed zymosan particles in at least 50 assigned macrophages in different fields in a Neubauer chamber under light microscopy (960× magnification, with a water immersion objective in an inverted motorized semi-confocal fluorescence microscope Olympus ix81). This gave the number of total phagocytosed zymosan particles (Z T) and the percentage of phagocytosing cell (P T) in relation to the total number of cells scored (N T). Phagocytic index (phI, aka Hishikawa’s index), as previously established [24], was calculated as follows: number of phagocytosed particles (Z T) divided by the number of cells scored (N T) multiplied by the percent phagocytosing cells (PT), so that, phI = (Z T/N T) × 100 % × (P T/N T) = [(Z T × P T)/(N T)2] × 100 %. Data were gathered as the mean phI values ± sem from three separate experiments.
Assessment of hydrogen peroxide production capacity
As the prompt production of hydrogen peroxide takes part of macrophage arsenal against microbes, we investigated the effects of exercise on this activity. Immediately after preparation, mononuclear cells were directly resuspended in 560 μM phenol red (phenolsulfonphthalein sodium salt, Sigma) dissolved in pre-warmed HBSS buffer. Cells (2–5 × 105/well) were then left to attach to 96-well plates at 37 °C, as described above, and the capacity of hydrogen peroxide production was evaluated by the stoichiometric oxidation of phenol red by macrophage-emanated hydrogen peroxide in the presence of 19 U/mL horseradish peroxidase (HRP, type II salt free, Sigma P6782) dissolved in HBSS for 1 h, as described previously [2].
Assessment of nitric oxide production capacity
Since the ability of producing the cytotoxic free radical nitric oxide (NO) is of importance for macrophage antimicrobial activity [8], the effects of exercise on NO production was evaluated. After mononuclear preparation, the cells (2–5 × 106 cells/well) were suspended in RPMI 1640 culture medium, as described above, and left to attach to 24-well plates for 2 h at 37 °C in a humidified 5 % (v/v) CO2 atmosphere in air. The cells were afterward washed 3 times with pre-warmed PBS, resuspended in 200 μL of HBSS and incubated for an additional 1 h period to allow for NO production and conversion into nitrites within the incubation media, as described by Rossato and co-workers [3]. Total nitrites accumulated in the medium were measured by incubating equal amounts of macrophage supernatants and Griess’ reagent [0.1 % (w/v) naphthalene diamine dihydrochloride/1 % (w/v) sulfanilamide in 5 % (v/v) H3PO4, (Sigma)] for 10 min at room temperature. The samples were then spectrophotometrically assessed at 540 nm in a microplate reader.
Glutathione status in peripheral blood erythrocytes
Glutathione disulfide (GSSG)-to-glutathione (GSH) ratio, the “glutathione status,” is an index of intracellular redox status [25]. Since red blood cells (RBC) circulate throughout the body, their intracellular glutathione metabolism (2GSH ⇌ GSSG + 2H+ + 2e−) reveals a “snapshot” of the overall redox equilibria existent in all the tissues. Therefore, we evaluated the effects of exercise bouts on erythrocyte glutathione status. Peripheral RBC were prepared from heparinized blood, either immediately or 12 h after exercise sessions, to be assessed for GSSG and GSH by the spectrophotometric GSSG reductase recycling method, essentially as previously described [26]. Then, [GSSG]-to-[GSH] ratios were calculated. Data were expressed as micromoles of either GSH or GSSG per milliliter of red blood cell (RBC) mash.
Oxidative stress markers: TBARS and lipid hydroperoxide (xylenol orange) measurements in the serum
For the estimative assessment of oxidative stress-induced lipoperoxide formation, thiobarbituric acid-reactive substances (TBARS) assay was performed in serum samples diluted to 1 mg of protein per milliliter with 10 mM Tris-HCl (pH 7.4) containing the antioxidant butylated hydroxytoluene (BHT, 450 μM final concentration) to prevent further oxidation of the samples. After protein precipitation with trichloroacetic acid (TCA, 10 % w/v final concentration), the samples were spectrophotometrically assayed, as previously described [27]. Since the TBARS technique is not entirely specific for the assessment of oxidative stress-induced formation of lipid peroxides and derivatives, we also assessed ferrous ion oxidation in the presence of xylenol orange, which is specific for the evaluation of lipid hydroperoxides, in an adaptation for serum [28] of the original protocol [29], as modified for triphenylphosphine blanks [30]. For this purpose, sera were treated with BHT, as described above, and processed for xylenol orange complexing with ferric ions using a t-butylhydroperoxide standard curve (0–20 μM) as reported elsewhere [3]. There are major concerns presented throughout the literature on the use of the TBARS technique for the assessment of oxidative stress-induced formation of lipid peroxides and derivatives (please, see, for instance, [31, 32]). Undoubtedly, polyunsaturated fatty acids with three or more double bonds, when subjected to an oxidative milieu, may undergo the formation of lipid peroxides (including hydroperoxides) which, in turn, rapidly generate various aldehydic byproducts, including malondialdehyde (MDA); all of them are TBA reactants in acid media. Although MDA may be frequently measured as an indicative of lipoperoxidation (by HPLC, for example), MDA measures per se may underestimate lipoperoxidation, since fatty acids carrying one or two unsaturations do not form MDA, but do produce TBA-reactive monofunctional aldehydes. Hence, measurements of TBARS in acidic medium allow for a good estimative assessment of oxidative stress-induced lipoperoxidation. Since other aldehydes (e.g., glucose), that can also react with TBA under the same conditions, suffer no influence of oxidative stress under physiological situations, these aldehydes tend to react at the same extent in both control and oxidative-stressed tissue samples. Therefore, we are absolutely convinced that the simultaneous observation of TBARS and xylenol orange reactions provides a confident view of oxidative stress status. For the analysis of TBARS, data were expressed in terms of nanomoles of malondialdehyde (MDA) equivalents per milligram of serum protein. Total serum lipoperoxides were assessed in terms of nanomoles of t-butylhydroperoxide (TBHP) per milligram of serum protein.
