Abstract
Context
A landscape is defined as a “system of ecosystems” and this is a model in which karst areas can easily be integrated. In karst areas, much of the connectivity between the units of the landscape is underground, with aquifers and caves forming a continuous layered tissue. However, underground environments are among the least studied landscapes on Earth because of limited accessibility and the difficulty of performing surveys.
Objectives
The aim of this paper is to provide a conceptual framework for applying principles of landscape ecology to research on karst environments.
Methods
By adapting the standard patch-corridor-matrix model to a 3d model, the main issues that need to be addressed were identified. These include identifying the main morphological (surface and underground) karst features; determining the landscape structure through its features, composition, and configuration; and developing adequate indices.
Results
The landscape spatial structure of different karst areas influences fundamental ecological functions and biodiversity patterns. Determining how structure, biodiversity, and functions relate reveals important insights into the functioning of karst systems. Emphasizing the provisioning of ecosystem services is essential in supporting the concept that karst regions are vital for human well-being because they host valuable resources and fundamental ecosystem processes. The paper discusses how this framework helps address anthropogenic impacts and conservation issues on karst.
Conclusions
The potential of applying a landscape approach to karst systems lies in developing models that provide ecological information relevant to understanding karst systems and understanding their implications for natural resources management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Karst environments are systems with peculiar geomorphological and hydrogeological characteristics and are considered some of Earth's most fragile natural systems (Brinkmann and Parise 2012). Karst areas represent approximately 15% of the world’s terrestrial zones, and they host valuable resources such as water, soil, and vegetation, providing habitats for several animal species, both epigean and hypogean, many of them being rare or endemic (Ford and Williams 2007; Williams 2008; Mammola et al. 2019). Simultaneously, almost 17% of the human population lives in karst areas, and 25% of them rely on groundwater (Ford and Williams 2007; Goldscheider et al. 2020), making these areas very valuable.
Terrestrial systems are generally represented as a mosaic of surface elements, but in karst areas the three-dimensional development of underground environments has a strong ecological relationship with the surface. In karst environments, a large part of the connectivity between the landscape units extends underground, with aquifer systems and empty spaces forming a continuous tissue developed on several levels (Helf and Olson 2017). A “system of ecosystems,” as the landscape is defined (Forman 1995a), is a model in which karst systems, and in particular underground karst, can easily be integrated. Accepting this model would allow the development of a holistic approach that involves rethinking the protection of caves, which should not be considered isolated environmental units, as defined in many environmental policies. For example, the EU Habitats Directive is the main European legislative framework for the conservation of habitats (Directive 1992/43/EEC) and governs the protection of caves as a distinct and self-contained habitat, distinguishing them from the rest of the karst landscape (“Caves not open to the public,” Natura 2000 code: 8310; “Fields of lava and natural excavations,” Natura 2000 code: 8320; “Submerged or partially submerged sea caves,” Natura 2000 code: 8330).
National or regional cave registers are a typical tool used to designate caves and are sometimes available as online databases or publications (see, e.g., Price 2014; Ferrario and Tognini 2016). Cave registers are usually systematic collections of information about the location and characteristics of caves, and they are the basis for protection measures in the territory. The term “cave,” however, is variably defined in different countries and by different authors. The International Union of Speleology (https://www.uis-speleo.org/) defines caves eligible to be cataloged in official registers as cavities with a horizontal or vertical development exceeding 5 m and a planimetric development /entrance width ratio > 1, provided they are large enough for human beings to enter. This is a human-based, or cavers’ definition; from a geological point of view, caves are connected voids formed by different “underground processes” (excluding rock primary porosity), whatever their dimensions. Caves can therefore be defined by their genesis (i.e., created by mechanical processes such as collapse or erosion, by chemical dissolution, by volcanic processes, etc.). Despite these differences, the common theme linking the various kinds of cavities is their interest to human explorers and their use as habitat by cave-adapted organisms (White and Culver 2011). Whatever definition of caves is adopted, considering caves as “single elements” is insufficient for their protection, as this hampers the capacity to implement effective conservation of these environments and associated resources.
It has sometimes been assumed that caves are isolated elements because populations of cave-adapted organisms can be extremely isolated (Culver 1970; Snowman et al. 2010; Balogh et al. 2020), although there is growing evidence of extensive gene flow between karst systems (Buhay and Crandall 2005). Cave entrances are critical for human access, they typically occur as a chance intersection of an evolving underground environment with the surface (Culver and Pipan 2019) and represent only a small portion of a cave system. In fact, cave entrances can be too small for human access or be absent. As an extreme example, caves without entrances include the Scot Hollow Cave in West Virginia (Lane et al. 2018) and the Pestera Movile Cave in Romania (Sarbu et al. 2019), and many other caves with no entrance have been discovered by drilling or mining activities. The network of fissures, joints, and bedding planes, the epikarst, the interstitial habitats, the shallow subterranean habitats, and the “milieu souterrain superficiel” (see Box 1 Glossary) should be considered together with caves and other underground voids, as they contribute to the complex system of a karst landscape.
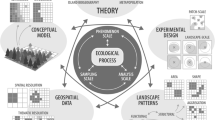
Given the vulnerability of these environments and the complex interconnections between karst landscape elements, it is crucial to shift the attention to the landscape level. This paper discusses how landscape ecology can contribute to the study and conservation of karst areas, paying specific attention to the underground domain. It addresses the classification of karst landscape elements and how landscape metrics could be further developed for a better description of karst landscapes and considers the fundamental aspect of the relationships between landscape structure, biodiversity, and ecosystem functioning. Finally, it discusses the ecosystem services of karst areas and the implications for karst conservation.
Applying principles of landscape ecology to karst environments
Defining elements, mosaics and spatial patterns
A first step in describing a landscape is to identify its elements (Zonneveld 1989; Table 1). Karst landscapes occur when dissolution is the primary agent modeling the landscape (Culver and Pipan 2019). Because of dissolution, these landscapes have distinct features such as caves, sinkholes, springs, blind and dry valleys, and many others (Fig. 1, Box 1). Although karst elements are mainly created by chemical processes of rock dissolution, physical, biological, and microbiological processes also contribute to karst evolution. Indeed, karst environments are not the passive result of a reaction between water and rock, but they are the product of dynamic interactions between rock and a continuous flux of energy and matter in and out of caves (water, air, nutrients, etc.) at varying scales of impact. When describing a karst area, each element in the landscape can be characterized by recording specific features such as type, size, shape, origin, location, and function. The characterization of elements and their location in space leads to the definition of a mosaic of elements composing the landscape. By characterizing the karst landscape as an arrangement of various elements, fundamental landscape properties can be determined, such as composition, diversity of patch types, spatial configuration, fractal dimension, and arrangement complexity (Fig. 1). This helps describe landscape and elements patterns, scale, connectivity, networks, circuitry, or mesh size (McGarigal 2014), which are important features when analyzing landscape-scale ecological processes in an environment. For example, water drainage in karst is affected by several elements in the landscape, such as topographical features, water and topographic gradients, characteristics of input in the catchment area (diffuse/concentrated inflow) and the output zone (springs), and the characteristics and development of caves systems. Species dispersal is another landscape-scale process affected by the connectivity of elements and the presence and position of barriers (Verhoeven et al. 2017). Ecosystems are not isolated systems and cannot be understood without considering the flow of energy and material across their boundaries. Considering ecosystems as “open” systems requires an understanding of how mosaics of ecosystems interact and are spatially organized to affect ecological exchanges and ecosystem processes.
