Abstract
Context
The metacommunity concept helps to understand how local and regional processes regulate species distributions in landscapes. Metacommunity structure is often assumed as static, but may be rather dynamic, following temporal changes along environmental gradients.
Objectives
We present an empirical test of the temporal dynamics of metacommunity structure, using small mammals in an Atlantic Forest landscape as a model system.
Methods
We analyzed incidence matrices using the Elements of Metacommunity Structure framework and evaluated whether local, landscape, and spatial factors structured the metacommunity during different climatic seasons (HS = humid; SHS = super-humid) and time periods (1 = 1999–2001; 2 = 2005–2009). We compared HS-1 and SHS-1 to evaluate if metacommunity structure varies between seasons, and HS-1 and HS-2 to evaluate if it varies between time periods.
Results
Metacommunity structure changed from Clementsian (HS-1) to random (SHS-1), but during HS-2 it was Clementsian again. This suggests that groups of species are responding similarly to the major gradient of variation during the HS only. Patch size structured the metacommunity during both humid periods, and local habitat structure only during HS-1. We suggest that during the SHS these gradients are lost due to increased matrix permeability to movement, which homogenizes local communities resulting in a random structure.
Conclusions
Species habitat requirements and specializations determined metacommunity structure, but only during the HS. The Clementsian structure indicates that forest disturbances may result in the loss of whole groups of species during the HS. Alternating patterns of metacommunity structure may be associated to changes on matrix suitability between seasons.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Community ecology traditionally explores local patterns of interactions between species and their abiotic environment to predict species abundances and distributions (Vellend 2010). However, the importance of regional processes to understand co-occurrence patterns of species has been increasingly acknowledged (Ricklefs 1987; Vellend 2010). In this sense, the metacommunity concept, defined as a set of local communities potentially linked by dispersal of interacting species, provides a useful unifying framework (Leibold and Mikkelson 2002; Leibold et al. 2004). This framework helps to understand how interactions between local (e.g., species interactions) and regional (e.g., dispersal) processes regulate species distributions in landscapes (Leibold and Mikkelson 2002; Leibold et al. 2004).
One key advantage of the metacommunity framework is the possibility to distinguish among several patterns of species distribution along environmental gradients that have historically been recognized by ecologists. Such distribution patterns include the Clementsian (Clements 1916), Gleasonian (Gleason 1926), checkerboard (Diamond 1975), evenly-spaced (Tilman 1982), and nested patterns (Patterson and Atmar 1986). To help identify the most likely pattern for a given metacommunity, Leibold and Mikkelson (2002) proposed the elements of metacommunity structure (EMS) framework, which is based on three statistics (coherence, turnover, and boundary clumping). By assessing the significance of these statistics and their values, it is possible to determine which metacommunity structure best describes the major pattern of species distribution (Leibold and Mikkelson 2002; Presley et al. 2010). For example, Gleasonian and Clementsian structures differ regarding whether species respond independently or similarly to the major gradient of variation, respectively (Clements 1916; Gleason 1926; Leibold and Mikkelson 2002).
Metacommunity structure is often treated as static, i.e. stable in time, thus assuming that only a single pattern or structure would describe a given metacommunity during all time periods (e.g., Hylander et al. 2005; Presley et al. 2009). However, metacommunity structure may be rather dynamic, if local communities are affected by temporal changes that occur along environmental gradients (Datry et al. 2016; Sarremejane et al. 2017). Such dynamism may be driven by several factors, such as environmental disturbances (e.g., Bloch et al. 2007), long-term climate change (e.g., Gilman et al. 2010), and intra-annual climatic seasonality (e.g., Fernandes et al. 2014). These factors may cause changes in the abiotic environment, resource availability, population densities, and species interactions (Bloch et al. 2007; Gilman et al. 2010). These changes, by their turn, may favor some species while disfavoring others, ultimately increasing turnover or nestedness in species composition, potentially changing the dominant pattern of metacommunity structure. For instance, Bloch et al. (2007) documented temporal variability in the nestedness of terrestrial gastropods from Puerto Rico, the metacommunity being less nested immediately following a hurricane and becoming more nested during subsequent years.
Climatic seasonality, i.e. intra-annual variation in abiotic factors, is a potentially important factor driving temporal variation in metacommunity structure (e.g., Fernandes et al. 2014). Climatic seasonality may cause variation in the availability of food resources (Develey and Peres 2000; Naxara et al. 2009), habitat heterogeneity and complexity (Wojciechowski et al. 2017), and habitat stability (e.g., frequency of disturbances; Altermatt et al. 2009). Recent studies have evaluated the role of seasonality in structuring metacommunities at different spatial scales and for different taxa, including bats (e.g., Cisneros et al. 2015), small mammals (e.g., de la Sancha et al. 2014), arthropods (e.g., Altermatt et al. 2009; Sarremejane et al. 2017), and phytoplankton (e.g., Wojciechowski et al. 2017). Cisneros et al. (2015), for example, found season-specific and guild-specific distributional patterns for a bat metacommunity in a fragmented landscape, driven by an interaction between landscape characteristics and seasonal variation in resources. The gleaning animalivore group exhibited checkerboard and random structures during the dry and wet seasons, respectively (Cisneros et al. 2015). This change in metacommunity structure was explained by differences in resource availability between seasons and associated changes in the use of landscape by species (Cisneros et al. 2015). The effects of seasonality on metacommunity parameters have also being demonstrated using other frameworks different from EMS. For example, climatic seasonality was associated with temporal variation in nestedness and turnover components of beta diversity of phytoplankton across subtropical reservoirs (Wojciechowski et al. 2017). Ruhí et al. (2017) also demonstrated seasonal variation on richness and replacement components of beta diversity in metacommunities of benthic invertebrates in intermittent rivers.