SDS-PAGE and immunoblot analysis of HSPA
Sera and peripheral blood monocytes from the animals were prepared either immediately or 12 h after exercise bouts, as described above. The cells were left to attach to the bottom of 6-well plates for 2 h in RPMI 1640 culture medium at 37 °C in a humidified 5 % (v/v) CO2 atmosphere in air. Then, for the preparation of whole-cell lysates to be analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), macrophages were washed 3 times with PBS and harvested in ice-cold PBS using a polyethylene cell scraper (Sigma). After pelleting (15,000×g for 10 s at room temperature), cells were disrupted on an ice bath in a model UIS250 V ultrasonic processor (Hielscher Ultrasonics GmbH, Germany) equipped with an LS24d3 sonotrode operating at 24 kHz and 75 % of maximal potency (=9 W final power in the tubes), for 30 s in pulses of 0.5 s each. Lysis buffer consisted of 0.1 % (w/v) SDS containing protease inhibitor cocktail (Sigma, freshly prepared) comprising (final concentrations) leupeptin (4.2 μM), aprotinin (0.31 μM), TLCK (N-Tosyl-l-Lysine Chloromethyl Ketone, hydrochloride; 20 μM) and PMSF (Phenyl Methyl Sulfonyl Fluoride, 100 μM). Cell lysates and serum samples were stored at −86 °C until use, when they were defrosted and processed for SDS-PAGE as described elsewhere [33]. Briefly, after protein determination, equivalent amounts of protein from whole-cell lysates (~6 μg) and sera (~40 μg) were mixed with 2× Laemmli loading buffer [50 mM Tris, 10 % (w/v) SDS, 10 % (v/v) glycerol, 10 % (v/v) 2-mercaptoethanol, and 2 mg/mL bromphenol blue, final concentrations] in a ratio of 1:1, boiled for 5 min, and electrophoresed in a 10 % polyacrylamide minigel for 4 h 15 mA/gel. After that, proteins were transferred onto nitrocellulose membranes (GE HealthCare) according to the electrotransfer (Bio-Rad) manufacturer’s instructions (2 h, 100 V) and transferred bands were visualized with 0.3 % (w/v) Red Ponceau S (Sigma) in 3 % (w/v) trichloroacetic acid solution to be photodocumented (ImageQuant 350, GE HealthCare). For immunoblotting procedures, SNAP i.d. (Merck Millipore) quick immunoblot vacuum system was used [34]. After washings, the membranes were incubated with mouse antihuman HSPA monoclonal antibody (Sigma H5147, clone BRM-22 ascites fluid), which recognizes both the 73 kDa constitutive HSC70 (hspa8 gene) and the 72 kDa inducible HSPA (hspa1a gene) forms, at 1:1000 dilution. The membranes were then probed with horseradish peroxidase (HRP)-labeled secondary antibody (anti-mouse IgG whole molecule, Sigma A9044, 1:10,000) for 10 min and protein detection was performed by the enhanced chemiluminescence method using a mixture containing 0.018 % (w/w, final concentration) hydrogen peroxide (Merck), DMSO-solubilized p-coumaric acid (Sigma, 200 μM final concentration), DMSO-solubilized luminol (3-aminophthalhydrazide, Sigma, 1.2 mM final concentration) in 1.5 M Tris-HCl, pH 8.8. Final DMSO concentration in the working reagent was 0.7 % (v/v). As a gel loading control, peroxidase-labeled mouse anti-human β-actin monoclonal antibody (Sigma A3854) was used at 1:25,000 dilution. Protein bands were photodocumented for 600 s (60 frames, 1 photo each 10 s) and quantified in ImageQuant™ 350 chemiluminescence system (GE HealthCare) and the accompanying online stacking imaging software ImageQuant TL 7.0. The data are given as the means ± sd of immunocontents normalized in terms of β-actin. Serum samples were normalized in terms of albumin, detected upon staining of the membranes with Red Ponceau S solution [35].
Reverse transcription and real-time quantitative-polymerase chain reaction (RT-qPCR) analyses of RNA
Twelve hours after exercise session and mononuclear separation, the cells (105/well) were plated in six well dishes in 2 mL of RPMI 1640 medium and left to attach for 2 h at 37 °C in a humidified 5 % (v/v) CO2 atmosphere in air. After removal of non-attached cells, macrophages were harvested and flash frozen in liquid nitrogen to be stored at −86 °C until use, when total RNA extraction (ca. 1–1.5 μg total yield/well) was performed with Illustra RNAspin Mini RNA Isolation Kit (GE Healthcare, 25-0500-72), following the manufacturer’s instructions. Reverse transcription was carried out using High-Capacity cDNA Reverse Transcription kit (Thermo) from total RNA (100–150 ng) in Mx3005-P (Stratagene) qPCR system (25 °C for 10 min, 37 °C for 120 min and 85 °C for the last 5 min incubation). After reverse transcription, 340 ng of cDNA from each sample were subjected to real-time PCR performed with Brilliant II qPCR Master Mix 2-step SYBR® Green kit (Agilent, #600828), using 1 denaturing cycle (95 °C for 10 min) and 50 cycles of annealing (95 °C for 30 s)/extension (60 °C for 60 s). Primers (designed for the rat mRNA), and their respective final concentrations in the reaction mixture, were: hspa1a (400 nM, forward: 5′-GCT GCG GAT CAT CAA CG-3′; reverse: 5′-AGA TGA GCA CGT TGC GC-3′), hspa8 (400 nM, forward 5′-GAT TTG CTG CTC TTG GAT GTC-3′; reverse: 5′-GCT TGA TGA CGA CAG TCA TGA C), nos2 (500 nM, forward: 5′-CAA CAT CCT GGA GGA AGT GG-3′; reverse: 5′-GGT CCA TGA TGG TCA CAT TCT-3′), and actb, used as reference gene (400 nM, forward: 5′-GTG CTA TGT TGC CCT AGA CTT CG-3′; reverse: 5′-GAT GCC ACAGGA TTC CAT ACC C-3′). MgCl2 was always 1.2 mM final concentration. The purity and concentration of total RNA and cDNA samples were spectrophotometrically checked in a twin CCD array detector system (Nanovue, GE Health Care).
