Abstract
Researchers are increasingly tasked with identifying biological recovery targets as ecosystems recover from anthropogenic stressors. Attempts to define such recovery targets are often hampered by two problems: (1) a lack of long-term monitoring data; and (2) the confounding influence of multiple stressors, especially regional stressors like climate warming. Paleolimnological studies of minimally disturbed reference sites can help address both these issues. Using paleoecological techniques, researchers can isolate the long-term impact of regional stressors like climate change on species assemblages largely independent of other confounding stressors such as acidification, eutrophication, and land-use change, thereby providing a framework to assess biological recovery in lakes that are recovering from acid deposition or other stressors. This manuscript provides a theoretical paleolimnological framework for the use of reference lakes in studying biological recovery from acidification, and provides an example of how assemblages of scaled-chrysophytes have changed in Adirondack-region reference lakes (NY, USA) from pre-ca. 1900 to present. The thirty-one reference lakes were selected from a database of over 1400 lakes, using criteria to minimize the influence of acidification, eutrophication, road-salt seepage, and piscivore introductions. As such, these lakes provide a unique opportunity to examine the effects of regional stressors in the Adirondack ecological region, which can inform biological recovery in lakes that have acidified historically. The modern chrysophyte assemblages from the reference lakes were significantly related to modern limnological variables including pH, dissolved organic carbon and ionic concentration as well as important physical variables including lake depth, which were used to help understand changes in the chrysophyte assemblages over the last century. Changes in chrysophyte assemblages from pre-1900 to present were determined by comparing the modern surface assemblages to a sediment interval representing pre-1900 conditions, revealing significant increases in the abundance of colonial chrysophyte taxa, especially S. petersenii, S. sphagnicola, and S. echinulata, and corresponding decreases in the relative abundance of many Mallomonas taxa. These changes suggest that regional warming and/or oligotrophication have influenced the species assemblages of minimally disturbed reference lakes, suggesting that lakes currently recovering from acidification are unlikely to return to their pre-disturbance assemblages.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Adirondack Park (NY, USA) covers over 24,300 km2 (6 million acres) and is thought to contain more protected lakes and streams than any other area in the continental United States (Jenkins et al. 2007). Unfortunately, the region has been heavily impacted by acid deposition. A 1980’s survey of 1469 Adirondack lakes found that 27 % of the lakes were chronically acidic and 24 % had pH values less than 5 (Driscoll et al. 1991). Recent long-term monitoring studies in the region have shown that some lakes have begun to undergo chemical recovery from acidification since the implementation of the 1990 US Clean Air Act Amendments (CAAA), characterized by decreases in SO4 2−, NO3 −, inorganic monomeric aluminum, and/or increases in pH and acid neutralizing capacity (ANC) (Waller et al. 2012). With chemical recovery progressing, ecologists and lake management professionals are now tasked with defining biological recovery targets for acid-impacted lakes.
It is expected that chemical recovery will promote biological recovery from acidification, characterized by a decrease in the abundance of acid-tolerant species and an increase in the abundance of acid-sensitive species (Driscoll et al. 2001). However, a growing number of studies have demonstrated that lakes recovering from stressors such as acidification or eutrophication may not return to a pre-disturbance state due to the influence of recent climate change (Arseneau et al. 2011; Battarbee et al. 2012, 2014). For example, warming surface waters may increase the abundance of warm-water species and small-bodied taxa, and longer periods of thermal stratification may increase the abundance of taxa with a high surface-to-volume ratio, buoyant taxa, and motile taxa (Daufresne et al. 2009; Winder and Sommer 2012).
The phenomenon where a recovering ecosystem does not return to its pre-disturbance state due to the influence of a novel stressor such as climate change is sometimes referred to as a ‘change in baseline conditions’ or, more simply, a ‘shifting baseline’(Bennion et al. 2011a; USEPA 2012). The concept of a ‘shifting baseline’ refers to the fact that climate change may alter fundamental environmental conditions in aquatic ecosystems such as the length of the ice-covered season and/or other thermal/chemical characteristics of a lake that cause a change in assemblage characteristics. These altered environmental conditions may then prevent lakes from returning to pre-disturbance assemblages as they recover from stressors such as acidification or eutrophication. Given the pronounced effects of climate change on aquatic ecosystems, a recovery target should consider the influence that climate change (or another regional stressor) may have on recovering species assemblages. Thus, in order to define appropriate recovery targets, we need to first characterize the shifting baseline, i.e. what kinds of species assemblage changes may occur in an area undergoing regional change, independent of other stressors? One approach to obtaining this information is to examine long-term species changes in a regional set of minimally disturbed reference sites.
The concept of a reference site is frequently applied in limnological work, but different studies apply the term in different ways, often resulting in confusion. First, a reference site is not typically a control site (i.e., a site that is identical to the impacted site in physical, chemical, and/or biological characteristics, except for the presence of a stressor being studied, Downes et al. 2002). True controls are rare in most non-manipulative limnological studies, and so limnologists generally rely on ‘reference’ frameworks for comparison between impacted and non-impacted sites. In its simplest application, a reference site is defined by the absence of a stressor of interest (e.g. acidification, eutrophication). This approach is simple to implement and can be effective at detecting the influence of stressors on species assemblages (Chessman et al. 2008). However, unknown and uncontrolled-for stressors may influence species assemblages in the reference lakes, affecting study results. A more rigorous reference approach is to designate a ‘reference’ condition based on the absence of multiple anthropogenic stressors (e.g., acidification, eutrophication, introduced species, road salt seepage) (Bailey et al. 2004). A variety of methods can be used to identify minimally disturbed reference sites (Soranno et al. 2011). However, the criteria must be clearly defined; otherwise, the sites have little interpretative or comparative value (Brucet et al. 2013). Arguably, the ideal reference site would be a “pristine” site that has never experienced any form of anthropogenic impact over time but, given the pervasiveness of anthropogenic influence on even remote alpine and arctic waterbodies, such a site is unlikely to exist (Bennion et al. 2011a; Catalan et al. 2013). While it is difficult to determine the minimum number of sites required in a regional reference site study, given the variation in lake physical/chemical characteristics, a minimum of 25 lakes is likely needed (Bailey et al. 2004).
The USEPA has called for long-term monitoring at reference sites to track the ‘shifting baseline’ effect associated with climate change to inform recovery studies (USEPA 2012). However, long-term monitoring data for aquatic ecosystems are rare or occur on relatively short timescales (<50 years). In such cases, paleolimnological studies can play an essential role as paleolimnological techniques can be used to reconstruct the environmental histories of lakes over decades or centuries using information from the physical, chemical, and biological characteristics of lake sediments.