Scale
The identification of the appropriate scale of analysis is a core question in subterranean biology. The theory of scale and hierarchy is a key framework for understanding pattern-process relationships and became the basis for landscape ecology (Turner et al. 2001; Cushman et al. 2010), but it has rarely been applied to karst environments. Most studies focus on fine-scale features such as springs or caves (Herrando-Pea et al. 2008) and only rarely consider the whole karst basin as the scale of analysis. The relevant scale depends on the research aims and the system itself, as different problems require distinct scales of analysis and a multi-scale method is often required. Cave or microhabitat approaches may be useful for answering questions related to specific fauna requirements or adaptation (e.g., Ficetola et al. 2018), but an examination at the drainage system scale is required to address hydrological and macroecological issues and to understand how the links between multiple elements determine the processes occurring across the whole system.
Researchers in subterranean biology have been slow to take up the conceptual framework of the ecosystem (Odum 1953) mainly because of uncertainty related to the definition of size, inputs, and outputs of the subterranean ecosystems (Culver and Pipan 2019). This barrier has been partially overcome by studies showing the importance of drainage system-level analyses (Rouch 1977; Simon et al. 2007; Schneider et al. 2011). In a pioneering study, Gibert (1986) estimated the hydrological balance of an entire karst basin in France by reporting water evapotranspiration, runoff, infiltration, and output as a percentage of precipitation in the study area, revealing that infiltration was more than twice the surface runoff and was related to the ratio of basin covered by soluble or insoluble rock. He also estimated the yearly flux of components such as organic matter, indicating the relationship between the organic carbon entering and leaving the system (Gibert 1986). Nevertheless, studies assessing the flow across the whole drainage system remain scarce (examples include Jones 1997, on the karst hydrologic budget and Simon et al. 2007, on organic carbon flow).
Three-dimensional spatial metrics and their representation
The landscape is modeled as a patch-corridor-matrix planimetric surface (Forman 1995b), and it is therefore difficult to fit aspects of three-dimensional patterns into this concept. The necessity of considering a third vertical dimension in landscape analysis has been often highlighted (Hoechstetter et al. 2008; Wu et al. 2017). The two-dimensional representation limits the analysis because it does not allow the inclusion of ecologically meaningful structures, with a consequent loss of valuable information about landscape heterogeneity. Considering the three-dimensional geometries of areas opens up perspectives for a more realistic representation of structure elements (Hoechstetter et al. 2006). However, current studies mainly refer to surface terrain features such as roughness, landform, relief, or the vertical structure of vegetation (topography- or elevation-related features) (Dorner et al. 2002; Mücke et al. 2010). In karst areas, key information may be lost due to an inability to describe the landscape structure with appropriate three-dimensional metrics, and this is a particular problem for underground environments. For example, a flooded gallery should be characterized not just by its length and width but also by its height, sinuosity (i.e., curvilinear, rectilinear, ramiform—see Palmer 2012), volume (which may stock water), and wall roughness, and by the sediments and deposits that totally or partly choke it. Furthermore, elements of the underground environment occur at a given depth, and a z-value associated with the x and y coordinates is needed to determine their location in the space. Hence, it is essential to develop adequate metrics—which do not yet exist for karst areas—to capture the 3D-features of the elements. These metrics would provide a more realistic assessment of the landscape’s spatial structure, thus assisting a better understanding of karst patterns and processes (Stupariu et al. 2010). Subterranean environments require more sophisticated analysis methods than the 3D-landscape characterizations of the Earth’s surface performed using remote sensing (Blaschke et al. 2004; Hoechstetter et al. 2008). Remote sensing is impossible in the subterranean domain, and new techniques are needed to overcome this limitation. Some of these have been tested, such as geophysical exploration combined with 3D laser scanning (Ba et al. 2020) or electrical resistivity tomography (Sono et al. 2020), but these techniques are generally expensive and need refinement before they can be broadly adopted.
Advantages and limitations in the study of underground environments
Karst landscapes have some peculiarities that assist or limit the investigation of the systems themselves. Factors making underground environments easier to study than their surface counterparts include the stability of many features and the fact that biotic characteristics are determined by a limited number of factors, such as distance from the surface and water availability. The distance from the surface determines temperature, light, and nutrient availability, while water is a primary driver for the occurrence of organisms and determines the movements of both biotic (species) and abiotic (sediments, nutrients) masses (Schneider et al. 2011; Lunghi et al. 2017; Ficetola et al. 2018). Moreover, underground environments have species-poor biotic communities because only a limited number of specialized species can thrive in these extreme environments (Romero 2009), which can make their description easier. Consequently, trophic networks are simpler (Gibert and Deharveng 2002), with primary producers and herbivores often missing and decomposers, predators, or parasites well represented (Mohr and Poulson 1966).
Limitations in investigating underground environments are related to difficulties in exploring, mapping, and collecting accurate data on geology, hydrology (Jeannin et al. 2007), and biodiversity (Ficetola et al. 2019). Mapping these environments is the first prerequisite for a reliable representation of the landscape and its characteristics. Direct explorations of underground realm may be costly and challenging because they require sustained efforts and complex organizational structures to support the activities and can be time-consuming, with even caves of a modest size requiring multiple trips (White 2019; see Box 2 Cave surveying).
In addition to mapping the underground elements, correlating their elevation with aerial photographs and using elevation controls such as geographic surface benchmarks help build the final map projected onto topographic overlays (Kambesis 2007). Direct mapping of underground environments also allows the recording of important information such as groundwater drainage, water flow rate, streams confluence, and cave morphologies and the occurrence of animals, fossils, speleothems, cave minerals, and sediments, allowing a greater understanding of cave origins and evolution, which is essential for underground systems investigation.
