Abstract
Isoconversional kinetic method (model-free kinetics) was used in this study to determine the activation energies (E a) of the combustion process of five different biomass samples, namely pine sawdust, sugarcane bagasse, coffee husk, rice husk and tucumã seeds, widely available in Brazil. Two different atmospheres with 20 % O2:N2/O2 (conventional combustion) and CO2/O2 (typical oxy-fuel combustion) were studied. Thermogravimetric (TG) and derivative thermogravimetric (DTG) curves were used to obtain experimental data on the thermal degradation behavior of the biomasses, and the activation energy values were obtained for hemicellulose, cellulose and residual lignin separately. The results show that the E a obtained for N2/O2 ranged from 68 to 236 kJ mol−1 for hemicellulose, 119 to 209 kJ mol−1 for cellulose and 87 to 205 kJ mol−1 for residual lignin, depending on the type of biomass. Under CO2/O2 atmosphere, E a showed decreases, in average, 35 % for hemicellulose and 26 % for cellulose, in comparison with N2/O2 atmosphere. However, a 6 % increase was observed for the residual lignin. These changes can be understood by differences between CO2 and N2 gas properties. However, the results show that the variation in the E a is more dependent on the type of biomass than on the atmosphere at which the combustion takes place.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Urbanization, population growth and the development of new technologies in both developed and developing countries have increased the demand for electrical power. A direct consequence of such a demand is the increase in the consumption of fossil fuels (oil, coal and natural gas) and gradual atmospheric pollutant emissions [1]. There has been a growing global consensus that the environmental damages (global warming, acid rain and urban smog) are caused by gaseous emissions, which intimately affect both climate changes and human health [2]. The development of new technologies aims at mitigating this situation, considering the reduction in the environmental impacts and also the CO, CO2 and NO emissions.
A promising alternative to reduce gaseous emissions is the use of renewable fuels for the production of energy [3]. In this sense, Brazil has advanced programs for the implementation of different types of biomass. Their utilization appears as an opportunity to collaborate with power supply systems [4]. In some regions, large parts of biomass wastes are still used only for boiler feeding in power plants for vapor generation. Among the several forms of biomass thermal conversion for energy, the most used ones are direct combustion, gasification and pyrolysis [4]. The knowledge on the biomass thermal behavior is a fundamental issue for the development and optimization of technological processes.
Biomass is an organic solid matrix, and its thermal decomposition involves a large number of parallel reactions, related to heat and mass transfer processes. The decomposition of lignocellulosic materials has attracted considerable attention from the scientific community because it is a major step in its combustion process and also the key step in thermal processing methods, such as fast pyrolysis to produce chemical products and bio-oil [5, 6]. The release rate, quantity and composition of the volatile materials present in a biomass influence its flame ignition, stability and temperature profile in the radiant furnace part. Such factors are important in the design of burners for pulverized fuel power stations and impact on NO x generation mechanisms [6].
Many researchers have conducted studies to elucidate several thermal conversion processes during biomass thermal degradation [3, 5–11]. According to Yang et al. [11], the mass losses of the main components of biomass (hemicellulose, cellulose and lignin) occur in a determined temperature range, which depends on experimental conditions, such as heating rate, heat transfer and biomass type. The chemical differences between these components directly influence their chemical reactivity during the combustion process [12].
The combustion process of biomasses comprises the burnout in an ambient rich in oxygen or air, which considers types and fuel properties, particles size, air flow rate and fuel moisture. Such characteristics affect combustion, reaction rates and generation and transfer of heat [13, 14]. Although the interest in the use of renewable fuels has been growing worldwide, combustion is a process that inherently emits CO2. An interesting technology to solve this problem is the oxy-fuel combustion, which comprises the recycling of combustion gases, mainly CO2, which is ready to be captured with increase in its concentration. Although this technology represents a significant advance of decreasing CO2 emission and can be used as retrofit technology in conventional boilers, the costs are still a barrier for its commercial application [15–19].
