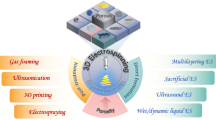
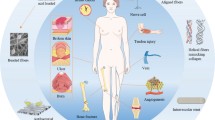
Abstract
Repairing large tissue defects often represents a great challenge in clinics due to issues regarding lack of donors, mismatched sizes, irregular shapes, and immune rejection. Three-dimensional (3D) printed scaffolds are promising for growing cells and manufacturing tissue constructs because of the intricate control over pore size, porosity, and geometric shape, but the lack of biomimetic surface nanotopography and restricted biomolecule presenting capacity render them less effective in regulating cell responses. Besides, electrospinning is a rapid and simple manner to fabrication of different forms of nanofibrous scaffolds and cause the novel and complex scaffold constructs for many tissue engineering applications. In this review article, we begin with a thorough discussion on the method of producing 1D, 2D, and 3D electrospun nanofiber materials. In particular, we insisted on the techniques which are developed to fabricate 3D scaffolds. We categorized these techniques into four groups, direct fabrication, post-processing method, the combination of textile method and electrospinning, and a combination of electrospinning and 3D printing. Then, we tried to explain their application in many clinical scopes, especially in drug delivery, regenerative medicine, and cancer treatment. Ultimately, we discussed future actions which could be done to improve nanomedicine and nanotechnology researches.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Various in-vitro studies have been conducted to improve clinical issues related to drug delivery and tissue engineering, such as cell therapy. For over a century, two-dimensional (2D) cell cultures have been employed as in vitro models to determine cellular responses to biophysical and pharmacological stimuli. However, cell seeding in the two-dimensional (2D) microenvironment cannot accurately mimic the in-vivo microenvironment due to irregular cell-cell interaction in the 2D environment. Besides, an improved microenvironment is required for optimum tissue engineering to mimic the extracellular matrix (ECM). Scaffolds/substrates play an important role in this scope. Scaffolds not only provide improved ECM-like structures but also help the cells to better viability, attachment, proliferation, migration, and differentiation. Studies show scaffolds could promote gene expression to a specific lineage. Also, scaffold-cell interaction with controlling cell signaling is very critical in tissue engineering and regeneration of damaged organs [1, 2]. Various scaffolds, such as hydrogel scaffolds, fibrous scaffolds, microsphere scaffolds, and so on, have been used in tissue engineering; however, these nanofiber-based scaffolds are superior to the others because they have the architecture of natural tissue, making them well-known as the best tissue imitative. Recently, several methods have been used to fabricate nanofiber-based scaffolds, including template synthesis, drawing, self-assembly, phase separation, and electrospinning. Among them, electrospun nanofiber-based scaffolds have acceptable features that make them practical such as simplicity, availability, high surface area to volume ratio and diversity of used biomolecules. Despite these promising features, electrospun nanofiber-based scaffolds usually have a 2D structure with tight pores that couldn’t promote better cellular infiltration. For the solution to this problem, researchers introduced 3D scaffolds for better in-vitro cell culture research. These scaffolds play a variety of roles in the construction of artificial structures [1, 3,4,5,6]. 3D scaffold structures have large pores and can trigger immunological responses to provide better cell-cell interactions and high cell affinity, which promote cell proliferation and improve cell morphogenesis [7]. Recently, very important attempts have been dedicated to the construction of 3D nanofibrous scaffolds, such as using a modified collector, orthogonal weaving technique, vibration, and ultra-sonication, using a sacrificial template, gas-foaming technique, etc. Besides, the progress of 3D printing technology especially in pluripotent stem cells and genome editing platforms provides a very important improvement in tissue regeneration. Interestingly, 3D printing method can fabricate shape-specific fiber scaffolds to repair injured tissues. Recently, researchers combine the 3D printing method with electrospinning to use the features of both techniques, such as the hybrid process. In this review, we will review the recent efforts which have been used for the fabrication of 3D electrospun nanofiber scaffolds (3D ENS) by several approaches, including direct fabrication techniques, post-processing methods, and a combination of 3D printing and electrospinning. Furthermore, we will highlight the use of these scaffolds in a variety of contexts such as drug delivery, regeneration medicine, and cancer treatment.
2 Electrospun Nanofiber
Drawing-processing, template-assisted synthesis, self-assembly, solvent casting, phase separation, and electrospinning techniques have all been documented for the fabrication of nanofiber-based scaffolds [8]. Electrospinning is an attractive technique because of its simplicity of technique, ease of process control, high surface area, high porosity [8, 9]. This process, which began in the late 1890s, was upgraded to electrospinning in 1900. In the 1930s, Formhals explained its basic principle [10, 11]. Generally, electrospinning has been done with a wide range of natural and synthetic polymers. Alginates collagen, and gelatins, as well as newly synthesized materials that mimic certain properties of natural materials, with improved properties such as the ability to signal to cells, enable cells to infiltrate the generated fibers/scaffolds [12, 13]. The electrospinning process works base on the electrostatic principle. Generally, an electrospinning instrument has three parts; (a) high voltage power supply (DC or AC) with two electrodes, one of these is connected to the spinneret and another to the collector which is usually grounded (b) syringe and needle assembly (collectively called as spinneret in needleless type) and (c) grounded metal collector. This technique consists of three stages; (a) initiation of the jet and its elongation on the straight line; (b) bending instability (whipping instability) occurs and jet elongates, which could be associated with branching or splitting or not; (c) fiber deposition and solidification on the metal collector [3]. Despite the simplicity of the electrospinning process, many parameters can influence the electrospun nonwoven mat’s morphology, like solution parameter, instrumental parameter and ambient parameter. We are going to explain the effect of these parameters on the diameter and morphology of the electrospun nanofiber (NFs) in Table 1.
3 1D Nanofiber Scaffold
Although continuous long fibers can be made using electrospinning, short nanofibers or one dimensional (1D) nanofiber (fragments and yarns) have attracted a lot of interest in medical applications due to their high mechanical properties, high tensile strength, high thermal stability, high conductivity, and injectability [1].
3.1 1D Nanofiber Fragments
Some studies have focused on the electrospinning-based fabrication method because of its merits for fabricating short nanofiber [19]. For example, Stoiljkovic and Agarwal developed the UV-cutting method which irradiated electrospun fibers with UV light then treated them with an appropriate solvent to remove soluble parts [20]. Yoshikawa and their co-worker fabricated short nanofiber with a concentrated polymer brush. After the electrospinning process, atom transfer radical polymerization forms the brushes on the electrospun nanofiber surface. Then, fibers were cut by a homogenizer [21]. In another study, Yabuki and coworkers developed a new system that used an electric spark. When electrospun nanofibers pass through the gap between the tips of two electrodes, an electric spark is generated, which cuts them into short nanofibers [19]. In another study, they used a solution composed of cellulose acetate, acetone, and dimethyl acetamide with a specific concentration. A quick increase in the repulsive force from surface charges and the longitudinal forces from the applied voltage causes short nanofibers formation [22]. Razorblade cutting and cutting with cryostat microtome are the other methods for the formation of short nanofibers which are better for brittle polymers and ductile polymers [23, 24]. Ultrasonication of nanofibers introduces another way to fabricate short nanofibers, especially brittle polymers. Ductile polymers need pretreatment before the ultrasonication process [25]. Li et al. demonstrated increasing NaCl nanoparticles to the solution could reduce lengths of short nanofiber [26]. Table 2 shows these techniques and their merits as well as demerits.