Seasonality can also change the relative importance of local (e.g., habitat structure) and landscape (e.g., patch size and isolation) factors on colonization-extinction dynamics in fragmented landscapes. For example, seasonality can mitigate or accentuate edge effects and disturbances on habitat structure inside small forest fragments (Machado and Oliveira-Filho 2010; Tscharntke et al. 2012). The effects of patch isolation can also vary seasonally, if dispersal rates among forest fragments vary between seasons, as detected for some mammals (Pires et al. 2002) and birds (Silva et al. 1996). In addition, matrix suitability may vary seasonally in many agricultural landscapes, as the planting and harvesting periods are often coupled with seasonal changes (e.g., Uzêda et al. 2011). Many studies have shown that matrix suitability regulates the movement of individuals across landscapes (e.g. Vandermeer and Carvajal 2001; Haynes and Cronin 2006), thus affecting species occurrence and abundance (e.g. Umetsu et al. 2008) and community structure within local habitat patches (e.g. review in Prevedello and Vieira 2010; Brady et al. 2011). Yet, it is still unclear whether temporal variations in matrix suitability affect overall metacommunity structure in agricultural landscapes. More generally, despite these previous studies, the interaction of climatic seasonality with local and landscape factors and their effects on species occurrences remain understudied.
Here we present an empirical test of the importance of temporal, local and landscape factors on metacommunity structure in fragmented landscapes. More specifically, we tested how metacommunity structure is affected by climatic seasonality, and if the pattern detected for a season (humid season) is consistently maintained during different time periods. For these purposes, we performed two complementary analyses for a small-mammal metacommunity in a fragmented landscape of the Atlantic Forest biodiversity hotspot, using a comprehensive empirical dataset spanning 10 years. In the first analysis, we tested for changes in metacommunity structure between climatic seasons (humid versus super-humid), comparing the EMS between seasons. In the second analysis, we tested for consistency in metacommunity structure between time periods (1999–2001 vs. 2005–2009). We also identified the environmental gradients structuring the metacommunity during each season and time period.
Materials and methods
Study area
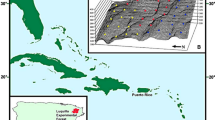
The Macacu River river basin is located within the Brazilian Atlantic Forest and is dominated by forest fragments smaller than 160 ha (Vieira et al. 2009; Delciellos et al. 2016). These forest fragments are surrounded by a heterogeneous matrix composed mostly of pastures and plantations (Vieira et al. 2009; Delciellos et al. 2016). A total of 22 forest sites were sampled at the base of Serra dos Órgãos mountain range, in the municipalities of Guapimirim (22°20′S and 42°59′W), Cachoeiras de Macacu (22°28′S and 42°39′W), and Itaboraí (22°44′S and 42°51′W), state of Rio de Janeiro, Brazil (Fig. 1; Online Resource 1—ESM 1). Twenty sites were located in forest fragments, and two sites in continuous forest in the surroundings of the Serra dos Órgãos National Park. Samplings extended over an area of 189 km2 (Fig. 1).
The vegetation of all 22 sites is dense evergreen forest (IBGE 2012). Vegetation of forest fragments is characterized by a canopy ca. 20 m high, with a higher presence of palms (e.g., Astrocaryum aculeatissimum), Cecropia spp., and lianas (Finotti et al. 2012; Delciellos et al. 2016) than in continuous forest sites. These species are frequently used as indicators of secondary forest succession (e.g., Oliveira-Filho et al. 2004; Castello et al. 2017). The vegetation of the two continuous forest sites is less disturbed than the vegetation in forest fragments (Freitas et al. 2005).
The climate in the study region is mild humid-mesothermic (Nimer 1989). The average annual rainfall is 1553.5 mm and the average annual temperature is 21.4 °C (data available at https://www.climatempo.com.br/). From October to March the climate is super-humid (super-humid season; hereafter SHS), with average rainfall of 175.0 ± 55.8 mm, and from April to September the climate is humid (humid season; hereafter HS), with average rainfall 65.0 ± 19.8 mm (https://www.climatempo.com.br/). There are no months that are water deficit (https://www.climatempo.com.br/). In tropical forests productivity and fruit production are influenced by the variation in rainfall and length of the HS (Davis 1945; Morellato et al. 2000). Arthropod availability is frequently correlated with variation in rainfall, temperature and productivity; all of which are higher in the SHS (e.g., Naxara et al. 2009). Fruit production is also frequently higher in the SHS (e.g., Develey and Peres 2000). Both fruits and arthropods are the major food items of most non-volant small mammal species occurring in tropical forests (e.g., Naxara et al. 2009). Consequently, population dynamics of many of these species is coupled with seasonality (e.g., Gentile et al. 2004; Ferreira et al. 2016), potentially affecting metacommunity structure.
Field methods
From 1999 to 2001, 10 forest fragments (A1–A4, A7–A12) and two continuous forest sites (A5, A6) were sampled once during the humid (HS-1) and once during the super-humid (SHS-1) seasons, with approximately four to six months interval between samples at the same site (Fig. 1; Online Resource 1—ESM 1). Ten other forest fragments (B1–B10) in the same landscape were sampled once during the humid season (HS-2) between 2005 and 2009 (Fig. 1; Online Resource 1—ESM 1).
In each sampling session a standardized effort of 800 trap-nights was used to capture small mammals, belonging to the orders Rodentia and Didelphimorphia. Four transects were established from the matrix to the center of forest fragments, each transect with four trap stations placed 10 m apart in the matrix, one trap station on the forest edge, and 15 trap stations 20 m apart inside the forest fragment. Each trap station had two live traps on the ground, one Tomahawk® and one Sherman®. In six of these trap stations one of the live traps was set 1–2 m above the ground in tree branches, rather than on the ground. Transects were sampled for five consecutive nights. Captures in the matrix were rare and had a negligible effect on the species composition of forest fragments, as most species captured in the matrix were also captured inside forest fragments. The exceptions were two invasive species (Mus musculus and Rattus rattus), which were not included in the analysis, and the rodent Euryzygomatomys spinosus, which was captured near the forest edge and also inhabits forests (Catzeflis et al. 2016). In the two continuous forest sites, methods and effort were the same as in the forest fragments, except that each transect was entirely within the forest, with the 20 trap stations 20 m apart. For further details, see Vieira et al. (2009) and Delciellos et al. (2016). Trapping and handling conformed to guidelines sanctioned by the American Society of Mammalogists (Sikes 2016).
Landscape variables
Four variables were measured to identify the environmental gradients related to landscape structure that may structure the metacommunity: (i) habitat amount, (ii) patch size, (iii) patch isolation, and (iv) distance to nearest continuous forest. The percentage of forest cover was used as a proxy of habitat amount, measured within a 900 m radius buffer surrounding the central coordinates of each site. The size of the buffer area (900 m radius) was chosen based on average maximum inter-fragment movement distances reported for didelphids in Atlantic Forest landscapes (Pires et al. 2002; Prevedello and Vieira 2010). Patch isolation was measured as the mean distance (in meters) between the focal (sampled) forest fragment and all other forest fragments present within a 900 m radius buffer (as in Crouzeilles et al. 2014; Delciellos et al. 2016). When no neighboring habitat patches were within the search buffer, we used the radius of the buffer (900 m) as the value of isolation. The landscape variables were obtained from the map produced by SOS Mata Atlântica and Instituto Nacional de Pesquisas Espaciais (2005). Data were converted to UTM projection to assure accurate area and distance calculations. Habitat amount and patch isolation were measured using the software ArcGis 9.3 (ESRI 2008). The distance to nearest continuous forest, Serra dos Órgãos mountain range, was measured as the Euclidean distance between the forest fragment and the nearest continuous area, in Google Earth software (Google Inc. 2015).
For the two continuous forest sites we attributed a relatively large ‘forest fragment’ size (400 ha), and a low value of isolation (0 m). Variation in landscape metrics was similar for the forest fragments sampled during the two time periods (1999–2001 and 2005–2009). For the ten forest fragments sampled from 1999 to 2001, habitat amount ranged from 10.5 to 36%, patch size from 13 to 78 ha, patch isolation from 170 to 925 m, and distance to nearest continuous forest from 250 to 8215 m. For the ten forest fragments sampled from 2005 to 2009, habitat amount ranged from 5 to 35%, patch size from 15 to 160 ha, patch isolation from 145 to 1195 m, and distance to nearest continuous forest from 597 to 7724 m.
Local variables
Nine habitat variables were measured to describe aspects of local habitat structure inside forest that could affect species incidence patterns across local communities. All habitat variables were measured within a 3 m radius circle around the center of each trapping station, except for those trapping stations located in the matrix. Six variables were measured qualitatively as present (1) or absent (0) (water course, Cecropia spp., lianas, grass, palm A. aculeatissimum, and fallen logs), and three were ranked in three levels (1–3; overstory vertical density, understory horizontal density, and tree size). Values recorded at all trap stations of the same forest site were summed, yielding a single value for each variable per site, which was used in subsequent statistical analyses (as in Delciellos et al. 2016). These nine variables were measured once in each site, assuming that the chosen variables do not vary between seasons, or that the variation would not be detected by the sampling method used. Local habitat structure was used as proxy of disturbance related to the history of anthropogenic use of each site, with more disturbed forest sites having higher abundance of lianas, A. aculeatissimum palms, trees of the genus Cecropia, grasses, reduced overstory vertical density and a more open understory, and predominance of small diameter trees (see Delciellos et al. 2016).
Prior to analysis, the nine habitat variables were standardized and then combined and reduced in a Principal Components Analysis (PCA), using the function prcomp in R package vegan (Oksanen et al. 2017). The PCA was used to obtain uncorrelated variables (principal components) that preserve most of the original variation in the data. The number of ‘meaningful’ principal components was selected by comparing the eigenvalues obtained in PCA to values given by the broken stick distribution (Borcard et al. 2011). For the 12 sites sampled from 1999 to 2001, the first four principal components (PC1 to PC4) were selected, which together explained 91% in data variation (Table 1). The PC1 was associated with sites with increased overstory vertical density, understory horizontal density, abundance of Cecropia spp., and abundance of grass; PC2 with decreased tree size and abundance of lianas; PC3 with increased abundance of fallen logs and palms; and PC4 with increased presence of water courses (Table 1). For the ten sites sampled from 2005 to 2009, the first two principal components (PC1 and PC2) were selected, which together explained 67.7% in data variation (Table 1). The PC1 was associated with sites with increased abundance of palms and Cecropia spp., and decreased abundance of lianas; and PC2 with decreased tree size and increased overstory vertical density (Table 1).
Spatial variables
A principal coordinate analysis of neighbor matrices (PCNM) was used to evaluate if the environmental gradients structuring the metacommunity were related to the spatial relationships among studied sites, using the function pcnm in R package vegan (Oksanen et al. 2017). PCNM method consists of a principal coordinate analysis of a truncated pairwise geographic Euclidean distance matrix between sampling sites (Dray et al. 2006). The chosen threshold to construct the truncated distance matrix was 21.5 km. In this method, only the principal coordinates associated with positive eigenvalues are used as spatial predictors, and retained to be used in further analysis (Dray et al. 2006). Eight positive eigenvectors were selected in the PCNM analyses for the 12 sites sampled from 1999 to 2001, and seven for the ten sites sampled from 2005 to 2009.
Data analysis
We used the analytical methods of Leibold and Mikkelson (2002), and the conceptual framework of Presley et al. (2010), to determine the most likely structure or quasi-structure for the studied metacommunity. The EMS framework is based on the significance of three statistics (coherence, turnover, and boundary clumping), which indicates the most likely pattern of species variation among local communities (Leibold and Mikkelson 2002; Presley et al. 2010). Significance of each statistic was tested with the “r1” moderately-conservative null model (Presley et al. 2010). This null model permutes species incidence across sites 1,000 times, preserving the original species richness of each site, but maintaining their probability of occurrence proportional to their incidence in the original matrix. This permutation procedure avoids that rare species enhance coherence (Presley et al. 2010), and has acceptable levels of type I error (Gotelli and Graves 1996; Presley et al. 2009).
Reciprocal averaging (Correspondence Analysis, CA) was used to ordinate the species-by-sites incidence matrices. We used incidence rather than abundance matrices because we performed snapshot surveys in all sites (see Field methods section), rather than repeated sampling which is necessary to properly estimate small mammal abundance. The CA maximizes the correspondence between the position of sites and species along the axes based on the degree to which their communities share similar species composition and ranges, respectively, reducing the number of interruptions in species’ ranges (Leibold and Mikkelson 2002). Species and sites were ranked according to their position along the primary CA axis.
Coherence was evaluated by counting the number of embedded absences in all species ranges and community composition for each site. Significant negative coherence (i.e. more embedded absences than the null distribution) indicates a checkerboard distribution (Diamond 1975). If coherence is non-significant, the metacommunity is randomly structured regarding the environmental gradient analyzed (Leibold and Mikkelson 2002). A significant positive coherence (i.e. less embedded absence than the null distribution) suggests that species are distributed according to the same gradient (Leibold and Mikkelson 2002), which is further differentiated by evaluating species range turnover and boundary clumping.
Range turnover was calculated by counting the number of replacements, i.e. the number of times a species replaces another at the edge of their ranges (see Presley et al. 2010 for details). The observed number of replacements was then compared to the ones generated by each null metacommunity. A negative or lower number of observed replacements suggest that nestedness (Patterson and Atmar 1986) characterizes the metacommunity structure. If the observed metacommunity exhibits a positive or higher number of replacements, the data are further contrasted by analysis of range boundary clumping. If the range turnover is non-significant the metacommunity exhibits quasi-structures that have the same characteristics as their associated idealized structures, but with weaker structuring processes (see Presley et al. 2010).
In the last step, range boundary clumping was tested using the Morisita’s Index, which has an expected value of 1. If the observed index is not significantly different from 1, range boundaries are randomly distributed, indicating a Gleasonian gradient (Leibold and Mikkelson 2002). Conversely, if the observed value is lower or higher than 1, it shows that species range boundaries are overdispersed or clumped, respectively (Leibold and Mikkelson 2002). Nested metacommunities may exhibit clumped, stochastic, or hyperdispersed species loss among sites, which are analogous to Clementsian, Gleasonian and evenly-spaced gradients, respectively, with the difference that these patterns of range dispersion are found only at one side of the distributional gradient (see Presley et al. 2010 for details). Significance of the observed Morisita Index was evaluated using a Chi squared goodness-of-fit test, which compares the observed distribution to an expected distribution of range boundary locations (Presley et al. 2009).
Analyses of EMS were conducted with algorithms written in Matlab R2017b 9.3.0.713579. Site scores for primary CA axes were derived using the function “ca” in Matlab. Script files are available at https://faculty.tarleton.edu/higgins/metacommunity-structure.html. If a positive significant coherence was found, Spearman correlation was used to evaluate the correlation between the site scores for each primary CA axis and the landscape, local, and spatial variables to identify which variables compose the environmental gradient. As the number of sites used in each correlation analysis was relatively low (n = 10 or 12), thus reducing the statistical power of the analysis, we did not perform any adjustment of P-values for multiple comparisons.
We analyzed EMS for three datasets as follows: (1) Humid season for the first time period (HS-1), using the data from 12 sites sampled from 1999 to 2001; (2) Super-humid season for the first time period (SHS-1), using data from 12 sites sampled from 1999 to 2001; and (3) Humid season for the second time period (HS-2), using data from other ten sites sampled from 2005 to 2009 (Online Resource 1—ESM 1). We first compared HS-1 and SHS-1 to evaluate if metacommunity structure varied between climatic seasons. Secondly, we analyzed whether metacommunity structure during a same season (humid) changed between time periods by comparing metacommunity structure during HS-1 and HS-2. All sites sampled the same metacommunity during different time periods and were randomly located across the landscape (Fig. 1). Consequently, each set of sites reflects a representative sample of the metacommunity and should reflect structure of the metacommunity regardless of the particular sites chosen.
Results
A total of 786 individuals of 19 small mammal species were captured during the first time period (from 1999 to 2001). At the metacommunity level, total species richness and abundance were slightly higher during the HS-1 (399 individuals from 18 species) than during the SHS-1 (387 individuals from 16 species) (Online Resource 1—ESM 2). Species richness across local communities varied from three to 11 species during HS-1 and three to eight species during SHS-1. The metacommunity during the first time period included seven species of marsupials and 12 species of rodents (Online Resource 1—ESM 2). Akodon cursor and Didelphis aurita were the most abundant species during this first time period. During the second time period (HS-2), 351 individuals of 11 species were captured (Online Resource 1—ESM 2). Species richness across local communities varied from three to seven species. The HS-2 metacommunity included seven species of marsupials and four species of rodents (Online Resource 1—ESM 2). Philander frenatus was the most abundant species during this period.
Small mammal metacommunity structure was non-coherent during SHS-1, resulting in a random structure (Table 2), indicating that species did not respond to the same environmental gradient during this season. For both humid periods (HS-1 and HS-2) coherence and turnover were positive and significant, and species range boundaries were significantly clumped (Table 2). Therefore, the small mammal metacommunity was structured in a Clementsian pattern during each humid season (Table 2).
For HS-1, the PC1 of local habitat structure and patch size were identified as the main variables structuring the metacommunity (Table 3). Positive turnover indicated that sites at opposite ends of the environmental gradient had different species compositions. Sites at one end of the PC1 gradient presented the arboreal Caluromys philander, Guerlinguetus ingrami, and Phyllomys sp. nov., as well as the terrestrial E. spinosus, and were associated with increased overstory and understory vegetation, and increased abundance of grass and Cecropia spp. (Figure 2). The sites at the opposite end of the gradient were associated with the more terrestrial species Euryoryzomys russatus, Marmosops incanus, Monodelphis sp., Oecomys catherinae, Oligoryzomys nigripes, and Trinomys dimidiatus, and the arboreal Phyllomys pattoni (Fig. 2). Also, some species occurred in larger patches as E. russatus, Monodelphis sp., Nectomys squamipes, O. nigripes, P. pattoni, and T. dimidiatus, whereas E. spinosus and G. ingrami occurred at the opposite end of gradient (Fig. 3). This ordination suggested the existence of at least two compartments in the metacommunity, the first composed by sites A3–A6, and the second by sites A7, A8, A11 and A12 (Fig. 4a). A third, intermediate compartment may also be identified, which contains mostly generalist species, composed by the remaining sites (Fig. 4a).
Ordination of incidence of small mammals along the first principal component (PC1) of habitat structure during the first humid period in 12 sites sampled from 1999 to 2001 in the Macacu River basin, Brazil. PC1 represented a gradient of increased overstory vertical density, understory horizontal density, and abundance of Cecropia spp. and grass (from left to right)
First axis of the Correspondence Analysis for the matrix of species-by-site incidence of small mammals during the humid season for a the 12 sites sampled from 1999 to 2001, and b the 10 sites sampled from 2005 to 2009, in the Macacu River basin, Brazil, both revealing a Clementsian metacommunity structure. Ac = Akodon cursor, Cp = Caluromys philander, Da = Didelphis aurita, Er = Euryoryzomys russatus, Es = Euryzygomatomys spinosus, Gi = Guerlinguetus ingrami, Gm = Gracilinanus microtarsus; M = Monodelphis sp., Mi = Marmosops incanus, Mn = Metachirus nudicaudatus, Mp = Marmosa paraguayana, Ns = Nectomys squamipes, Oc = Oecomys catherinae, Od = Oxymycterus dasytrichus, On = Oligoryzomys nigripes, Pn = Phyllomys sp. nov., Pf = Philander frenatus, Pp = Phyllomys pattoni, Ri = Rhipidomys itoan, Td = Trinomys dimidiatus
For HS-2, the ordination of the species along the first CA axis was related only to patch size variation, with M. nudicaudatus occurring in the larger patches, and C. philander, E. russatus, M. incanus, and O. nigripes occupying the opposite end of gradient (Fig. 5). This indicated the existence of at least two compartments during the HS-2, the first composed by sites B2, B3, and B8, and the second by sites B4 to B6 (Fig. 4b). Again, a third compartment may be identified in between the first two compartments.
Discussion
The structure of the studied small mammal metacommunity is seasonally dynamic, which changes from Clementsian during the humid season to random during the super-humid season. These results corroborate previous studies showing that metacommunity structure is dynamic, rather than constant through time (e.g., Fernandes et al. 2014; Cisneros et al. 2015; Wojciechowski et al. 2017). We also detected a consistency in the Clementsian structure of the metacommunity during humid periods 5 years apart. This consistency occurred even though different sets of sites (forest fragments) were used for the two periods, indicating that a Clementsian structure does indeed apply to the small mammal community. A cycle in metacommunity structure has rarely been demonstrated, i.e. the returning to the previous idealized structure after the homogenization of structuring environmental gradients. Temporal changes from idealized to random structures have been described for other metacommunities both within and between seasons, and attributed to responses to the homogenization of structuring environmental gradients (Fernandes et al. 2014; Cisneros et al. 2015; Wojciechowski et al. 2017). For example, the change from checkerboard (dry season) to random structure (wet season) of the gleaning animalivore bat metacommunity in a tropical forest was attributed to more abundant prey during the wet season, that reduces competition among species and homogenizes their distribution (Cisneros et al. 2015). Also, a change from idealized to random structure in a metacommunity of phytoplankton was associated with periods of low environmental heterogeneity in limnological characteristics of water in subtropical reservoirs during the spring season of two subsequent years (Wojciechowski et al. 2017). A cycle in metacommunity structure was also detected for the floodplain-fish metacommunity in the Pantanal of South America (Fernandes et al. 2014). The floodplain-fish metacommunity structure changed from nestedness to quasi-Clementsian throughout the wet season, following the progress of flood levels. During the beginning of the flood season the water level rises, increasing the connectivity and dispersal among habitat patches (Fernandes et al. 2014). The flood-plain fish metacommunity remained structured during the same month (March) across four subsequent years (Fernandes et al. 2014). Similarly, we found consistency in metacommunity structure within a season across time periods for the small mammals.
Variation in functional connectivity between seasons may be a possible explanation for the dynamic cycle in metacommunity structure detected in our study, i.e., the changing from a random to an idealized structure between seasons. The studied forest fragments are embedded in an agricultural matrix, part of which varies in composition between climatic seasons following agricultural activities. The main agricultural products in the studied landscape are corn (Zea mays) and cassava (Manihot esculenta), which are cultivated from November (beginning of super-humid season) to June (beginning of humid season), when they are harvested (Uzêda et al. 2011). Thus, during more than half of the humid season, part of the matrix in the landscape is likely to be depleted of any type of vegetation cover. This change in matrix quality may reduce the offer of potentially supplementary (Braga et al. 2015) or complementary (Dunning et al. 1992) resources, and increase exposure of dispersing individuals to predators, thus affecting the movement rates of individuals between forest fragments.
Matrix permeability to movement is generally higher when the matrix is structurally more similar to the habitat patches (Prevedello and Vieira 2010). During the super-humid season, matrix permeability is probably higher than during the humid season, allowing more frequent inter-fragment dispersal. The expected result may be more similar community compositions in different forest fragments, leading to a random pattern of metacommunity structure. On the other hand, when plantations are harvested during the humid season, matrix suitability is likely to decrease and the metacommunity may become structured. The hypothesis that variation in functional connectivity drives the differences in metacommunity structure between seasons may be tested in the future, for example by quantifying inter-fragment movement and functional connectivity during different seasons.
The positive turnover and high and significant Morisita index during both humid seasons indicate that groups of species are associated with different parts of the environmental gradient in the landscape (Leibold and Mikkelson 2002). These groups form distinct communities from the regional species pool during this season, based on similarities in species habitat requirements or specialization (Leibold and Mikkelson 2002; Presley et al. 2009). Many species of non-volant small mammals of the Atlantic Forest have species-specific habitat requirements (Püttker et al. 2008; Delciellos et al. 2016), thus are likely to vary in their occurrence or abundance in the landscape according to gradients of habitat structure. For example, species of the genus Akodon are frequently associated to open habitats, such as natural clearings inside the forest or open matrix habitats where the species is more abundant (e.g., Umetsu et al. 2008), and the semi-aquatic rodent N. squamipes is found more frequently or in higher abundance near water courses in forested habitats (e.g., Briani et al. 2001). Also, several species of non-volant small mammals have particular specializations in their diet or locomotion, such as the arboreal marsupial C. philander, which has adaptations to locomotion in the forest canopy (e.g., Delciellos and Vieira 2006).
The Clementsian structure detected during the humid season was structured by patch size and habitat structure. Patch size appeared as the only common factor structuring the metacommunity during both time periods (HS-1 and HS-2). Some species were restricted to smaller or larger patches only (see Figs. 3, 5), which may be related to the increased habitat heterogeneity and resource availability in larger forest fragments (Ewers and Didham 2007; Delciellos et al. 2016), and a higher probability of dispersing individuals to find larger patches (Coleman et al. 1982). In addition to patch size, local habitat structure also structured the metacommunity only during the first time period (HS-1). During the HS-1 most species with arboreal and terrestrial habits were found in sites associated to different degrees of complexity of habitat structure. The Clementsian structure detected implies that disturbances acting more strongly on one end of the structuring environmental gradient will affect the group of species of that compartment. For example, deforestation or selective logging inside forest fragments act on the forested end of the gradient, where the compartment composed mostly by arboreal species probably will be the most negatively affected. Habitat structure, considered a proxy of habitat quality in some studies, is also frequently found as one of the main determinants of species composition for several taxa, such as lepidopterans (Summerville and Crist 2004) and birds (Uezu and Metzger 2011). Our results highlight the importance of local habitat structure for structuring metacommunities of marsupials and rodents in fragmented landscapes.
Climatic seasonality affects the structure of the metacommunity of non-volant small mammals, and both local and landscape factors are important to determine species composition. Our results provide reinforcing evidence that metacommunity structure may be dynamic, rather than constant through time, as previously suggested (e.g., Fernandes et al. 2014; Cisneros et al. 2015). This result has important implications for species inventory and conservation, landscape management, and detection of regional patterns in community structure. First, we suggest that caution is necessary when extrapolating metacommunity patterns detected during snapshot surveys, as metacommunity structure may vary between seasons, at least for non-volant small mammals in agricultural landscapes. Thus, we recommend conducting repeated sampling of local communities in different climatic seasons, whenever possible. Second, we suggest that matrix suitability may determine metacommunity structure even in terrestrial landscapes, as matrix suitability directly affects functional connectivity. There is a growing consensus that management of matrix may be a more feasible and less expensive way to benefit local populations and communities in fragmented landscapes compared to other actions, such as increasing habitat amount (Tscharntke et al. 2012; Driscoll et al. 2013). Here we suggest that management of matrix may also affect metacommunity structure. This suggestion may be tested in future studies by comparing inter-patch dispersal rates and metacommunity structure among landscapes composed of different matrix types or land uses, or at a same landscape during different seasons. Explicit tests of this hypothesis may contribute to identify the underlying mechanisms shaping metacommunity structure in fragmented landscapes, allowing a better understanding of how different anthropogenic activities impact biodiversity.
Data accessibility
Data are presented in ESM 1–2.
References
Altermatt F, Pajunen VI, Ebert D (2009) Desiccation of rock pool habitats and its influence on population persistence in a Daphnia metacommunity. PLoS ONE 4(3):e4703
Bloch CP, Higgins CL, Willig MR (2007) Effects of large scale disturbance on metacommunity structure of terrestrial gastropods: temporal trends in nestedness. Oikos 116(3):395–406
Borcard D, Gillet F, Legendre P (2011) Numerical ecology with R. Springer Science & Business Media, New York
Brady MJ, McAlpine CA, Possingham HP, Miller CJ, Baxter GS (2011) Matrix is important for mammals in landscapes with small amounts of native forest habitat. Landscape Ecol 26(5):617–628
Braga CAC, Prevedello JA, Pires MRS (2015) Effects of cornfields on small mammal communities: a test in the Atlantic Forest hotspot. J Mamm 96(5):938–945
Briani DC, Vieira EM, Vieira MV (2001) Nests and nesting sites of Brazilian forest rodents (Nectomys squamipes and Oryzomys intermedius) as revealed by a spool-and-line device. Acta Theriol 46(3):331–334
Castello ACD, Coelho S, Cardoso-Leite E (2017) Lianas, tree ferns and understory species: indicators of conservation status in the Brazilian Atlantic Rainforest remnants, southeastern Brazil. Braz J Biol 77(2):213–226
Catzeflis F, Patton J, Percequillo A, Weksler M (2016) Euryzygomatomys spinosus. The IUCN Red List of Threatened Species 2016: e.T8418A22205855. http://dx.doi.org/10.2305/IUCN.UK.2016-2.RLTS.T8418A22205855.en
Cisneros LM, Fagan ME, Willig MR (2015) Season specific and guild specific effects of anthropogenic landscape modification on metacommunity structure of tropical bats. J Anim Ecol 84(2):373–385
Clements FE (1916) Plant Succession: an analysis of community functions, vol 242. Carnigie Institution Washington Publications, Washington DC
Coleman BD, Mares MA, Willig MR, Hsieh YH (1982) Randomness, area, and species richness. Ecology 63:1121–1133
Crouzeilles R, Prevedello JA, Figueiredo MSL, Lorini ML, Grelle CEV (2014) The effects of the number, size and isolation of patches along a gradient of native vegetation cover: how can we increment habitat availability? Landscape Ecol 29:479–489
Datry T, Bonada N, Heino J (2016) Towards understanding the organization of metacommunities in highly dynamic ecological systems. Oikos 125(2):149–159
Davis ED (1945) The annual cycle of plants, mosquitoes, birds, and mammals in two Brazilian forests. Ecol Monogr 15(3):243–295
de la Sancha NU, Higgins CL, Presley SJ, Strauss RE (2014) Metacommunity structure in a highly fragmented forest: has deforestation in the Atlantic Forest altered historic biogeographic patterns? Divers Dist 20(9):1058–1070
Delciellos AC, Vieira MV (2006) Arboreal walking performance in seven didelphid marsupials as an aspect of their fundamental niche. Austral Ecol 31(4):449–457
Delciellos AC, Vieira MV, Grelle CEV, Cobra P, Cerqueira R (2016) Habitat quality versus spatial variables as determinants of small mammal assemblages in Atlantic Forest fragments. J Mamm 97(1):253–265
Develey PF, Peres CA (2000) Resource seasonality and the structure of mixed species bird flocks in a coastal Atlantic forest of southeastern Brazil. J Trop Ecol 16(01):33–53
Diamond JM (1975) Assembly of species communities. Ecol Evol Communities 342:444
Dray S, Legendre P, Peres-Neto PR (2006) Spatial modelling: a comprehensive framework for principal coordinate analysis of neighbour matrices (PCNM). Ecol Model 196(3):483–493
Driscoll DA, Banks SC, Barton PS, Lindenmayer DB, Smith AL (2013) Conceptual domain of the matrix in fragmented landscapes. Trends Ecol Evol 28:605–613
Dunning JB, Danielson BJ, Pulliam HR (1992) Ecological processes that affect populations in complex landscapes. Oikos 65(1):169–175
ESRI (2008) ARCGIS, V-late tool. Environmental System Research Institute Inc, Redlands
Ewers RM, Didham RK (2007) The effect of fragment shape and species sensitivity to habitat edges on animal population size. Conserv Biol 21:926–936
Fernandes IM, Henriques-Silva R, Penha J, Zuanon J, Peres-Neto PR (2014) Spatiotemporal dynamics in a seasonal metacommunity structure is predictable: the case of floodplain fish communities. Ecography 37(5):464–475
Ferreira MS, Vieira MV, Cerqueira R, Dickman CR (2016) Seasonal dynamics with compensatory effects regulate populations of tropical forest marsupials: a 16-year study. Oecologia 182(4):1095–1106
Finotti R, Kurtz BC, Cerqueira R, Garay IG (2012) Variação na estrutura diamétrica, composição florística e características sucessionais de fragmentos florestais da bacia do rio Guapiaçu (Guapimirim/Cachoeiras de Macacu, RJ, Brasil). Acta Bot Bras 26:464–475
Freitas SR, Mello MC, Cruz CB (2005) Relationships between forest structure and vegetation indices in Atlantic Rainforest. For Ecol Manage 218(1):353–362
Gentile R, Finotti R, Rademaker V, Cerqueira R (2004) Population dynamics of four marsupials and its relation to resource production in the Atlantic Forest in southeastern Brazil. Mammalia 68(2):5–15
Gilman SE, Urban MC, Tewksbury J, Gilchrist GW, Holt RD (2010) A framework for community interactions under climate change. Trends Ecol Evol 25(6):325–331
Gleason HA (1926) The individualistic concept of the plant association. Bull Torrey Bot Club 53(1):7–26
Gotelli NJ, Graves GR (1996) Null models in ecology. Smithsonian Institution, Washington, DC
Haynes KJ, Cronin JT (2006) Interpatch movement and edge effects: the role of behavioral responses to the landscape matrix. Oikos 113(1):43–54
Hylander K, Nilsson C, Jonsson BG, Göthner T (2005) Differences in habitat quality explain nestedness in a land snail meta community. Oikos 108(2):351–361
IBGE (2012) Manual Técnico da Vegetação Brasileira. Instituto Brasileiro de Geografia e Estatística, Rio de Janeiro
Leibold MA, Mikkelson GM (2002) Coherence, species turnover, and boundary clumping: elements of metacommunity structure. Oikos 97(2):237–250
Leibold MA, Holyoak M, Mouquet N, Amarasekare P, Chase JM, Hoopes MF, Holt RD, Shurin JB, Law R, Tilman D, Loreau M (2004) The metacommunity concept: a framework for multi-scale community ecology. Ecol Lett 7(7):601–613
Machado ELM, Oliveira-Filho ATD (2010) Spatial patterns of tree community dynamics are detectable in a small (4 ha) and disturbed fragment of the Brazilian Atlantic forest. Acta Bot Bras 24(1):250–261
Morellato LPC, Talora DC, Takahasi A, Bencke CC, Romera EC, Zipparro VB (2000) Phenology of Atlantic rain forest trees: a comparative study 1. Biotropica 32(4):811–823
Naxara L, Pinotti BT, Pardini R (2009) Seasonal microhabitat selection by terrestrial rodents in an old-growth Atlantic Forest. J Mamm 90(2):404–415
Nimer E (1989) Climatologia no Brasil, 2nd edn. Fundação IBGE, Rio de Janeiro
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MH, Szoecs E, Wagner H (2017) Package ‘vegan’—community ecology package: ordination, diversity and dissimilarities. v.2.4-2. L. http://cran.r-project.org/, http://r-forge.r-project.org/projects/vegan/
Oliveira-Filho AT, Carvalho DA, Vilela EA, Curi N, Fontes MAL (2004) Diversity and structure of the tree community of a fragment of tropical secondary forest of the Brazilian Atlantic Forest domain 15 and 40 years after logging. Rev Bras Bot 27(4):685–701
Patterson BD, Atmar W (1986) Nested subsets and the structure of insular mammalian faunas and archipelagos. Biol J Linn Soc 28:65–82
Pires AS, Lira PK, Fernandez FAS, Schittini GM, Oliveira LC (2002) Frequency of movements of small mammals among Atlantic Coastal Forest fragments in Brazil. Biodivers Conserv 108(2):229–237
Presley SJ, Higgins CL, López-González C, Stevens RD (2009) Elements of metacommunity structure of Paraguayan bats: multiple gradients require analysis of multiple ordination axes. Oecologia 160(4):781–793
Presley SJ, Higgins CL, Willig MR (2010) A comprehensive framework for the evaluation of metacommunity structure. Oikos 119(6):908–917
Prevedello JA, Vieira MV (2010) Does the type of matrix matter? A quantitative review of the evidence. Biodivers Conserv 19(5):1205–1223
Püttker T, Pardini R, Meyer-Lucht Y, Sommer S (2008) Responses of five small mammal species to micro-scale variations in vegetation structure in secondary Atlantic Forest remnants, Brazil. BMC Ecol 8(1):9
Ricklefs RE (1987) Community diversity: relative roles of local and regional processes. Science 235(4785):167–171
Ruhí A, Dtary T, Sabo JL (2017) Interpreting beta-diversity components over time to conserve metacommunities in highly dynamic ecosystems. Conserv Biol 31(6):1459–1468
Sarremejane R, Cañedo-Argüelles M, Prat N, Mykrä H, Muotka T, Bonada N (2017) Do metacommunities vary through time? Intermittent rivers as model systems. J Biogeogr 44(12):2752–2763
Sikes RS, ACAUC of the AS of Mammalogists (2016) Guidelines of the American Society of Mammalogists for the use of wild mammals in research and education. J Mamm 97:663–688
Silva JMC, Uhl C, Murray G (1996) Plant succession, landscape management, and the ecology of frugivorous birds in abandoned Amazonian pastures. Conserv Biol 10(2):491–503
SOS Mata Atlântica, INPE (2005) Atlas dos remanescentes florestais da Mata Atlântica. Fundação SOS Mata Atlântica and Instituto Nacional de Pesquisas Espaciais, São Paulo
Summerville KS, Crist TO (2004) Contrasting effects of habitat quantity and quality on moth communities in fragmented landscapes. Ecography 27(1):3–12
Tilman D (1982) Resource competition and community structure. Princeton University Press, New Jersey
Tscharntke T, Tylianakis JM, Rand TA, Didham RK, Fahrig L, Batáry P, Bengtsson J, Clough Y, Crist TO, Dormann CF, Ewers RM, Fründ J, Holt RD, Holzschuh A, Klein AM, Kleijn D, Kremen C, Landis DA, Laurance W, Lindenmayer D, Scherber C, Sodhi N, Steffan-Dewenter I, Thies C, van der Putten WH, Westphal C (2012) Landscape moderation of biodiversity patterns and processes-eight hypotheses. BiolRev 87(3):661–685. https://doi.org/10.1111/j.1469-185X.2011.00216.x
Uezu A, Metzger JP (2011) Vanishing bird species in the Atlantic Forest: relative importance of landscape configuration, forest structure and species characteristics. Biodivers Conserv 20(14):3627–3643
Umetsu F, Metzger JP, Pardini R (2008) Importance of estimating matrix quality for modeling species distribution in complex tropical landscapes: a test with Atlantic forest small mammals. Ecography 31(3):359–370
Uzêda MC, Fidalgo ECC, Iguatemy MA, Alves RC, Rouws JRC (2011) Explorando as relações entre estrutura da paisagem e atributos de qualidade de fragmentos em região de Mata Atlântica no Estado do Rio de Janeiro. Bol Pesqui Desenvolv 136:1–31
Vandermeer J, Carvajal R (2001) Metapopulation dynamics and the quality of the matrix. Am Nat 158(3):211–220
Vellend M (2010) Conceptual synthesis in community ecology. Q Rev Biol 85(2):183–206
Vieira MV, Olifiers N, Delciellos AC, Antunes VZ, Bernardo LR, Grelle CEV, Cerqueira R (2009) Land use vs. fragment size and isolation as determinants of small mammal composition and richness in Atlantic Forest remnants. Biol Conserv 142:1191–1200
Wojciechowski J, Heino J, Bini LM, Padial AA (2017) Temporal variation in phytoplankton beta diversity patterns and metacommunity structure across subtropical reservoirs. Freshwater Biol 62:751–766
Acknowledgements
We thank the students and staff of the Laboratório de Vertebrados (Universidade Federal do Rio de Janeiro), particularly Angela Marcondes, Maycon Belarmino, Natalie Olifiers, and Nélio Barros. We also thank Carlos Grelle for comments on previous statistical analyses, and Renato Crouzeilles for providing the measures of forest fragments isolation and habitat amount. Two anonymous reviewers provided valuable suggestions to the manuscript. Landowners of the study sites at the Macacu River basin that allowed access to their lands for scientific research. Financial support was provided by grants from Projetos Demonstrativos/Ministério do Meio Ambiente, Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ; to JAP, MVV, and RC), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq; to JAP, MVV, and RC), PPBIO/CNPq/Rede Bio.Ma, PROBIO I/MMA/GEF, and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). ACD has a postdoctoral scholarship from CAPES/FAPERJ (PAPD- E-26/202.144/2015); VNTBJ from CNPq/PDJ (157750-9) and CNPq/CAPES/PELD (88887.137536/2017-00); and CB from CNPq/CAPES/FAPs/PELD (88887.140649/2017-00). SER has a doctoral fellowship from CAPES/PROEX (1343229). Permissions to handle animals were given by Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis/Ministério do Meio Ambiente (Authorization numbers 87/05-RJ, 099/06-RJ, 13861-1, 13861-2, and 16703).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Delciellos, A.C., Borges-Júnior, V.N.T., Prevedello, J.A. et al. Seasonality in metacommunity structure: an empirical test in the Atlantic Forest. Landscape Ecol 33, 1769–1783 (2018). https://doi.org/10.1007/s10980-018-0701-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-018-0701-5