However, the successful implementation of the oxy-fuel technology requires fundamental knowledge on the combustion under a CO2 atmosphere. The determination of kinetics parameters of the thermal degradation of solid fuels is essential for the design of conversion plants with efficient operating systems [7, 20] and for an accurate prediction of their behavior under different conditions.
Several studies have reported different approaches of kinetic methods to describe thermal degradations [7, 21–26]. The determination of the E a based on the Arrhenius’s equation and using thermogravimetric data has been widely applied to the evaluation of the reaction mechanisms in solid phases.
This study investigates the thermal decomposition of five different types of biomass in natura (pine sawdust, sugarcane bagasse, coffee husk, rice husk and tucumã seed) under both atmospheres: N2/O2 (conventional combustion) and CO2/O2 (oxy-fuel combustion). Experiments were performed in a thermogravimetric balance, and data were applied to a mathematical model (isoconversional kinetic method—model-free kinetics) for the determination of the activation energies of the combustion processes.
Materials and methods
Origin of biomasses
Five in natura biomass samples, namely pine sawdust, sugarcane bagasse, coffee husk (from São Paulo State—Southeast region), rice husk (from Maranhão State—Northeast region) and tucumã seed (from Pará State—Northern region), were selected for the study. Physicochemical characteristics of the in natura samples after preparation are shown in Table 1.
Samples preparation
The samples were received in natura and underwent pre-treatments that consisted in washing for the removal of impurities, dried at 90 °C for 24 h, chopped in a household blender and sieved. The average particle size of 460 µm was selected and obtained by the arithmetic mean of two consecutive ASTM laboratory sieves whose mesh apertures ranged between 420 and 500 µm. Particles of 460 µm average size were used for all the samples and determined based on our previous study [27].
Thermal analysis (TG/DTG)
TG/DTG non-isothermal tests were performed in a Shimadzu thermogravimetric analyzer, TGA-50H model for the obtaining of the experimental data. Three steps of thermal degradation related to hemicellulose, cellulose and lignin were evaluated.
The following conditions were used: atmospheres of synthetic air (N2/O2–80/20) to simulate conventional combustion and CO2/O2 (80/20) to simulate oxy-fuel combustion; 100 mL min−1 flow rate; 10.0 ± 0.5 mg samples mass; and five heating rates: 10, 15, 20, 25 and 30 °C min−1 from room temperature up to 700 °C.
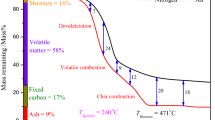
Figure 1 shows the TG/DTG curves only for the sugarcane bagasse sample in N2/O2 atmosphere. For each biomass and atmosphere, the TG and DTG curves exhibited similar behaviors, but with their own characteristics.
Activation energies (E a) and conversion degree (α) of the thermal degradation steps for hemicellulose (first step), cellulose (second step) and residual lignin (third step) were obtained by model-free kinetics. Each thermal degradation step was considered based on the peak separation methods of DTG curves [26].
Determination of the activation energies by model-free kinetics
The activation energies for the combustion processes of five biomasses (pine sawdust, sugarcane bagasse, coffee husk, rice husk and tucumã seed) were obtained using non-isothermal thermogravimetric experiments and model-free kinetics.
The model-free kinetics method enables the evaluation of the Arrhenius parameters. It is based on isoconversional techniques and calculates the activation energy as a function of conversion (E a = f(α)) [28–32]. The method is supported on the assumption that:
where T is the temperature, t is the time, f(α) is the reaction model, k(T) is the constant of reaction rate, R is the universal gas constant, A is the pre-exponential factor, E a is the activation energy and (α) is the conversion degree.
Conversion degree is defined as α = (m 0 − m)/(m 0 − m ∞ ), where m is the local sample mass that varies with time, m 0 is the initial sample mass and m ∞ is the final sample mass. For simple reactions, the function of conversion, f(α), can be determined, but for complex reactions it is generally unknown [28].