3.2 1D Nanofiber Bundle/Yarn
Many approaches have been developed for Yarn fabrication. For example, Jha et al. used the air gap electrospinning process. This process forms fibers into linear bundles that are aligned in parallel with the long [27]. Teo et al. used a steel blade to form a bundle that is highly aligned with nanofiber. Then, the fiber bundle could be twisted or braided [28]. Dual collection rings electrospinning is another method that forms fibers between two ring electrodes. Finally, for the Yarn formation, the ground on the right collector is rotated, while the left ground collector remains stationary [29]. Besides, Sanderson et al. developed an efficient method based on wet electrospinning. In this process, a liquid bath has been used as a collector. The non-woven web of fibers that formed on the surface of the water was collected using an external mandrel [30]. Furthermore, Lotus et al. developed a new system with two collectors: a rotating hollow hemisphere for twisting the fibers together and a rotating mandrel for collecting the twisted yarns [31].
4 2D Nanofiber Scaffold
One of the most important purposes of the nanotechnology process is controlled drug delivery. For this purpose, many approaches have been developed. First, blend electrospinning provides a straightforward technique for the various composite fabrications [32]. Then, for the purposive drug delivery, core/sheath nanofibers were developed which could be fabricated via emulsion electrospinning [33] or coaxial electrospinning [34]. The core-sheath structured nanofibers could provide a prolonged release pattern because of the outer layer which acts as a barrier [35]. In another attempt, researchers fabricated side-by-side nanofibers with two different polymers orienting parallel in one fiber to control the drug release from the scaffolds which were made via dual electrospinning [14, 36].
5 3D Nanofiber Scaffold
While two-dimensional electrospun nanofiber has many advantages, such as being cost-effective and commercially accessible, we need three-dimensional nanofiber for cell growth due to the low pore size and two-dimensional environment. Despite having optimized pores in three-dimensional scaffolds, we face other challenges, such as reduced nutrient diffusion and hypoxia; hence, much attention has been devoted to constructing 3D nanofiber scaffolds, and major improvements have been made, as described in the approaches below [1].
5.1 Direct Fabrication
5.1.1 Multilayer Electrospinning
One simple way of fabricating 3D nanofiber is multi-layering electrospinning or sequential electrospinning. In this technique, one layer of electrospun nanofiber deposits another layer. This deposition continues to reach the thickness around hundreds of microns (Fig. 1ia) [37]. It seems that researchers often use an external electric field to collect fibers at a definite site to fabricate 3D scaffolds [38]. It is important to note that the thickness described above is approximately 1 mm. This technique has many advantages, such as controllable pore size and diameter, fiber construction, and the number of layers of scaffolds required for cell adherence, development, and differentiation on the scaffold, but it may take a long time [3]. To dissolve this problem, Sherif Soliman et al. developed a new multimodal structure where the distribution of fibers is not simply juxtaposed. This procedure has only one step and this will reduce the time of the electrospinning process. There is an important point about this technique that copper ring electrode was used to control the spinning jet and the deposition area of polymer solutions from two syringes. These polymer solutions have different concentrations that are pumped from separate pumps (Fig. 1ib) [39].
5.1.2 Using Modified Collector
Among many methods for the fabrication of scaffolds, electrospinning is popular because it has a simple process the fabrication of biocompatible scaffolds for different applications. However, this technique has a serious limitation due to the tightly packaged nanofiber layers with low porosity. This is a significant obstacle to cell penetration and migration into the scaffolds [40]. As a result, there is a critical need for new electrospinning techniques to produce porous electrospun scaffolds by adjusting fiber deposition and orientation for tissue engineering applications. Recently, researchers investigated different collectors and examined their impact on scaffold porosity and diameters. Zhu et al. designed a rotating metal frame cylinder as a collector which increases porosity as high as 92.4% and average pore size of 132.7 μm was fabricated [41]. Blakeney et al. used a non-conductive spherical dish instead of the traditional collector. This collector has an array of metal probes that have been connected to the ground. Deposited NFs can be settled next to the previous fiber and embedded between probes and their mid-air dish. This spherical dish generates fluffy and cotton ball-like electrospun scaffolds with high porosity and high stability. This technique has another feature that took less time (Fig. 1 iia) [40]. In another study, Vaquette et al. used several patterns of collectors such as wire form, round, ladder and star pattern of collectors, then described their porosity [42]. Besides, Leong et al. used cryogenic mandrel or low-temperature under humid conditions. When fibers are deposited on the cryogenic collector in the humid environment of the instrument, ice crystals are formed. The formation of these crystals caused many pores after post fabrication drying. The formation of ice crystals ratio determines fiber porosity and density [43] (Fig. 1iib).
5.1.3 Wet Electrospinning
Besides the techniques which are mentioned above, there is another technique for the fabrication of 3D nanofiber-based scaffolds, which was developed by ki et al. They use non-solvent baths as collectors to fabricate sponge-like scaffolds that are called wet electrospinning. When electrospun nanofiber is deposited in a non-solvent bath, this collector inhibits pack densely and creates a highly porous structure of scaffolds [44]. A considerable point about wet electrospinning is the porosity of the wet electrospun NFs. The electrical conductivity and surface tension of the solvent bath used as the collector and play an important role in the morphology of 3D scaffolds [45]. Ethanol, methanol, tertiary-butyl alcohol, and acetone which have low surface tension are suitable for better deposition and porosity increasing, while high surface tension causes fibers to deposit densely in non-solvent baths [1, 46, 47]. Electrical conductivity is one of the most influential features of fiber porosity in wet electrospinning. No fibers were observed when nonpolar solvents with low conductivity and high electrical resistance, such as Hexane, were used as collectors [48]. Shin et al. used this technique to provide a 3D electrospun P (TMC-co-CL)-b-PPDO scaffold. In this technique, after solution preparation, they used a stainless dish which has methanol that plays the collector’s role. After deposition of fiber into the bath, the next step is fiber recovering from the methanol bath by substitution with distilled water. To extract excess non-solvent, they first immersed the fibers in liquid N2 and allowed them to freeze. The following phase was freeze-drying. These NFs have a cotton-like appearance. Thus, they should compress these NFs into sponge-like. To dissolve this problem, they placed cotton-like nanofiber in a 1ml syringe and compressed it, then placed it in a warm water bath (70 °C) for 30 min. After these processes, compressed NFs were placed in N2 liquid to be frozen, and disks were cut for use [49].