Indirect methods can help underground mapping and partially overcome surveying difficulties. For example, information on hydrology can be derived from dye-tracing techniques: a non-toxic dye (typically a fluorescent dye such as Tinopal or Uranine) is injected into a sink or a cave stream, and its arrival time and concentration are recorded at specific output points (springs). This technique, combined with geological and hydrological data, allows the catchment area and the output points of a karst system to be defined, together with throughput rates and recharge and storage amounts, to evaluate the system’s vulnerability to pollutants. Indirect investigations may also involve air tracing, air pressure, temperature, and flow measurements in multi-entrance caves systems, to evaluate fluxes throughout the cave and energy exchanges with the surface. Indirect methods usually require physical cave exploration for sampling. Integrating direct exploration and indirect approaches may be helpful, and thus the possibility of using landscape surface characteristics to infer underground structures needs to be investigated. For example, spring outflow hydrograph and chemograph analysis, which correlates discharge, temperature, and water chemistry variations of a karst spring with input/rainfall in the catchment area, can help evaluate the degree of karstification and the main characteristics of a karst drainage system (e.g., large karst conduits drainage, i.e., well-karstified aquifers, versus diffuse drainage through poorly karstified joints, i.e., fissured aquifers) (Ford and Williams 2007).
Integrating landscape ecology and assessment of ecosystem services in the study of karst areas
Landscape ecology is based on the principle that ecosystem composition, structure, and function partially depend on the spatial and temporal context of the ecosystem (i.e., its landscape context) so that ecological observations at any location are affected by its boundary conditions—that is, by what is around. This approach has been applied to various natural and anthropogenic landscapes, from tropical regions to agricultural areas and urban areas to deep oceans (Naveh and Lieberman 2013; Young et al. 2017). This paper proposes a framework to integrate landscape ecology principles (elements, mosaics, patterns, disturbances) with the study of karst areas, particularly the subterranean dimension (Fig. 2).
Biodiversity in karst landscapes
Ecological and evolutionary factors determine biodiversity in karst environments. Karst age, the cave’s origin, past climate events (e.g., glaciation), and biogeographic processes have shaped the distribution of organisms, and the interplay between these factors is complex (see Mammola et al. 2015). The current distribution of organisms in underground environments largely depends on nutrient availability, water supply, light, or niche differentiation (Christman and Culver 2001; Lunghi et al. 2014). Subterranean species generally have narrow distribution ranges, which results in high spatial turnover in species composition across regions, with clusters of spatially structured populations that may evolve into new species (Zagmajster et al. 2018; Ficetola et al. 2020). The high endemism levels are related to the fragmentation of the subterranean habitats in karst landscapes and the long-term persistence and relative stability of subterranean environments (Gibert and Deharveng 2002). In this context, the analysis of habitat patch distribution can illuminate the evolutionary processes caused by the isolation of populations (Chiari et al. 2012).
Much of underground biodiversity is yet to be described (Manenti and Pezzoli 2019; Ficetola et al. 2019). In addition to identifying species, the modeling of their distribution in subterranean environments is a further task of primary importance (Mammola 2018). How organisms interact with the landscape depends on their needs and on the characteristics of the landscape itself. Landscape elements can represent both barriers and corridors for movement. Some cave organisms need to live in underground habitats for their entire life cycle (troglobites and stygobites), while others enter or live in caves for specific needs (troglophiles). The movements of these organisms are determined by landscape characteristics. The composition of the matrix and how patches are arranged within the space may determine isolation (Chiari et al. 2012) or aggregation (Biswas 2010) of animal populations, and this influences genetic exchanges and interspecific interactions, which have consequences for the survival of populations. It has been observed that the extinction of cave-dwelling metapopulations depends on the complexity of the network, particularly on the size and spatial arrangement of habitat patches, together with species movement (Campbell Grant 2011).
There is a growing interest in relationships between subterranean habitats and biodiversity (Zagmajster et al. 2018). The diversity of subterranean species is determined by the interplay between productivity, habitat availability, spatial heterogeneity, energy production, and climate suitability (Eme et al. 2015). The overall diversity tends to be higher in regions characterized by high surface productivity (Culver et al. 2006) and high density of caves (this can be an effect of higher habitat availability, or of better sampling) (Christman and Culver 2001; Christman and Zagmajster 2012; Ficetola et al. 2014; Niemiller et al. 2021). However, additional factors can increase the richness of subterranean species, including habitat heterogeneity (Sket et al. 2004) and regional species richness (Malard et al. 2009), highlighting the importance of landscape context in biodiversity patterns. Nevertheless, biodiversity patterns are also influenced by subterranean dispersal (Culver and Pipan 2019), which is determined by the arrangement and types of landscape elements. Understanding the connectivity of landscapes requires data on specific dispersal behaviors and pathways in subterranean systems that are often lacking. For large animals such as bats, the general capacity of permeation in a landscape is known, and barriers are readily detectable (Furey and Racey 2016), but for terrestrial arthropods, water-dwelling animals and microorganisms, there is not enough knowledge available.
Landscape structure and ecosystem function in karst environments
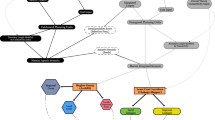
The relationship between ecological functions and spatial patterns is a key theme of landscape ecology that helps inform land management practices. This approach may also shed light on the functioning of karst landscapes. In karst landscapes, material and energy flows follow complex pathways that are not always fully understood. Generally, water occurs at the surface and enters the subterranean system at the rock-soil interface, following vertical and horizontal pathways (e.g., Helf and Olson 2017). The biophysical structure of habitats influences many ecological processes. Water flow, water storage, rock erosion and dissolution, speleothems and sediment deposition, organic matter accumulation, nutrient flow, rate of photosynthesis close to the entrances, air-flow, organisms’ niche availability, and organisms’ movements (Lunghi et al. 2017) are all examples of these processes. A general pattern of the source of energy and its destination in subterranean habitats is presented in Fig. 3. Temporal and spatial dynamics are also fundamental factors (Turner 1990). For example, flooding dynamics determine community changes and affect the overall flux of materials such as sediments or organic matter (Simon and Benfield 2001). Moreover, karst landscapes are formed by geochemical processes that are generally ongoing because of water flow, and the morphology of the rocks is therefore reshaped continuously as a result of continuous dissolution, new rock formation, or changes in hydrological regimes.