There is a temporal dependence on non-isothermal conditions, which can be eliminated dividing f(α) by the heating rate β = dT/dt. After rearranging Eq. (1), one obtains:
Integrating Eq. (2) up to the conversion at the local temperature, one has:
Provided that E a/2RT ≫ 1, the temperature integral of the right-hand side can be approximated by
Inserting Eq. (4) in Eq. (3), rearranging Eq. (3) and taking the logarithm of the generated expression, one has:
where subscript α represents the values related to a given conversion degree. The activation energies can be obtained graphically by plotting ln (β/T 2) versus 1/T.
Results and discussion
Values of the activation energies for combustion under synthetic air atmosphere
Figure 2 shows the profiles of the E a versus conversion degree (α) for three thermal decomposition steps under synthetic air atmosphere of the five biomasses (pine sawdust, sugarcane bagasse, coffee husk, rice husk and tucumã seed).
Table 2 shows the activation energy (E a) values obtained by model-free kinetics under N2/O2 (80/20) atmosphere for the three thermal degradation steps.
The first, second and third steps correspond to the thermal degradation of hemicellulose, cellulose and residual lignin, respectively. Moisture release, which occurs below 110 °C, was not considered in this study.
Among the samples, only pine sawdust showed two distinct events of mass loss. The first and second events correspond to the hemicellulose and cellulose decomposition together, and the third is related to the decomposition of the residual lignin.
E a versus (α) for coffee husk (Fig. 2c) of hemicellulose degradation (first step) shows a variation between 3 and 176 kJ mol−1 related to a very heterogeneous thermal degradation. Such a behavior may be indicative of the occurrence of different mechanisms during the thermal decomposition process, which can be explained by the high volatile content (≈74 %) in the sample [23, 33, 34].
Regarding the thermal degradation of cellulose (second step), pine sawdust and tucumã seed samples (Fig. 2a, e) showed a more complex decomposition mechanism with variations of E a between 105 and 400 kJ mol−1, and 46 and 282 kJ mol−1, respectively. The profiles of the E a versus conversion curves show that the thermal decomposition of a biomass is considerably influenced by its structural and chemical composition. According to Poletto et al. [7], the crystallinity index and the extractive content can vary from 34 to 43 % and from 4 to 14 %, respectively; such factors can either accelerate or slow the thermal degradation process at low temperatures.
The high variation in the E a values for pine sawdust, which is a woody sample, can be associated with the high content of organic compounds [35]. For tucumã seeds, the high variation of E a may have been caused by its complex structure, which can be confirmed by means of TG/DTG curves that showed several mass loss events.
The average E a values achieved in this study are in agreement with values reported in the literature regarding order of magnitude. For example, these E a values reported to the biomasses ranged between 160 and 280 kJ mol−1 for pine sawdust [23], 76–220 kJ mol−1 and 75–116 kJ mol−1 for sugarcane bagasse [25, 36], 160–250 kJ mol−1 for coffee husk [37] and 120–250 kJ mol−1 for rice husk samples [26, 38].
However, it is worth mentioning that those authors employed TG experiments, but with different kinetic methods and considered the entire process of thermal degradation as one step. Regarding the tucumã seed samples, no E a value for thermal decomposition has been reported in the literature.
Influence of the atmosphere type on the thermal degradation of biomasses
In order to compare the influence of the atmosphere type on the thermal decomposition of biomasses, an atmosphere of 80 % CO2–20 % O2 was used, i.e., N2 was replaced by CO2.
Under 80 % CO2–20 % O2 condition, three different events of mass loss for all samples could be observed in the DTG curves. Under synthetic air condition (80 % N2–20 % O2), the pine sawdust samples showed two events and the others showed three events.
The DTG curves are shown in Fig. 3. The presence of CO2 causes a displacement of the thermal degradation to a higher temperature for two samples (pine sawdust and coffee husk). However, such an effect was not observed for other samples (sugarcane bagasse, rice husk and tucumã seed). It seems that the degree displacement depends more on the biomass type than on the atmosphere type.