The goal of this manuscript is to use a paleolimnological approach to examine changes in scaled chrysophyte assemblages (Synurophyceae and Chrysophyceae) since pre-1900 in a set of 31 reference lakes in the Adirondack Park (NY, USA). The reference lakes were identified from an online database of 1469 Adirondack lakes surveyed as part of the Adirondack Lakes Survey (ALS) using a set of stringent selection criteria (Kretser et al. 1989). The reference lakes have been largely protected from several common stressors in the Adirondack Park, including: acidification (i.e., decreases in lake pH and ANC), eutrophication from watershed/shoreline development, leaching of road salt, and piscivore introductions. As the reference lakes have been protected from several common anthropogenic stressors, they can be used to identify what influence, if any, regional stressors such as climate change are having on Adirondack lakes. This information, in turn, can be used to help define targets for Adirondack lakes recovering from stressors such as acidification. For example, if the reference sites document a recent increase in warm-water taxa or small planktonic algae, changes predicted to occur with regional warming (Winder and Sommer 2012), a recovery target for an impacted site should reflect such regional shifts in species composition. Effectively, the recovery target would be a change to a “novel” state that is similar to suitable reference systems (Hobbs et al. 2006). The ‘recovered’ species assemblages of the impacted site would contain elements of the pre-disturbance assemblage, such as previously abundant acid-sensitive taxa, as well as ‘novel’ additions as seen in the reference sites (Fig. 1). With this concept in mind, the following fundamental questions are addressed in this paper: 1) What chrysophyte assemblages are present in Adirondack reference lakes today, and what environmental variables are associated with their distribution? 2) How have assemblages of scaled chrysophytes changed since ca. pre-1900 times, and what are the plausible mechanisms for the observed changes? We hope that answers to these questions will help address the importance of identifying changes over time in minimally disturbed reference lakes and demonstrate their potential utility for current studies of biological recovery from anthropogenic stressors.
Conceptual diagram outlining the biological response of two lakes (Lake A and Lake B) to acidification and climate warming since pre-industrial times. The lake response is shown across two gradients: one of increasing acidity and one of increasing air temperature. Lake A is a higher-pH/ANC ‘reference lake’ that is resistant to acidification. Lake B is a lake susceptible to acidification. The purpose of including a reference lake (Lake A) is to identify trends in species composition caused by regional warming (i.e., to characterize the ‘shifting baseline’). Historically, the influences of regional warming and acid deposition on the lakes are minimal. The lakes are similar in species composition (Ahist, Bhist), though the taxa of Lake A are characteristic of higher pH/ANC conditions. With the onset of acid deposition, Lake B acidifies (solid arrow). Acid-sensitive taxa are lost and replaced with acid-tolerant ones (Bacid). Lake A does not acidify; its species assemblages do respond, however, to regional warming (solid arrow). There is an increase in warmer-water taxa, taxa that benefit from shorter-ice covered seasons, etc. (Apres). When the stress of acid deposition is reduced, there are four possible recovery trajectories for Lake B (dashed arrows): (1) return to pre-disturbance state: this is only possible if Lake B does not respond to warming, which is unlikely; (2) no recovery: Lake B responds to warming but shows no decline in acid-tolerant taxa; (3) partial recovery: Lake B responds to warming and there is some decline in acid-tolerant taxa and/or increase in acid-sensitive taxa; (4) recovery to a novel state: Lake B responds to warming and the abundance of acid-tolerant/acid-sensitive taxa returns to historic levels. However, regional warming has prevented the lake from returning to its pre-acidification state. Lake A and B are again similar in species composition, though the taxa of lake A remain characteristic of higher pH/ANC conditions. Conceptual diagram based on ideas presented in Vinebrooke et al. (2004), Hobbs et al. (2009), and Battarbee et al. (2012)
Study sites
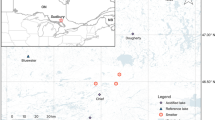
The lakes in this investigation are a set of 31 minimally disturbed reference lakes in the Adirondack Park (Arseneau 2014) (Fig. 2). This study builds on the work of Stager and Sanger (2003) who introduced the concept of “Heritage Lakes”, or minimally disturbed lakes in the Adirondacks. Our primary interest was to identify a set of Adirondack lakes that had been largely protected from several stressors common in the Adirondack Park, namely: acidification, eutrophication from watershed/shoreline development, road salt seepage, and the introduction of non-native piscivores. The reference lakes were identified from an online database of 1469 Adirondack lakes sampled from 1984 to 1987 as part of the Adirondack Lakes Survey (ALS) (Kretser et al. 1989). In total, the 1469 lakes sampled in the ALS survey are considered a representative sample of the 2759 lakes greater than 0.2 ha that fall within the Adirondack Ecological Zone.
Map of the Adirondack Park (NY, USA) showing the locations of a set 31 Adirondack reference lakes minimally impacted by anthropogenic stressors. Map shows the position of the Adirondack Park in the northeastern United States (enlarged area); inset shows lakes in the St. Regis Canoe area in detail—all other reference lakes are shown as points
The six selection criteria used to identify reference lakes in the ALS are listed below. The criteria are based on the findings of previous limnological or paleolimnological studies of the Adirondack Park and elsewhere.
-
1.
Public access: The search was restricted to lakes with public access. Lakes that were not accessible via hiking trails and lakes with excessively long or difficult hikes were also excluded.
-
2.
1980s pH ≥ 6.5; 1980s ANC ≥ 50 μeq L−1: These paired criteria were used to exclude lakes that were likely to have acidified. Cumming et al. (1992b) demonstrated that few Adirondack lakes with a pH ≥ 6.5 in the late 1970s and early 1980s underwent significant chronic acidification. Additionally, an acid neutralizing capacity (ANC) ≥ 50 μeq L−1 largely protects acid-sensitive lakes from episodic acidification (Driscoll et al. 2001).
-
3.
Watershed and shoreline development ≤5 %: These paired criteria were used to exclude lakes that were likely to have experienced eutrophication due to development along their shorelines or in their watersheds. A 5 % development cut-off was used so that Adirondack lakes with small camps would not be excluded.
-
4.
Cl− ≤ 2 mg L−1: In the ALS, lakes were considered ‘salt impacted’ based on a cut-off criterion of 20 µmol L−1 (~0.71 mg L−1) (Newton and Driscoll 1990). We applied a less restrictive selection criterion, allowing lakes with a larger concentration of salt to be included in the survey. In general, it was found that lakes excluded from the reference set based on high Cl− concentration were also excluded based on other criteria, primarily shoreline or watershed development.
-
5.
No introduced piscivores: A number of invertebrates are potentially sensitive to changes in fish predation (e.g., Cladoceran zooplankton, Korhola and Rautio 2001). As such, lakes that had non-indigenous piscivores (e.g., largemouth bass, Micropterus salmoides Lacepède) in the ALS survey and lakes with rainbow smelt (Osmerus mordax Mitchill), a species introduced in the Adirondack uplands that can compete with lake trout fry, were excluded. Non-native piscivores were identified using George (1981), Kraft et al. (2006), and USGS (2013).
-
6.
Maximum depth ≥5 m; surface area ≥4 ha: These selection criteria were included because some of the paleo-indicators of interest (scaled-chrysophytes, the Chrysophyceae and Synurophyceae; and Daphnia taxa) are planktonic taxa that are rare or absent in small, shallow waterbodies (Dixit et al. 1999; Ginn et al. 2010; Jeziorski et al. 2012). Recognizing that many Adirondack lakes are shallow and ≤4 ha in size (Kretser et al. 1989), the size selection criteria used in this study were chosen to maximize the total number of possible reference sites while minimizing the inclusion of sites unlikely to contain the paleo-indicators of interest.