Protein quantitation
The amount of protein in serum and cell lysates was assessed by the Bradford’s technique [36] using type V bovine serum albumin (Sigma) as standard.
H-index of immunoinflammatory status
Extracellular-to-intracellular HSPA ratio index (H-index) measured in monocytes/macrophages in relation to serum values has been recently described as novel and overall index of immunoinflammatory status of an individual [10, 11, 37]. The rationale for this is that the higher eHSPA amounts, the more inflammatory signals are coming into play, because eHSPA is pro-inflammatory in nature. On the other hand, for each particular situation, if cells are able to respond to stressful stimuli by enhancing iHSPA, then they are, at the same time, entering into a state of anti-inflammation. Therefore, if one takes R c = [eHSPA]c/[iHSPA]c as the HSPA ratio in a control situation, whatever the techniques used to assess each eHSPA and iHSPA, then H-index can be calculated as the quotient of any R j = [eHSPA]j/[iHSPA]j by R c, which will be therefore considered as the unity (R c = 1), normalizing all the remaining results in this situation “j.” Hence, H-index = R j/R c may allow for the comparisons between any stressful situation “j” and the situation assumed as the control one. H-index can be applied to estimate immunoinflammatory status in many different situations, as immune responses, diabetes and immunological impacts of exercise. As previously argued [11], assuming H-index for the controls (resting, unstimulated) as the unity, exercise produces a shift in H-indices to up to ca. 5, which is paralleled by an elevation in inflammatory markers and stimulation of cell proliferation. H values higher than 5 denote an exacerbated pro-inflammatory response. Conversely, H-indices between 0 and 1 indicate a predominantly anti-inflammatory status. Thus, changes in H-index emerge as a potentially new biomarker for inflammation, and as a very sensitive indicator of inflammatory status.
Statistics analysis
After Kolmogorov–Smirnov test of normality, one-way linear mode analysis of variance (ANOVA) was performed to track between-group differences, which were identified by Bonferroni post hoc test. Immunoblotting analyses were also evaluated by unequal variance (Welch’s) unpaired bitailed t test. As demanded in each case, correlations between exercise load and the parameters analyzed were tested using the parametric Pearson’s product moment correlation followed by bitailed Student’s t test. Pearson’s r coefficients were also calculated and given appropriately. P values for type I errors were always given alongside data presentation. GraphPad InStat software was used throughout the statistics.
Results
Phagocytosis
The influence of exercise on the phagocytic capacity of monocytes/macrophages was assessed by the Hishikawa’s phagocytic index (phI) by counting cells of the same fields under both DIC and fluorescence microscopy (Fig. 1) in order to confirm internalization of the zymosan particles, as described in the Methods section. Immediately after the 20 min sessions of exercise, phagocytosis was found to be strongly correlated with exercise load (r = 0.8331, p = 0.0001), which was confirmed by ANOVA (Fig. 2a). Although presenting a different pattern of response after 12 h, phagocytosis was also correlated with exercise load (r = 0.7580, p = 0.0027) peaking at the high-intensity exercise load (6 % of overweight), as shown in Fig. 2b. Contrarily to that observed soon after exercise, phagocytic capacity of macrophages from rats of the strenuous exercise group (8 %) was lower in comparison with the one at 6 % load (p = 0.0015, by ANOVA).
Functional analyses of monocytes/macrophages obtained from rats submitted to exercise bouts at the indicated intensities (or resting controls). The animals were killed either immediately (a, c, and e) or 12 h (b, d, f) after exercise sessions. At the indicated times, the capacity to phagocytose zymosan particles during 30 min (a, b), to produce hydrogen peroxide in 1 h incubation (c, d) and to produce nitric oxide, as assessed by nitrite accumulation within incubation medium in 1 h (e, f). For the intergroup comparisons of Hishikawa’s index of phagocytosis, by ANOVA, p = 0.0015 in a and p = 0.0008 in b; for multiple consecutive comparisons with increasing exercise load groups: A = p < 0.05; for the comparison between 8 and 6 % group animals, B = p < 0.05. For multiple consecutive comparisons of hydrogen peroxide production capacity in c, p = 0.0188 by ANOVA; A = p < 0.05 relative to controls. For multiple consecutive comparisons of nitric oxide production capacity in f, p = 0.0001 by ANOVA; A = p < 0.05 relative to controls. Data are presented as means ± sem from three separate experiments
Hydrogen peroxide production
Similar to that encountered for the phagocytic pattern, the capacity of macrophages to enhance hydrogen peroxide production when incubated for 1 h was found to be dramatically correlated with exercise load up to 6 % (r = 0.6395, p = 0.0057) when the cells were taken for analyses just after the exercise. This high H2O2-producing profile was maintained at 8 % overload (Fig. 2c). On the other hand, 12 h after the exercise sessions (Fig. 2d), no alteration in hydrogen peroxide accumulation was found (p = 0.2095).
