Abstract
Context
Power line corridors have been repeatedly assessed as habitat for wild bees; however, few studies have examined them as bee habitat relative to nearby crop fields and surrounding landscape context.
Objectives
We surveyed bee communities in power line corridors near to and isolated from lowbush blueberry fields in two landscape contexts in Maine, U.S.A. We examined the influences of blooming plant abundance and diversity and bee life-history traits including sociality, nesting preference, and body size.
Methods
We surveyed wild bees and blooming plants in power line corridors from 2013 to 2015. We calculated landscape composition surrounding sites at multiple scales and gathered bee trait information from the literature. We assessed differences in bee communities owing to landscape context with generalized linear models.
Results
We collected 125 wild bee species and observed a rare plant-pollinator relationship within power line corridors. We found greater bee abundance and species richness throughout a complex, resource-rich landscape, while mass-flowering lowbush blueberry fields enhanced bee species richness only in a simple, resource-poor landscape. Landscape composition and blooming plant diversity varied with landscape context, though only landscape composition influenced bee communities. Solitary and ground-nesting species were more sensitive to landscape context than social or cavity-nesting species.
Conclusions
Power line corridors provide crucial refugia for crop pollinating wild bees in agricultural landscapes with resource-poor natural habitat, while bees may selectively forage in power line corridors within agricultural landscapes containing resource-rich natural habitat. We found high-quality forage within corridors; quantifying nesting resources could clarify corridor use by wild bees.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lowbush blueberry (Vaccinium angustifolium Aiton) is a North American native plant that is commercially harvested in Maine, U.S.A., and New Brunswick, Prince Edward Island, and eastern Quebec, Canada. Lowbush blueberry requires insect pollination to set fruit, and Maine lowbush blueberry growers invest heavily in managed pollinator species (Hanes et al. 2015; Asare et al. 2017). Managed pollinators are typically honeybees (Apis mellifera L.) and sometimes include commercial bumble bees (Bombus impatiens Cresson) (Drummond 2012). However, more than 100 wild bee species, some of which are more efficient pollinators than managed species (Kevan et al. 1990; Javorek et al. 2002; Drummond 2016; Asare et al. 2017), have been observed in blooming crop fields (Bushmann and Drummond 2015). By increasing reliance on wild bee pollination, growers could reduce costs, though wild bees require consistent access to diverse floral resources throughout the growing season to provide pollination services (Kremen et al. 2002). Growers may be hesitant to supplement their crop fields with wildflowers for pollinators given uncertainty regarding the return of investment on crop yield (Hanes et al. 2015). Many of the most abundant wild bee pollinators of lowbush blueberry are small-bodied with short foraging distances typically less than 500 m (Bushmann and Drummond 2015; Groff et al. 2016), meaning that supplemental wildflower plantings should be located within the maximum foraging distances of crop-pollinating wild bees to be beneficial. These distances are linked to bee body size (Greenleaf et al. 2007). In lowbush blueberry, wild bee foraging distance ranges from less than 100 m for small sweat bees (Family Halictidae) to approximately 1 km for very large bumble bees (Groff et al. 2016). Field margins and planted floral strips have been explored as floral resources for lowbush blueberry pollinators beyond crop bloom (Venturini et al. 2017; McCallum and McLean 2017), and natural habitat surrounding crop fields may also provide these resources (Groff et al. 2016; Du Clos et al. 2020).
Another potential source of floral resources beyond lowbush blueberry bloom are power line corridors, which are managed as open, herb and shrub-dominated early-successional habitat to prevent trees and tall shrubs from reaching power lines. This early succession condition provides nesting and floral resources for wild bees (Russell et al. 2005, 2018; Steinert et al. 2020) and a consistent source of bee habitat in forest-dominated landscapes, which often have few other forage resources for wild bees (Hill and Bartomeus 2016; Eldegard et al. 2017; Steinert et al. 2018, 2020). The plant communities within power lines create novel habitat comparable to extensively studied semi-natural grasslands (Hill and Bartomeus 2016; Eldegard et al. 2017; Russo et al. 2021), though they are sensitive to environmental context, particularly site productivity (Eldegard et al. 2017; Steinert et al. 2018; Sydenham et al. 2020). Although power line corridors have been assessed as wild bee habitat (Russell et al. 2005, 2018; Wagner et al. 2014a, b, 2019; Sydenham et al. 2015, 2016, 2017; Steinert et al. 2020; Russo et al. 2021), few studies have assessed their bee communities relative to surrounding landscape context (but see Sydenham et al. 2020).
In Maine’s forest-dominated lowbush blueberry production landscape, the late spring mass-flowering crop provides substantial floral resources for wild bees over a three- to four-week period from early-mid May to early-mid June (Bushmann and Drummond 2015). Though wild bees can be found in crop fields with little available floral resources post-crop bloom (Du Clos et al. 2020), a consistent source of food for blueberry-pollinating wild bees outside of crop fields for the remainder of the growing season has yet to be identified. Mass-flowering crops can promote the density and diversity of wild bees in nearby semi-natural habitats that provide floral resources pre- and post-crop bloom (Westphal et al. 2003; Diekötter et al. 2013; Holzschuh et al. 2013; Kovács-Hostyanski et al. 2013); however, other studies have found diluted wild bee abundance and reduced wild plant pollination in the surrounding landscape (Holzschuh et al. 2011, 2016; but see Magrach et al. 2018). Early season mass-flowering crops lead to greater bee abundance in both nearby semi-natural habitat and late season flowering crops when there is more semi-natural habitat in the surrounding landscape (Diekötter et al. 2010; Riedinger et al. 2014); further, bee density is greater in blooming crop fields the following year (Riedinger et al. 2015). Therefore, both local- and landscape-scale effects determine how bees use habitat near mass-flowering crop fields. Broadly, more complex landscapes with more semi-natural habitat and greater heterogeneity promote bee abundance and species richness (Steffan-Dewenter 2003; Holzschuh et al. 2007; Ricketts et al. 2008).
The distribution of life-history traits of wild bees in power line corridors, including body size, social habit, and nesting preference, can provide an assessment of habitat quality (Russell et al. 2018; Steinert et al. 2020). Diversity in social habit (solitary or social) and nesting preference (cavity or ground nesting) is reduced in agricultural land when compared to natural habitat (Williams et al. 2010; Forrest et al. 2015); further, trait diversity also decreases when there is less natural habitat in the surrounding landscape (Martins et al. 2015). Wild bee communities with diverse life-history traits increase fruit and seed set of apples (Malus domestica Borkh; Martins et al. 2015), a specialty crop with a pollinator community in northeastern North America similar to that of lowbush blueberry (Stubbs et al. 1992; Bushmann and Drummond 2015). Assessing life-history traits of wild bees observed in power line corridors may clarify the role of this habitat in supporting pollinator communities within Maine’s lowbush blueberry production landscape.