In both atmospheres, a shoulder around 310 °C was observed for the sugarcane bagasse and rice husk samples. Such a characteristic is indicative of higher hemicellulose content in the samples [23]. For the sugarcane bagasse and rice husk samples, the distinction between hemicellulose and cellulose is possible by means of DTG curves, while for the other samples, such a distinction is not possible due to the overlapping of hemicellulose and cellulose during the thermal process [25].
For most biomasses (pine sawdust, sugarcane bagasse, rice husk and tucumã seed) of this study, no significant effect was observed by replacing N2 by CO2 during the combustion process. However, only for the coffee husk sample there was a decrease in the combustion reaction rate accompanied by a displacement to a higher temperature in the second degradation step.
Values of the activation energies for combustion under 80 % CO2 atmosphere
The activation energies (E a) were determined for each thermal degradation step separately so that the effect of CO2 could be understood.
Figure 4 shows the E a versus (α) curves under 80 % CO2–20 % O2 atmosphere for the thermal decomposition steps of the biomasses studied (pine sawdust, sugarcane bagasse, coffee husk, rice husk and tucumã seed). All the biomasses showed the three distinct thermal degradation steps.
A homogeneous behavior related to the hemicellulose thermal degradation for all biomasses was observed under 80 % CO2 atmosphere in the first step. A more heterogeneous behavior was observed for the rice husk and tucumã seed samples in the second step (Fig. 4d, e). Such heterogeneity is attributed to the high variation in the activation energy, i.e., for rice husk the values are between 150 and 320 kJ mol−1, and for tucumã seed the E a varied from 80 to 150 kJ mol−1.
In the second stage and only for rice husk, there was a significant change in the E a versus (α) curve when N2 was replaced by CO2. In the third thermal degradation step, related to the residual lignin content, the higher activation energy values were observed for pine sawdust and coffee husk samples, as shown in Fig. 4a, c. The E a values ranged from 120 to 350 kJ mol−1 for pine sawdust and from 100 to 250 kJ mol−1 for coffee husk.
Table 3 shows the E a under 80 % CO2 atmosphere for the three thermal degradation steps obtained by model-free kinetics.
No trend was observed when N2 was replaced by CO2, and the behavior of each sample is independent of the atmosphere in which the degradation takes place.
Conclusions
This study investigated the effects of two atmospheres on the activation energy values of the thermal degradation steps of five different biomass samples (pine sawdust, sugarcane bagasse, coffee husk, rice husk and tucumã seed) at the same oxygen concentrations: N2/O2 and CO2/O2.
The isoconversional kinetic method (model-free kinetics) was used, and experimental conditions were performed in a thermobalance.
Three different steps were considered, and each decomposition region corresponded to the hemicellulose, cellulose and lignin contents in the samples. Each region was delimited in the TG curves by the peak separation method based on DTG curves.
The results show that the activation energy values varied for each thermal degradation step, atmosphere type and biomass type, and no trend was observed regarding the replacement of N2 by CO2 in the environment. For the rice husk sample, the activation energy values in CO2 atmosphere increased 56 % on average, in comparison with synthetic air atmosphere, which is in agreement with E a values reported in the literature.
On the other hand, in average, the E a values for sugarcane bagasse, pine sawdust, coffee husk and tucumã seed are lower in CO2 than in synthetic air atmosphere, i.e., 44, 26, 6 and 36 %, respectively.
Biomass is a complex lignocellulosic matrix and involves several steps during its thermal decomposition process. According to the results, each constituent component of the different biomasses showed a specific thermal degradation mechanism. Such difference was shown in the E a versus α curves for each step defined. In comparison with the thermal processes conducted at two atmospheres, the mechanisms were distinct, as indicated by the curves profiles and average E a values.