Nitric oxide production
During the 2 h incubation period examined, NO production by macrophages did not respond to exercise when the cells were obtained just after the sessions (Fig. 2e). Nevertheless, when macrophages were collected 12 h after exercise bouts, a conspicuous correlation (r = 0.7267, p = 0.0021) was noted from rest to 6 % group (p = 0.0001, by ANOVA) with respect to nitrite accumulation in the medium, which continued at 8 % overload (Fig. 2f). Hence, the capacity to produce NO upon the exercise stimulus is not manifested immediately after the exercise “stress,” but later.
Glutathione status and oxidative stress
Soon after exercise, none of the overloads tested, up to 6 %, were able to impose any change in GSH, GSSG, or GSSG-to-GSH ratio in red blood cells (RBC), thus suggesting that exercise of moderate-to-high intensity per se did not induce any redox imbalance, a priori (Table 2). This is confirmed by the absence of alterations in TBARS (p = 0.1234) and lipid hydroperoxide (p = 0.4451) evaluations. Intriguingly, however, at the strenuous exercise level, there was a manifest rise in GSSG-to-GSH ratio (p = 0.0202), suggesting that the animal is facing some sort of redox imbalance due to a reaction that produced GSSG from GSH. The picture 12 h after the exercise sessions was not much distinct: RBC glutathione status remained unaltered up to the 6 % overload, this being paralleled by no changes in TBARS and lipid hydroperoxide results (Table 2). Once again, at 8 % exercise load, a noticeable rise in GSSG-to-GSH ratio (r = 0.5242, p = 0.0384) accompanied by increased TBARS values (p = 0.0068) were detected.
Extracellular HSPA
Extracellular HSPA is produced by circulating monocytes as well as by other tissues during exercise and acts as a danger signal to the remaining tissues [10]. Because of this, we tested whether exercise intensity could interfere in such response. The results showed a marked correlation between exercise load and the delivery of both hspa1a (r = 0.7580, 0.0027) and hspa8 (r = 0.8331, p = 0.0001) just after exercise (Fig. 3). This led to an overall correlation between exercise load and total HSPA (r = 0.8331, p = 0.0001). However, no correlation was observed 12 h after the exercise bouts.
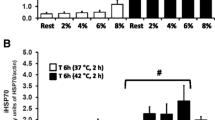
Immunoblotting detection of plasma extracellular HSPA (eHSPA) from animals killed either immediately (filled bars) or 12 h (gray-shaded bars) after exercise sessions. a Total eHSPA (=eHSPA1A + eHSPA8), b eHSPA1A (the ‘inducible’ form of HSPA) and c HSPA8 (the cognate form of HSPA) values normalized in terms of serum albumin are shown. d Representative gels from t = 0 and t = 12 h groups are given. Data are presented as means ± sem from three separate experiments. For consecutive multiple comparisons by ANOVA, significant differences are depicted as their respective p values
Intracellular HSPA in monocytes/macrophages
Since previous works from this laboratory have shown that HSPA protein expression in circulating monocytes/macrophages peaks 6 h after moderate exercise [2], we were interested in knowing whether this situation persists for longer periods. The results showed, however, that exercise was not able to change total HSPA protein levels in monocytes/macrophages 12 h after the exercise sessions, so that the amount of total HSPA remained exactly as in the time zero point (Fig. 4).
H-index of immunoinflammatory status
Extracellular-to-intracellular HSPA ratio index (H-index) was measured in monocytes/macrophages in relation to the serum of the animals for each of the test situations essentially as described in the Methods section. When the samples for H-index assessment were taken just after exercise (t = 0), the results indicate that exercise positively impacts H-index of the animals in a workload-dependent way (Fig. 5a). This assumption is based on the fact that exercise increased H-index to the 1–5 (immunostimulatory) range. Furthermore, when the samples were collected 12 h after the bouts, H-index remained unchanged between test groups (Fig. 5b).
Extracellular-to-intracellular HSPA ratio index (H-index) in monocytes/macrophages in relation to serum obtained from rats killed either immediately (a) or 12 h (b) after exercise sessions. Indices were calculated from the results presented in Figs. 3 and 4 as described in the “Methods” section. For consecutive multiple comparisons by ANOVA, data are significantly different from the controls (p < 0.05) in the groups 4, 6, and 8 % in time zero, but not in t = 12 h. Data are presented as means ± sem from three separate experiments
Long-term effects of exercise on monocyte/macrophage mRNA levels of HSPA’s and iNOS
In order to gain further insight into the molecular mechanisms associated with monocyte/macrophage response to exercise, we examined mRNA levels of hspa1a (encoding the ‘inducible’ form of HSPA), hspa8 (encoding the cognate HSPA), and nos2 (encoding the inducible, NF-κB-dependent iNOS) 12 h after exercise. The results confirmed that exercise did not appreciably influence long-term expression of mRNA encoding the above proteins (Fig. 6). Indeed, only at strenuous level (8 %), there was some augmentation of hspa1a (p = 0.0304 vs remaining groups) but the overall correlation between exercise level and mRNA level is solely mild (r = 0.4743, p = 0.0740) and considered not statistically significant.
Reverse-transcriptase quantitative-polymerase chain reaction (RT-qPCR) analysis of hspa1a, hspa8, and nos2 (iNOS) mRNA obtained from monocytes/macrophages of rats killed 12 h after exercise sessions. For the comparisons relative to controls, *p = 0.0302. Data are presented as means ± sem from 3–6 separate experiments
Discussion
Exercise has been reputed to be immunostimulating when performed at moderate intensities but could be immunosuppressive at strenuous loads [1, 38], particularly in long-duration schedules (e.g., marathon). Particularly for training (i.e., regular physical activity) in humans, the assumed “inverted U-shape” response to exercise has been recently challenged [39]. However, short-duration exercise bouts have also proven to exert long-lasting impacts on immune function. For instance, we have shown that circulating monocytes/macrophages from rats submitted to moderate (5 % overload) swimming exercise bouts of 60 min increased phagocytic activity, hydrogen peroxide liberating capacity, and iNOS-dependent NO production that was maximal 6 h after exercise sessions [2, 3]. Nevertheless, the effects of increasing exercise loads have never been studied in rat monocytes/macrophages in a longer time frame or with increasing workloads.