Materials and methods
Field collection
Sediment cores were collected from the deepest basin of the study lakes in either summer 2010 or 2011 using a 7.6-cm (ID) Glew gravity corer, and were sectioned on-site into 0.25-cm intervals using a vertical Glew extruder. Water samples were collected by hand at ~0.5 m below the water’s surface in glass and polypropylene jars. All chemistry analyses were performed by the Center for Environmental Systems Engineering (Syracuse University, NY) using standard US EPA methods.
Sample preparation
In a top–bottom paleolimnological investigation, a surface-sediment sample is taken to represent the present-day species assemblage and compared to a sediment sample taken from further down in the sediment core to represent pre-industrial times (Cumming et al. 1992b). The technique is an effective way to obtain a regional ‘snap-shot’ of species changes over time. In this investigation, the ‘top’ sample is an integrated sample from 0–1 cm in the sediment core and is estimated to represent the past 2–3 years. The ‘bottom’ sample is a 0.25 cm sediment subsection from ~20 cm (in 4 cases where the sediment core length was <20 cm, the bottommost sediment interval was used; 3 cases ~17–18 cm, 1 case ~15 cm). A single 0.25 cm subsection was analyzed at ~20 cm, rather than a 1-cm subsection, as a decrease in water content in the sediment occurs downcore which increases the amount of ‘time’ represented per sediment subsection at this depth. A depth of 20 cm was chosen because 210Pb-dated cores from over 25 lakes suggests that sediment at a depth of >20 cm is greater than 100 year of age in the vast majority of Adirondack region lakes (Arseneau 2014; Arseneau et al. 2011; Cumming et al. 1992b, 1994).
Sediment samples were prepared for enumeration of chrysophyte scales using standard methods (Battarbee et al. 2001). Briefly, sediment samples were digested in a 1:1 molar mixture of strong H2SO4:HNO3. Samples were heated to ~70 °C for 6–7 h and were then rinsed repeatedly until samples reached the pH of distilled water (litmus test). Samples were plated as a series of four 100× dilutions on microscope slides using Naphrax®. Chrysophyte scales were identified using DIC optics under oil-immersion using a 100X Fluotar objective with a numerical aperture of 1.3. Counts in this study were typically enumerated to a minimum count of 400 scales but in cases where scales were sparse, a minimum of 200 scales were counted (Ginn et al. 2010). Five of the 31 pre-1900 samples from the reference lakes were considered uncountable due to low scale concentration (i.e. <10 scales encountered per transect). The principle taxonomic references used for identifications were: Siver (1991), Nicholls and Gerrath (1985), and Kling and Kristiansen (1983), as well as unpublished photo reference materials. Small Mallomonas spp. scales that could not be identified using light microscopy were grouped into a Mallomonas ‘small’ category (Cumming et al. 1992a).
Modern distribution of scaled chrysophytes
A Redundancy Analysis (RDA) was used to examine the influence of water chemistry and lake morphometric variables on the chrysophyte assemblages present in the surface sediments in the Adirondack reference lakes. RDA, a linear direct ordination method, was selected because an initial Detrended Correspondence Analysis (DCA) of the present-day samples for the 31 reference lakes indicated that the length of the species turnover gradient was short (<3 s.d.) and so linear ordination techniques were deemed most appropriate (Lepš and Šmilauer 2003). Because of the relatively small number of surface samples in this investigation (31 samples), the environmental data matrix was summarized as PCA axis scores of the first four PCA axes (Lepš and Šmilauer 2003). It is necessary to reduce the number of explanatory variables in the RDA so as not to inflate the variance explained by including a large number of correlated environmental variables. This approach allows for the main patterns of variation to be established while avoiding a spurious inflation of the variation explained. The normality of environmental variables was assessed using the Shapiro–Wilks test using SYSTAT v. 11. Non-normal environmental variables were transformed using either square-root or log transformations, and were not included in the PCA if transformation did not result in a normal distribution. In all of the analyses, taxa that reached ≥2 % relative abundance in more than two sediment samples were included in the analyses. This cut-off criterion was used primarily to exclude rare taxa, as they can have a large influence in ordinations with a small number of samples. Forward selection with Monte Carlo permutation tests was used to identify a minimum set of environmental variables that could explain a significant proportion of the species assemblage data using the computer program CANOCO v. 5.0 (ter Braak and Šmilauer 2012). The species percentage data were square-root transformed to reduce the importance of the dominant taxon Mallomonas duerrschmidtiae Siver, Hamer and Kling, a common Adirondack taxon with a wide tolerance to many environmental variables (Cumming et al. 1992a).
Changes in scaled-chrysophyte assemblages since pre-1900
Changes in species composition from pre-1900 to present were analyzed in the reference lakes using both univariate and multivariate techniques. Changes in scaled chrysophytes were summarized using a PCA ordination, because an initial DCA of both the present-day and pre-1900 samples for the 26 reference lakes (i.e., those with sufficient chrysophyte scales in both the surface and the pre-1900 samples) indicated that the species turnover was short (<3 s.d.), suggesting that a linear ordination technique was appropriate (Lepš and Šmilauer 2003). The PCA of the modern and pre-1900 chrysophyte assemblages was based on square-root transformed species data using CANOCO v. 5.0 (ter Braak and Šmilauer 2012). The significance of the changes in species assemblages between pre-1900 and present-day was assessed using a multivariate Analysis of Similarity (ANOSIM) on square-root transformed species data, using Bray–Curtis similarities (Clarke 1993). The Similarities Percentages test (SIMPER) was used to identify which species make an important contribution to the difference evident between groups (Clarke and Warwick 2001).
Previous paleolimnological investigations have identified recent increases in colonial chrysophyte taxa in Ontario (Flear 2011; Paterson et al. 2004) and Maritime Canada (Ginn et al. 2010), increases which the authors suggest may be due, at least in part, to regional warming. As such, a Wilcoxon signed-rank test (a non-parametric analog of a paired t test) was used to identify if there has been a significant increase in the relative abundance of colonial taxa in the reference lakes from pre-1900 to present. Finally, changes in the pH of the reference lakes were assessed. This was done using a 71-lake Adirondack lake training set that spanned a pH gradient from just over 4 to ~8 (Cumming et al. 1992a), using a simple weighted-averaging model based on a ln(x + 1) species transformation with tolerance down-weighting and inverse deshrinking (Cumming et al. 1994) using the program C2 v. 1.72.
Results
Reference lakes and the contemporary distribution of scaled-chrysophyte assemblages
In general, the reference lakes are small (mean SA: 20.5 ha), moderately deep (mean Zmax: 10.9 m), and are located at elevations greater than 300 m (Table 1 and Electronic Supplementary Material 1). The mean pH of the reference lakes was approximately 6.4 when sampled in 2010 and 2011 and 28 lakes (90 %) had ANC values greater than 50 μeq L−1. The lakes were calcium-poor (mean Ca2+ = 2.3 mg L−1) with a DOC range of ~2.1 mg C L−1 to 8.8 mg C L−1. All lakes had Cl− concentrations less than 0.5 mg L−1 and TP data available for 20 lakes suggests the sites are oligotrophic (mean TP = 1.3 µg L−1). Most of the reference lakes are protected in either ‘wild forest’ or ‘wilderness’ preserves in the Adirondack Park. The two most dominant land-cover types in the watersheds of the reference lakes, excluding open water, are deciduous forest and coniferous forest. The least common land-cover type is developed land (Table 1). A PCA ordination of the physical and/or chemical variables of the 1469 lakes in the ALS dataset showed that the reference lakes broadly encompass the characteristics of between 24 to 36 % of the physical and/or chemical characteristics of Adirondack lakes (Arseneau 2014).