Although the properties of CO2 and N2 gases are different, our findings indicate that the variation in the E a is more dependent on the biomass type than on the atmosphere in which the combustion takes place.
References
Toftegaard MB, Brix J, Jensen PA, Glarborg P, Jensen AD. Oxy-fuel combustion of solid fuels. Prog Energy Combust Sci. 2010;36:581–625.
Saidur R, Abdelazoz EA, Demirbas A, Hossain MS, Mekhilef S. A review on biomass as a fuel for boilers. Renew Sust Energy Rev. 2011;15:2262–89.
Babu BV. Biomass pyrolysis: a state-of-the-art review. Biof Bioprod Bioref. 2008;2:393–414.
Foletto EL, Hoffman R, Hoffaman RS, Portugal UL, Jahn SL. Applicability of rice husk ash. Quim Nova. 2005;28:1055–60.
Sanchez-Silva L, López-González D, Villaseñor J, Sánchez P, Valverde JL. Thermogravimetric-mass spectrometric analysis of lignocellulosic and marine biomass pyrolysis. Bioresour Technol. 2012;109:163–72.
Saddawi A, Jones J, Williams A, Wojtowicz M. Kinetics of the thermal decomposition of biomass. Energy Fuels. 2010;24:1274–82.
Poletto M, Zaterra AJ, Santana RMC. Thermal decomposition of wood: kinetics and degradation mechanisms. Bioresour Technol. 2012;126:7–12.
Leroy V, Cancellieri D, Leoni E. Chemical and thermal analysis of lignocellulosic fuels. For Ecol Manage. 2006;234S:S125.
Magdziarz A, Wilk M. Thermal characteristics of the combustion process of biomass and sewage sludge. J Therm Anal Calorim. 2013;114:519–29.
Braga RM, Costa TR, Freitas JCO, Barros JMF, Melo DMA, Melo MAF. Pyrolysis kinetics of elephant grass pretreated biomasses. J Thermal Anal Calorim. 2014;117:1341–8.
Yang H, Yang H, Yan R, Chen H, Lee DH, Zheng C. Characteristics of hemicellulose, cellulose and lignin pyrolysis. Fuel. 2007;86:1781–8.
Carrier M, Loppinet-Serani A, Denux D, Lasnier JM, Ham-Pichavani F, Cansel F, Aymonier C. Thermogravimetric analysis as a new method to determine the lignocellulosic composition of biomass. Biomass Bioenergy. 2011;35:298–307.
Zhao W, Li Z, Wang D, Zhu Q, Sun R, Meng B, Zhao G. Combustion characteristics of different parts of corn straw and NO formation in a fixed bed. Bioresource Technol. 2008;99:2956–63.
Senneca O. Kinetics of pyrolysis, combustion and gasification of three biomass fuels. Fuel Process Technol. 2007;88:87–97.
Álvarez L, Yin C, Riaza J, Pevida C, Pis JJ, Rubiera F. Biomass co-firing under oxy-fuel conditions: a computational fluid dynamics modeling study and experimental validation. Fuel Process Technol. 2014;120:22–33.
Álvarez L, Gharebaghi M, Pourkashanian M, Williams A, Riaza J, Pevida C, Pis JJ, Rubiera F. CFD modeling of oxy-coal combustion in an entrained flow reactor. Fuel Process Technol. 2011;92:1489–97.
Riaza J, Álvarez L, Gil MV, Pevida C, Pis JJ, Rubiera F. Effect of oxy-fuel combustion with steam addition on coal ignition and burnout in an entrained flow reactor. Energy. 2011;36:5314–9.
Arias B, Pevida C, Rubiera F, Pis JJ. Effect of biomass blending on coal ignition and burnout during oxy-fuel combustion. Fuel. 2008;87:2753–9.
Wang G, Silva RB, Azevedo JLT, Martins-Dias S, Cost M. Evaluation of the combustion behavior and ash characteristics of biomass waste derived fuels, pine and coal in a drop tube furnace. Fuel. 2014;117:809–24.
Moltó J, Font R, Conesa JA, Martin-Gution I. Thermogravimetric analysis during the decomposition of cotton fabrics in an inert and air environment. J Anal Appl Pyrol. 2006;76:124–31.
Wilson L, Yang W, Blasiak W, John GR, Mhilu CF. Thermal characterization of tropical biomass feedstocks. Energy Convers Manage. 2011;52:191–8.
Harun NY, Afzal MT. Thermal decomposition kinetics of forest residue. J Appl Sci. 2010;10:1122–7.
Shen DK, Gu S, Luo KH, Bridgwater AV, Fang MX. Kinetic study on thermal decomposition of woods in oxidative environment. Fuel. 2009;88:1024–30.
Yao F, Wu Q, Lei Y, Guo W, Xu Y. Thermal decomposition kinetics of natural fibers: activation energy with dynamic thermogravimetric analysis. Polym Degrad Stab. 2008;93:90–8.
Ramajo-Escalera B, Espina A, Garcia JR, Sosa-Arnao JH, Nebra SA. Model-free kinetics applied to sugarcane bagasse combustion. Thermochim Acta. 2006;448:111–6.
Shuangning X, Zhihe L, Baoming L, Weiming Y, Xueyuan B. Devolatilization characteristics of biomass at flash heating rate. Fuel. 2006;85:664–70.
Pécora AAB, Ávila I, Cruz G, Lima C, Crnovic PM. Prediction of the combustion process in fluidized bed based on physical-chemical properties of biomass particles and their hydrodynamic behavior. Fuel Proc Technol. 2014;124:188–97.
Vyazovkin S, et al. ICTAC Kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta. 2011;520:1–19.
Vyazvkin S, Wight CA. Model-free and model-fitting approaches to kinetic analyses of isothermal and non-isothermal data. Thermochim Acta. 1999;340:53–68.
Vyazovkin S, Dollimore D. Linear and nonlinear procedures in isoconversional computations of the activation energy of non-isothermal reaction in solids. J Chem Inform Comp Sci. 1996;36:42–5.
Vyazovkin S. Advanced isoconversional method. J Therm Anal Calorim. 1997;49:1493–9.
Vyazovkin S, Sbirrazzuoli N. Confidence intervals for the activation energy estimated by few experiments. Anal Chim Acta. 1997;355:175–80.
Nassar MM, Ashour EA, Wahid SS. Thermal characteristics of bagasse. J Appl Polym Sci. 1996;61:885–90.
García R, Pizarro C, Lavin A, Bueno JL. Characterization of Spanish biomass wastes for energy use. Bioresour Technol. 2012;103:249–58.
Aghamohammadi N, Sulaiman NMN, Aroua MK. Combustion characteristics of biomass in Southeast Asia. Biomass Bioenergy. 2011;35:3884–90.
Munir S, Daood SS, Nimmo W, Cunliffe AM, Gibbs BM. Thermal analysis and devolatilization kinetics of cotton stalk, sugarcane bagasse and shea meal under nitrogen and air atmospheres. Bioresour Technol. 2009;100:1413–8.
Wilson L, John GR, Mhilu CF, Yang W, Blasiak W. Coffee husks gasification using high temperature air/steam agent. Fuel Process Technol. 2010;91:1330–7.
Sonobe T, Worasuwannarak N. Kinetic analyses of biomass pyrolysis using the distributed activation energy model. Fuel. 2008;87:414–21.
Acknowledgements
The authors gratefully acknowledge CAPES (DS00011/07-0) and FAPESP (2012/00639-9) for the financial support. The assistance from Lilian Torquato and Angela P. Giampedro is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cruz, G., Crnkovic, P.M. Investigation into the kinetic behavior of biomass combustion under N2/O2 and CO2/O2 atmospheres. J Therm Anal Calorim 123, 1003–1011 (2016). https://doi.org/10.1007/s10973-015-4908-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-015-4908-2