In the present study, we showed that phagocytosis and hydrogen peroxide production capacity markedly correlate with exercise load, reaching a maximum at the highest intensities tested herein, when the cells were taken just after exercise (Fig. 2). Yet, 12 h after the bouts, phagocytosis exhibited an inverted U-shape pattern (maximum at 6 % load), whereas H2O2 production seemed to be insensitive. Contrarily, macrophage capacity to produce NO when cultured for 2 h was enhanced only if the cells were taken 12 h after exercise, which agrees with the notion that macrophage NO production depends on the de novo synthesis of iNOS [2, 3]. As a whole, these findings indicate that single bouts of exercise may influence innate immune response for at least 12 h.
iNOS expression is NF-κB dependent and the activation of this nuclear transcription factor is redox modulated [40]. Beside of this, monocyte/macrophage activation leads to oxidative burst [41, 42]. Therefore, we tested whether such observed alterations could be associated with some redox imbalance, by assessing erythrocyte glutathione status and oxidative stress markers. Red blood cell (RBC) glutathione (GSH) plays an important role in attenuating possible oxidative damage evoked by circulating reactive oxygen/nitrogen species (ROS/RNS), particularly because erythrocytes circulate throughout the body, thus having an overall glutathione metabolism that operates in equilibrium with whole-body tissue redox status [43, 44]. In addition, RBC hemoglobin also responds to systemic NO by changing intra-erythrocyte redox status [45, 46].
As we have previously debated [47], glutathione disulfide (GSSG) is a primary glutathione S-conjugate physiologically produced within the cells from the oxidation of the thiol moieties of GSH molecules and its accumulation induces a severe state of oxidative stress. This is because intracellular redox potential, i.e., the redox “voltage,” given by the Nernst equation, is described as ε = εo−(RT/nℱ)·ℓn (aGSSG/aGSH), where aGSSG and aGSH are the chemical activities of GSSG and GSH, respectively. Assuming that intracellular concentrations of GSSG and GSH (respectively [GSSG] and [GSH]) are sufficiently diluted, it is reasonable to write ε = εo−(RT/nℱ)·ℓn ([GSSG]/[GSH]). Because of this, [GSSG]/[GSH] ratio is considered the best index of cellular redox status [25] and gives a scenario of how intracellular redox reactions may be affected if one or both of the components of this quotient have changed as in the case of exercise [3]. However, in the present study, none of the workloads tested, up to 6 %, were able to impact RBC glutathione status when the cells were taken just after exercise, thus suggesting that exercise of moderate-to-high intensity per se did not induce any redox imbalance (Table 2). Similar to that previously reported for rat swimming exercise bouts of 60 min at 5 % overload [2], this immutable glutathione status was paralleled by changeless patterns of TBARS and lipid hydroperoxides in the sera of the rats. Remarkably, however, strenuous exercise was able to elevate both GSSG/GSH ratio and TBARS, thus suggesting that the capacity of the animal to blunt a dangerous oxidative stress milieu is limited. In any way, the rise in phagocytosis soon after exercise does not appear to be related to oxidative stress; perhaps to other physiological alterations, such as enhanced exercise-elicited sympathetic activity [3].
Extracellular HSPA (eHSPA) is produced by circulating monocytes and other tissues during exercise and acts as a danger signal to the remaining tissues [10]. Because of this, we tested whether exercise intensity could interfere in such response. Consistent with a rapid signal role during stressful situations, the results showed that exercise was able to increase eHSPA (both eHSPA1A and eHSPA8) only if the blood was collected just after the bouts, as this pattern was not depicted 12 h later at all (Fig. 3). It is also of note that the irrefutable presence of the cognate form of HSPA (eHSPA8) in the sera of the animals. This is striking because the vast array of studies on eHSPA have pointed out to the detection of the inducible (eHSPA1A) form of HSPA. This is particularly true concerning eHSPA1A in the plasma of exercised subjects [48, 49]. Although some reports have addressed the increase in tissue HSPA8 evoked by exercise in animal models [50–52], none has shown evidence of HSPA8 release into the blood after exercise. Hence, to the best of our knowledge, this is the first study reporting that exercise is able to increase eHSPA8 in a workload-dependent fashion. This finding, although apparently obvious (since HSPA8 is constantly expressed and stored inside cells as a housekeeping protein chaperone), has important repercussions in the field of stress research because investigators use to check eHSPA with ELISA kits specific for the HSPA1A gene (not for HSPA8). Hence, choosing eHSPA1A alone as a sole indicator of HSPA status after exercise may obfuscate some possible as yet unidentified HSPA functions.
eHSPA1A has been found to act as a pro-inflammatory signal influencing phagocytosis after its binding to Toll-like receptors (e.g., TLR2, TLR4, and TLR7) [10, 53] which triggers membrane changes, rearranging the cytoskeleton, and eventually phagocytosis. Whether eHSPA8 and/or other forms of HSPA are also capable of triggering the same is an open question that is currently under investigation in our laboratory.
Independent of HSPA nature, however, calculated H-indices for serum to monocyte/macrophage HSPA ratio were found to increase within the 1–5 (immunostimulatory) range with exercise in a workload-dependent fashion (Fig. 5a). Confirming its usefulness as a sensitive tool to provide information on immunoinflammatory status, H-index responded to exercise only if samples were obtained just after the bouts. This implies that, although macrophages are apparently still activated 12 h after exercise, as inferred from phagocytic and NO-producing capacities (Fig. 2), the state of whole-body alertness to stressful challenges and immunoinflammatory surveillance ceased, as judged by the unaltered H-index at this time point, irrespective of workload (Fig. 5b).
Finally, it is remarkable that HSPA1A mRNA expression continued 12 h after exercise, in a workload-dependent way (Fig. 6a). On the other hand, the cognate HSPA8 form of HSPA, which is used to be “constitutive” in cells, presented no change in its pattern of expression in monocytes/macrophages 12 h after exercise (Fig. 6b). Interestingly, however, it was surprising a priori that iNOS mRNA did not vary (Fig. 6c) because a marked correlation with exercise intensity and NO (nitrite) accumulation in medium was found (Fig. 2f). Since NO production by macrophages is entirely dependent on the de novo synthesis of iNOS, we speculate that the synthesis of new iNOS molecules in response to the exercise stress might have sufficed iNOS to account for late NO generation. Nevertheless, iNOS expression is, in turn, wholly dependent on NF-κB activation [2, 40], and this may explain why iNOS mRNA expression was unchanged 12 h after exercise with respect to resting controls. Accordingly, as HSPAs exert their anti-inflammatory effects through different intracellular pathways, including the blockade NF-κB activation [6, 54], the high levels of monocyte/macrophage HSPA (Fig. 6a) may render macrophages anti-inflamed, presenting diminished levels of iNOS mRNA, even in the presence of iNOS protein amounts enough to produce a conspicuous NO liberation in culture (Fig. 2f). Thus, it is concluded that macrophage NO production in response to exercise is self-limiting, which avoids excess oxidative/nitrosative stress that can be deleterious to the animal.
Taken as a whole, the present findings suggest that short-duration exercise bouts are able to produce stimulatory effects over circulating monocyte/macrophage innate responses that last over 12 h after exercise, this being independent but accompanied by oxidative stress at strenuous intensities. These positive effects are workload-dependent and are correlated with H-index of HSPA ratios that can be of value in assessing immunoinflammatory status in response to exercise in medical clinic and sport.
References
Walsh NP, Gleeson M, Shephard RJ, Gleeson M, Woods JA, Bishop NC, Fleshner M, Green C, Pedersen BK, Hoffman-Goetz L, Rogers CJ, Northoff H, Abbasi A, Simon P (2011) Position statement. Part one: immune function and exercise. Exerc Immunol Rev 17:6–63
Silveira EMS, Rodrigues MF, Krause MS, Vianna DR, Almeida BS, Rossato JS, Oliveira LP Jr, Curi R, Homem de Bittencourt PI Jr (2007) Acute exercise stimulates macrophage function: possible role of NF–κB pathways. Cell Biochem Funct 25:63–73
Rossato JS, Krause M, Fernandes AJ, Fernandes JR, Seibt IL, Rech A, Homem de Bittencourt PI Jr (2014) Role of alpha- and beta-adrenoreceptors in rat monocyte/macrophage function at rest and acute exercise. J Physiol Biochem 70:363–374. doi:10.1007/s13105-013-0310-3
Dimauro I, Mercatelli N, Caporossi D (2016) Exercise-induced ROS in heat shock proteins response. Free Radic Biol Med. doi:10.1016/j.freeradbiomed.2016.03.028
Chen HW, Kuo HT, Wang SJ, Lu TS, Yang RC (2005) In vivo heat shock protein assembles with septic liver NF-κB/I-κB complex regulating NF-κB activity. Shock 24:232–238
Gutierrez LL, Maslinkiewicz A, Curi R, Homem de Bittencourt PI Jr (2008) Atherosclerosis: a redox-sensitive lipid imbalance suppressible by cyclopentenone prostaglandins. Biochem Pharmacol 75:2245–2262
Walsh RC, Koukoulas I, Garnham A, Moseley PL, Hargreaves M, Febbraio MA (2001) Exercise increases serum Hsp72 in humans. Cell Stress Chaperones 6:386–393
Campisi J, Fleshner M (2003) Role of extracellular HSP72 in acute stress-induced potentiation of innate immunity in active rats. J Appl Physiol 9:43–52
Gelain DP, de Bittencourt Pasquali MA, Comim CM, Grunwald MS, Ritter C, Tomasi CD, Alves SC, Quevedo J, Dal-Pizzol F, Moreira JCF (2011) Serum heat shock protein 70 levels, oxidant status, and mortality in sepsis. Shock 35:466–470. doi:10.1097/SHK.0b013e31820fe704
Heck TG, Schöler CM, Homem de Bittencourt PI Jr (2011) HSP70 expression: does it a novel fatigue signalling factor from immune system to the brain? Cell Biochem Funct 29:215–226. doi:10.1002/cbf.1739
Krause MS, Heck TG, Bittencourt A, Scomazzon SP, Newsholme P, Curi R, Homem de Bittencourt PI Jr (2015) The chaperone balance hypothesis: the importance of the Extracellular to Intracellular HSP70 Ratio (eHSP70/iHSP70) to inflammation-driven type 2 diabetes, the effect of exercise and the implications for clinical management. Mediat Inflamm 2015:249205. doi:10.1155/2015/249205
Kregel KC, Allen DL, Booth FW, Fleshner M, Henricksen E, Musch TI, O’Leary DS, Parks CM, Poole DC, Ra’anan AW, Sheriff DD, Sturek MS, Toth LA (2006) Resource book for the design of animal exercise protocols. American Physiological Society, Bethesda
Brown ET, Umino Y, Loi T, Solessio E, Barlow R (2005) Anesthesia can cause sustained hyperglycemia in C57/BL6J mice. Vis Neurosci 22:615–618
Saha JK, Xia J, Grondin JM, Engle SK, Jakubowski JA (2005) Acute hyperglycemia induced by ketamine/xylazine anesthesia in rats: mechanisms and implications for preclinical models. Exp Biol Med 230:777–784
Chang Y, Chen TL, Sheu JR, Chen RM (2005) Suppressive effects of ketamine on macrophage functions. Toxicol Appl Pharmacol 204:27–35
Febbraio MA, Mesa JL, Chung J, Steensberg A, Keller C, Nielsen HB, Krustrup P, Ott P, Secher NH, Pedersen BK (2004) Glucose ingestion attenuates the exercise-induced increase in circulating heat shock protein. Cell Stress Chaperones 9:390–396
Gobatto CA, de Mello MA, Sibuya CY, de Azevedo JR, dos Santos LA, Kokubun E (2001) Maximal lactate steady state in rats submitted to swimming exercise. Comp Biochem Physiol A: Mol Integr Physiol 130:21–27
Voltarelli FA, Gobatto CA, de Mello MA (2002) Determination of anaerobic threshold in rats using the lactate minimum test. Braz J Med Biol Res 35:1389–1394. doi:10.1590/S0100-879X2002001100018
Bϕyum A, Lϕvhaug D, Tresland L, Nordlie EM (1991) Separtaion of leucocytes: improved cell purity by fine adjustment of gradient medium density and osmolality. Scand J Immunol 34:697–712
El-Sayed MS (1998) Effects of exercise and training on blood rheology. Sports Med 26:281–292
Brun JF, Khaled S, Raynaud E, Bouix D, Micallef JP, Orsetti A (1998) The triphasic effects of exercise on blood rheology: which relevance to physiology and pathophysiology? Clin Hemorheol Microcirc 19:89–104
Pires de Melo M, Curi TC, Miyasaka CK, Palanch AC, Curi R (1998) Effect of indole acetic acid on oxygen metabolism in cultured rat neutrophil. Gen Pharmacol 31:573–578
Grunwald MS, Pires AS, Zanotto-Filho A, Gasparotto J, Gelain DP, Demartini DR, Schöler CM, Homem de Bittencourt PI Jr, Moreira JCF (2014) The oxidation of HSP70 is associated with functional impairment and lack of stimulatory capacity. Cell Stress Chaperones 19:913–925. doi:10.1007/s12192-014-0516-5
Hishikawa T, Cheung JY, Yelamarty RV, Knutson DW (1991) Calcium transients during Fc receptor-mediated and nonspecific phagocytosis by murine peritoneal macrophages. J Cell Biol 115:59–66
Akerboon TPM, Sies H (1981) Assay of glutathione, glutathione disulfide, and glutathione mixed disulfides in biological samples. Methods Enzymol 77:373–382
Schaun MI, Dipp T, Rossato JS, Wilhelm EN, Pinto R, Rech A, Plentz RD, Homem de Bittencourt PI Jr, Reischak-Oliveira A (2011) The effects of periodized concurrent and aerobic training on oxidative stress parameters, endothelial function and immune response in sedentary male individuals of middle age. Cell Biochem Funct 29:534–542. doi:10.1002/cbf.1781
Miragem AA, Ludwig MS, Heck TG, Baldissera FG, dos Santos AB, Frizzo MN, Homem de Bittencourt PI Jr (2015) Estrogen deprivation does not affect vascular heat shock response in female rats: a comparison with oxidative stress markers. Mol Cell Biochem 407:239–249. doi:10.1007/s11010-015-2472-5
Arab K, Steghens JP (2004) Plasma lipid hydroperoxides measurement by an automated xylenol orange method. Anal Biochem 325:158–163
Jiang ZY, Woollard AC, Wolff SP (1991) Lipid hydroperoxide measurement by oxidation of Fe2+ in the presence of xylenol orange. Comparison with the TBA assay and an iodometric method. Lipids 26:853–856
Södergren E, Nourooz-Zadeh J, Berglund L, Vessby B (1998) Re-evaluation of the ferrous oxidation in xylenol orange assay for the measurement of plasma lipid hydroperoxides. J Biochem Biophys Methods 37:137–146
Lapenna D, Cuccurullo F (1993) TBA test and “free” MDA assay in evaluation of lipid peroxidation and oxidative stress in tissue systems. Am J Physiol 265(3 Pt 2):H1030–H1032
Ceconi C, Cargnoni A, Pasini E, Condorelli E, Curello S, Ferrari R (1991) Evaluation of phospholipid peroxidation as malondialdehyde during myocardial ischemia and reperfusion injury. Am J Physiol 260(4 Pt 2):H1057–H1061
Kolberg A, Rosa TG, Puhl MT, Scola G, da Rocha Janner D, Maslinkiewicz A, Lagranha DJ, Heck TG, Curi R, Homem de Bittencourt PI Jr (2006) Low expression of MRP1/GS-X pump ATPase in lymphocytes of Walker 256 tumour bearing rats is associated with cyclopentenone prostaglandin accumulation and cancer immunodeficiency. Cell Biochem Funct 24:23–39
Di Naso FC, Porto RR, Fillmann HS, Maggioni L, Padoin AV, Ramos RJ, Mottin CC, Bittencourt A, Marroni NA, Homem de Bittencourt PI Jr (2015) Obesity depresses the anti-inflammatory HSP70 pathway, contributing to NAFLD progression. Obesity (Silver Spring) 23:120–129. doi:10.1002/oby.20919
Fortes MA, Marzuca-Nassr GN, Vitzel KF, da Justa Pinheiro CH, Newsholme P, Curi R (2016) Housekeeping proteins: how useful are they in skeletal muscle diabetes studies and muscle hypertrophy models? Anal Biochem 504:38–40. doi:10.1016/j.ab.2016.03.023
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Krause M, Bock PM, Takahashi HK, Homem de Bittencourt PI Jr, Newsholme P (2015) The regulatory roles of NADPH oxidase, intra- and extra-cellular HSP70 in pancreatic islet function, dysfunction and diabetes. Clin Sci (Lond) 128:789–803. doi:10.1042/CS20140695
Pedersen BK, Rohde T, Ostrowski K (1998) Recovery of the immune system after exercise. Acta Physiol Scand 162:325–332
Eijsvogels TM, Thompson PD (2015) Exercise is medicine: at any dose? JAMA 314:1915–1916. doi:10.1001/jama.2015.10858
Moynagh PN (2005) The NF-kappaB pathway. J Cell Sci 118(Pt 20):4589–4592
Babior BM, Kipnes RS, Curnutte JT (1973) Biological defense mechanisms. The production by leukocytes of superoxide, a potential bactericidal agent. J Clin Invest 52:741–744
Gwinn MR, Vallyathan V (2006) Respiratory burst: role in signal transduction in alveolar macrophages. J Toxicol Environ Health B Crit Rev 9:27–39
Schafer FQ, Buettner GR (2001) Redox environment of the cell as viewed through the redox state of the glutathione disulfide/glutathione couple. Free Radic Biol Med 30:1191–1212
Raftos JE, Whillier S, Kuchel PW (2010) Glutathione synthesis and turnover in the human erythrocyte: alignment of a model based on detailed enzyme kinetics with experimental data. J Biol Chem 285:23557–23567
Gladwin MT, Lancaster JR, Freeman BA, Schechter AN (2003) Nitric oxide’s reactions with hemoglobin: a view through the SNO-storm. Nat Med 9:496–500
Jia L, Bonaventura C, Bonaventura J, Stamler JS (1996) S-nitrosohaemoglobin: a dynamic activity of blood involved in vascular control. Nature 380:221–226
Krause MS, Oliveira Junior LP, Silveira EMS, Vianna DR, Rossato JS, Almeida BS, Rodrigues MF, Fernandes AJ, Costa JA, Curi R, Homem de Bittencourt PI Jr (2007) MRP1/GS-X pump ATPase expression: is this the explanation for the cytoprotection of the heart against oxidative stress-induced redox imbalance in comparison to skeletal muscle cells? Cell Biochem Funct 25:23–32
Milne KJ, Noble EG (2002) Exercise-induced elevation of HSP70 is intensity dependent. J Appl Physiol 93:561–568
Whitham M, Laing SJ, Jackson A, Maassen N, Walsh NP (2007) Effect of exercise with and without a thermal clamp on the plasma heat shock protein 72 response. J Appl Physiol 103:1251–1256
Demirel HA, Powers SK, Naito H, Tumer N (1999) The effects of exercise duration on adrenal HSP72/73 induction in rats. Acta Physiol Scand 167:227–231
González B, Manso R (2004) Induction, modification and accumulation of HSP70 s in the rat liver after acute exercise: early and late responses. J Physiol 556(Pt 2):369–385
McGivney BA, Eivers SS, MacHugh DE, MacLeod JN, O’Gorman GM, Park SD, Katz LM, Hill EW (2009) Transcriptional adaptations following exercise in thoroughbred horse skeletal muscle highlights molecular mechanisms that lead to muscle hypertrophy. BMC Genomics 10:638. doi:10.1186/1471-2164-10-638
Wang R, Town T, Gokarn V, Flavell RA, Chandawarkar RY (2006) HSP70 enhances macrophage phagocytosis by interaction with lipid raft-associated TLR-7 and upregulating p38 MAPK and PI3K pathways. J Surg Res 136:58–69
Homem de Bittencourt PI Jr, Curi R (2001) Antiproliferative prostaglandins and the MRP/GS-X pump role in cancer immunosuppression and insight into new strategies in cancer gene therapy. Biochem Pharmacol 62:811–819
Acknowledgements and funding
PIHBJ is responsible for grant support with respect to Brazilian National Council for Scientific and Technological Development (CNPq, Grants #402626/2012–5 and #402364/2012–0). TGH was supported by CNPq [#382692/2011–0] and State of Rio Grande do Sul Foundation for Research Support (FAPERGS, grant # 002106–2551/13–5). CMS was recipient of a scholarship from CAPES (The Brazilian Ministry of Education Coordination for the Improvement of Higher Education Personnel).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Authors’ contributions
CMS, TGH, LPOJ, and PIHBJ designed the study. CMS, CVM, GSS, and TGH completed all the experiments described in this manuscript. CMS, LPOJ, TGH, and PIHBJ analyzed the results. CMS and PIHBJ co-wrote the article. PIHBJ provided experimental advice and revised the final version of the manuscript. All the authors had final approval of the submitted and published versions.
Conflicts of interest
The authors declare no conflict of interest and no competing interests such as consultancies, financial involvement, patent ownership, etc. in relation to the work described herein. CNPq, FAPERGS and CAPES (the funding organisms) had no involvement in the propositions presented in this manuscript.
Ethical approval
All the procedures performed in studies involving the animals followed the ethical rules established by Arouca’s Act (Federal Law 11794/2008) and the Guide for Care and Use of Experimental Animals published by the National Institutes of Health (NIH publication no. 85-23, revised in 1996). The procedures were approved by the Federal University of Rio Grande do Sul Ethics Committee on Animal Experimentation (CEUA #23451), according to the guidelines of the Brazilian National Council for the Control of Animal Experimentation (CONCEA).
Rights and permissions
About this article
Cite this article
Schöler, C.M., Marques, C.V., da Silva, G.S. et al. Modulation of rat monocyte/macrophage innate functions by increasing intensities of swimming exercise is associated with heat shock protein status. Mol Cell Biochem 421, 111–125 (2016). https://doi.org/10.1007/s11010-016-2791-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-016-2791-1