Analysis of historic maps (1890 and 1916) of the watersheds surrounding the 31 reference lakes revealed that in 1890, 24 lakes (77 %) were located in minimally disturbed forests. In 1916, 13 lakes (42 %) were classified as 100 % green timber (termed Class-1 reference lakes), 6 (19 %) had experienced at least 1 form of disturbance in less than 50 % of their watershed (termed Class-2 reference lakes), and 12 (39 %) had experienced at least 1 form of disturbance in ≥50 % of their watershed (termed Class-3 reference lakes) (Arseneau 2014). Forest fires were the most common form of anthropogenic disturbance experienced by the reference lakes in 1916, followed by selective logging, and agricultural clearance. Available data on fisheries, from a variety of government sources, showed that, as of 2011, 20 lakes (65 %) had a record of some level of fisheries management involving rotenone use and subsequent stocking of fish such as lake trout. These historical analyses demonstrated that our 31 reference lakes have experienced some varying degree of historic landscape disturbance and fisheries alterations, as is typical of lakes in the region (Stager and Sanger 2003). Despite these early disturbances, the reference sites provide the opportunity to examine regional changes in species composition independent of many common contemporary stressors: acidification, eutrophication due to watershed/shoreline development, road salt seepage, and non-native piscivores introductions.
The gradients in water chemistry present in the 31-lake reference dataset were summarized by the first four axes of a PCA, which represented ~72 % of the variation measured in physical/chemical environmental variables (Fig. 3a, b). The first two axes of the PCA are strong (λ1 = 0.27, and λ2 = 0.21) and axis-1 scores broadly represent a gradient positively related to lake-water pH, ANC and cation/anion concentration, whereas PCA axis-2 is positively associated with lake depth and low concentrations of DOC/TN, with pH loading positively on both axis 1 and 2 (Fig. 3a). The third and fourth PCA axes account for half of the variance of the first two axes (λ3 = 0.14, and λ2 = 0.10), with PCA axis-3 positively related to pH and ANC, and negatively related to SO4 2−, and PCA axis-4 is positively related to DOC and negatively related to elevation and K+ (Fig. 3b).
Principal components analysis (PCA) of environmental variables. a Biplot of PCA axes 1 and 2. b Biplot of PCA axes 3 and 4. Input variables are: pH, acid neutralizing capacity (ANC, μeq L−1), K, Na, SO4 2− (μmol L−1), Cl−, Mg2+ (μmol L−1, log transformed), dissolved organic carbon (DOC, μmol C L−1, log transformed), total nitrogen (TN, μmol L−1, log transformed), Elevation (elv, m), and maximum depth (Zmax, m, log transformed). Environmental variables are closed arrows and samples are shown as points. Eigenvalues (λ) are given
The chemical/physical PCA axis scores of the first four axes were used as a reduced set of possible explanatory variables to understand the relationship of these variables to the contemporary distribution of scaled-chrysophytes in the reference lakes. The initial RDA using all environmental variables explained a significant proportion of the species variance along all canonical axes (p = 0.002, variance explained = 24.7 %). An RDA using forward selection and permutation tests identified PCA axis-1 and axis-2 scores as significant predictors of the variation in the chrysophyte assemblages (PCA axis-2: F = 3.6, p = 0.002; PCA axis-1: F = 2.8, p = 0.007), accounting for 19.1 % of the variance (Fig. 4). The first axis of the RDA ordination (λ1 = 0.14) separated higher abundances of positively-loading species including Synura petersenii Korshikov (and to a lesser extent Mallomonas elongata Reverdin and Mallomonas crassisquama (Asmund) Fott) from higher abundances of negatively loading species including Synura sphagnicola Korshikov (and to a lesser extent Mallomonas acaroides sensu Iwanoff, Mallomonas ‘small’, M. duerrschmidtiae, and Synura echinulata Korshikov). As such, S. petersenii achieved higher abundances on average in deeper lakes with circumneutral to higher pH values, with M. crassisquama achieving higher abundances in high ANC lakes of intermediate depth. Conversely, higher abundances of S. sphagnicola were found in shallower, slightly acidic to circumneutral sites with higher concentrations of DOC. However, S. sphagnicola did achieve higher abundances in some deeper lakes with lower concentrations of DOC (e.g., Deer Pond). RDA axis-2 was much weaker (Fig. 4, λ2 = 0.05), and weakly separated a number of chrysophyte taxa. Together with RDA axis-1, M. crassisquama tended to be found in higher ANC sites that were slightly deeper (positive loading from PCA axis-1 and axis-2 scores), whereas a number of taxa (e.g. S. echinulata, and M. ‘small’, amongst others) tended to be relatively more abundant in circumneutral to higher ANC sites, which were shallower with higher DOC (Fig. 4).
Redundancy analysis (RDA) using PCA axes scores as input. a RDA of present-day samples from 31 reference lakes. PCA axes (environmental variables) are open arrows, species are closed arrows, and samples are shown as points. Eigenvalues (λ) are given. b The same as above, but with the input variables from the PCA of environmental variables plotted passively in the ordination (grey arrows). See Fig. 3 caption for environmental variable units and transformations
Pre-1900 to present-day change in scaled chrysophytes
A total of 17 chrysophyte taxa, from three genera, were present at above the cut-off of achieving ≥2 % relative abundance in the 31 reference lakes, although scales were absent from the 20-cm interval of 5 lakes. There were 10 taxa from the genus Mallomonas, 6 taxa from the genus Synura, and 1 taxon from the genus Chrysosphaerella (Electronic Supplementary Material 2). The PCA ordination of both the modern and pre-1900 samples from the 26 lakes summarizes the changes in species assemblages (Fig. 5a), representing ~46 % of the variation in the 26 lakes since pre-1900 times. In these 26 lakes, there is a shift in assemblage composition in the PCA that can be summarized as a shift from a Mallomonas-dominated assemblage to one more dominated by increasing abundances of colonial Synura taxa (i.e., a movement generally upward and to the right on the PCA ordination diagram, Fig. 5b, c). Unicellular taxa such as M. duerrschmidtiae, Mallomonas pseudocoronota Prescott, M. crassisquama, and Mallomonas lychenensis Conrad (lower left quadrant of the PCA, Fig. 5a) have generally declined in relative abundance from pre-1900 to present. In contrast, Mallomonas punctifera Korshikov (bottom-right quadrant, Fig. 5a) increased in some lakes since the 1900s, as did M. elongata (upper-left quadrant, Fig. 5a). The ANOSIM test supports the shift shown in the PCA, with a significant difference in species composition from pre-1900 to present (R = 0.12; p = 0.002), with SIMPER showing that the changes were largely driven by increases in S. petersenii, S. sphagnicola, and S. echninulata, and decreases in unicellular taxa including M. duerrschmidtiae, M. pseudocoronata and other taxa (Table 1). Overall, there has been a significant increase in the total relative abundance of colonial taxa since pre-industrial times (Fig. 6, Wilcoxon signed-rank test, Z = 3.9; p < 0.0001). The only colonial chrysophyte taxon that generally had a higher relative abundance in the pre-1900 samples was Synura curtispina (Peter and Hansen) Asmund (Electronic Supplementary Material 2).
Principal components analysis (PCA) of chrysophyte assemblages in top (present-day) and bottom (pre-1900) samples in 26 reference lakes. The PCA is summarized in multiple panels to reduce crowding in biplots. a Species vectors. b Present-day (filled circle) and pre-1900 (empty square) samples. Dashed lines show trajectory of species change in ordination space from pre-1900 to present. c Present-day samples only (labelled). Eigenvalues (λ) are given
Total relative abundance (%) of colonial chrysophyte taxa in top (present-day) and bottom (pre-1900) samples in 26 reference lakes that had both ‘top’ and ‘bottom’ assemblages. Grey line represents a one-to-one line. All samples above the grey line have shown an increase in the total relative abundance of colonial chrysophytes from pre-1900 to present
Inferences of lake-water pH based on changes in the chrysophyte assemblages show that chrysophyte-inferred pH (CI-pH) has increased, decreased, or remained unchanged between the pre-1900 and present-day samples (See Electronic Supplementary Material 2). Only two lakes (Middle Branch Lake and Bass Lake) showed a decline in CI-pH greater than the RMSEP (0.51) (Table 2).
Discussion
Present-day distribution of scaled chrysophytes in reference lakes and change over time
Numerous studies have shown the importance of pH for determining chrysophyte species distribution in the Adirondacks and elsewhere (Cumming et al. 1992a; Hyatt et al. 2010). In this investigation, even though the pH gradient in the reference lakes was relatively narrow (~5.9–6.8), the Redundancy Analysis demonstrated that lakes with higher pH values and deeper maximum depths (higher PCA axis-1 and PCA axi-2 scores) were characterized by a higher relative abundance of S. petersenii. Flear (2011) found a similar separation in a study of 40 minimally disturbed lakes in the Experimental Lakes Area in northwest Ontario. In the Adirondack lakes, deep, higher-pH lakes (i.e., generally pH > 6.5) were characterized by a higher relative abundance of S. petersenii and shallower, lower-pH lakes were characterized by a higher relative abundance of S. sphagnicola, M. acaroides, and M. punctifera. S. sphagnicola was abundant in shallower, high-DOC sites (low PCA axis-2 scores) but did occur at relatively high relative abundances in deeper low-DOC sites, suggesting that pH could be more important in determining the abundance of S. sphagnicola than DOC/depth. In contrast, a close examination of the DOC data reveals that S. petersenii was less abundant (<~10 % relative abundance) in lakes with a DOC concentration greater than ~5.2 mg C L−1 (Arseneau 2014). Therefore, high DOC levels may be related to the lower abundance of S. petersenii in some reference lakes.
The fact that the RDA explained only approximately 19.1 % of the variation in the present-day chrysophyte assemblages suggests that important explanatory variables were not included in the analysis. Lake thermostructure, for example, is known to influence chrysophyte species composition (Siver et al. 1995; Pla-Rabes and Catalan 2011). Some chrysophyte taxa are considered warm-water species (e.g. M. punctifera) and during long periods of thermal stratification, large metalimnetic populations of both colonial (Synura, Chrysosphaerella) and unicellular (Mallomonas caudata Ivanov em. Krieger, M. acaroides, M. crassisquama) taxa can occur (Siver 1991, 2003). The fact that maximum depth was important for structuring chrysophyte assemblages in the reference lakes suggests that lake thermostructure may be an important control on chrysophyte distribution in the region. Biological factors may also be an important control on chrysophyte species distribution. Large colonial chrysophytes likely have a size refuge from small herbivores but may be susceptible to predation by large daphniids (Sandgren and Walton 1995).
The Adirondack reference lakes have experienced a significant shift in species composition from pre-1900 to present, with the present-day samples characterized by an increased abundance of colonial taxa. Both univariate and multivariate analyses found that there has been a significant increase in the relative abundance of colonial taxa from pre-1900 to present, along with increases in some unicellular taxa in some lakes (e.g., M. punctifera, M. elongata). Because of the regional distribution of the reference lakes, we can conclude that regional rather than local stressors are likely responsible for the changes in chrysophyte species composition. Furthermore, because of the reference lake study design, we can conclude that these increases are not due to: acidification, eutrophication (from shoreline or watershed development), salinization, or introduced piscivores. Additionally, comparisons of lake classes and lakes that had or had not experienced reclamation suggested that both historic fisheries disturbance and watershed disturbance have minimal influence on the present-day chrysophyte taxa in the reference lakes (Arseneau 2014). The RDA results suggest that both physical and chemical characteristics may control the distribution of chrysophyte taxa. Of particular interest are the controls on S. petersenii, a taxon that has been shown to cause taste and odour issues in lakes (Nicholls and Gerrath 1985). The results of this investigation suggest that S. petersenii is more common in deeper lakes with higher pH values and DOC concentrations less than ~5.2 mg C L−1. Nuisance S. petersenii blooms are unlikely to be a concern in the many Adirondack lakes which are small (SA < 4 ha, Zmax < 5 m) or relatively acidic (pH < 6) (Kretser et al. 1989). However, lakes which support sportfish like lake trout are often fairly large, deep, and characterized by higher pH and hence may provide suitable habitat for S. petersenii. Increases in colonial chrysophyte taxa (and S. petersenii in particular) may pose a management concern (Paterson et al. 2004), and so it is important to discuss the possible causal mechanisms behind the increases in colonial chrysophyte taxa noted in this investigation.
Given the large impact atmospheric deposition has had in the Adirondack region, it is important to consider whether or not the changes in chrysophyte species composition noted in this investigation could be due to long-term effects of acid deposition. CI-pH reconstructions suggest that the majority of the reference lakes are unlikely to have acidified (i.e. experienced decreases in pH/ANC to low values) but atmospheric deposition may have resulted in other changes. Increased leaching of cations with acid deposition is unlikely to be an important driver of the species changes noted from pre-1900 to present as chrysophyte taxa are broadly tolerant of a range of cations, though increased long-distance transport and deposition of certain trace metals may be important (i.e. Se, a micronutrient related to chrysophyte blooms, Sandgren 1988). Long-term exposure to acid deposition may also increase the ionic strength of soil solutions, decreasing DOC flux to nearby waterbodies (Monteith et al. 2007). Decreases in DOC flux, in turn, would increase light penetration in the water column and improve light conditions for metalimnetic colonial chrysophytes (Healy 1983). However, while DOC may influence the distribution of S. petersenii in the Adirondack reference lakes, S. sphagnicola was present in both high-DOC and low-DOC sites (Fig. 4). Long-term exposure to acid deposition may also decrease the flux of phosphorus to lakes by increasing the retention of TP in watershed soils (Eimers et al. 2009). Furthermore, logging and subsequent forest regrowth may also decrease TP export to nearby lakes, and many of the reference sites experienced early logging disturbances (Hall and Smol 1996). The fact that M. lychenensis and S. curtispina decreased in abundance from pre-1900 to present in some lakes (Electronic Supplementary Material 2) suggests that total phosphorus (TP) concentrations may have decreased in those sites, as both taxa are associated with higher lake trophic status (Siver and Marsicano 1996). Under the low-TP conditions characteristic of Adirondack lakes (Crowley et al. 2012), the metalimnetic position of colonial chrysophytes may provide a competitive advantage by allowing access to the more nutrient-rich hypolimnetic waters (Nicholls 1995). Interestingly, though unicellular taxa generally declined in relative abundance from pre-1900 to present, a few lakes showed increases in the relative abundance of M. caudata and M. elongata, both of which can form metalimnetic blooms (Siver 2003). Thus, oligotrophication may have contributed to the changes in chrysophyte assemblages noted in the reference lakes.