5.2 Combination of Textile Method and Electrospinning
5.2.1 Three Dimensional (3d) Orthogonal Weave Method
The combination of electrospinning and textile methods provides new avenues for fabrication of 3D scaffolds like twisting [50], braiding [51], and weaving/noobing [52] methods. Among them, weaving/noobing, a novel process made from stripes, yarns, and bundles with anisotropic properties and normal pores [1]. The term “noobing” is an abbreviation for these terms (Non-interlacing, Orientating Orthogonally and Binding). The “noobing” method is also known as the orthogonal weave method. The combination of electrospinning and the “noobing” process allows for the fabrication of 3D scaffolds by assembling three orthogonal sets of yarns/ bundles/ strips with no interlacing. In other words, the fabric’s constituent strips are crimp-free. In this method, aligned NFs sheets were first cut into fine strips along the direction of fiber alignment after being produced by high-speed rotating mandrel (collector) electrospinning; the next step is the noobing step. Then, cutting and thermal treatment are necessary to seal the edge of 3D fibrous scaffolds without interlacing (Fig. 1 iii) [52]. Although this method is the most adaptable for creating macro- or mesoscale pores, there have been only a few reports of electrospun fibers being used to create porous woven constructions. Because of the complexity of fabricating continuous electrospun fibers for industrial weaving machines, weaving electrospun filaments presents many challenges [53]. Xie et al. have proved human adipose-derived stem cells seeded on incorporated woven nanofiber (basket-weaved architecture) scaffolds with a significant amount of vascular endothelial growth factor (VEGF) can potentially simulate the construction of native cardiac tissue and act as implants to provide an inherent structural capacity to conduct the in situ regeneration of cardiac tissue after grafting [52, 54].
3D scaffolds fabricated by direct fabrication (i-ii) and weaving and noobing method (iii).
(i) multilayer electrospinning; (a) conventional multilayer electrospinning, (b) novel multi electrospinning. (ii) Modified collector; a) cotton ball-like nanofiber scaffold which fabricated with the modified spherical dish. (iii) schematic diagram of weaving and noobing method; (a) electrospun nanofiber, (b) cutting, (c) noobing
5.3 Post Processing Method
Although defined pre- or co-fabrication strategies aim to increase the pores of scaffolds and are widely applicable, some drawbacks exist, such as the need for significant changes to the setup. Thus, many efforts were made to eliminate these limitation [55].
5.3.1 Vibration and Sonication Method
Ultra-sonication is a straightforward technique to develop electrospun nanofibrous scaffolds into 3D constructions with preserving topographic cues. The ultrasonic vibrations can upsurge porosity [1]. Lee et al. identified an ultrasonication procedure that improved the thickness and pore size of a synthetic NF. In this process, after collecting the membrane and placing it under vacuum conditions at room temperature for one day. They were cut and immersed in 70% ethanol and DDW before beginning the sonication and lyophilization [56]. Gu et al. confirmed that ultrasonication of electrospun chitosan NFs increased pore sizes from 79.9 to 97.2%, making it suitable for wound dressing due to its high porosity and improved water absorption time. This scaffold’s coagulation effectiveness was superior to that of chitosan sponge controls, especially for surgical applications. Furthermore, normal human dermal fibroblast viability was found to be 1.4-fold higher than non-sonicated scaffolds [57]. Despite the fact that ultrasonication has no impact on molecular weight, mechanical properties in ultrasonicated scaffolds are reduced. For example, after ultrasonic treatment with 60 J·mL − 1 of ultrasonic power, the mechanical properties of PLLA NFs scaffolds around 11-fold decreased [56]. The tensile strength and pore size subsequently can be adjusted via changing the ultrasonic power and exposure times [55]. Sawawi et al. proved the sonication process has no effect on the chemical or physical structure of the fibers or their constituent polymers [25].
5.3.2 Gas Foaming Method and Modified Gas Foaming
The techniques that are noted above, have some drawbacks like insufficient thickness and uncontrolled porosity. Furthermore, the preceding 3D scaffolds achieved were related to unordered organizations and the absence of nano-topographic cues that are crucial for regenerating organized tissues such as nerve, tendon and muscle [58, 59]. The Gas foaming technique is broadly used to construct 3D sponge scaffolds for tissue engineering applications. This technique includes three basic phases: (1) gas/polymer solution creation and generation of gas bubbles via chemical reaction (2) gas bubble (pore) nucleation (3) gas bubble growth and volume expansion [60]. Nam et al. created dual-porosity nanofiber scaffolds using a salt leaching/gas foaming process. As a gas foaming agent, NH4CO3 was used. High temperature (90 °C) was used to leach out the NH4CO3 particles, resulting in gaseous ammonia and CO2 within the solidified polymeric matrix [61]. Jiang et al. presented another way to fabricate a 3D fibrous scaffold by an in situ gas foaming process. In this process, NaBH4 solution was applied as a gas foaming reagent, and the nonwoven scaffold fabricated through electrospinning was immersed in a NaBH4 solution for different time intervals. The following steps were taken to complete the expansion process: (i) NaBH4 aqueous solution penetrated into the aligned nanofibrous mats by capillary effect; (ii) different nucleation of gas bubbles on the surface of NFs or at intersections of aligned NFs; (iii) growth of the nucleated bubbles because of production of hydrogen gas; (iv) coalescence of adjacent gas bubbles and subsequently formation of a ‘bubble layer’. To maintain the scaffold integrity, the mats were freeze-dried using a lyophilizer at the end of this process. It was confirmed that an increase in the concentration of NaBH4 as well as in keeping time has a crucial effect on the thickness of the NFs. Surprisingly, after just 24 h of exposure to the 1 M NaBH4 aqueous solution, the thickness of nanofibrous scaffolds increased from 1 to 35.6 mm. Notably, the maximum expansion of randomly oriented nanofibrous mats is lower than that of aligned NFs due to a close interaction between intertwined NFs [60]. In this process, NaBH4 cannot penetrate into the hydrophobic polymers like PCL or PVDF. To dissolve this issue and expansion of this technique, researchers used methanol solution [62]. Nevertheless, controlling the width, length, and thickness of the scaffold remains a challenge. To solve this problem, a study combined a customized mold with a glass spacer and a gas-foaming approach. Using this approach, expanded mats with precise density and thickness can be adapted to the size of the glass spacer (Fig. 2i) [4]. Despite several advantages, the gas foaming technique has some drawbacks. For example, a leak of gas blowing agent near to the surface causes a dense skin layer on the scaffolds. The gas foaming process produces pores from the inner of the materials, but the selective solvent etching method has solved this drawback that generates pores from the surface [63, 64].