source: Culver and Pipan 2019)
Classification of sources and origins of energy and their destinations in subterranean environments (
Biodiversity influences ecosystem functioning and determines many fundamental ecosystem processes, including water purification or nutrient cycling (Mace et al. 2012). However, it is still largely unknown how biodiversity sustains ecosystem functions and services in karst areas. Certainly, functional diversity is central to understand ecosystems functioning. It can be measured by the diversity of functional traits of a community and is of primary ecological importance because it influences ecosystem dynamics, stability, productivity, nutrient balance, and other aspects of ecosystem functioning (Tilman 2001; Cadotte et al. 2011). Functional diversity can explain variation in ecosystem function even when species diversity does not, thus offering crucial insights (Cadotte et al. 2011). The functional diversity of subterranean environments has rarely been studied, but it can reveal unexpected patterns. Confounding the expectation of lower functional diversity in such a harsh environment, Fernandes et al. (2016) demonstrated that cave isopods (Oniscidea) show higher functional diversity compared to surface taxa, possibly because they find more suitable conditions, including lower predation pressures and greater water availability, and this promotes their distribution and diversification. Knowing the functional diversity of organisms is fundamental because it is one of the most effective predictors of ecosystem functioning (Song et al. 2014). The growing availability of theoretical and technical frameworks and the development of trait databases for animals and microorganisms enable a greater understanding of underground functional diversity (Moretti et al. 2017; Nguyen et al. 2016; White et al. 2020), but there is still a significant gap in knowledge concerning the traits and ecology of cave organisms.
Karst ecosystem services
Ecosystem services (ES) are defined as the goods and services deriving from ecosystems that contribute, directly or indirectly, to human well-being (MEA 2005). The recognition, evaluation, and monitoring of these benefits may offer new empirical and conceptual tools that can be combined with more traditional approaches (e.g., the establishment of protected areas and endangered species protection) to support the management of natural systems and promote sustainable human development (Müller et al. 2010). For these reasons, an integrated assessment of the ES of karst environments should be a primary goal for conservation. The almost total absence of such studies makes such assessments urgent (however, see ES approach to karst areas by Žujo and Marinšek 2012; Quine et al. 2017; Wang et al. 2019). This paper provides a list of potential ES provided by karst areas, indicated separately for surface and underground environments (Table 2). Underground karst supports many services, providing water and genetic material from species, regulating water fluxes and chemical and biological conditions, transforming biochemical and physical inputs, and regulating and maintaining abiotic and biotic factors overall. Other important services deriving from underground environments are related to the cultural dimension of humans and, although undervalued, cultural ecosystem services are essential for human health and well-being (Bratman et al. 2019). Cultural benefits derived from the exploitation of natural environments include outdoor and recreational activities and the aesthetic appeal of calcareous forms, fossils, and underground spaces. Furthermore, cave settings encourage social gatherings and human interactions both for sport and scientific purposes, and interactions with the natural environment shape people’s sense of personal identity. The physical, mental, and cultural enrichment that can be achieved in caves makes them some of the most intriguing environments on Earth (Fig. 4).
a Surface limestone landscape with typical karst morphology (Monte Grignone, Italy; photo by C. Canedoli); b Underground environment with narrow passages and calcite speleothems (Antro delle Gallerie, Valganna, Italy; photo by D. Corengia); c Cave diving exploration in a flooded passage about 300 m from the cave entrance (Sorgente del Torregione, Italy; photo by D. Corengia); d Cave fauna (gen. Polydesmus, photo by D. Corengia); e Cave polluted with garbage (grotta della Selva, Italy; photo by F. Merisio); f Tourism activity in caves (Buco del corno, Italy; photo by A. Ferrario)
Several studies have reported that the evaluation of ES is an effective practical strategy for environment conservation. It is used to prioritize key biodiversity areas for conservation (Shrestha et al. 2021), to identify conflicts between nature conservation and human societies (Setälä et al. 2014; Bezák et al. 2017), and to inform conservation planning (Mitchell et al. 2021). These insights can be included in strategic environmental assessments (tools supporting decision-making to make sustainable territorial plans; Semeraro et al. 2021) or can even be integrated into economic decision-making (Banerjee et al. 2020; Yang et al. 2020). Finally, the ES approach ensures that the complex relationships between nature and humans are clearly understood and explicitly stated, promoting solutions that balance the existence of human societies with nature (Luck et al. 2009; Beaumont et al. 2017). However, the current measures of services fail to capture adequately the benefits humans derive from karst areas.
Human impact and opportunities for karst conservation
Landscape dynamics occur over temporal and spatial scales: evolution and geological processes act over long timespans, colonization and reproductive processes act over medium to short timescales, and local disturbance processes can have immediate consequences. Among local disturbances there is the human impact that can alter landscape context and biodiversity. Impacts on underground environments and processes include land use and land cover change, pollution of soil and water, water pumping, mining and quarrying exploitation, rock excavation for underground infrastructures, modifications of conditions of underground water drainage, and disturbance and poaching of fauna.
While human activities can foster conservation in the subterranean environment, they can also determine impacts if not correctly managed. For example, cave tourism and caving entail people entering caves, which are extremely fragile environments. Direct experience of subterranean environments enhances people’s awareness, ecological knowledge, and connection to nature, and this can result in respectful behavior and environmental stewardship. At the same time, tourists may negatively impact cave habitats by changing microhabitat availability (e.g., with light or the creation of pathways), continuous treading, the introduction of alien species, acoustic pollution, and direct disturbance resulting from people touching speleothems and animals (Mammola et al. 2017). Similarly, caving is fundamental for exploring the underground environment, recording biodiversity data or enhancing speleologists’ knowledge, and caving associations are often the first promoters of cave protection. However, this activity can be invasive if cavers’ behavior is not regulated. Luckily, most cavers and speleologists regulate their activities, abandoning explorations if there is a danger of damaging speleothems or preventing visits to bat-inhabited caves when the bats are hibernating or nursing. However, the trade-offs between cave exploitation by humans and cave protection can be complex and require careful evaluation.
Despite the close dependence humans on karst, protection policies are often absent, incomplete, or ineffective at the landscape scale. As karst systems are intrinsically fragile environments with high connectivity among their elements, they would benefit from a landscape conservation approach that goes beyond the conservation of single caves or single cave species. Local conservation actions are effective on small scale but do not prevent landscape-level threats. This shift in perspective from “the site” to “the site embedded in a landscape” has profound implications for management. Landscape knowledge is the fundamental requirement when defining conservation priorities and regulating activities that may influence the landscape, and this can only be achieved by undertaking comprehensive landscape studies. For example, the European Landscape Convention has established landscape quality objectives and consequent recommended actions that could serve as models for other countries (Déjeant-Pons 2006). It is important to include social perceptions of landscapes and manage trade-offs between human activities and karst protection, ensuring the safeguarding of both. This aspect is of particular importance, as strengthening relationships between populations and their surroundings underpins sustainable development (Makhzoumi et al. 2011).
Conclusion
The potential of applying a landscape approach to karst systems lies both in developing models that provide ecological information relevant to the understanding of karst systems (spatial heterogeneity, ecological connectivity, ecosystem functionality) and in understanding the possible implications for resource management. A landscape ecology approach enables an understanding of the dynamics of the karst regions and provides a rationale for improving their management and conservation. An adequate understanding of structure, biodiversity, and functioning of karstic systems and a greater awareness of their value through the quantification of benefits derived by humanity is of paramount importance for addressing the sustainable exploitation of the resources associated with them and promoting effective and large-scale conservation choices. The valorization of ecosystem services is here indicated as a way to implement karst protection for conservationists seeking to combine conservation and human development successfully.
References
Algeo K (2004) Mammoth Cave and the making of place. Southeast Geogr 44(1):27–47
Allocca V, Marzano E, Tramontano M, Celico F (2018) Environmental impact of cattle grazing on a karst aquifer in the southern Apennines (Italy): quantification through the grey water footprint. Ecol Ind 93:830–837
Ba X, Li L, Wang J, Zhang W, Fang Z, Sun S, Liu Z, Xiong Y (2020) Near-surface site investigation and imaging of karst cave using comprehensive geophysical and laser scanning: a case study in Shandong, China. Environ Earth Sci 79(12):1–14
Balogh A, Dixon GB, Ngo L, Zigler KS (2020) Testing the caves-as-islands model in two cave-obligate invertebrates with a genomic approach. BioRxiv 2020.04.08.032789
Banerjee O, Cicowiez M, Malek Ž, Verburg PH, Vargas R, Goodwin S (2020) The value of biodiversity in economic decision making: applying the IEEM+ ESM approach to conservation strategies in Colombia.
Beaumont NJ, Mongruel R, Hooper T (2017) Practical application of the Ecosystem Service Approach (ESA): lessons learned and recommendations for the future. Int J Biodivers Sci Ecosyst Serv Manag 13(3):68–78
Bezák P, Mederly P, Izakovičová Z, Špulerová J, Schleyer C (2017) Divergence and conflicts in landscape planning across spatial scales in Slovakia: an opportunity for an ecosystem services-based approach? Int J Biodivers Sci Ecosyst Serv Manag 13(2):119–135
Blaschke T, Tiede D, Heurich M (2004) 3D landscape metrics to modelling forest structure and diversity based on laser scanning data. Int Arch Photogram Remote Sens Spatial Inf Sci 36(8/W2):129–132
Biswas J (2010) Kotumsar Cave biodiversity: a review of cavernicoles and their troglobiotic traits. Biodivers Conserv 19(1):275
Bonsall C, Mlekuž D, Bartosiewicz L, Pickard C (2016) Early farming adaptations of the northeast Adriatic Karst. In: The origins and spread of domestic animals in Southwest Asia and Europe. Routledge, London, pp 145–160
Bratman GN, Anderson CB, Berman MG, Cochran B, De Vries S, Flanders J, Folke C, Frumkin H, Gross JJ, Hartig T, Kahn PH, Kuo M, Lawler JJ, Levin PS, Lindhal T, Meyer-Linderberg A, Mitchell R, Ouyang Z, Roe J, Scarlett L, Smith JR, Van den Bosch M, Wheeler BW, White MP, Zheng H, Daily GC (2019) Nature and mental health: an ecosystem service perspective. Sci Adv 5(7):eaax0903
Brinkmann R, Parise M (2012) Karst environments: problems, management, human impacts, and sustainability. An introduction to the special issue. J Cave Karst Stud 74(2):135–136
Buffetrille K (1998) Reflections on pilgrimages to sacred mountains, lakes and caves. In: McKay A (ed) Pilgrimage in Tibet. Routledge, London, pp 18–34
Buhay JE, Crandall KA (2005) Subterranean phylogeography of freshwater crayfishes shows extensive gene flow and surprisingly large population sizes. Mol Ecol 14(14):4259–4273
Cadotte MW, Carscadden K, Mirotchnick N (2011) Beyond species: functional diversity and the maintenance of ecological processes and services. J Appl Ecol 48(5):1079–1087
Campbell Grant EH (2011) Structural complexity, movement bias, and metapopulation extinction risk in dendritic ecological networks. J N Am Benthol Soc 30(1):252–258
Cao J, Hu B, Groves C, Huang F, Yang H, Zhang C (2016) Karst dynamic system and the carbon cycle. Z Geomorpholog Suppl Issues 60(2):35–55
Cao JH, Wu X, Huang F, Hu B, Groves C, Yang H, Zhang CL (2018) Global significance of the carbon cycle in the karst dynamic system: evidence from geological and ecological processes. China Geology 1(1):17–27
Chiari Y, Van Der Meijden A, Mucedda M, Lourenco JM, Hochkirch A, Veith M (2012) Phylogeography of Sardinian cave salamanders (genus Hydromantes) is mainly determined by geomorphology. PLoS ONE 7(3):e32332
Christman MC, Culver DC (2001) The relationship between cave biodiversity and available habitat. J Biogeogr 28(3):367–380
Christman MC, Zagmajster M (2012) Mapping subterranean biodiversity. In Encyclopedia of caves. Academic Press, New York, pp 474–481
Coxon C (2011) Agriculture and karst. In Karst management. Springer, Dordrecht, pp 103–138
Cushman SA, Evans JS, McGarigal K (2010) Landscape ecology: past, present, and future. In Spatial complexity, informatics, and wildlife conservation. Springer, Tokyo, pp 65–82
Culver DC (1970) Analysis of simple cave communities I. Caves as islands. Evolution, 463–474.
Culver DC, Pipan T (2014) Shallow subterranean habitats: ecology, evolution, and conservation. Oxford University Press
Culver DC, Pipan T (2019) The biology of caves and other subterranean habitats. Oxford University Press
Culver DC, Deharveng L, Bedos A, Lewis J, Madden J, Reddell R, Sket B, Trontelj P, White D (2006) The mid-latitude biodiversity ridge in terrestrial cave fauna. Ecography 29(1):120–128
Déjeant-Pons M (2006) The European landscape convention. Landsc Res 31(4):363–384
Dorner B, Lertzman K, Fall J (2002) Landscape pattern in topographically complex landscapes: issues and techniques for analysis. Landsc Ecol 17:729–743. https://doi.org/10.1023/A:1022944019665
Doorne S (2000) Caves, cultures and crowds: carrying capacity meets consumer sovereignty. J Sustain Tour 8(2):116–130
Eme D, Zagmajster M, Fišer C, Galassi D, Marmonier P, Stoch F, Cornu JF, Oberdorff T, Malard F (2015) Multi-causality and spatial non-stationarity in the determinants of groundwater crustacean diversity in Europe. Ecography 38(5):531–540
Ferrario A, Tognini P (2016) Catasto Speleologico Lombardo (Progetto Tu.Pa.Ca.), Federazione Speleologica Lombarda
Fernandes CS, Batalha MA, Bichuette ME (2016) Does the cave environment reduce functional diversity? PLoS ONE 11(3):e0151958
Ficetola GF, Cagnetta M, Padoa-Schioppa E, Quas A, Razzetti E, Sindaco R, Bonardi A (2014) Sampling bias inverts eco-geographical relationships in island reptiles. Glob Ecol Biogeogr 23:1303–1313
Ficetola GF, Lunghi E, Canedoli C, Padoa-Schioppa E, Pennati R, Manenti R (2018) Differences between microhabitat and broad-scale patterns of niche evolution in terrestrial salamanders. Sci Rep 8(1):1–12
Ficetola GF, Canedoli C, Stoch F (2019) The Racovitzan impediment and the hidden biodiversity of unexplored environments. Conserv Biol 33(1):214–216
Ficetola GF, Lunghi E, Manenti R (2020) Microhabitat analyses support relationships between niche breadth and range size when spatial autocorrelation is strong. Ecography 43(5):724–734
Ford DC, Williams P (2007) Karst hydrogeology and geomorphology. Wiley, Chichester, p 562
Forman RT (1995a) Some general principles of landscape and regional ecology. Landsc Ecol 10(3):133–142
Forman RTT (1995b) Land mosaics: the ecology of landscapes and regions. Cambridge University Press, Cambridge
Furey NM, Racey PA (2016) Conservation ecology of cave bats. In Bats in the anthropocene: conservation of bats in a changing world. Springer, Cham, pp 463–500
Gibert J, Deharveng L (2002) Subterranean ecosystems: a truncated functional biodiversity. Bioscience 52(6):473–481
Gibert J (1986) Ecologie d’un systeme kartsique jurassien. Hydrogéologie, dérive animale, transits de matièrese, dynamique de la population de Niphargus (Crustacé Amphipode). Mémoire De Biospéleologie 13:1–379
Goldscheider N, Chen Z, Auler AS, Bakalowicz M, Broda S, Drew D, Hartmann J, Jiang G, Moosdorf N, Stevanovic Z, veni G (2020) Global distribution of carbonate rocks and karst water resources. Hydrogeol J 28:1661–1677
Griebler C, Avramov M (2015) Groundwater ecosystem services: a review. Freshwater Science 34:355–367
Groves C, Groves D, Hawkins W (2015) An education is not all derived from textbooks: a century of interactions between Mammoth Cave and Western Kentucky University.
Groves C, Webb CJ, Turner AB, Bledsoe LA, Toomey RS, Carson B, Algeo K (2018) How is mammoth cave national park doing? The 2018 MACA natural resource condition assessment. In: GSA annual meeting in Indianapolis, Indiana, USA-2018. GSA.
Guerra AJT, Fullen MA, Jorge MCO, Bezerra JR, Shokr MS (2017) Slope processes, mass movement and soil erosion: a review. Pedosphere 27:27–41
Haines-Young R, Potschin-Young M (2018) Revision of the common international classification for ecosystem services (CICES V5. 1): a policy brief. One Ecosyst 3:e27108
Heinz B, Birk S, Liedl R, Geyer T, Straub KL, Andresen J, Bester K, Kappler A (2009) Water quality deterioration at a karst spring (Gallusquelle, Germany) due to combined sewer overflow: evidence of bacterial and micro-pollutant contamination. Environ Geol 57(4):797–808
Helf KL, Olson RA (2017) Subsurface aquatic ecology of mammoth cave. In: Mammoth cave. Springer, Cham, pp 209–226
Herrando-Peâ S, Baratti M, Messana G (2008) Subterranean ecological research and multivariate statistics: a review (1945±2006). J Cave Karst Stud 70(2):120–128
Hoechstetter S, Thinh NX, Walz U (2006) 3D-indices for the analysis of spatial patterns of landscape structure. Proc InterCarto InterGIS 12:108–118
Hoechstetter S, Walz U, Thinh NX (2008) Effects of topography and surface roughness in analyses of landscape structure-a proposal to modify the existing set of landscape metrics. Landsc Online 3:1–14
Huang Q, Cai Y, Xing X (2008) Rocky desertification, antidesertification, and sustainable development in the karst mountain region of Southwest China. Ambio 37(5):390–392
Iannace G, Trematerra A (2014) The acoustics of the caves. Appl Acoust 86:42–46
Jeannin PY, Groves C, Häuselmann P (2007) Speleological investigations. In: Goldscheider N, Drew D (eds) Methods in karst hydrogeology. Taylor & Francis, New York, pp 25–44
Jianhua C, Daoxian Y, Groves C, Fen H, Hui Y, Qian L (2012) Carbon fluxes and sinks: the consumption of atmospheric and soil CO2 by carbonate rock dissolution. Acta Geol Sin Engl Edn 86(4):963–972
Jones WK (1997) Karst hydrology Atlas of West Virginia: Karst Waters Institute Special Publication 4, Charles Town, WV
Juan C, Guzik MT, Jaume D, Cooper SJ (2010) Evolution in caves: Darwin’s “wrecks of ancient life” in the molecular era. Mol Ecol 19:3865–3880
Juberthie C, Delay B (1981) Ecological and biological implications of the existence of a “superficial underground compartment”. In: Beck BF (ed) Proceedings of the 8th international congress of speleology, Bowling Green, KY, Georgia, USA 1, pp 203–206
Kambesis PN (2007) The importance of cave exploration to scientific research. J Cave Karst Stud 69(1):46–58
Kim SS, Kim M, Park J, Guo Y (2008) Cave tourism: tourists’ characteristics, motivations to visit, and the segmentation of their behavior. Asia Pac J Tourism Res 13(3):299–318
Kunz TH, Braun de Torrez E, Bauer D, Lobova T, Fleming TH (2011) Ecosystem services provided by bats. Ann N Y Acad Sci 1223:1–38
Lane SB, Bishop MR, Dore MJ, Sasowsky ID (2018) Scott Hollow Cave. In Caves and Karst of the Greenbrier Valley in West Virginia. Springer, Cham, pp 339–357
Luck GW, Chan KM, Fay JP (2009) Protecting ecosystem services and biodiversity in the world’s watersheds. Conserv Lett 2(4):179–188
Lunghi E, Manenti R, Ficetola GF (2014) Do cave features affect underground habitat exploitation by non-troglobite species? Acta Oecologica 55:29–35
Lunghi E, Manenti R, Ficetola GF (2017) Cave features, seasonality and subterranean distribution of non-obligate cave dwellers. PeerJ 5:e3169
Mace GM, Norris K, Fitter AH (2012) Biodiversity and ecosystem services: a multilayered relationship. Trends Ecol Evol 27(1):19–26
Magnabosco C, Lin LH, Dong H, Bomberg M, Ghiorse W, Stan-Lotter H, Pedersen K, Kieft TL, van Heerden E, Onstott TC (2018) The biomass and biodiversity of the continental subsurface. Nat Geosci 11:707
Malard F, Boutin C, Camacho AI, Ferreira D, Michel G, Sket B, Stoch F (2009) Diversity patterns of stygobiotic crustaceans across multiple spatial scales in Europe. Freshw Biol 54(4):756–776
Makhzoumi J, Egoz S, Pungetti G (2011) The right to landscape: contesting landscape and human rights. Ashgate Publishing Ltd, New York
Mammola S (2018) Finding answers in the dark: Caves as models in ecology fifty years after Poulson and White. Ecography 41:1–21
Mammola S, Isaia M, Arnedo MA (2015) Alpine endemic spiders shed light on the origin and evolution of subterranean species. PeerJ 3:e1384
Mammola S, Di Piazza S, Ziotti M, Badino G, Marco I (2017) Human-induced alterations of the mycobiota in an alpine show cave (Italy, SW-Alps). Acta Carsologica, 46(1).
Mammola S, Cardoso P, Culver DC, Deharveng L, Ferreira RL, Fišer C, Galassi DMP, Griebler C, hales S, Humphreys WF, Isaia M, Malard F, Martinez A, Moldovan OT, Niemiller ML, pavlek M, Reboleira ASPS, Souza-Silva M, Teeling EC, Wynne JJ, Zagmajster M (2019) Scientists’ warning on the conservation of subterranean ecosystems. Bioscience 69(8):641–650
Manenti R, Pezzoli E (2019) Think of what lies below, not only of what is visible above, or: a comprehensive zoological study of invertebrate communities of spring habitats. Eur Zool J 86(1):272–279
McGarigal K (2014) Landscape pattern metrics. Wiley StatsRef: Statistics Reference Online
Millennium Ecosystem Assessment, MEA, (2005) Ecosystems and human well-being, vol 5. Island Press, Washington, DC
Medellín RA, Wiederholt R, Lopez-Hoffman L (2017) Conservation relevance of bat caves for biodiversity and ecosystem services. Biol Conserv 211:45–50
Mitchell MG, Schuster R, Jacob AL, Hanna DE, Dallaire CO, Raudsepp-Hearne C, Bennet EM, Lehner B, Chan KMA (2021) Identifying key ecosystem service providing areas to inform national-scale conservation planning. Environ Res Lett 16(1):014038
Mohr CE, Poulson TL (1966) The life of the cave. MacGraw-Hill, New York
Moretti M, Dias ATC, De Bello F, Altermatt F, Chown SL, Azacrate FM, Bell JR, Fournier B, Hedde M, Hortal J, Ibanez S, Ockinger E, Sousa JP, Ellers J, Berg M (2017) Rothamsted repository download. Funct Ecol 31:558–567
Mücke W, Hollaus M, Prinz M (2010) Derivation of 3D landscape metrics from airborne laser scanning data. In: Silvilaser 2010.
Müller F, de Groot R, Willemen L (2010) Ecosystem services at the landscape scale: the need for integrative approaches. Landsc Online 23:1–11
Naveh Z, Lieberman AS (2013) Landscape ecology: theory and application. Springer Science & Business Media, Berlin
Nguyen NH, Song Z, Bates ST, Branco S, Tedersoo L, Menke J, Schilling JS, Kennedy PG (2016) FUNGuild: an open annotation tool for parsing fungal community datasets by ecological guild. Fungal Ecol 20:241–248
Niemiller ML, Helf K, Toomey RS (2021) Mammoth cave: a hotspot of subterranean biodiversity in the United States. Diversity 13(8):373
Odum HT (1953) Fundamentals of Ecology. W.B. Saunders Company Ltd, Philadelphia
Palmer AN (2012) Passage growth and development. In: White WB, Culver DC (eds) Encyclopedia of caves, 2nd edn. Elsevier/Academic Press, Amsterdam, The Netherdlands, pp 598–603
Parise M (2011) Some considerations on show cave management issues in Southern Italy. In Karst management. Springer, Dordrecht, pp 159–167
Price L, (2014) Caves and Karst of Peninsular Malaysia: a register. In: Berliner Höhlenkundliche Berichte, vol 54. Publisher Speläoclub, Berlin
Pronk M, Goldscheider N, Zopfi J (2009) Microbial communities in karst groundwater and their potential use for biomonitoring. Hydrogeol J 17:37–48
Quine T, Guo D, Green SM, Tu C, Hartley I, Zhang X, Dungait J, Wen X, Song Z, Liu H, Buss H, Barrows T, Evershed R, Johnes P, Meersmans J (2017) Ecosystem service delivery in Karst landscapes: anthropogenic perturbation and recovery. Acta Geochim 36(3):416–420
Reboleira AS, Borges PAV, Gonçalves F, Serrano A, Oromí P (2011) The subterranean fauna of a biodiversity hotspot region - Portugal: an overview and its conservation. Int J Speleol 40(1):23–37
Riddle MR, Aspiras AC, Gaudenz K, Peuß R, Sung JY, Martineau B, Peavey M, Box AC, Tabin JA, McGaugh S, Borowsky R, Tabin Cj, Rohner N (2018) Insulin resistance in cavefish as an adaptation to a nutrient-limited environment. Nature 555(7698):647–651
Robert E (2017) The role of the cave in the expression of prehistoric societies. Quatern Int 432:59–65
Romero A (2009) Cave biology: life in darkness. Cambridge University Press, Cambridge
Rouch R (1977) Considérations sur l’écosystème karstique. Compte Rendu Acad Sci, Paris 284:1101–1103
Safarabadi A, Shahzeidi SS (2015) Tourism silence in geomorphosites: a case study of Ali-Sadr Cave (Hamadan, Iran). GeoJournal Tourism Geosites 21(1):49–60
Sarbu SM, Lascu C, Brad T (2019) Dobrogea: movile cave. In Cave and Karst systems of Romania. Springer, Cham, pp 429–436
Schneider K, Christman MC, Fagan WF (2011) The influence of resource subsidies on cave invertebrates: results from an ecosystem-level manipulation experiment. Ecology 92(3):765–776
Semeraro T, Radicchio B, Medagli P, Arzeni S, Turco A, Geneletti D (2021) Integration of ecosystem services in strategic environmental assessment of a Peri-urban development plan. Sustainability 13(1):122
Setälä H, Bardgett RD, Birkhofer K, Brady M, Byrne L, De Ruiter PC, de Vries FT, Gardi C, Hedlund K, hemerik L, Hotes S, Liiri M, Mortimer SR, Pavao-Zuckerman M, Pouyat R, tsiafouli M, van der Putter WH (2014) Urban and agricultural soils: conflicts and trade-offs in the optimization of ecosystem services. Urban Ecosyst 17(1):239–253
Shrestha M, Piman T, Grünbühel C (2021) Prioritizing key biodiversity areas for conservation based on threats and ecosystem services using participatory and GIS-based modeling in Chindwin River Basin, Myanmar. Ecosyst Serv 48:101244
Simon KS, Benfield EF (2001) Leaf and wood breakdown in cave streams. J N Am Benthol Soc 20(4):550–563
Simon KS, Pipan T, Culver DC (2007) A conceptual model of the flow and distribution of organic carbon in caves. J Cave Karst Stud 69(2):279–284
Siebert C, Rosenthal P, Möller P, Rödiger T, Meiler M (2012) The hydrochemical identification of groundwater flowing to the Bet She’an-Harod multiaquifer system (Lower Jordan Valley) by rare earth elements, yttrium, stable isotopes (H, O) and Tritium. Appl Geochem 27:703
Sket B, Paragamian K, Trontelj P (2004) A census of the obligate subterranean fauna of the Balkan Peninsula. In Balkan biodiversity. Springer, Dordrecht, pp 309–322
Smith D (2004) Beyond the cave: Lascaux and the prehistoric in post-war French culture. Fr Stud 58(2):219–232
Snowman CV, Zigler KS, Hedin M (2010) Caves as islands: mitochondrial phylogeography of the cave-obligate spider species Nesticus barri (Araneae: Nesticidae). J Arachnol 38(1):49–56
Soares D, Niemiller ML (2013) Sensory adaptations of fishes to subterranean environments. Bioscience 63:274–283
Song Y, Wang P, Li G, Zhou D (2014) Relationships between functional diversity and ecosystem functioning: a review. Acta Ecol Sin 34(2):85–91
Sono P, Nthaba B, Shemang EM, Kgosidintsi B, Seane T (2020) An integrated use of induced polarization and electrical resistivity imaging methods to delineate zones of potential gold mineralization in the Phitshane Molopo area. Southeast Botswana. J Afr Earth Sci 174:104060
Stokes T, Griffiths P, Ramsey C (2010) Karst geomorphology, hydrology, and management. Compend Forest Hydrol Geomorphol Br Columb 66:373
Stupariu MS, Pàtru-Stupariu IG, Cuculici R (2010) Geometric approaches to computing 3D-landscape metrics. Landsc Online 24:1–12
Tilman D (2001) Functional diversity. Encyclopedia Biodivers 3(1):109–120
Turner MG (1990) Spatial and temporal analysis of landscape patterns. Landsc Ecol 4(1):21–30
Turner MG, Gardner RH, O'neill RV, O'Neill RV (2001) Landscape ecology in theory and practice (Vol. 401). Springer New York.
Verhoeven JT, Beltman B, Janssen R, Soons MB (2017) Delineating landscape-scale processes of hydrology and plant dispersal for species-rich fen conservation: the Operational Landscape Unit approach. Wetlands Ecol Manage 25(6):761–774
Wang KL, Zhang CH, Chen HS, Yue YM, Zhang W, Zhang MY, Qi XK, Fu ZY (2019) Karst landscapes of China: patterns, ecosystem processes and services. Landsc Ecol 34:2743–2763
White WB (2019) Exploration of caves - general. In Encyclopedia of caves. Academic Press, New York, pp 407–413
White WB, Culver DC (2011) Encyclopedia of caves. Academic Press, New York
White HJ, León-Sánchez L, Burton VJ, Cameron EK, Caruso T, Cunha L, Dirilgen T, jutrburg SD, Kelly R, Kumaresan D, Ochoa-Hueso R, ordones A, Phillips HRP, Prieto I, Schimdt O, Caplat P (2020) Methods and approaches to advance soil macroecology. Glob Ecol Biogeogr 29(10):1674–1690
Williams P (2008) World heritage Caves and Karst: a thematic study. IUCN World Heritage Studies 2:50
Wu Q, Guo F, Li H, Kang J (2017) Measuring landscape pattern in three dimensional space. Landsc Urban Plan 167:49–59
Yang H, Lupi F, Zhang J, Liu J (2020) Hidden cost of conservation: a demonstration using losses from human-wildlife conflicts under a payments for ecosystem services program. Ecol Econ 169:106462
Yoshizawa M, Settle A, Hermosura M, Tuttle L, Centraro N, Passow CN, McGaugh SE (2018) The evolution of a series of behavioral traits is associated with autism-risk genes in cavefish. BMC Evol Biol 18:89
Young MA, Wedding LM, Carr MH (2017) Applying landscape ecology for the design and evaluation of marine protected area networks. Seasc Ecol, 429–462
Zagmajster M, Malard F, Eme D, Culver DC (2018) Subterranean biodiversity patterns from global to regional scales. In Cave ecology. Springer, Cham, pp 195–227
Zhang W, Chen HS, Wang KL (2007) The heterogeneity and its influencing factors of soil nutrients in peak-cluster depression areas of karst region. Agric Sci China 6:101–108
Zhang M, Wang K, Liu H, Wang J, Zhang C, Yue Y, Qi X (2016) Spatio-temporal variation and impact factors for vegetation carbon sequestration and oxygen production based on rocky desertification control in the karst region of southwest China. Remote Sens 8:102
Zonneveld IS (1989) The land unit A fundamental concept in landscape ecology, and its applications. Landsc Ecol 3:67–86. https://doi.org/10.1007/BF00131171
Žujo J, Marinšek M (2012) Ecosystem services evaluation in the Škocjan Caves Regional Park. Protected Areas for a Living Planet - Dinaric Arc Eco-region
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Canedoli, C., Ficetola, G.F., Corengia, D. et al. Integrating landscape ecology and the assessment of ecosystem services in the study of karst areas. Landsc Ecol 37, 347–365 (2022). https://doi.org/10.1007/s10980-021-01351-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-021-01351-2