Regional warming may have also contributed to the changes in species composition noted in the reference lakes from pre-1900 to present. The Adirondack region has warmed at a rate of 1.3 °C per 100 years, a warming trend associated with an increased ice-free period in Adirondack lakes (Jenkins 2010). The increases in M. punctifera (a warm-water taxon, Siver 1991) noted in some reference lakes may therefore be due to warming surface waters. Similarly, an extended ice-free season may benefit taxa like S. sphagnicola which persists in the water column through summer and fall and is lost at ice-on (Siver and Hamer 1992). Longer ice-free periods can also lead to a longer period of summer stratification, which may provide motile algae like chrysophytes a competitive advantage over non-motile algae, like diatoms, because they can seek out optimal temperature/nutrient/light conditions in the water column (Winder and Sommer 2012). At the assemblage level, longer periods of stratification may provide a competitive advantage to colonial chrysophyte taxa over unicellular ones because large flagellates tend to be superior swimmers, with faster swim velocities and larger migration amplitudes (Sommer 1988). Longer periods of stratification can also result in increased nutrient limitation in the epilimnion, providing chrysophytes that form metalimnetic blooms with a competitive advantage because of their access to nutrient-rich hypolimnetic waters. Thus, the increases in colonial taxa noted in this investigation, along with increases in certain unicellular taxa like M. punctifera, are likely related at least in part to regional warming.
It is possible that the long-term effects of regional warming and/or acid deposition may have also resulted in changes in biotic conditions that could have contributed to the increased abundance of colonial chrysophytes. Large colonial chrysophytes likely have a size refuge from small zooplankton (Bosmina, calanoid and cyclopoid copepods) but can be predated upon by large Daphnia taxa (i.e. >1 mm in size) (Sandgren and Walton 1995). It has been hypothesized that regional warming may benefit small zooplankton (Daufresne et al. 2009). Furthermore, long-term exposure to acid deposition may lead to depletion of soil available Ca in watershed soils, a phenomenon that may negatively affect Ca-sensitive Daphnia (Jeziorski et al. 2008). A subsequent paleolimnological study of cladoceran assemblages in the reference lakes could determine whether or not cladoceran assemblages have undergone a significant shift in species composition from pre-1900 to present and what link, if any, those changes may have to changes in colonial chrysophyte abundance in Adirondack lakes.
Methodological considerations: reference site approaches in non-protected areas
Our work to identify minimally disturbed reference sites was supported by the fact that the research was being undertaken in a large state park with lands designated for protection over a century ago. It may not be possible for researchers and managers working in developed areas to identify ‘minimally disturbed’ reference sites, but alternative frameworks exist. In highly developed areas of the United States, it is difficult if not impossible to identify minimally disturbed reference sites. In such cases, managers can use the least disturbed sites in a region to define recovery targets (Stoddard et al. 2006). For example, the ‘best attainable condition’ for a lake in a region with intensive agriculture may not necessarily be one without agrarian development (a minimally disturbed condition) but, rather, one characterized by low levels of agricultural development (e.g. <10 %). Similarly, it is well-recognized that minimally disturbed reference sites are rare in parts of Europe (Bjerring et al. 2008). As an alternative, Bennion et al. (2011b) used a modern analogue approach to identify reference sites. In that study, the historical (baseline) diatom species assemblages of 9 lakes were compared to the present-day sedimentary diatom assemblages of 347 European lakes. Lakes that had a square-chord distance measure of <0.75 were considered modern analogues of the study lakes’ historic condition, and so were taken to represent reference sites. While a useful approach, there are two important limitations. First, although a lake may have a present-day species composition similar to the historic condition of another site, such a reference site could have experienced significant human impact over time, complicating an attempt to characterize a shifting baseline due to climate change. Secondly, if climate warming results in particular ‘syndromes’ of changes in species composition (i.e. an increase in warm-water species, small-bodied taxa, taxa with a high surface-to-volume ratio, etc., Daufresne et al. 2009; Winder and Sommer 2012), then the likelihood of identifying modern analogues that can serve as reference sites may decrease over time.
Importance of reference lake results for recovery studies
Arguably, the most important finding of this study is simply that minimally disturbed reference lakes in the Adirondack Park show a pronounced shift in species composition from pre-1900 to present, likely due to climate warming and/or oligotrophication. Thus, it should be expected that lakes in an area recovering from acidification will not return to their pre-disturbance state. Rather, the recovery endpoints for acidified lakes will be different from their pre-disturbance condition—a state characterized by an increased abundance of colonial and/or warm-water taxa. Repeating the top–bottom analysis of the reference sites with other paleo-indicators (diatoms, cladocera, chironomids, etc.) will allow the assessment of shifting baselines in other groups of aquatic organisms. Moreover, by pairing both acidified and reference lakes in subsequent paleolimnological studies, we will be able to determine whether or not the recovery trajectories of impacted sites suggest that they are moving towards a novel ‘recovered’ condition similar to the reference sites. While a growing number of studies have shown chemical recovery from acidification, lakes are increasingly influenced by multiple anthropogenic stressors, making the return of biological assemblages to a pre-disturbance state unlikely. Paleolimnology can, and should, play an important role in recovery studies because paleolimnological techniques can both identify species changes in impacted sites and, as we have illustrated, be applied in reference lakes to identify shifting baselines, thereby informing recovery targets.