5.3.3 Porous Nanofiber with Adding Porogen Substance
The electrostatic repulsion between many substrates and deposited NFs may be altered by adding ionic salts to the electrospinning solution, allowing sponge-like NFs to be developed [65]. This strategy can produce limited thickness of NF matrix and requires the application of additives (e.g., ionic salts) that may lead to safety issues and side effects during tissue regeneration [66, 67]. Another method for addressing these issues is to use sacrificial fibers, which are soluble fibers that have been removed by a suitable dissolution medium [66]. Gualandi et al. proved fibers consist of 60% PEO have better cell infiltration than 5% PEO to the center of the scaffold [68]. Aghajanpoor et al. developed a set of sacrificial co-electrospinning with PEO and subsequent ultrasonication of PCL/nanohydroxyapatite electrospun mats to improve pore size by 1.9 times. This resulted in major improvements in hMSC penetration, proliferation, and osteogenic differentiation [69].
5.3.4 Depressurization Subcritical co2
There are various disadvantages of adding Porogen material, including the requirement for numerous processes to remove sacrificial fibers, a complicated procedure, a restricted number of materials, the inability to use randomly oriented NFs, the need for an additive substrate, and the need for an aqueous solution [70]. Jiang et al. developed another expansion method by utilization of subcritical CO2 fluid to dissolve these problems. They discovered that CO2-expanded 3D nanofibrous scaffolds enhance cellular penetration and have a positive host response. In this technique at first NFs were cut into 1 cm × 1 cm squares in liquid nitrogen to avoid deformation on the edges. Then, 1 g of dry ice and one piece of nanofiber mat were placed in a 30 mL Oak Ridge centrifuge tube. In the high-pressure condition of the centrifuge tube, the dry ice changed into CO2 fluid versus low pressure. This fluid penetrates the fiber. Then, when the cap was loosened rapidly, the pressure reduced, and CO2 fluid trapped in the pores of NFs changed into a gas. The result of these events is the puffy structure of NFs. This expanding process was repeated again and again to reach the desired thickness (Fig. 2ii) [70].
3D scaffolds fabricated with post-processing method. (i) modified gas foaming technique with glass spacer. (a) design of glass spacer and putting nanofiber scaffold in glass spacer, (b) expanding and rinsing steps continue after mold assembly, (c) freeze-drying, (d) remove mold. (ii) schematic diagram of depressurization subcritical CO2. (a) electrospun nanofiber, (b) nanofiber was cut into 1 cm× 1 cm, (c) piece of nanofiber with 1 gr of dry ice {CO2(s)} in the 30 mL Oak Ridge centrifuge tube, (d) in high pressure, dry ice change into CO2(L), (e) then open the cap rapidly, pressure decrease, fluid CO2 change into gaseous CO2, (f) 3D scaffold
5.4 Combination of 3D Printing and Electrospinning
Fabrication of 3D scaffolds for mimicking the extracellular matrix is a significant issue in tissue regeneration because ECM has an essential role in growing the cells, diffusion of nutrients, metabolites, and essential soluble factors [71]. The combination of 3D printing and electrospinning can endow hierarchical architectures composed of macrostructures and microstructures. In this part we will try to explain these methods in more detail.
5.4.1 Hybrid Process and Rapid Prototyping (RP) Method
3D nanofibrous scaffolds have the potential for cell adhesion and migration as well as natural ECM, while the measurements of fabricated scaffolds for tissue engineering should be potential for cell adhesion and migration as well as natural ECM. One of the most important technologies for fabricating tissue structures with dimensions ranging from micrometers to millimeters is a hybrid process [72, 73]. Park et al. developed this method for the fabrication of experimental 3D scaffolds that mimic the biophysical milieu. These scaffolds were made up of two parts: the first microfibrous structure created using direct polymer melt deposition (DPMD) and the second part nanofibrous structure created using electrospinning. The DPMD process produces a 3D structure to promote tissue ingrowth, as well as functions associated with surface affinity and an increased surface area for improved cell adhesion and proliferation. The system hybrid process consists of a micronozzle (stainless steel syringe), a device heating syringe, a compressed air dispenser and a three-dimensionally moving micropositioning system. The heating device melts the polymer in the syringe before applying compressed air to the polymer melts. The woodpile-like structure was fabricated via 3D movement of a micronozzle. The NF networks were then inserted between the layers of the microfibrous structure. The parameters that affect the quality of fibrous scaffolds in this process are as follows: (1) diameter of the micro nozzle, (2) the temperature of the material process; the coiled heater raises the temperature above the melting point, (3) the compressed air pressure, and (4) the nozzle’s velocity. After one day of culture, Park et al. discovered that the cell attachment rate in hybrid scaffolds is 2.2 times higher than in previous scaffolds (Fig. 3i) [74]. We can see the advanced hybrid processes in the RP method. RP method has proven to be an effective tool for dramatically reducing the time and expense involved in the realization of a new products and for overcoming the bottlenecks of existing manufacturing processes [75]. In this process, 3D plotting scaffolds are made by using the Bioplotter to manufacture biofunctional scaffolds for tissue engineering. Because of the computer-aided design that produces complex scaffolds with a variety of biomaterials and fabricating conditions, such as form, thickness, and strand spacing, rapid prototyping is a very simple and practical process [71].
5.4.2 Near Field Electrospinning
The Near Field Electrospinning (NFES) process is one of the most well-known techniques which is used in various applications, including 3D scaffold fabrication for biomedical applications, drug delivery systems, and tissue engineering [76]. Sun et al. introduced the concept of near-field electrospinning in 2006 to bridge the gap between solution electrospinning and direct-write additive manufacturing [77]. The NFES technique consists of two types just like electrospinning. One of these is solution near field electrospinning and the another is near-field melt electrospinning [78]. More specifically, in solution near field electrospinning, the spinneret-to-collector distance frequently ranges from 500 m to 2 mm, allowing for proper fiber deposition on the collector. Another part of the instrument has two motional axes, a programmable x-y translational motion stage which deposits fibers repetitively and a z-axis regulates deposition distance from spinneret to collector. A sheet of printing paper is placed on top of the grounded conductive plate to accumulate fibers and absorb excess solvent that does not evaporate due to the short distance. Because of the excessive solvent penetration, the local resistance of the paper near the fibers decreases, which improves electrical charge transfers between the deposited fibers and the conductive ground plate through the fibrous network within the paper. This transfer can guide other fibers to deposit on preferred site. While Melt Near Field Electrospinning (MNFES) with molten polymer has some drawbacks, such as a short spinning distance, it has many advantages, such as being eco-friendly, avoiding solvent evaporation, having a high yield, and its ability to fabricate high aspect-ratio structures. This process can produce various 3D structure with pre-designed shapes [78, 79]. The voltage used in the NFES method is low since the distance is short, and using external forces such as a magnetic field will also reduce it, which is another advantage [80, 81]. Despite encouraging achievements about NFES, Little study has evaluated the effect of parameters on uniformity of NEFS printed fibers. Zhao et al. studied these parameters and reported results. The parameters that were studied are applied voltage, speed of silicon substrate travelling, flow rate and distance between the needle and silicon substrate. They proved that with increasing voltage and flow rate, the width of fiber increased, but when travelling speed and distance increased, the width of fiber decreased. Relative standard deviation (RSD) is a unit that is connected to fiber uniformity; as width uniformity improves, RSD value decreases. For example, as the applied voltage and travel speed to the silicon substrate increase, this unit decreases, and as the flow rate of the syringe pump and distance between the needle and silicon substrate increase, RSD value increases, and thus fiber uniformity decreases. The effects of distance between the needle and the silicon substrate, as well as applied voltage to the needle, on width and its uniformity, can be attributed to variations in electric field strength. Relatively uniform ribbons were printed under high electric field strength, while bead-on-string lines with poor uniformity were obtained under low electric field intensity, possibly due to insufficient polymer expanding force in the polymer jets (Fig. 3ii) [82]. In summary, Table 3 shows advantages and disadvantages of 3D nanofiber based scaffolds approaches and their application.
6 Biomedical Application
6.1 Drug Delivery
Tissue engineering and its regeneration is a complicated biological process that requires the assembly and delivery of numerous biomolecules such as cytokines, growth factors, and other proteins as well as DNA, siRNA at a specific site and time [89,90,91,92]. As a result, we must place a greater emphasis on drug delivery. We have various local drug delivery vehicles (e.g., nanoparticles and NFs) that can provide managed release while potentially reducing undesirable systemic side effects [93,94,95,96]. But among them, NFs are more useful because they have many features like various material choices, simple drug encapsulation, use of various drug formulations, and high controlled release of drugs [1]. Drug release from NFs might be due to drug desorption from the surface, diffusion from pores, or matrix degradation [97]. There are various drug release patterns such as zero-order release, biphasic release, sequential release, stimuli-triggered and spatiotemporal release that are utilized in many applications [1]. Burst release involves a high concentration of drug at the time of injection or implantation, followed by a long period of drug maintenance at an acceptable concentration. This pattern of drug release is more useful for local treatment, such as lesion sites, but burst release of drug can be destructive in some cases; therefore, drug release must have a sustained release pattern. Stimuli-triggered release is a new pattern of drug release with few side effects. Various stimuli responsive polymers have been reported upon exposure to exogenous stimuli such as magnetic fields, ultrasounds, light, temperature, and electric fields or endogenous variations such as pH value, ionic strength, and enzyme concentration. Thermoresponsive drug delivery is one of the most studied stimuli-responsive strategies, and it has received a lot of attention in oncology [1, 98]. Demirci and colleagues investigated PH sensitive polymers as another stimuli responsive strategy. They designed a poly (4-vinylbenzoic acid-co-(ar-vinylbenzyl) trimethylammonium chloride) nanofiber scaffold which is sensitive to the PH alteration in order to control the release of ciprofloxacin in acidic, neutral and basic medium [99]. Generally, in addition to these techniques, there are many factors that influence drug release rate. Drug dependent parameters include molecular weight, drug loading, physical state, solubility and interaction between drug and polymers [100]. For example, as drug loading increases, so does the rate of drug release. Low molecular weight drugs have a high rate of release. The crystalline drugs have a burst release pattern because these are often loaded on the fiber surface and an amorphous form of drug is located deep site of the scaffold and is released in a sustained pattern [101]. Another parameter that affects the rate of release of drug is polymer dependent parameters like polymer composition, crystallinity and polymer molecular weight. Results demonstrate that when we use amphiphilic and hydrophilic polymers, drug loading increases and its burst release decreases. Highly crystalline polymers have a lower drug release rate than other polymers, because the three-dimensional structure of crystalline doesn’t allow water to enter the polymer easily. In other words, water uptake in these polymers is very low. Porosity, alignment and mechanical property of nanofiber are other parameters that influence drug release rate. Various studies have often demonstrated that porous NFs have faster drug release than fibers which have low porosity. Alignment of nanofiber is another parameter that influences drug release rate. We see faster release in randomized pattern of nanofiber, because the tendency of water entrance increases into the polymer. Chou and colleagues have proved the effect of the mechanical property. They showed stretched nanofiber of PCL/PLGA (20:80) had faster drug release than unstretched nanofiber at the same period of time [102,103,104].
6.2 Regenerative Medicine
6.2.1 Bone
The bone is a highly complex, dynamic, and vascularized tissue that can remodel and regenerate itself in a limited area during life [105]. Despite this potential, large bone disorders like trauma, osteoporosis or tumor removal are major challenges which bone can’t treat itself [1]. Autogenous bone graft is the gold standard in bone dysfunction treatment. However, this approach has some drawbacks, including donor site pain and reduced movement [106]. Another technique for treating bone disorders is allogenic bone grafting, but it is not suitable due to immune responses and disease transmission [107]. Because of these drawbacks, bone regeneration is a viable option. 3D nanofiber scaffolds have a promising role in bone regeneration. Phase separation technique with the porogen leaching technique (TIPS & P) fabricate 3D scaffolds with interconnected pores [108]. In other work, the combination of thermally induced self-agglomeration (TISA technique) and freeze-drying method produces 3D short nanofiber. Yao et al. proved this scaffold can support BMP2 to bone regeneration [109]. Wang et al. fabricate biomimetic bone scaffold with a 3D printing method. These scaffolds have two parts; RGD-phage attaches to chitosan and a 3D composite of biphasic calcium phosphate (BCP), hydroxyapatite (HA) and β-tricalcium phosphate (β-TCP). The results showed a novel strategy for formation of vascularized bone and proposed new ideas for further research [110]. He et al. confirmed a novel scaffold which was fabricated with NFES technique has pores ~ 167 μm that guaranties better MT3T3-E1 cell line differentiation which leads to better bone regeneration [88].
6.2.2 Nerve
Nerve injury is an important clinical and social concern which requires essential intervention to restore their functions since it has limited regeneration capacity. Although small nerve defects can be improved by formation of tunnels along the axons, larger defects in PNS can’t regenerate themselves. Nowadays, using autografts is a gold standard therapeutic approach for the treatment of neural defects; however, it has several limitations, such as functional disorders, neuroma formation, and so on. Scientists follow new approaches to the treatment of neural disorders [111]. Many strategies for neural engineering, such as electrospun NFs, have recently been developed, however 3D scaffolds are the best option for 3D environments [112]. Melissinaki et al. achieved a new structure to improve neural disorders and developed neuronal cell guidance devices by femtosecond direct laser writing (DLW) to fabricate 3D scaffolds [113]. Gunay et al. demonstrated the 3D peptide nanofiber scaffolds was superior than its 2D structure for the induction of neural differentiation, even in the absence of a critical biological inducer in nerve growth factor (NGF) [114]. The organ-on-chips system is another technique for neural regeneration. In this technique, microfluidic technology caused better regeneration of neural tissue by the culture of different types of cells into the 3D model of tissue [115]. Serrano et al. described a 3D GOx scaffold with a novel ISISA method (ice segregation self-assembly). They observed ENPC cultures have high differentiation of neural network on these scaffolds [116]. In other work, results show 3D scaffolds based on MWCNTs and CS provide different signals for the improvement of neural damage [117].
6.2.3 Cartilage
Cartilage injuries cause joint instability and pain in damaged sites which can cause dysfunctionality. Cartilage injures can worsen over time and cause severe consequences like homeostasis and biomechanics problems in the joints. Human cartilage tissue can be damaged in exercise, accidents, or during autoimmune diseases such as arthritis. The results show OA in the population is 22.7% and by 2020, around 50 million people will struggle with OA in the U.S [118, 119]. Treatment of cartilage disorders associated with some limitations, including the poor regenerative capacity of cartilage tissue and biomechanical changes in the damaged site. Using tissue-engineered cartilage (TEC) has many advantages in this scope. The tissue-engineered cartilage requires a combination of 3D construction and chondrogenic cells and chondrocyte-related growth factors. Lee et al. assessed a microstereolithography system for fabrication of 3D scaffolds. They found chondrocyte adhesion and cellular activity over 2 weeks increased. These scaffolds have great potential for cartilage regeneration [118]. Moutos et al. used polycaprolactone (PCL) as a 3D-printed thermoplastic which showed success in cartilage and intervertebral disc regeneration [120]. In another study, Rosenzweig et al. confirmed 3D-printed acrylonitrile butadiene styrene (ABS) and polylactic acid (PLA) scaffolds have good features that provide a suitable condition for chondrogenic induction of chondrocytes or NP cells [121]. Also, Hung et al. created a new 3D printing scaffolds that can promote cartilage regeneration through MSC self-aggregation and confirmed the results of this research with rabbit knee implantation [122].
6.3 Treatment of Cancer
The World Health Organization has reported that cancer caused 10 million deaths in 2020, and it is one of the leading causes of death worldwide. Breast, lung and colorectal cancer are the most common cancers in the world [123, 124]. Thus, scientists are trying to develop new improvements in cancer diagnosis and treatment to increase life expectancy in these patients [125,126,127]. 3D NFs have great potential for diagnosis and treatment of cancer. Cancer models in the laboratory provide a powerful device for cancer research in several scopes, such as studying of carcinogens and their effects on cell signaling pathway of cancers or metastasis of tumors, studying drug effects, and cancer therapy. Previously, cells were cultured on 2D polystyrene surface to study, but this technique has a great disadvantage that cells, especially cancer cells, can’t match properly with this condition and don’t show their real face. Electrospun fibers are useful, because of their high similarity to collagen and other components of the tumor ECM. Kim et al. designed a gastric cancer model consisting of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) 3D electrospun scaffolds and collagen to test chemosensitivity of the tumor [128]. Besides, Mikos et al. created 3D scaffolds made of PCL nanofiber as an ex vivo ewing sarcoma model with better phenotype features than traditional monolayer cultures [129]. Mikos et al. developed novel 3D scaffolds that mimic ewing sarcoma, based on previous studies. This cancer model has a flow perfusion bioreactor that creates mechanical stimulation to mimic cell mechanical stress [130]. This method has better cell–cell signaling, tumor angiogenesis, biomechanical stimulation that are necessary for cancer prognosis, especially biomechanical stimulation [32].
7 Conclusion and Future Perspective
The manufacturing of a scaffold as the important subunit that provides mechanical properties, cell adherence, proliferation, and differentiation, could be achieved by altering important critical aspects such as material selection and ECM-like structure formation methods that are appropriate for medical application. 3D fiber scaffolds opened new opportunities for drug delivery, regenerative medicine, cancer treatment, and other disorders that affect everyone’s life. As a result, significant progress has been made in the development of 3D structures that are compatible with organ conditions and can enhance drug delivery ratio. Accordingly, scientists can employ both electrospinning and 3D printing technologies to develop 3D structures and apply the resulting 3D fiber scaffolds in clinics which we had short perspective reviews in this literature. Recently, four-dimensional (4D) printing has been used for fabricating smart scaffolds which can mimic the dynamic and structure of injured tissues. These scaffolds change their shape according to schedule. In other words, their shape alters after specific stimuli. More efforts should be made to evaluate the use of 4D printing nanofiber in drug delivery and regenerative medicine because these smart scaffolds require additional information for use in clinics.
Data Availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
References
S. Chen et al., Electrospinning: an enabling nanotechnology platform for drug delivery and regenerative medicine. Adv. Drug Deliv. Rev 132, 188–213 (2018)
Z. Liu et al., Looking into the future: toward advanced 3D biomaterials for stem-cell‐based Regenerative Medicine. Adv. Mater 30(17), 1705388 (2018)
B. Sun et al., Advances in three-dimensional nanofibrous macrostructures via electrospinning. Prog. Polym. Sci 39(5), 862–890 (2014)
J. Jiang et al., Expanded 3D nanofiber scaffolds: cell penetration, neovascularization, and host response. Adv. Healthc. Mater. 5(23), 2993–3003 (2016)
B. Dhandayuthapani et al., Polymeric scaffolds in tissue engineering application: a review International journal of polymer science, 2011. 2011
M. Dadashpour et al., Watercress-based electrospun nanofibrous scaffolds enhance proliferation and stemness preservation of human adipose-derived stem cells Artificial cells, nanomedicine, and biotechnology, 2018. 46(4): p. 819–830
L. Weng et al., Novel 3D hybrid nanofiber aerogels coupled with BMP-2 peptides for cranial bone regeneration. Adv. Healthc. Mater. 7(10), 1701415 (2018)
A. Haider, S. Haider, I.-K. Kang, A comprehensive review summarizing the effect of electrospinning parameters and potential applications of nanofibers in biomedical and biotechnology. Arab. J. Chem. 11(8), 1165–1188 (2018)
D.I. Braghirolli, D. Steffens, P. Pranke, Electrospinning for regenerative medicine: a review of the main topics. Drug discovery today 19(6), 743–753 (2014)
A. Formhals, Process and apparatus for preparing artificial threads. US Patent: 1975504 vol, 1934. 1: p. 7
J.F. Cooley, Apparatus for Electrically Dispersing Fluids (Google Patents, 1902)
S.N. Jayasinghe, Cell electrospinning: a novel tool for functionalising fibres, scaffolds and membranes with living cells and other advanced materials for regenerative biology and medicine. Analyst 138(8), 2215–2223 (2013)
R. Zamani et al., Recent advances in cell electrospining of natural and synthetic nanofibers for regenerative medicine. Drug Res. 68(08), 425–435 (2018)
T. Jiang et al., Electrospinning of polymer nanofibers for tissue regeneration. Prog. Polym. Sci 46, 1–24 (2015)
S. Thenmozhi et al., Electrospun nanofibers: new generation materials for advanced applications. Mater. Sci. Engineering: B 217, 36–48 (2017)
C.H. Kim et al., Effect of collector temperature on the porous structure of electrospun fibers. Macromol. Res 14(1), 59–65 (2006)
J. Lasprilla-Botero, M. Alvarez-Lainez, J. Lagaron, The influence of electrospinning parameters and solvent selection on the morphology and diameter of polyimide nanofibers. Mater. Today Commun. 14, 1–9 (2018)
J. Pelipenko et al., The impact of relative humidity during electrospinning on the morphology and mechanical properties of nanofibers. Int. J. Pharm 456(1), 125–134 (2013)
I.W. Fathona, A. Yabuki, One-step fabrication of short electrospun fibers using an electric spark. J. Mater. Process. Technol 213(11), 1894–1899 (2013)
A. Stoiljkovic, S. Agarwal, Short electrospun fibers by UV cutting method. Macromol. Mater. Eng 293(11), 895–899 (2008)
C. Yoshikawa et al., A Novel Shortened Electrospun Nanofiber Modified with a ‘concentrated’Polymer Brush (Science and Technology of Advanced Materials, 2011)
I.W. Fathona, A. Yabuki, A simple one-step fabrication of short polymer nanofibers via electrospinning. J. Mater. Sci 49(9), 3519–3528 (2014)
O. Kriha et al., Connection of hippocampal neurons by magnetically controlled movement of short electrospun polymer fibers—a route to magnetic micromanipulators. Adv. Mater 19(18), 2483–2485 (2007)
J. Wei et al., Spatial distribution and antitumor activities after intratumoral injection of fragmented fibers with loaded hydroxycamptothecin. Acta Biomater 23, 189–200 (2015)
M. Sawawi et al., Scission of electrospun polymer fibres by ultrasonication. Polymer 54(16), 4237–4252 (2013)
H. Zhang et al., Shape effects of electrospun fiber rods on the tissue distribution and antitumor efficacy. J. Controlled Release 244, 52–62 (2016)
B.S. Jha et al., Two pole air gap electrospinning: fabrication of highly aligned, three-dimensional scaffolds for nerve reconstruction. Acta Biomater 7(1), 203–215 (2011)
W. Teo, S. Ramakrishna, Electrospun fibre bundle made of aligned nanofibres over two fixed points. Nanotechnology 16(9), 1878 (2005)
P.D. Dalton, D. Klee, M. Möller, Electrospinning with dual collection rings. Polymer 46(3), 611–614 (2005)
E. Smit, U. Bűttner, R.D. Sanderson, Continuous yarns from electrospun fibers. Polymer 46(8), 2419–2423 (2005)
A. Lotus et al., Electrical, structural, and chemical properties of semiconducting metal oxide nanofiber yarns. J. Appl. Phys 103(2), 024910 (2008)
S. Chen et al., Emerging roles of electrospun nanofibers in cancer research. Adv. Healthc. Mater. 7(6), 1701024 (2018)
X. Xu et al., Preparation of core-sheath composite nanofibers by emulsion electrospinning. Macromol. Rapid Commun 27(19), 1637–1642 (2006)
I. Liao, S. Chew, K. Leong, Aligned core–shell nanofibers delivering bioactive proteins 2006
H. Yu et al., Regulation of biphasic drug release behavior by graphene oxide in polyvinyl pyrrolidone/poly (ε-caprolactone) core/sheath nanofiber mats. Colloids Surf., B 146, 63–69 (2016)
M. Cai et al., Efficient synthesis of PVDF/PI side-by-side bicomponent nanofiber membrane with enhanced mechanical strength and good thermal stability. Nanomaterials 9(1), 39 (2019)
S. Kidoaki, I.K. Kwon, T. Matsuda, Mesoscopic spatial designs of nano-and microfiber meshes for tissue-engineering matrix and scaffold based on newly devised multilayering and mixing electrospinning techniques. Biomaterials 26(1), 37–46 (2005)
Q.P. Pham, U. Sharma, A.G. Mikos, Electrospun poly (ε-caprolactone) microfiber and multilayer nanofiber/microfiber scaffolds: characterization of scaffolds and measurement of cellular infiltration. Biomacromolecules 7(10), 2796–2805 (2006)
S. Soliman et al., Multiscale three-dimensional scaffolds for soft tissue engineering via multimodal electrospinning. Acta Biomater 6(4), 1227–1237 (2010)
B.A. Blakeney et al., Cell infiltration and growth in a low density, uncompressed three-dimensional electrospun nanofibrous scaffold. Biomaterials 32(6), 1583–1590 (2011)
X. Zhu et al., Electrospun fibrous mats with high porosity as potential scaffolds for skin tissue engineering. Biomacromolecules 9(7), 1795–1801 (2008)
C. Vaquette, J.J. Cooper-White, Increasing electrospun scaffold pore size with tailored collectors for improved cell penetration. Acta Biomater 7(6), 2544–2557 (2011)
M.F. Leong et al., In vitro cell infiltration and in vivo cell infiltration and vascularization in a fibrous, highly porous poly (D, L-lactide) scaffold fabricated by cryogenic electrospinning technique Journal of Biomedical Materials Research Part A: An Official Journal of The Society for Biomaterials, The Japanese Society for Biomaterials, and The Australian Society for Biomaterials and the Korean Society for Biomaterials, 2009. 91(1): p. 231–240
C.S. Ki et al., Electrospun three-dimensional silk fibroin nanofibrous scaffold. J. Appl. Polym. Sci 106(6), 3922–3928 (2007)
Y. Yokoyama et al., Novel wet electrospinning system for fabrication of spongiform nanofiber 3-dimensional fabric. Mater. Lett 63(9–10), 754–756 (2009)
M.B. Taskin et al., Three-dimensional polydopamine functionalized coiled microfibrous scaffolds enhance human mesenchymal stem cells colonization and mild myofibroblastic differentiation. ACS Appl. Mater. Interfaces 8(25), 15864–15873 (2016)
W. Yang et al., In vivo bone generation via the endochondral pathway on three-dimensional electrospun fibers. Acta Biomater 9(1), 4505–4512 (2013)
H. Bagheri, F. Manshaei, O. Rezvani, Three-dimensional nanofiber scaffolds are superior to two-dimensional mats in micro-oriented extraction of chlorobenzenes. Microchim. Acta 185(7), 322 (2018)
T.J. Shin et al., Development of 3-D poly (trimethylenecarbonate-co-ε-caprolactone)-block-poly (p-dioxanone) scaffold for bone regeneration with high porosity using a wet electrospinning method. Biotechnol. Lett 32(6), 877–882 (2010)
S.F. Fennessey, R.J. Farris, Fabrication of aligned and molecularly oriented electrospun polyacrylonitrile nanofibers and the mechanical behavior of their twisted yarns. Polymer 45(12), 4217–4225 (2004)
B.B. Rothrauff et al., Braided and stacked electrospun nanofibrous scaffolds for tendon and ligament tissue engineering. Tissue Eng. Part A 23(9–10), 378–389 (2017)
J. Xie, B. Ma, P.L. Michael, Fabrication of novel 3D nanofiber scaffolds with anisotropic property and regular pores and their potential applications. Adv. Healthc. Mater. 1(5), 674–678 (2012)
R.E. Abhari, A.J. Carr, P.-A. Mouthuy, Multifilament electrospun scaffolds for soft tissue reconstruction, Electrofluidodynamic Technologies (EFDTs) for Biomaterials and Medical Devices. 2018, Elsevier. pp. 295–328
N. Baharivand et al., Relationship between vitreous and serum vascular endothelial growth factor levels, control of diabetes and microalbuminuria in proliferative diabetic retinopathy. Clinical Ophthalmology, 2012: p. 185–191
J.M. Ameer, A.K. PR, N. Kasoju, Strategies to tune electrospun scaffold porosity for effective cell response in tissue engineering. J. Funct. biomaterials 10(3), 30 (2019)
S. Lee, H.-S. Yun, S.-H. Kim, The comparative effects of mesoporous silica nanoparticles and colloidal silica on inflammation and apoptosis. Biomaterials 32(35), 9434–9443 (2011)
B.K. Gu et al., Fabrication of sonicated chitosan nanofiber mat with enlarged porosity for use as hemostatic materials. Carbohydr. Polym 97(1), 65–73 (2013)
W. Liu, S. Thomopoulos, Y. Xia, Electrospun nanofibers for regenerative medicine. Adv. Healthc. Mater. 1(1), 10–25 (2012)
B. Ma et al., Rational design of nanofiber scaffolds for orthopedic tissue repair and regeneration. Nanomedicine 8(9), 1459–1481 (2013)
J. Jiang et al., Expanding two-dimensional electrospun nanofiber membranes in the third dimension by a modified gas-foaming technique. ACS Biomaterials Science & Engineering 1(10), 991–1001 (2015)
Y.S. Nam, J.J. Yoon, T.G. Park, A novel fabrication method of macroporous biodegradable polymer scaffolds using gas foaming salt as a porogen additive. J. Biomedical Mater. Research: Official J. Soc. Biomaterials, The Japanese Society for Biomaterials, and The Australian Society for Biomaterials and the Korean Society for Biomaterials, 2000. 53(1): 1–7
M.K. Joshi et al., Multi-layered macroporous three-dimensional nanofibrous scaffold via a novel gas foaming technique. Chem. Eng. J 275, 79–88 (2015)
H. Zhang, S. Takeoka, Morphological evolution within spin-cast ultrathin polymer blend films clarified by a freestanding method. Macromolecules 45(10), 4315–4321 (2012)
A. Puiggalí-Jou et al., Nanoperforations in poly (lactic acid) free-standing nanomembranes to promote interactions with cell filopodia. Eur. Polymer J 75, 552–564 (2016)
G. Jin et al., SpONGE: spontaneous Organization of numerous-layer generation by Electrospray. Angew. Chem. Int. Ed 54(26), 7587–7591 (2015)
B.M. Baker et al., The potential to improve cell infiltration in composite fiber-aligned electrospun scaffolds by the selective removal of sacrificial fibers. Biomaterials 29(15), 2348–2358 (2008)
B.M. Baker et al., Sacrificial nanofibrous composites provide instruction without impediment and enable functional tissue formation Proceedings of the National Academy of Sciences, 2012. 109(35): p. 14176–14181
C. Gualandi et al., Nanovascularization of polymer matrix: generation of nanochannels and nanotubes by sacrificial electrospun fibers. Nano Lett 13(11), 5385–5390 (2013)
M. Aghajanpoor et al., The effect of increasing the pore size of nanofibrous scaffolds on the osteogenic cell culture using a combination of sacrificial agent electrospinning and ultrasonication. J. Biomedical Mater. Res. Part A 105(7), 1887–1899 (2017)
J. Jiang et al., CO2-expanded nanofiber scaffolds maintain activity of encapsulated bioactive materials and promote cellular infiltration and positive host response. Acta Biomater 68, 237–248 (2018)
G. Kim et al., Hybrid process for fabricating 3D hierarchical scaffolds combining rapid prototyping and electrospinning. Macromol. Rapid Commun 29(19), 1577–1581 (2008)
S. Khorshidi et al., A review of key challenges of electrospun scaffolds for tissue-engineering applications. J. Tissue Eng. Regen. Med 10(9), 715–738 (2016)
F. Klein et al., Elastic fully three-dimensional microstructure scaffolds for cell force measurements. Adv. Mater 22(8), 868–871 (2010)
S.H. Park et al., Development of dual scale scaffolds via direct polymer melt deposition and electrospinning for applications in tissue regeneration. Acta Biomater 4(5), 1198–1207 (2008)
W. Lee, C. Wei, S.-C. Chung, Development of a hybrid rapid prototyping system using low-cost fused deposition modeling and five-axis machining. J. Mater. Process. Technol 214(11), 2366–2374 (2014)
A. Repanas, S. Andriopoulou, B. Glasmacher, The significance of electrospinning as a method to create fibrous scaffolds for biomedical engineering and drug delivery applications. J. Drug Deliv. Sci. Technol 31, 137–146 (2016)
D. Sun et al., Near-field electrospinning. Nano Lett 6(4), 839–842 (2006)
X.-X. He et al., Near-field electrospinning: progress and applications. J. Phys. Chem. C 121(16), 8663–8678 (2017)
G. Luo et al., Direct-write, self-aligned electrospinning on paper for controllable fabrication of three-dimensional structures. ACS Appl. Mater. Interfaces 7(50), 27765–27770 (2015)
R. Tan, X. Yang, Y. Shen, Robot-aided electrospinning toward intelligent biomedical engineering. Rob. biomimetics 4(1), 1–13 (2017)
Y. Yang et al., Effect of electric field distribution uniformity on electrospinning. J. Appl. Phys 103(10), 104307 (2008)
L. Zhao et al., Evaluation of width and width uniformity of near-field electrospinning printed micro and sub-micrometer lines based on optical image processing. J. Micromech. Microeng 28(3), 035010 (2018)
T. Okuda, K. Tominaga, S. Kidoaki, Time-programmed dual release formulation by multilayered drug-loaded nanofiber meshes. J. Controlled Release 143(2), 258–264 (2010)
S. Sankar et al., Electrospun fibers for recruitment and differentiation of stem cells in regenerative medicine. Biotechnol. J 12(12), 1700263 (2017)
Y. Li et al., Cell-free 3D wet-electrospun PCL/silk fibroin/Sr2 + scaffold promotes successful total meniscus regeneration in a rabbit model. Acta Biomater 113, 196–209 (2020)
S.A. Poursamar et al., Potential application of gelatin scaffolds prepared through in situ gas foaming in skin tissue engineering. Int. J. