Abstract
Density dependent numerical response is considered advantageous for biocontrol agents. Although density dependent behaviors have been examined in aphidophagous ladybirds, they haven’t been studied for coccidophagous ladybirds. The relationship of prey density and prey oviposition stage on the aggregating behavior and oviposition of the coccidophagous predator Nephus includens (Coleoptera: Coccinellidae) was empirically investigated. In a semi-field experiment potted citrus trees were artificially infested with Planococcus citri (Hemiptera: Pseudococcidae) prey in order to obtain two densities, one mealybug and ten mealybugs per tree. Released N. includens adults were found to aggregate at citrus trees with high prey density. In a subsequent laboratory experiment four prey densities of P. citri were used ranging from 1 to 8 adult mealybugs at 3 ovipositional stages, a) at the young gravid female stage, b) at mid oviposition (half of egg load oviposited) and c) at the end of the ovipositional period (full egg load oviposited). Nephus includens females increased their oviposition rate as prey density increased and as prey oviposition progressed. It is hypothesized that this behavior of the predator might be advantageous for offspring survival and the efficacy of N. includens as a biological control agent against mealybugs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Predators that respond numerically by aggregating attacks or by ovipositing in patches with high prey density are promising biocontrol agents (Mills and Lacan 2004). However, the outcome of this aggregating behavior in stabilizing predator–prey dynamics has created considerable debate (Godfray and Pacala 1992; Hassell and May 1973; Murdoch and Briggs 1996; Murdoch et al. 1985). Several studies have examined aggregating responses of predators to prey density using ladybirds (Coccinellidae). In some studies positive numerical responses by coccinellid species have been reported (Elliott and Kieckhefer 2000; Evans and Youssef 1992; Kareiva 1990) while other studies on the same or other species have not shown a similar response (Coderre et al. 1987; Schellhorn and Andow 2005).
The optimal foraging theory is also applicable to ovipositing female insects that seek suitable sites to lay eggs (Mangel 1987). A foraging female seeking oviposition sites should accept a patch if it is above a certain level of quality or she might choose to leave the patch in seeking for future fitness increment (Mangel 1987). This assumes that foraging females are omniscient to their environment at some extent (Bernstein et al. 1991). Upon arriving to a specific patch ovipositing females should be capable of assessing its quality in terms of supporting survival of their offspring. This is an important decision that can greatly affect their fitness. For aphidophagous ladybirds an important aspect of patch quality that determines the survival of their offspring is prey density and the developmental stage of the prey colony.
Prey density determines the magnitude of interspecific and intraspecific competition for prey and thus the probability of offspring survival. Apart from prey density, prey colony structure has also been shown to be an important factor for ovipositing females (Hodek and Honěk 2009). Prey quality assessed as prey age and prey mixtures versus single prey instars, affects the behavior of an ovipositing female (Seagraves 2009).
For aphidophagous coccinellids oviposition has been shown to occur above a certain level of prey density (Dixon 1959, 2000). Additionally, aphidophagous coccinellids avoid egg-laying when aphid colony abundance has reached its peak (Dixon 2000; Hemptinne et al. 1992). As a consequence female coccinellids lay most of their eggs within a short period of time in the early stages of an aphid colony, which has been named the egg window (Kindlmann and Dixon 1993).
The present study addresses patch quality assessments by a coccidophagous predator based on prey colony density and age structure of its prey. Our system consists of the mealybug Planococcus citri Risso (Hemiptera: Pseudococcidae) and the coccinellid predator Nephus includens (Kirsch) (Coleoptera: Coccinellidae). Planococcus citri is a widespread mealybug pest species of many economically important plants (horticultural to ornamental) worldwide (Franco et al. 2004). Yield losses occur when mealybugs infest fruit or excrete honeydew that covers the fruits and leaves, often resulting in sooty mold growth, defoliation and sunburned fruit (Walton and Pringle 2005). Nephus includens is a predatory ladybird that preys upon P. citri and other mealybug species (Katsoyannos 1996). Nephus includens is a native widely distributed species in Greece, Italy, Spain, and Portugal (Kontodimas et al. 2007; Magro et al. 1999; Milonas et al. 2011).
In this study we (1) compared the aggregative response of N. includens to P. citri densities, (2) examined the effects of P. citri density on oviposition by the predator and (3) determined how prey oviposition time affected the oviposition behavior of the predator.
Materials and methods
Study organisms
Planococcus citri mealybugs were collected from Citrus sp. (Sapindales: Rutaceae) at Galata (Troizinia-Greece) and reared in the laboratory on potato sprouts (Solanum tuberosum L., Solanales: Solanaceae) at 25 ± 1 °C, 16L: 8D photoperiod and 65 ± 2 % RH, in large plastic boxes (30 × 40 × 15 cm) tightly covered with fine muslin.
Nephus includens was originally collected in 2007 from citrus plants in Preveza County (Northwestern Greece), infested with P. citri. The predator cultures were maintained in the laboratory at 25 ± 1 °C, 16L: 8D photoperiod and 65 ± 2 % RH, in large cylindrical Plexiglass cages (50 cm height × 30 cm diam.) tightly covered with fine muslin that contained 8–12 potato sprouts infested with P. citri prey.
Experiment A: Aggregation behavior of N. includens
To test the effect of prey density on the aggregation of N. includens adults, a semi-field trial was designed. Potted Citrus aurantium L. (Sapindales: Rutaceae) trees (1–1.5 m height) were infested using ovisacs from P. citri mealybugs reared under laboratory conditions to create two levels of prey density. A high prey density tree was infested with 10 ovisacs and a low prey density tree with 1 ovisac. Mealybugs were allowed to establish and when young individuals reached adulthood, infested trees were used in the experiments. Six trees, 3 of each prey density level were placed in a field cage of 2 × 2 × 2 m covered with wired net. Trees were placed in two rows randomly. In each of the three cages 100 N. includens adults were released in the morning at 8 am. Starting 2 h later and at hour intervals afterwards till 6:00 pm the number of adults found on each plant was recorded. Additional counts before sunset and in the morning of the next day were taken. Experiments were conducted during the summer of 2009 at the Benaki Phytopathological Institute in Athens, Greece.
Experiment B: Oviposition by N. includens adults
Experiments were conducted on squash plants (Cucurbita pepo L.) (Cucurbitales: Cucurbitaceae). Seeds were sown in a half and half mixture of perlite and turf. Seeds were placed at 2 cm depth in plastic pots of 5 cm diameter and 8 cm height. Pots were kept in a greenhouse until the seedlings reached the 2-leaf stage. All plants used were at the 2-leaf stage and about 8 cm height. Experiments took place in a laboratory room with constant temperature of 25 °C and RH approximately 50 % with a long day photoperiod 16:8 (L:D). Plants were covered with a plastic cup (10 cm height, 5 cm diam.) that was sealed on top with a plastic lid with a hole of 2 cm diameter covered with fine muslin. A factorial design with two factors was used to examine P. citri density and oviposition status. Plants were allocated to four adult females P. citri densities (1, 2, 4, 8) and at each density P. citri females were either young gravid P. citri females (1) before starting oviposition (day 1), or (2) females at the 4th day of their oviposition period or (3) females at the 7th day of their oviposition period. An adult P. citri female in our cultures had an oviposition period of almost 1 week.
On each plant with the various combinations of mealybug densities and oviposition period, a single mated N. includens female was placed at the beginning of the photophase. Mated females 5 to 10 days old, were collected by observing mating pairs and collecting the females immediately after mating. Female predators were kept in plastic Petri dishes without access to mealybugs for 1 day before being used in the experiments to ensure a high level of oviposition rate. The next day the predator was removed and the eggs laid were counted. Fifteen replicates were established for each treatment combination however in order to get the final numbers for the experiment many more females were allowed to oviposit.
Statistical Analysis
A repeated measures ANOVA was used to determine the influence of prey density and time of the day on the aggregation behavior of N. includens adults with time as the within subject factor and density the between subject factor (Sokal and Rohlf 2012).
The influence of adult P. citri prey density and P. citri oviposition period on the ratio of ovipositing predators was tested with generalized linear model for binomial data with density and oviposition period as predictor variables. An ANOVA model was applied in order to discriminate effects of prey density and mealybug oviposition period on the total number of eggs laid by N. includens females and the number of eggs laid by N. includens females per individual mealybug. ANOVA was followed by a simple main affect analysis for each main factor to interpret the influence of density within each level of oviposition period and vice versa. Data were sqrt (x +1) transformed in order to meet ANOVA requirements for normality and homogeneity of variances. Interaction term was removed from the final model when found not significant (Sokal and Rohlf 2012).
Results
Aggregation experiment
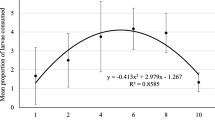
Density significantly influenced the aggregation of N. includens adults (F = 6.9; df = 1, 16; P = 0.018) and time of the day (F = 3.03; df = 10, 160; P = 0.002) while interaction of time and density did not have a significant effect (F = 0.25, df = 10, 160; P = 0.99) (Fig. 1).
Adult prey density and oviposition stage
The ratio of ovipositing N. includens females ranged from 0.33 to 0.87 (Fig. 2). The overall test was found highly significant (χ 2 = 15.4; df = 5; P = 0.009). There was a significant effect of P. citri prey density on the ratio of ovipositing female N. includens predators (χ 2 = 12.5; df = 3; P = 0.006) but not of mealybug oviposition period (χ 2 = 3.5; df = 2; P = 0.178). The interaction term was also not significant (χ 2 = 10.9; df = 6; P = 0.09).
There was a significant interaction effect of prey density and oviposition period of P. citri females (F = 3.59, df = 6, 168; P = 0.0022) on the mean number of eggs that predators laid. However, a simple main effect analysis revealed that at later prey oviposition stages (4th and 7th) the number of predator eggs increased significantly with prey density (F4th = 5.19; df = 3, 168; P = 0.002; (F7th = 13.9; df = 3, 168; P < 0.001) (F1st = 1.39; df = 3, 168; P = 0.248). At higher prey densities (4 and 8) the predator laid a significantly larger number of eggs on prey at more advanced oviposition stages (F4 = 4.44; df = 2, 168; P = 0.013), (F8 = 6.97; df = 2, 168; P = 0.001). At the density of eight mealybugs, a higher number of predator eggs was oviposited when they were at the 4th and 7th day of their oviposition period. At the density of four mealybugs, a higher number of predator eggs was oviposited when they were at 7th day of their oviposition period (Fig. 3). No differences were detected when there were present one or two mealybugs.
The average number of eggs laid per prey is shown in Fig. 4. There was a significant interaction effect of prey density and oviposition period (F = 3.14; df = 6, 97; P = 0.007) on the number of eggs laid per prey. The simple main effect analysis showed that the number of predator eggs per mealybug, on the density of one and four mealybugs, was affected by the oviposition period of the mealybug (F1 = 3.49; df = 2, 97; P = 0.034) (F4 = 4.14; df = 2, 97; P = 0.019). At the densities of 2 and 8 mealybugs, no significant influence of the mealybug’s oviposition period was found (F2 = 1.45; df = 2, 97; P = 0.239) (F8 = 2.73; df = 2, 97; P = 0.07). In addition, simple main effect analysis showed that mealybug density had a significant influence on the number of eggs per mealybug oviposited by a N. includens female, for each oviposition period of the P. citri females (F1 = 5.6; df = 3, 97; P = 0.001) (F4 = 18.2; df = 3, 97; P < 0.001), (F7 = 8.2; df = 3, 97; P < 0.001). For each oviposition period, the number of predator eggs laid per mealybug decreases with prey density. Higher number of eggs per mealybug is found at a late oviposition stage at the density of a single mealybug.
Discussion
Following our study, N. includens adults aggregate in patches with high prey density even at a small spatial scale. In general, coccinellids are known to aggregate in areas of high prey density as a result of local search patterns (Seagraves 2009). This general belief is based on numerous studies on aphidophagous coccinellids (Dixon 2000; Evans and Toler 2007; Ives et al. 1993; Schellhorn and Andow 2005). However, this is also driven by the magnitude of prey specialization of the predator. Specialists are known to thrive in lower prey densities than generalist (Sloggett and Majerus 2000). Our data show that similar aggregative behavior is likely to be exhibited also by coccidophagous predators. Aggregative responses of aphidophagous ladybirds have been shown to occur at the level of individual plants (Schellhorn and Andow 2005) and also at large-scale habitat units (Brown 2004). Here, the aggregative response was only examined at individual plant level. It is likely that N .includens adults respond different at larger scales like an entire field. In the field, coccids tend to form smaller colonies than aphids and more aggregated (Borges et al. 2006) and coccidophagous ladybirds like N. includens are expected to follow a similar pattern like their prey as life histories of ladybirds are believed to have evolved in response to those of their prey (Borges et al. 2011). In any case, aggregative responses of coccidophagous predators would have important implications for the pest management of mealybugs (Murdoch and Briggs 1996).
In the present study, P. citri prey density and oviposition period had an effect on the oviposition behavior of the predator. Nephus includens females showed increased egg laying in response to increased prey density and progress of the prey oviposition period. Although the later could be viewed as an increase of prey density as well. In any case, prey density had a positive influence on the number of eggs an individual predator laid. This density dependent numerical response is advantageous for the successful suppression of mealybug prey under biological control programmes. In combination with the aggregative behavior observed under the semi-field conditions, N. includens adults can be a potentially effective biological control agent against the citrus mealybug.
In aphidophagous coccinellids there is accumulating evidence that there is a density of aphids below which ladybirds are unlikely to lay eggs (Dixon 2000). Such a response had not been tested for coccidophagous coccinellids (Dixon 2000). In the current study N. includens females showed a density-dependent tendency in egg-laying behavior. In addition, egg-laying was affected by the oviposition period of the ovipositing female mealybugs. In aphidophagous ladybirds, females will lay their eggs when an aphid colony is rather young and the density of aphids is adequate to sustain the development of the ladybird offspring. Aphidophagous females avoid laying eggs when their egg could be consumed by conspecifics (Doumbia et al. 1998; Hemptinne et al. 1992). This is driven by the relatively rapid growth of an aphid colony (Dixon 2000). In contrast, coccid colonies are stable over time and space and provide more reliable food resources to their predators. Nephus includens oviposit one to two eggs per adult mealybug even when there is no prior egg-laying by the mealybugs. For a first instar N. includens larva it will be difficult to prey upon an adult mealybug. However, hatching for N. includens eggs takes as many as 5 days under constant temperature of 25 °C (Kontodimas et al. 2004). In the meantime, female mealybugs will start ovipositing and the young coccinellid larva will have enough to prey upon. Therefore, N. includens females exhibit strong numerical responses in relation to prey density. Moreover, at a late stage of mealybugs’ ovipositional progress, predators are more likely to lay more eggs than when mealybugs are at the onset of their oviposition period.
A predator is considered as a potentially good biocontrol agent when many individuals aggregate and oviposit in areas of high prey densities. Our results showed that N. includens exhibit an aggregation behavior towards prey density. In addition, N. includens females oviposit at different stages as a mealybug colony develops and exhibit higher oviposition rates as mealybug density increases. This behavior has direct implications in biological control by coccinellids (Oliver et al. 2006). Aggregative response at high prey densities is desirable for stabilizing prey abundance at low levels. Although, other factors such as oviposition deterring pheromones by conspecific larvae may hamper the aggregative response of a coccinellid predator (Doumbia et al. 1998).
References
Bernstein C, Kacelnik A, Krebs JR (1991) Individual decisions and the distribution of predators in a patchy environment. II. The influence of travel costs and structure of the environment. J Anim Ecol 60:205–225
Borges I, Soares AO, Hemptinne JL (2006) Abundance and spatial distribution of aphids and scales select for different life histories in their ladybird beetle predators. J Appl Entomol 130:356–359
Borges I, Soares AO, Magro A, Hemptinne JL (2011) Prey availability in time and space is a driving force in life history evolution of predatory insects. Evol Ecol 25:1307–1319
Brown MW (2004) Role of aphid predator guild in controlling spirea aphid populations on apple in West Virginia, USA. Biol Control 29:189–198
Coderre D, Provencher L, Tourneur JC (1987) Oviposition and niche partitioning in aphidophagous insects on maize. Can Entomol 119:195–203
Dixon AFG (1959) An experimental study of the searching behaviour of the predatory coccinellid beetle Adalia decempunctata (L.). J Anim Ecol 28:259–281
Dixon AFG (2000) Insect predator–prey dynamics: ladybird beetles and biological control. Cambridge University Press, Cambridge
Doumbia M, Hemptinne JL, Dixon AFG (1998) Assessment of patch quality by ladybirds: role of larval tracks. Oecologia 113:197–202
Elliott N, Kieckhefer RW (2000) Response by coccinellids to spatial variation in cereal aphid density. Popul Ecol 42:81–90
Evans EW, Toler TR (2007) Aggregation of polyphagous predators in response to multiple prey: ladybirds (Coleoptera: Coccinellidae) foraging in alfalfa. Popul Ecol 49:29–36
Evans EW, Youssef NN (1992) Numerical responses of aphid predators to varying prey density among Utah alfalfa fields. J Kansas Entomol Soc 65:30–38
Franco JC, Suma P, Da Silva EB, Blumberg D, Mendel Z (2004) Management strategies of mealybug pests of citrus in mediterranean countries. Phytoparasitica 32:507–522
Godfray HCJ, Pacala SW (1992) Aggregation and the population dynamics of parasitoids and predators. Am Nat 140:30–40
Hassell MP, May RM (1973) Stability in insect host parasite models. J Anim Ecol 42:693–726
Hemptinne JL, Dixon AFG, Coffin J (1992) Attack strategy of ladybird beetles (Coccinellidae): factors shaping their numerical response. Oecologia 90:238–245
Hodek I, Honěk A (2009) Scale insects, mealybugs, whiteflies and psyllids (Hemiptera, Sternorrhyncha) as prey of ladybirds. Biol Control 51:232–243
Ives AR, Kareiva P, Perry R (1993) Response of a predator to variation in prey density at three hierarchical scales: lady beetles feeding on aphids. Ecology 74:1929–1938
Kareiva P (1990) The spatial dimension in pest-enemy interactions. In: Mackauer M, Ehler L, Roland J (eds) Critical issues in biological control. Hants, Andover, pp 213–217
Katsoyannos P (1996) Integrated insect pest management for citrus in northern Mediterranean countries. Benaki Phytopathological Institute, Athens
Kindlmann P, Dixon AFG (1993) Optimal foraging in ladybird beetles (Coleoptera: Coccinellidae) and its consequences for their use in biological control. Eur J Entomol 90:443–450
Kontodimas DC, Eliopoulos PA, Stathas GJ, Economou LP (2004) Comparative temperature-dependent development of Nephus includens (kirsch) and Nephus bisignatus (boheman) (coleoptera: coccinellidae) preying on Planococcus citri (risso) (homoptera: pseudococcidae): evaluation of a linear and various nonlinear models using specific criteria. Environ Entomol 33:1–11
Kontodimas DC, Milonas PG, Stathas GJ, Economou LP, Kavallieratos NG (2007) Life table parameters of the pseudococcid predators Nephus includens and Nephus bisignatus (Coleoptera: Coccinellidae). Eur J Entomol 104:407–415
Magro A, Araujo J, Hemptinne JL (1999) Coccinellids (Coleoptera: Coccinellidae) in citrus groves in Portugal: listing and analysis of geographical distribution. Bol San Veg Plagas 25:335–345
Mangel M (1987) Oviposition site selection and clutch size in insects. J Math Biol 25:1–22
Mills NJ, Lacan I (2004) Ratio dependence in the functional response of insect parasitoids: evidence from Trichogramma minutum foraging for eggs in small host patches. Ecol Entomol 29:208–216
Milonas PG, Kontodimas DC, Martinou AF (2011) A predator’s functional response: influence of prey species and size. Biol Control 59:141–146
Murdoch WW, Briggs CJ (1996) Theory for biological control: recent developments. Ecology 77:2001–2013
Murdoch WW, Chesson J, Chesson PL (1985) Biological control in theory and practice. Am Nat 125:344–366
Oliver TH, Timms JEL, Taylor A, Leather SR (2006) Oviposition responses to patch quality in the larch ladybird Aphidecta obliterata (Coleoptera: Coccinellidae): effects of aphid density, and con- and heterospecific tracks. Bull Entomol Res 96:25–34
Schellhorn NA, Andow DA (2005) Response of coccinellids to their aphid prey at different spatial scales. Popul Ecol 47:71–76
Seagraves MP (2009) Lady beetle oviposition behavior in response to the trophic environment. Biol Control 51:313–322
Sloggett JJ, Majerus MEN (2000) Habitat preferences and diet in the predatory coccinellidae (Coleoptera): an evolutionary perspective. Biol J Linn Soc 70:63–88
Sokal RR, Rohlf FJ (2012) Biometry: the principles and practice of statistics in biological research, 4th edn. W. H. Freeman and Co., New York
Walton VM, Pringle KL (2005) Developmental biology of vine mealybug, Planococcus ficus (Signoret) (Homoptera: Pseudococcidae), and its parasitoid Coccidoxenoides perminutus (Timberlake) (Hymenoptera: Encyrtidae). Afr Entomol 13:143–147
Acknowledgments
This work was conducted with the financial support from the General Secretariat of Research and Technology of the Greek Ministry of Research under a bilateral cooperation for research of Greece and France (GSRT 228 e). We would like to thank two anonymous reviewers for their helpful comments that greatly improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Milonas, P.G., Partsinevelos, G. & Martinou, A.F. Patch Assessment for Oviposition by a Predator: the Effect of Prey Density and Prey Oviposition Period. J Insect Behav 28, 535–543 (2015). https://doi.org/10.1007/s10905-015-9523-3
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10905-015-9523-3