References
Arseneau KMA (2014) Acidification and climate warming: Understanding the impact of multiple anthropogenic stressors on Adirondack (NY, USA) lakes. Ph.D. dissertation, Queen’s University, Kingston
Arseneau KMA, Driscoll CT, Brager LM, Ross KA, Cumming BF (2011) Recent evidence of biological recovery from acidification in the Adirondacks (New York, USA): a multiproxy paleolimnological investigation of Big Moose Lake. Can J Fish Aquat Sci 68:575–592
Bailey RC, Norris RH, Reynoldson TB (2004) Bioassessment of freshwater ecosystems: using the reference condition approach. Kluwer Academic Publishers, New York
Battarbee RW, Jones VJ, Flower RJ, Cameron NG, Bennion H, Carvalho L, Juggins S (2001) Diatoms. In: Smol JP, Birks HJB, Last WM (eds) Tracking environmental change using lake sediments, volume 3: terrestrial, algal, and siliceous indicators. Kluwer Academic Publishers, Dordrecht, pp 155–202
Battarbee RW, Anderson NJ, Bennion H, Simpson GL (2012) Combining limnological and palaeolimnological data to disentangle the effects of nutrient pollution and climate change on lake ecosystems: problems and potential. Freshw Biol 57:2091–2106
Battarbee RW, Simpson GL, Shilland EM, Flower RJ, Kreiser A, Yang H, Clarke G (2014) Recovery of UK lakes from acidification: an assessment using combined palaeoecological and contemporary diatom assemblage data. Ecol Indic Part B 37:365–380
Bennion H, Battarbee RW, Sayer CD, Simpson GL, Davidson TA (2011a) Defining reference conditions and restoration targets for lake ecosystems using palaeolimnology: a synthesis. J Paleolimnol 45:533–544
Bennion H, Simpson GL, John Anderson N, Clarke G, Dong X, Hobæk A, Guilizzoni P, Marchetto A, Sayer CD, Thies H, Tolotti M (2011b) Defining ecological and chemical reference conditions and restoration targets for nine European lakes. J Paleolimnol 45:415–431
Bjerring R, Bradshaw EG, Amsinck SL, Johansson LS, Odgaard BV, Nielsen AB, Jeppensen E (2008) Inferring recent changes in the ecological state of 21 Danish candidate reference lakes (EU Water Framework Directive) using palaeolimnology. J Appl Ecol 45:1566–1575
Brucet S, Poikane S, Lyche-Solheim A, Birk S (2013) Biological assessment of European lakes: ecological rationale and human impacts. Freshw Biol 58:1106–1115
Catalan J, Pla-Rabés S, Wolfe AP, Smol JP, Rühland KM, Anderson NJ, Kopáček J, Stuchlík E, Schmidt R, Koinig KA, Camarero L, Flower RJ, Heiri O, Kamenik C, Korhola A, Leavitt PR, Psenner R, Renberg I (2013) Global change revealed by palaeolimnological records from remote lakes: a review. J Paleolimnol 49:513–535
Chessman BC, Muschal M, Royal MJ (2008) Comparing apples with apples: use of limiting environmental differences to match reference and stressor-exposure sites for bioassessment of streams. River Res Appl 24:103–117
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Aust J Ecol 18:117–143
Clarke KR, Warwick RM (2001) Change in marine communities: an approach to statistical analysis and interpretation, 2nd edn. PRIMER-E Ltd., Plymouth
Crowley KF, McNeil BE, Lovett GM, Canham CD, Driscoll CT, Rustad LE, Denny E, Hallett RA, Arthur MA, Boggs JL, Goodale CL, Kahl JS, McNulty SG, Ollinger SV, Pardo LH, Schaberg PG, Stoddard JL, Weand MP, Weathers KC (2012) Do nutrient limitation patterns shift from nitrogen toward phosphorus with increasing nitrogen deposition across the Northeastern United States? Ecosystems 15:940–957
Cumming BF, Smol JP, Birks HJB (1992a) Scaled chrysophytes (Chrysophyceae and Synurophyceae) from Adirondack drainage lakes and their relationship to environmental variables. J Phycol 28:162–178
Cumming BF, Smol JP, Kingston JC, Charles DF, Birks HJB, Camburn KE, Dixit SS, Uutala AJ, Selle AR (1992b) How much acidification has occurred in Adirondack region lakes (New York, USA) since preindustrial times? Can J Fish Aquat Sci 49:128–141
Cumming BF, Davey KA, Smol JP, Birks HJB (1994) When did acid-sensitive Adirondack lakes (New York, USA) begin to acidify and are they still acidifying? Can J Fish Aquat Sci 51:1550–1568
Daufresne M, Lengfellner K, Sommer U (2009) Global warming benefits the small in aquatic ecosystems. P Natl Acad Sci USA 106:12788–12793
Dixit SS, Dixit AS, Smol JP (1999) Lake sediment chrysophyte scales from the northeastern USA and their relationship to environmental variables. J Phycol 35:903–918
Downes BJ, Barmuta LA, Fairweather PG, Faith DP, Keough MJ, Lake PS, Mapstone BD, Quinn GP (2002) Monitoring ecological impacts: concept and practice in flowing waters. Cambridge University Press, Cambridge
Driscoll CT, Newton RM, Gubala CP, Baker JP, Christensen S (1991) Adirondack mountains. In: Charles DF (ed) Acidic deposition and aquatic ecosystems: regional case studies. Springer, New York, pp 133–202
Driscoll CT, Lawrence GB, Bulger AJ, Butler TJ, Cronan CS, Eagar C, Lambert KF, Likens GE, Stoddard JL, Weathers KC (2001) Acidic deposition in the northeastern United States: sources and inputs, ecosystem effects, and management strategies. Bioscience 51:180–198
Eimers MC, Watmough SA, Paterson AM, Dillon PJ, Yao HX (2009) Long-term declines in phosphorus export from forested catchments in south-central Ontario. Can J Fish Aquat Sci 66:1682–1692
Flear K (2011) Changes in scaled-chrysophyte assemblages in response to recent climate change in Northwestern Ontario. M.Sc., dissertation, Queen’s University Kingston, Ontario
George CJ (1981) The fishes of the Adirondacks. New York State Department of Environmental Conservation, Albany
Ginn BK, Rate M, Cumming BF, Smol JP (2010) Ecological distribution of scaled-chrysophyte assemblages from the sediments of 54 lakes in Nova Scotia and southern New Brunswick, Canada. J Paleolimnol 43:293–308
Hall RI, Smol JP (1996) Paleolimnological assessment of long-term water-quality changes in south-central Ontario lakes affected by cottage development and acidification. Can J Fish Aquat Sci 53:1–17
Healy FP (1983) Effect of temperature and light intensity on the growth rate of Synura sphagnicola. J Plankton Res 5:767–774
Hobbs RJ, Arico S, Aronson J, Baron JS, Bridgewater P, Cramer VA, Epstein PR, Ewel JJ, Klink CA, Lugo AE, Norton D, Ojima D, Richardson DM, Sanderson EW, Valladares F, Vilà M, Zamora R, Zobel M (2006) Novel ecosystems: theoretical and management aspects of the new ecological world order. Glob Ecol Biogeogr 15:1–7
Hobbs RJ, Higgs E, Harris JA (2009) Novel ecosystems: implications for conservation and restoration. Trends Ecol Evol 24:599–605
Hyatt CV, Paterson AM, Cumming BF, Smol JP (2010) Factors related to regional and temporal variation in the distribution of scaled chrysophytes in northeastern North America: evidence from lake sediments. Nova Hedwigia 136:87–102
Jenkins J (2010) Climate change in the Adirondacks: the path to sustainability. Cornell University Press, Ithaca
Jenkins JC, Roy K, Driscoll C, Buerkett C (2007) Acid rain in the Adirondacks: an environmental history. Cornell University Press, Ithaca
Jeziorski A, Yan ND, Paterson AM, DeSellas AM, Turner MA, Jeffries DS, Keller B, Weeber RC, McNicol DK, Palmer ME, McIver K, Arseneau K, Ginn BK, Cumming BF, Smol JP (2008) The widespread threat of calcium decline in fresh waters. Science 322:1374–1377
Jeziorski A, Paterson A, Smol J (2012) Crustacean zooplankton sedimentary remains from calcium-poor lakes: complex responses to threshold concentrations. Aquat Sci 74:121–131
Kling HJ, Kristiansen J (1983) Scale-bearing Chrysophyceae (Mallomonadaceae) from Central and Northern Canada. Nord J Bot 3:269–290
Korhola A, Rautio M (2001) Cladocera and other branchiopod crustaceans. In: Smol JP, Birks HJB, Last WM (eds) Tracking environmental change using lake sediments, vol 4., Zoological indicatorsKluwer Academic Publishers, Dordrecht, pp 5–41
Kraft CE, Carlson DM, Carlson M (2006) Inland fishes of New York (online). Version 4.0. Department of Natural Resources, Cornell University, and the New York State Department of Environmental Conservation
Kretser W, Gallagher J, Nicolette J (1989) Adirondack lakes survey 1984–1987: an evaluation of fish communities and water chemistry. Adirondack Lakes Survey Corporation, Ray Brook
Lepš J, Šmilauer P (2003) Multivariate analysis of ecological data using CANOCO. Cambridge University Press, Cambridge
Monteith DT, Stoddard JL, Evans CD, de Wit HA, Forsius M, Høgasen T, Wilander A, Skjelkvåle BL, Jeffries DS, Vuorenmaa J, Keller B, Kopácek J, Vesely J (2007) Dissolved organic carbon trends resulting from changes in atmospheric deposition chemistry. Nature 450:537–540
Newton RM, Driscoll CT (1990) Classificiation of ALSC lakes. Adirondack lakes survey: an interpretive analysis of fish communities and water chemistry, 1984–1987. Adirondack Lakes Survey Corporation, Ray Brook
Nicholls KH (1995) Chrysophyte blooms in the plankton and neuston of marine and freshwater systems. In: Sandgren CD, Smol JP, Kristiansen J (eds) Chrysophyte algae: Ecology, phylogeny and development. Cambridge University Press, Cambridge, pp 181–213
Nicholls KH, Gerrath JF (1985) The taxonomy of Synura (Chrysophyceae) in Ontario with special reference to taste and odor in water-supplies. Can J Bot 63:1482–1493
Paterson AM, Cumming BF, Smol JP, Hall RI (2004) Marked recent increases of colonial scaled chrysophytes in boreal lakes: implications for the management of taste and odour events. Freshw Biol 49:199–207
Pla-Rabes S, Catalan J (2011) Deciphering chrysophyte responses to climate seasonality. J Paleolimnol 46:139–150
Sandgren CD (1988) The ecology of chrysophyte flagellates: their growth an dperennation strategies as freshwater phytoplankton. In: Sandgren CD (ed) Growth and reproductive strategies of freshwater phytoplankton. Cambridge Univeristy Press, Cambridge, pp 9–104
Sandgren CD, Walton WE (1995) The influence of zooplankton herbvory on the biogeography of chrysophyte algae. In: Sandgren CD, Smol JP, Kristiansen J (eds) Chrysophyte algae: ecology, phylogeny and development. Cambridge University Press, Cambridge, pp 269–302
Siver PA (1991) The biology of Mallomnas: morphology, taxonomy and ecology. Kluwer Academic Publishers, Dordrecht
Siver PA (2003) Synurophyte algae. In: Wehr JD (ed) Freshwater algae of North America: Ecology and classification. Academic Press, San Diego, pp 523–558
Siver PA, Hamer JS (1992) Seasonal periodicity of chrysophyceae and synurophyceae in a small New England lake: implications for paleolimnological research. J Phycol 28:186–198
Siver P, Marsicano L (1996) Inferring lake trophic status using scaled chrysophytes. Nova Hedwigia 114:233–246
Siver PA, Sandgren CD, Smol JP, Kristiansen J (1995) The distribution of chrysophytes along environmental gradients: their use as biological indicators. In: Sandgren CD, Smol J, Kristiansen J (eds) Chrysophyte algae: ecology, phylogeny and development. Cambridge University Press, Cambridge, pp 232–268
Sommer U (1988) Some size relationships in phytoflagellate motility. Hydrobiologia 161:125–131
Soranno PA, Wagner T, Martin SL, McLean C, Novitski LN, Provence CD, Rober AR (2011) Quantifying regional reference conditions for freshwater ecosystem management: a comparison of approaches and future research needs. Lake Reserv Manag 27:138–148
Stager JC, Sanger T (2003) An Adirondack “heritage lake”. AJES 10:6–10
Stoddard JL, Larsen DP, Hawkins CP, Johnson RK, Norris RH (2006) Setting expectations for the ecological condition of streams: the concept of reference condition. Ecol Appl 16:1267–1276
ter Braak CJF, Šmilauer P (2012) CANOCO reference manual and user’s guide: software for ordination (version 5.0). Microcomputer Power, Ithaca
United States Environmental Protection Agency (USEPA) (2012) Implications of climate change for state bioassessment programs and approaches to account for effects (final report). U.S. Environmental Protection Agency, Washington
United States Geological Survey (USGS) (2013) Nonindigenous aquatic species database. U.S. Geological Survey, Gainesville
Vinebrooke RD, Cottingham KL, Norberg M, Scheffer J, Dodson SI, Maberly SC, Sommer U (2004) Impacts of multiple stressors on biodiversity and ecosystem functioning: the role of species co-tolerance. Oikos 104:451–457
Waller K, Driscoll C, Lynch J, Newcomb D, Roy K (2012) Long-term recovery of lakes in the Adirondack region of New York to decreases in acidic deposition. Atmos Envion 46:56–64
Winder M, Sommer U (2012) Phytoplankton response to a changing climate. Hydrobiologia 698:5–16
Acknowledgments
Funding for this research comes from the Natural Sciences and Engineering Research Council (NSERC) Discovery grant awarded to Dr. B.F. Cumming, an NSERC CGS-D grant awarded to K.M.A. Arseneau, and support from the New York State Energy Research and Development Authority (NYSERDA) to C.T. Driscoll. The Adirondack Lakes Survey (ALS) dataset used in this manuscript is maintained online by the Adirondack Lakes Survey Corporation (ALSC). The ALSC receives support from NYSERDA, the New York State Department of Environmental Conservation, and the US Environmental Protection Agency. The authors would like to thank Dr. J.C. Stager (Paul Smith’s College, NY) for his informative discussion of Adirondack ‘Heritage Lakes’ that was inspirational in the design of this study, the summer field crews who help collect samples from the reference lakes (A. Fenton, M. Kelly, I. MacKenzie, C. Robinson, and J. Velasco), and the members of the Paleoecological Environmental Assessment and Research Laboratory (Queen’s University) for their support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Arseneau, K.M.A., Driscoll, C.T., Cummings, C.M. et al. Adirondack (NY, USA) reference lakes show a pronounced shift in chrysophyte species composition since ca. 1900. J Paleolimnol 56, 349–364 (2016). https://doi.org/10.1007/s10933-016-9922-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10933-016-9922-2