Here, we investigate power line corridors as semi-natural bee habitat relative to nearby mass-flowering lowbush blueberry fields in two landscape contexts in Maine. We examine body size, social habit, and nesting preference of wild bee species observed in power line corridors. We expect bee communities to be more diverse and abundant in corridors near mass-flowering crop fields owing to population spillover post-crop bloom; additionally, we expect greater bee abundance and diversity in corridors within a more complex landscape that provides more semi-natural habitat for bees pre- and post-crop bloom. Furthermore, we expect differences in life-history traits of bee communities in power line corridors, including body size, social habit, and nesting preference. Specifically, in power line corridors near lowbush blueberry fields, we expect more wild bee species exhibiting traits associated with common lowbush blueberry pollinators (Bushmann and Drummond 2015) including a diversity of body sizes, a solitary habit, and a preference for ground nesting. We surveyed bee communities throughout the growing season and over multiple growing seasons to assess seasonal and annual changes in diversity and abundance. Additionally, we surveyed blooming plant abundance and species richness in power line corridors to assess local-scale effects on bee communities.
Methods
Study area and spatial data
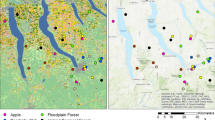
The Maine lowbush blueberry production landscape covers a substantial swath of coastal Maine, U.S.A (44–45° N, 67.5–69.5° W), and consists of two major growing regions with contrasting landscape contexts (Fig. 1). We evaluated the landscape context of each growing region and determined the patterns differed in Du Clos et al. (2020). The Downeast region (Hancock and Washington counties) contains the largest (0.05–1800 ha, average field size 21.4 ha) and most intensively managed lowbush blueberry fields in a matrix dominated by managed coniferous forest. Non-blueberry agriculture and developed land cover are scattered and comprise little of this relatively homogeneous, rural landscape. In contrast, the Midcoast region (Knox, Sagadahoc, and Waldo counties) is heterogeneous, containing smaller (0.05–15.6 ha, average field size 8.26 ha), less intensively managed lowbush blueberry fields interspersed with other agriculture, including pasture, orchards, and small, diversified farms. Developed land in small towns is more prevalent in the more complex Midcoast region than the simpler Downeast region. The matrix in the Midcoast region is deciduous forest-dominant and less intensively harvested.
Extent of the Downeast and Midcoast growing regions within the Maine, USA, lowbush blueberry production landscape. Map insets display representative landscape contexts of the a Downeast (light gray) and b Midcoast (dark gray) growing regions. Bar charts indicate proportion of eight land cover types in the Downeast (top) and Midcoast (bottom) growing regions
We developed a land cover map of the Maine lowbush blueberry production landscape that combines the 2004 Maine Landcover Dataset (https://www.maine.gov/megis/catalog/metadata/melcd.html) with ancillary data on roads, railroads, and wetlands. Full details on the classification of this map are provided in Groff et al. (2016) and Du Clos et al. (2020). The prepared map has 10 m pixel size and eight land cover classes representing different floral and nesting resources for wild bees: (1) agriculture/pasture, consisting of small, diversified farms, orchard crops, or pasture; (2) lowbush blueberry fields; (3) coniferous forest; (4) deciduous/mixed forest; (5) deciduous/mixed forest edge; (6) emergent wetland, an aggregation of forested wetland and scrub-shrub land cover; (7) wetlands/water; and (8) urban areas.
Field sampling
We conducted surveys during the 2013–2015 growing seasons. In 2013, we conducted a pilot study for which we sampled six power line corridor sites in the Downeast region; three were isolated from (≥ 1 km) and three were near (within 150–300 m) lowbush blueberry fields. In 2014 and 2015, we expanded sampling efforts, establishing six study sites in both the Downeast and Midcoast growing regions (12 sites total each year) within power line corridors 30–40 m wide; in each region, three sites were isolated from and three sites were near to lowbush blueberry fields. Sites were distributed within growing regions so that they could all be sampled on the same day, and all sites were at least 2.5 km apart (range 2.5–13.7 km, mean 6.1 km) to minimize overlap in bee communities (Du Clos et al. 2020). We relocated one isolated site Downeast owing to lost access between 2013 and 2014. We sampled the same isolated sites in both growing regions in 2014 and 2015 and sampled different near sites if the nearby crop field was in a non-bloom year (i.e., no bloom or subsequent fruit). This led to the relocation of two Downeast sites in 2014, one Downeast site in 2015, and one Midcoast site in 2015. Two of the three near sites sampled Downeast in 2013 were resampled in 2015. We followed this approach as lowbush blueberry fields are managed so that they bloom and produce a fruit crop every other year (Yarborough 2009). In total, we sampled 16 sites in power line corridors, nine near and seven isolated from lowbush blueberry fields. Nine sites were sampled in the Downeast growing region, and seven sites were sampled in the Midcoast growing region.
We sampled sites in early (27 May-12 June), mid-(7–18 July), and late (7 Aug-21 Sept) season each year to capture seasonal variability in bee communities. Sampling occurred on days that maximized bee activity with clear or bright cloudy skies, minimal wind, and early morning temperatures > 13 °C (Bushmann and Drummond 2015). Average air temperatures during our sampling periods were 17.9 °C, 18 °C, and 17.2 °C, respectively (NOAA 2022). We surveyed wild bee diversity and abundance by placing a set of three bowl traps (one each in fluorescent blue, yellow, and white; New Horizons Supported Services, Inc.) every 10 m along a 150 m transect placed in the center of the corridor. Each bowl contained approximately 85 ml of water and a drop of dish soap to break water tension. After 24 h, captured bees were removed from the traps, and we walked along the transect for one person-hour, live-netting foraging bees observed on blooming flowers. Netting occurred on the same day at all sites in each growing region. In the early season, the same two collectors conducted netting, and in the mid- and late season, the same three collectors conducted netting to reduce collector bias. The one person-hour period was either 20 or 30 min depending on the number of collectors. Using both bowl traps and live netting captures a more complete bee community (Wilson et al. 2008). All collected specimens were cleaned and stored in ethanol (bowl-trapped) or frozen (netted) until pinned. We obtained species-level verifications from Dr. Sara Bushmann, George Stevens Academy; Sam Droege, U.S. Geological Survey Native Bee Inventory and Monitoring Lab; Dr. Jason Gibbs, University of Manitoba; and Dr. Robert Jean, Environmental Solutions and Innovations, Inc. Specimens are stored at the Maine State Museum in Augusta, ME, U.S.A. We gathered nesting preference, social habit, and body size of each species from the literature.
We established two 25 m long transects parallel to our bee sampling transect at 10 m and 100 m to record blooming plant abundance and species richness in the power line corridors. We conducted blooming plant surveys after collecting bowl traps during each sampling period at all sites in 2014 and 2015; this protocol was implemented after our 2013 pilot study. For each blooming patch intersecting each transect up to 1 m on either side, we recorded species, patch size in m2 and percent bloom. Reference specimens of each blooming species were collected and pressed to confirm species identification, and all identifications were confirmed by a botanist (Dr. Alison C. Dibble, University of Maine).
We calculated landscape composition surrounding our corridor transects at four spatial scales (100, 250, 500, and 1000 m) by measuring the percentage of each land cover type present (PLAND) in Fragstats 4.2 (McGarigal et al. 2012). Landscape pattern at these spatial scales has been shown to influence wild bee abundance and species richness (Steffan-Dewenter et al. 2002).
Statistical analyses
We conducted all statistical analyses in R v.3.5.0 (R Core Team 2019). Bee communities are known to vary widely across and within growing seasons, therefore we evaluated annual and seasonal differences in bee abundance and species richness with Kruskal–Wallis tests and determined seasonal differences post-hoc with Dunn’s test of multiple comparisons with package dunn.test (Dinno 2017). We tested for temporal autocorrelation with the Durbin-Watson test with package car (Fox and Weisberg 2011). We tested for spatial autocorrelation with Mantel tests on bee abundance and species richness at all focal spatial scales (100, 250, 500, and 1000 m) with package ade4 (Dray and Dufour 2007). Owing to our different sampling scheme in 2013, we conducted all bee community data analyses on specimens collected in 2014 and 2015.
We compared landscape composition and blooming plant availability at our sampling sites across growing regions and site types. We compared the percentage of all eight land cover types at all four spatial scales with Mann–Whitney U tests. Blooming plant abundance was aggregated over the entire growing season. We multiplied patch size by percent bloom for each species, then summed those values for a cumulative value. We calculated blooming plant species richness as the total number of blooming species counted over the growing season. We used linear regression to model blooming plant abundance and species richness by growing region and site type, as the plant data were normally distributed and homoscedastic.
We sorted bee species into four body size classes based on mean body length: small (< 6 mm), medium (6–9 mm), large (9–12 mm), and extra-large (> 12 mm) (Russell et al. 2018) to represent bees that forage within our four focal spatial scales. We did not include males or queens owing to inconsistencies in intraspecies body size or specimens with an undetermined sex or species identification in our body size analyses. Bees were labeled as ground or cavity nesting, though we did not include Bombus spp., which may nest above or below ground (Kells and Goulson 2003), kleptoparasitic species, or bees with an undetermined species identification in our nesting preference analyses. Lastly, bees were labeled as social or solitary, and we did not include kleptoparastic species or bees with an undetermined species identification in our social habit analyses. We assessed possible associations between these traits with Cramér’s V, a correlation coefficient for categorical variables, with package DescTools (Russell et al. 2018; Signorell et al. 2021).
We assessed differences in bee communities across growing regions and site types with generalized linear models (GLMs) calculated with package MASS (Venables and Ripley 2002) and determined significant relationships with post-hoc analysis of deviance. We modeled the influence of site type, growing region, and a type by region interaction on bee abundance and species richness overall and with respect to nesting preference, social habit, and body size (Table S1). Models of overall bee abundance and species richness had negative binomial error distributions owing to overdispersion, whereas, with the exception of solitary bee species richness, models of abundance and species richness by life-history traits had Poisson error distributions. We then determined if landscape composition or blooming plant availability across growing regions or site types influenced bee communities with GLMs and analysis of deviance of bee abundance and species richness. We targeted this series of models to explain the overall and life-history-associated bee community model results, only modeling significant relationships with site characteristics (Table S2). We modeled bee communities associated with the percent of each land cover type at each spatial scale; solitary bees were modeled with a negative binomial error and all other models were based upon Poisson error distributions.
Results
Bee community summary
Over three sampling years, we collected 2121 bee specimens representing six families, 27 genera, and 125 species. We collected 1177 bees representing 107 species in sampling sites near lowbush blueberry fields and 944 bees representing 98 species in sites isolated from lowbush blueberry fields. Generally, we collected more bees in power line corridor sites near lowbush blueberry fields. The number of species was greater in power line corridor sites near lowbush blueberry fields in the Downeast growing region, while species numbers were similar across sites near to or isolated from lowbush blueberry fields in the Midcoast growing region (Table 1). A summary of sampling effort is provided in Table S3.
The most abundant bee species collected was the sweat bee Lasioglossum cressonii (Robertson) (535 individuals collected). Other common bees included the bumble bees Bombus ternarius (Say) (249) and B. vagans (Smith) (159) and the sweat bee Augochlorella aurata (Smith) (193). Uncommon bee genera included Colletes and Osmia, both of which contain species associated with lowbush blueberry. We collected two specimens of Macropis nuda (Provancher), a rare solitary bee that specializes on oils produced by Lysimachia spp., a plant group we found throughout our sites in both growing regions. We also collected one new bee species state record for Maine, represented by one specimen of Melitta americana (Smith). This species is a rare specialist of Vaccinium spp., particularly cranberry; our specimen was collected Downeast, where wild cranberry (Vaccinium macrocarpon Aiton) is abundant (Jones et al. 2014). A complete list of bee species, their size class, and the site types and growing regions in which they were collected is presented in Table S4.
We found no spatial autocorrelation of bee communities within growing regions across sampling years. Bee abundance and species richness changed throughout the sampling season (Kruskal–Wallis abundance χ2 = 19.19, df = 3, p < 0.001, richness χ2 = 23.30, df = 3, p < 0.001). Bees were less abundant and less species rich in the late season than early or mid-season, with no significant differences in abundance or species richness between early and mid-season. We found annual differences in bee communities between 2013 and 2014 (Dunn’s test abundance Z = − 2.14, p = 0.01) and 2013 and 2015 (abundance Z = 3.44, p < 0.001, richness Z = − 2.40, p = 0.001), with no differences between 2014 and 2015. Additionally, bee abundance was temporally autocorrelated when 2013 data were included in analyses (Durbin-Watson test statistic = 1.36, p = 0.03), though species richness was not (D-W = 1.77, p = 0.28). We found no temporal autocorrelation in our data for bee abundance (D-W = 2.07, p = 0.72) or species richness (D-W = 2.16, p = 0.98) between 2014 and 2015. This supports our decision to conduct community analyses on specimens collected in 2014 and 2015.
Blooming plant availability
We observed 62 blooming plant species within our corridor sites between 2014 and 2015, 10 of which were non-native. Blooming plant species richness was greater in the Midcoast region, where we recorded 51 species, than in the Downeast region, where we recorded 31 species (F(1,22) = 13.11, p = 0.001). There was no significant difference in blooming plant abundance between growing regions, and no difference in blooming plant abundance or species richness between corridor sites near to or isolated from lowbush blueberry fields. Eleven blooming plant species were recorded exclusively Downeast, whereas 31 occurred exclusively Midcoast. Downeast-exclusive species tended to be ericaceous and bog associated; in contrast, Midcoast-exclusive species were often disturbance-associated. A full list of floral species and the growing regions in which they were observed is provided in Table S5.
Landscape composition
We found more coniferous forest and wetland land cover at all spatial scales surrounding corridor sites in the Downeast growing region than in the Midcoast region (Table S6). In contrast, we found more deciduous/mixed forest, more urban land cover, and more deciduous/mixed forest edge and agriculture/pasture around corridor sites in the Midcoast growing region than in the Downeast region (Table S3). Corridor sites near lowbush blueberry fields were surrounded by more lowbush blueberry land cover at all spatial scales. We also found more coniferous forest around our sites isolated from lowbush blueberry fields at larger spatial scales (Table S6). We did not find a difference in the amount of lowbush blueberry surrounding corridor sites between growing regions.
Bee communities by growing region and site type
We found significant differences in both bee abundance (df = 1,21, deviance = 3.94, p = 0.05) and species richness (df = 1,21, deviance = 16.24, p < 0.001) between the two growing regions. Differences in bee communities across site types were not significant. However, we found a significant type x region interaction for bee species richness (df = 1,21, deviance = 5.21, p = 0.02). Bee species richness was significantly different between corridor sites near to and isolated from lowbush blueberry in the Downeast growing region (df = 1,10, deviance = 7.24, p = 0.007), while we found no difference in bee species richness between site types in the Midcoast growing region (Table 2).
Bee life-history traits and corridor context across growing regions
We found little association between body size and nesting habit (Cramér’s V = 0.13), moderate association between nesting habit and sociality (Cramér’s V = 0.25), and moderate to strong association between sociality and body size (Cramér’s V = 0.43). Strong associations are represented by Cramér’s V > 0.6; therefore, we analyzed all traits. We found significant differences in medium (df = 1,21, deviance = 15.10, p < 0.001) and extra-large sized (df = 1,21, deviance = 14.59, p = 0.008) bee species richness by growing region (Fig. 2). Medium sized bee species richness is greater in the Midcoast growing region, where it is associated with more deciduous/mixed forest edge (df = 1,22, deviance = 7.88, p = 0.004) and less coniferous forest (df = 1,22, deviance = 8.65, p = 0.003) surrounding corridor sites (Fig. S1a, b). Extra-large sized bee species richness is also greater in the Midcoast region, where it is influenced by more agriculture/pasture surrounding corridor sites (df = 1,22, deviance = 4.11, p = 0.042) (Fig. S2a). Extra-large sized bee species richness was also positively influenced by blooming plant abundance (m2 per transect) (df = 1,22, deviance = 17.79, p = 0.042) (Fig. S2b).
Bee species richness in power line corridor sites in two Maine, USA, lowbush blueberry growing regions within four body size classes: small (< 6 mm), medium (6–9 mm), large (9–12 mm), and extra-large (> 12 mm) (Russell et al. 2018), 2014–2015. **significant at p < 0.01 and ***significant at p < 0.001
We found significant differences in the species richness of social bees (df = 1,21, deviance = 7.12, p = 0.007) and solitary bees (df = 1,21, deviance = 10.97, p < 0.001) between growing regions. Solitary bee species richness was greater in the Midcoast region, where it is associated with less coniferous forest and wetland and more agriculture/pasture and deciduous/mixed forest edge surrounding corridor sites (Fig. 3a; Table S7a). We found a significant site type by growing region interaction for the species richness of solitary bees (df = 1,20, deviance = 6.98, p = 0.008) owing to an interactive effect of lowbush blueberry land cover surrounding our corridor sites at 250 (df = 1,20, deviance = 7.54, p = 0.006), 500 (df = 1,20, deviance = 6.46, p = 0.01), and 1000 m (df = 1,20, deviance = 8.68, p = 0.003). Lowbush blueberry had a negative association with solitary bee species richness in the Midcoast region and a positive association in the Downeast region (Fig. 4), though as a single variable, the amount of lowbush blueberry surrounding corridor sites did not significantly influence solitary bee species richness (Table S7a). Furthermore, we found that greater blooming plant abundance in corridor sites was associated with greater solitary bee species richness (df = 1,22, deviance = 6.62, p = 0.01) (Fig. S3a). Social bee species richness was also greater in the Midcoast region, increasing with less coniferous forest cover and more agriculture/pasture (Fig. 3a; Table S7b) in the surrounding landscape.
Interactive effect of percent lowbush blueberry surrounding power line sampling sites at three spatial scales on solitary bee species richness in the Midcoast (gray circles, dashed line) and Downeast (black triangles, solid line) growing regions of the Maine, USA lowbush blueberry production landscape, 2014–2015
Ground nesting (df = 1,21, deviance = 9.98, p = 0.001) and cavity nesting (df = 1,21, deviance = 4.45, p = 0.034) bee species richness was greater in corridors in the Midcoast region (Fig. 3b). Less coniferous forest and more agriculture/pasture influenced ground nesting bee species richness at multiple scales surrounding corridors, whereas more deciduous/mixed forest edge and urban area and less wetland were influential at small scales surrounding corridors (Table S7c). Greater blooming plant abundance in corridor sites led to greater ground nesting bee species richness (df = 1,22, deviance = 7.93, p = 0.004) (Fig. S3b). We found greater cavity nesting bee species richness in corridor sites surrounded by less coniferous forest, more deciduous/mixed forest edge, and more wetland (Table S7d).
We found significant differences in bee species richness associated with body size (df = 1,10, deviance = 13.00, p = 0.05), social habit (df = 1,10, deviance = 4.85, p = 0.027; Fig. 5a), and nesting preference (df = 1,10, deviance = 5.43, p = 0.019; Fig. 5b) between corridor sites near to and isolated from lowbush blueberry fields in the Downeast growing region. Large sized (500 m scale; df = 1,10, deviance = 4.73, p = 0.03), solitary (all spatial scales; Table S8a), and ground nesting (all spatial scales; Table S8b) bee species richness were positively associated with surrounding lowbush blueberry land cover at sites near crop fields. Ground nesting bee species richness decreased in both near and isolated sites with greater coniferous forest cover surrounding corridors at 500 and 1000 m, whereas greater emergent wetland land cover at 1000 m led to increased species richness of ground nesting and solitary bee species richness. Finally, more deciduous forest in the 1000 m surrounding corridor sites near lowbush blueberry fields reduced solitary bee species richness, and interestingly, there was no effect of coniferous land cover on bee species richness at any spatial scale surrounding near sites (Table S8a, b). We found no significant differences in bee life-history traits between site types in the Midcoast growing region. All modeling results are summarized in Tables S9a and b.
Discussion
Bee communities in Maine power line corridors
Power line corridors provide ample pre- and post-bloom habitat for crop-pollinating wild bees in Maine’s two major lowbush blueberry production regions. We collected 125 of the 278 recorded wild bee species in the state (Dibble et al. 2017) and observed diverse and abundant blooming plants in corridors throughout the growing season. Bee species richness, but not abundance, was affected by local and landscape scale characteristics of corridor sites in our study landscape (Table S9a, b). In the heavily forested Downeast region, where bee habitat is relatively scarce outside of lowbush blueberry fields and bee species richness in power line corridors was low, corridors with abundant forage may serve as refugia, providing more food for bees than is available in the surrounding landscape. Diverse and abundant bee communities have been documented in power line corridors in the forested landscape of southeastern Norway and in seismic lines in the boreal forests of Alberta, Canada, supporting our results (Steinert et al. 2020; Nelson et al. 2021). In contrast, the greater bee species richness found in Midcoast corridors may reflect the surrounding landscape diversity, where bee habitat is more prevalent. Bees may selectively forage in corridors with more abundant flowers along with other habitat types, including agriculture/pasture, deciduous/mixed forest edge, and urban areas (Krimmer et al. 2019; Du Clos et al. 2020).
Our findings suggest the intermediate landscape-complexity hypothesis may apply, which states that conservation efforts (in this case, power line vegetation management) may be more beneficial in structurally simple landscapes over more complex landscapes (Tscharntke et al. 2012; Krimmer et al. 2019), especially if the conservation activity creates quality habitat (Kleijn and vanLangevelde 2006). Power line corridors in Maine are not managed for conservation purposes; however, the habitat resulting from current management practices appears to have the same effect as conservation directed management techniques such as Integrated Vegetation Management (IVM; Russell et al. 2005, 2018; Russo et al. 2021) by supporting bee communities Downeast where there are few other sources of bee habitat (Du Clos et al. 2020). These practices work in concert to maintain early-successional treeless habitat; Wagner et al. (2019) found corridors managed with mowing only, without pesticide spot treatments, reduced bee abundance. Removing some woody debris from power line corridors after mowing may further enhance bee communities (Steinert et al. 2020).
Life-history traits
We observed that species richness of solitary bees and ground nesting bees was influenced by proximity to a mass-flowering crop and surrounding landscape composition. Common pollinators of our focal crop, lowbush blueberry, are often solitary and nest underground (Bushmann and Drummond 2015). Our findings contrast with Russell et al. (2018), who found greater diversity of cavity nesting bees in corridors that are managed long term to maintain early-successional habitat, owing to nesting resources in dead wood and woody shrubs. Nesting habitat assessments of similar land cover in Maine suggest that dead wood and woody shrubs should be plentiful in corridors and that suitable soil for ground nesting should be limited (Du Clos 2019); however, the bee communities we observed in our study, which were dominated by ground nesting species, indicate the opposite. We observed, but did not quantify, both cavity and ground nesting resources within power line corridors in Maine, and they varied widely in amount and quality across our sites. These findings align with Steinert et al. (2020), who found ample cavity nesting resources in corridors but few cavity nesting bees; further, removal of woody debris from corridors, a likely source of cavity nests, unexpectedly enhanced cavity-nesting bees in their study. Owing to their relative rarity compared to ground-nesting bees, less is known about cavity-nesting bees in semi-natural habitat such as that found in power line corridors, though they appear to be sensitive to changes in environmental and landscape context (Williams et al. 2010; Forrest et al. 2015; Harrison et al. 2018). Surveying nesting resources (tree snags, soft-pith stem resources, bare soil, soil moisture and texture) in corridors during future research and perhaps targeting cavity-nesting bees may clarify these relationships (Russell et al. 2018).
Blooming plants in power line corridors and other linear landscape features may support greater social bee species richness (Kallioniemi et al. 2017; Russell et al. 2018; Russo et al. 2021); however, our work revealed a link between blooming plant abundance and solitary bee species richness, not social bee species richness. Solitary bee species richness responded to more local and landscape scale habitat characteristics in our study than all other life-history traits. Social bee species vary widely in body size and therefore foraging range (Greenleaf et al. 2007), and larger social species can travel longer distances to obtain sufficient resources. In contrast, smaller solitary bees may be more dispersal-limited and thus more susceptible to limited resource availability (Sydenham et al. 2017). Blooming plants in nearby power line corridors may be the only accessible forage for solitary bees after crop bloom, particularly in the coniferous forest-dominant Downeast growing region. Additionally, the number of singleton solitary species occurring in our study sites may contribute to the observed effects of local and landscape resources on solitary bee species richness. Finally, species richness as a measure of diversity overlooks potential relationships between habitat resources and bee communities. Further analyses using functional traits (e.g., bumble bee tongue length) may provide more insight into how bees use and are distributed across our study landscape (Martins et al. 2015; Sydenham et al. 2015, 2016, 2017; Steinert et al. 2020).
Blooming plants
Our work demonstrates that power line corridors contain enough blooming plants to provision wild bee populations post-lowbush blueberry bloom through the remainder of the growing season (mid-June through October). Blooming plants are often correlated with bee species richness (Potts et al. 2003; Ebeling et al. 2008; Russo et al. 2021). In our study, blooming plant abundance but not species richness in corridor sites influenced bee species richness. Available forage in corridors may be comparable to grasslands (Hill and Bartomeus 2016), though in the Maine lowbush blueberry production landscape, corridor forage is more similar to wooded or emergent wetlands (Dibble et al. 2018; Du Clos et al. 2020). Further, wooded or emergent wetlands and early succession vegetation occurs in power line corridors throughout the New England states (Connecticut, Rhode Island, Massachusetts, Vermont, New Hampshire, and Maine, USA) (Wagner et al. 2014a, b; 2019). Indeed, we observed a similar progression of blooming plant communities in Maine corridors as that found in southern New England (Wagner et al. 2019): ericaceous flowers in the early season, brambles (Rubus spp.), in the mid-season, and goldenrods (Solidago spp.) in the late season. Power line corridors in the Maine lowbush blueberry production landscape support rare wild bee species. We found two specimens of the native loosestrife specialist Macropis nuda (Provancher). Native loosestrifes in southern New England power line corridors were attributed to the rediscovery of one of North America’s rarest wild bees, Epeoloides pilosulus (Cresson), which parasitizes Macropis spp. (Wagner and Ascher 2008; Wagner et al. 2019).
We found more herb-dominated plant communities in the Midcoast region and more ericaceous-dominated plant communities in the Downeast region; a similar trend in corridors within Norway boreal forest suggests that herb-dominant communities promote wild bee diversity (Sydenham et al. 2016, 2017). However, the role of ericaceous-dominant early successional vegetation in corridors is noteworthy. Ericaceous plant species have poricidal anthers with unique morphology that often inhibits generalist bees from obtaining pollen (Bell et al. 2009). Additionally, certain suites of bee species (e.g., Andrena spp. and Bombus spp.) are morphologically and behaviorally adapted to extracting pollen from and pollinating ericaceous flowers (Javorek et al. 2002; Drummond 2016); many of these bee species show floral constancy on ericaceous flowers, including our focal mass-flowering crop, lowbush blueberry (Stubbs et al. 1992, 1997; Bushmann and Drummond 2015). In our corridor surveys, 80% of the bee species we collected were ericaceous affiliated (Table S3). Indeed, Wagner et al. (2019) found greater wild bee diversity associated with greater ericaceous plant cover in power line corridors in southern New England. Additionally, Sydenham et al. (2015) concluded that ericaceous-dominant plant communities in Norway power line corridors contain more spring emerging bees, large-sized bees, and social bees, indicating that this plant community may support particular bee functional traits. While our work identified blooming plants available to wild bees, quantifying all vegetation by type (trees, shrubs, herbs, grasses) would provide a more complete assessment of corridor habitat (Wagner et al. 2014b; Russell et al. 2018).
Landscape context and mass-flowering crops
In both the simple Downeast and complex Midcoast growing regions, we found that bee species richness decreased in power line corridors surrounded by more coniferous forest, which offers little forage and has low bee abundance (Groff et al. 2016; Eldegard et al. 2017; Du Clos et al. 2020). Differences in landscape composition across the lowbush blueberry production landscape explain many of our results. In a mixed-use, primarily forested landscape, agricultural and urban land cover promote bee species richness (Winfree et al. 2007), as they do in the complex Midcoast growing region (Du Clos et al. 2020). Additionally, urban and agricultural land cover in Maine contain different bee species than found in the forested matrix (Du Clos et al. 2020; also see Harrison et al. 2018), contributing to the greater species richness we observed in power line corridors in the Midcoast growing region. Urban land cover in Maine’s lowbush blueberry production landscape is generally classified as exurban along an urban–rural land use gradient (Kaminski et al. 2021), meaning there is plenty of green space and natural area that provides bee habitat. Although we found more wetland land cover in landscapes of the simple Downeast growing region than the complex Midcoast growing region, we do not have evidence that wetlands influence Maine bee communities (Du Clos et al. 2020). Rather, it is lowbush blueberry fields that have an important influence on bee communities in corridors Downeast, an effect that is amplified by the dominance of coniferous forest in the surrounding landscape.
We found a positive influence of surrounding lowbush blueberry land cover on bee species richness in power line corridors near lowbush blueberry fields in the more homogeneous Downeast but not the heterogeneous Midcoast growing region. In fact, bee species richness decreased with increasing lowbush blueberry surrounding corridor sites Midcoast, contrary to our expectation. A similar contextual response to a mass-flowering crop has been observed on plant reproductive success associated with planted floral strips in southern Sweden; reproductive success increased near planted strips in homogeneous landscapes, whereas it decreased near planted strips in heterogeneous landscapes (Herbertsson et al. 2018). Our work supports the growing consensus that linear landscape features (e.g., floral strips, field edges, hedgerows, corridors) relative to a mass-flowering crop support different bee communities based on landscape context. Previous work has assessed bee communities of hedgerows in agriculturally-intense landscapes (Sardiñas and Kremen 2015; Garratt et al. 2017) and bumble bees in edge habitat across two landscape contexts in Sweden (Kallioniemi et al 2017). Conservation plantings, which are often linear, have a greater influence on bee communities in resource-poor landscapes than in more complex areas (Krimmer et al. 2019). Here, we reveal contextual influences on bee communities of power line corridors within heavily forested landscapes.
Conservation value of power line corridors in Maine
Power line corridors in Maine’s lowbush blueberry production landscape lie within a forest-dominant matrix. In the Midcoast growing region, the forest is transitional between the hardwood forests of southern New England and the boreal forest that dominates the Downeast growing region. Our work reveals that the open habitat provided by corridors in Maine contains early-successional vegetation typical of boreal forest and transition zones, including ericaceous species that many lowbush blueberry pollinators can use pre- and post-crop bloom. Owing to their constancy throughout Maine’s lowbush blueberry production landscape and the variety of floral resources found within, power line corridors may serve as refugia for both generalist and ericaceous-specialist pollinators, potentially benefitting lowbush blueberry pollination with little to no effort from crop growers. When managed for an early-successional vegetation community, power line corridors may be a beneficial, but overlooked, source of wild bee habitat in forest-dominant landscapes.
Data availability
Not applicable, see Acknowledgements.
Code availability
Not applicable, see Acknowledgements.
References
Asare E, Hoshide AK, Drummond FA, Criner GK, Chen X (2017) Economic risk of bee pollination in Maine wild blueberry Vaccinium angustifolium. J Econ Entomol 110(5):1980–1992.
Bell DJ, Rowland LJ, Smagula J, Drummond FA (2009) Recent advances in biology and genetics of lowbush blueberry. Maine Agric for Exp Station Misc Rep 203:1–28
Bushmann SL, Drummond FA (2015) Abundance and diversity of wild bees (Hymenoptera: Apoidea) found in lowbush blueberry growing regions of Downeast Maine. Environ Entomol 44(4):975–989.
Dibble AC, Drummond FA, Stubbs C, Bushmann SL, Ascher JS, Ryan J (2017) Bees of Maine, with a state species checklist. Northeastern Nat 24(15):1–48.
Dibble AC, Averill AL, Bickerman-Martens K, Bosworth SC, Bushmann S, Drummond FA, Fowler J, Hoshide AK, Leach ME, Skyrm K, Venturini E, White A (2018) Bee habitat in Northern New England. In: Maine agricultural and forest experiment station misc. report, vol 448. University of Maine, Orono, Maine, USA.
Diekötter T, Kadoya T, Peter F, Wolters V, Jauker F (2010) Oilseed rape crops distort plant-pollinator interactions. J Appl Ecol 47:209–214.
Diekötter T, Peter F, Jauker B, Wolters V, Jauker F (2013) Mass-flowering crops increase richness of cavity-nesting bees and wasps in modern agro-ecosystems. GCB Bioenergy 6(3):219–226.
Dinno A (2017) dunn.test: Dunn's test of multiple comparisons using rank sums. R package version 1.3.5. https://CRAN.R-project.org/package=dunn.test. Accessed 1 February 2017.
Dray S, Dufour AB (2007) The ade4 package: implementing the duality diagram for ecologists. J Stat Soft 22(4):1–20
Drummond FA (2012) Commercial bumble bee pollination of lowbush blueberry. Int J Fruit Sci 12(1–3):54–64
Drummond FA (2016) Behavior of bees associated with the wild blueberry agro-ecosystem in the USA. Int J Entomol Nematol 2(1):27–41
Du Clos B (2019) Landscape pattern and wild bee communities in Maine. Dissertation, University of Maine.
Du Clos B, Drummond FA, Loftin CS (2020) Noncrop habitat use by wild bees in a mixed-use agricultural landscape. Environ Entomol 49(2):502–515.
Ebeling A, Klein AM, Schumacher J, Weisser WW, Tscharntke T (2008) How does plant richness affect pollinator richness and temporal stability of flower visits? Oikos 117:1808–1815.
Eldegard K, Eyitayo DL, Lie MH, Moe SR (2017) Can powerline clearings be managed to promote insect-pollinated plants and species associated with semi-natural grasslands? Landsc Urban Plan 167:419–428.
Forrest JRK, Thorp RW, Kremen C, Williams NM (2015) Contrasting patterns in species and functional-trait diversity of bees in an agricultural landscape. J Appl Ecol 52(3):706–715.
Fox J, Weisberg S (2011) An R companion to applied regression, 2nd edn. Sage, Thousand Oaks
Garratt MPD, Senapathi S, Coston DJ, Mortimer SR, Potts SG (2017) The benefits of hedgerows for pollinators and natural enemies depends on hedge quality and landscape context. Agric Ecosyst Environ 247:363–370.
Groff SC, Loftin CS, Drummond F, Bushmann S, McGill B (2016) Parameterization of the InVEST crop pollination model to spatially predict abundance of wild blueberry (Vaccinium angustifolium Aiton) native bee pollinators in Maine, USA. Environ Modell Softw 79:1–9.
Greenleaf SS, Williams NM, Winfree R, Kremen C (2007) Bee foraging ranges and their relationship to body size. Oecologia 153:589–596.
Hanes SP, Collum KK, Hoshide AK, Asare E (2015) Grower perceptions of native pollinators and pollination strategies in the lowbush blueberry industry. Renew Agric Food Syst 30(2):124–131.
Harrison T, Gibbs J, Winfree R (2018) Forest bees are replaced in agricultural and urban landscapes by native species with different phenologies and life-history traits. Global Chang Biol 24(1):287–296.
Herbertsson L, Jönsson AM, Andersson GKS, Seibel K, Rundlöf M, Ekroos J, Stjernman M, Olsson O, Smith HG (2018) The impact of sown flower strips on plant reproductive success in Southern Sweden varies with landscape context. Agr Ecosyst Environ 259:127–134.
Hill B, Bartomeus I (2016) The potential of electricity transmission corridors in forested areas as bumblebee habitat. R Soc Open Sci 3:160525.
Holzschuh A, Steffan-Dewenter I, Kleijn D, Tscharntke T (2007) Diversity of flower-visiting bees in cereal fields: effects of farming system, landscape composition, and regional context. J Appl Ecol 44:41–49.
Holzschuh A, Dormann CF, Tscharntke T, Steffan-Dewenter I (2011) Expansion of mass-flowering crops leads to transient pollinator dilution and reduced wild plant pollination. Proc R Soc B 278:3444–3451.
Holzschuh A, Dormann CF, Tscharntke T, Steffan-Dewenter I (2013) Mass-flowering crops enhance wild bee abundance. Oecologia 172:477–484.
Holzschuh A, Dainese M, González-Varo JP, Mudri-Stonjić S, Riedinger V, Rundlöf M, Scheper J, Wickens JB, Wickens VJ, Bommarco R, Kleijn D, Potts SG, Roberts SPM, Smith HG, Vilà M, Vujić A, Steffan-Dewenter I (2016) Mass-flowering crops dilute pollinator abundance in agricultural landscapes across Europe. Ecol Lett 19(10):1228–1236.
Javorek SK, MacKenzie KE, Vander Kloet SP (2002) Comparative pollination effectiveness among bees (Hymenoptera: Apoidea) on lowbush blueberry (Ericaceae: Vaccinium angustifolium). Ann Entomol Soc Am 95(3):345–351.
Jones MS, Vanhanen H, Peltola R, Drummond FA (2014) A global review of arthropod-mediated ecosystem-services in Vaccinium berry agroecosystems. Terrestrial Arthropod Rev 7:41–78.
Kallioniemi E, Åström J, Rusch GM, Dahle S, Åström S, Gjershaug JO (2017) Local resources, linear elements and mass-flowering crops determine bumblebee occurrences in moderately intensified farmlands. Agr Ecosyst Environ 239:90–100.
Kaminski A, Bauer DM, Bell KP, Loftin CS, Nelson EJ (2021) Using landscape metrics to characterize towns along an urban-rural gradient. Landsc Ecol 36:2937–2956.
Kells AR, Goulson D (2003) Preferred nesting sites of bumblebee queens (Hymenoptera: Apidae) in agroecosystems in the UK. Biol Conserv 109(2):165–174.
Kevan PG, Clark A, Thomas VG (1990) Insect pollinators and sustainable agriculture. Am J Altern Agric 5(1):13–22.
Kleijn D, van Langevelde F (2006) Interacting effects of landscape context and habitat quality on flower visiting insects in agricultural landscapes. Basic Appl Ecol 7:201–214.
Kovács-Hostyánski A, Haenke S, Batáry P, Jauker B, Báldi A, Tscharntke R, Holzschuh A (2013) Contrasting effects of mass-flowering crops on bee pollination of hedge plants at different spatial and temporal scales. Ecol Appl 23(8):1938–1946.
Kremen C, Williams NM, Thorp R (2002) Crop pollination from native bees at risk from agricultural intensification. P Natl Acad Sci USA 99(26):16812–16816.
Krimmer E, Martin EA, Krauss J, Holzschuh A, Steffan-Dewenter I (2019) Size, age, and surrounding semi-natural habitats modulate the effectiveness of flower-rich agri-environment schemes to promote pollinator visitation in crop fields. Agr Ecosyst Environ 284:106590.
Magrach A, Holzschuh A, Bartomeus I, Riedinger V, Roberts SPM, Rundlöf M, Vujić A, Wickens JB, Wickens VJ, Bommarco R, González-Varo JP, Potts SG, Smith HG, Steffan-Dewenter I, Vilà M (2018) Plant-pollinator networks in semi-natural grasslands are resistant to the loss of pollinators during blooming of mass-flowering crops. Ecography 41:62–74.
Martins KT, Gonzalez A, Lechowicz MJ (2015) Pollination services are mediated by bee functional diversity and landscape context. Agr Ecosyst Environ 200:12–20.
McCallum RS, McLean NL (2017) Floral resources and bumble bee abundance in lowbush blueberry field margins. J Acad Entomol Soc 13:37–45
McGarigal K, Cushman SA, Ene E (2012) FRAGSTATS v4: spatial pattern analysis program for categorical and continuous maps. Computer software program produced by the authors at the University of Massachusetts, Amherst. http://www.umass.edu/landeco/research/fragstats/fragstats.html. Accessed 31 August 2017.
Nelson CJ, Frost CM, Nielsen SE (2021) Narrow anthropogenic linear corridors increase the abundance of abundance, diversity, and movement of bees in boreal forests. Forest Ecol Manag 489:119044.
NOAA National Centers for Environmental information, Climate at a Glance: Statewide Time Series, published January 2022. Accessed 28 Jan 2022 https://www.ncdc.noaa.gov/cag/
Potts SG, Vulliamy B, Dafni A, Ne’eman G, Willmer P (2003) Linking bees and flowers: how do floral communities structure pollinator communities? Ecology 84(10):2628–2642.
R Core Team (2019) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed 25 March 2019.
Ricketts TH, Regetz J, Steffan-Dewenter I, Cunningham SA, Kremen C, Bogdanski A, Gemmill-Herren B, Greenleaf SS, Klein AM, Mayfield MM, Morandin LA, Ochieng A, Viana BF (2008) Landscape effects on crop pollination services: are there general patterns? Ecol Lett 11(5):499–515.
Riedinger V, Renner M, Rundlöf M, Steffan-Dewenter I, Holzschuh A (2014) Early mass-flowering crops mitigate pollinator dilution in late-flowering crops. Landsc Ecol 29:425–435.
Riedinger V, Mitesser O, Hovestadt T, Steffan-Dewenter I, Holzschuh A (2015) Annual dynamics of wild bee densities: attractiveness and productivity effects of oilseed rape. Ecology 96(5):1351–1360.
Russell KN, Ikerd H, Droege S (2005) The potential conservation value of unmowed powerline strips for native bees. Biol Conserv 124:133–148.
Russell KN, Russell GJ, Kaplan KL, Mian S, Kornbluth S (2018) Increasing the conservation value of powerline corridors for wild bees through vegetation management: an experimental approach. Biodivers Conserv 27(10):2541–2565.
Russo L, Stout H, Roberts D, Ross BD, Mahan CG (2021) Powerline right-of-way management and flower visiting insects: how vegetation management can promote pollinator diversity. PLoS ONE 16(1):e0245146.
Sardiñas HS, Kremen C (2015) Pollination services from field-scale agricultural diversification may be context-dependent. Agr Ecosyst Environ 207:17–25.
Signorell A et al (2021) DescTools: tools for descriptive statistics. R package version 0.99.44. . Accessed 4 Feb 2022.
Steffan-Dewenter I, Münzenberg U, Bürger C (2002) Scale-dependent effects of landscape context on three pollinator guilds. Ecology 83(5):1421–1432.
Steinert M, Moe SR, Sydenham MAK, Eldegard K (2018) Different cutting regimes improve species and functional diversity of insect-pollinated plants in power-line clearings. Ecosphere 9(11):e02509.
Steinert M, Sydenham MAK, Eldegard K, Moe SR (2020) Conservation of solitary bees in power-line clearings: sustained increase in habitat quality through woody debris removal. Global Ecol Conserv 21:e00823.
Stubbs CS, Jacobson HA, Osgood EA, Drummond FA (1992) Alternative forage plants for native (wild) bees associated with lowbush blueberry, (Vaccinium spp.), in Maine. In: Maine agricultural and forest experiment station misc. report, vol 148. University of Maine, Orono, Maine, USA.
Stubbs CS, Drummond FA, Allard SL (1997) Bee conservation and increasing Osmia spp. Maine Wild Blueberry Fields Northeast Nat 4(3):133–144.
Sydenham MAK, Moe SR, Totland Ø, Eldegard K (2015) Does multi-level environmental filtering determine the functional and phylogenetic composition of wild bee species assemblages? Ecography 38:140–153.
Sydenham MAK, Moe SR, Stanescu-Yadav DN, Totland Ø, Eldegard K (2016) The effects of habitat management on the species, phylogenetic, and functional diversity of bees are modified by the environmental context. Ecol Evol 6(4):961–973.
Sydenham MAK, Moe SR, Kuhlmann M, Potts SG, Roberts SPM, Totland Ø, Eldegard K (2017) Disentangling the contributions of dispersal limitation, ecological drift, and ecological filtering to wild bee community assembly. Ecosphere 8(1):e01650.
Sydenham MAK, Moe SR, Eldegard K (2020) When context matters: spatial prediction models of environmental conditions can identify target areas for wild bee habitat management interventions. Landsc Urban Plan 193:103673.
Tscharntke T, Tylianakis JM, Rand TA, Didham RK, Fahrig L, Batáry P, Bengtsson J, Clough Y, Crist TO, Dormann CF, Ewers RM, Fründ J, Holt RD, Holzschuh A, Klein AM, Kleijn D, Kremen C, Landis DA, Laurance W, Lindenmayer D, Scherber C, Sodhi N, Steffan-Dewenter I, Thies C, van der Putten WH, Westphal C (2012) Landscape moderation of biodiversity patterns and processes—eight hypotheses. Biol Rev 87:661–685.
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, New York
Venturini EM, Drummond FA, Hoshide AK, Dibble AC, Stack LB (2017) Pollination reservoirs in lowbush blueberry (Ericales, Ericaceae). J Econ Entomol 110(2):333–346.
Wagner DL, Ascher JS (2008) Rediscovery of Epeoloides pilosula (Cresson) (Hymenoptera: Apidae) in New England. J Kan Entomol Soc 81(2):81–84.
Wagner DL, Ascher JS, Bricker NK (2014a) A transmission right-of-way as habitat for wild bees (Hymenoptera: Apoidea: Anthophila) in Connecticut. Ann Entomol Soc Am 107(6):1110–1120.
Wagner DL, Metzler KJ, Leicht-Young SA, Motzkin G (2014b) Vegetation composition along a New England transmission line corridor and its implications for other trophic levels. Forest Ecol Manag 327:231–239.
Wagner DL, Metzler KJ, Frye H (2019) Importance of transmission line corridors for conservation of native bees and other wildlife. Biol Conserv 235:147–156.
Westphal C, Steffan-Dewenter I, Tscharntke T (2003) Mass flowering crops enhance pollinator densities at a landscape scale. Ecol Lett 6:961–965.
Williams NM, Crone EE, Roulson TH, Minckley RL, Packer L, Potts SG (2010) Ecological and life-history traits predict bee species responses to environmental disturbances. Biol Conserv 143:2280–2291.
Wilson JS, Griswold T, Messinger OJ (2008) Sampling bee communities (Hymenoptera: Apiformes) in a desert landscape: are pan traps sufficient? J Kan Entomol Soc 81(3):288–300.
Winfree R, Griswold T, Kremen C (2007) Effect of human disturbance on wild bee communities in a forested ecosystem. Conserv Biol 21(1):213–223.
Yarborough D (2009) Wild blueberry culture in maine. Wild Blueberry Fact Sheet. No. 220, Bulletin No. 2088. https://extension.umaine.edu/blueberries/factsheets/production/wildblueberryculture-in-maine/
Acknowledgements
Thanks to J Eddy, S Kenney, R Jackson, C Thomes, and D Phipps for fieldwork assistance and to landowners for granting sampling permission. This work was funded by the USDA National Institute of Food and Agriculture—Specialty Crops Research Initiative [Grant Number 2011-51181-30673], Northeast Sustainable Agriculture and Research Education [Grant Number GNE14-076], the University of Maine and Maine Department of Inland Fisheries and Wildlife through the Cooperative Agreement with the U.S. Geological Survey Maine Cooperative Fish and Wildlife Research Unit. This project was partially supported by the USDA National Institute of Food and Agriculture, Hatch Project number ME0-50502 through the Maine Agricultural and Forest Experiment Station. This is Maine Agricultural and Forestry Experiment Station Journal Number 3884. This manuscript was improved with review by Dr. Kim Russell. At the time of publication, portions of data sets created in this study but omitted from the manuscript tables have limited availability (i.e., ongoing work, proprietary, or sensitive). Contact Brianne Du Clos for more information about availability of data sets. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Funding
Funding was provided by USDA National Institute of Food and Agriculture—Specialty Crops Research Initiative (2011-51181-30673), USDA National Institute of Food and Agriculture, Hatch through the Maine Agricultural and Forest Experiment Station (ME0-21505), University of Maine and Maine Department of Inland Fisheries and Wildlife through the Cooperative Agreement with the U.S. Geological Survey Maine Cooperative Fish and Wildlife Research Unit, and Northeast SARE (GNE14-076).
Author information
Authors and Affiliations
Contributions
BD, FD, and CL designed the experiment, BD conducted fieldwork and analyses and wrote the manuscript, FD and CL contributed to the analyses and manuscript and financially supported the research.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Du Clos, B., Drummond, F.A. & Loftin, C.S. Effects of an early mass-flowering crop on wild bee communities and traits in power line corridors vary with blooming plants and landscape context. Landsc Ecol 37, 2619–2634 (2022). https://doi.org/10.1007/s10980-022-01495-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-022-01495-9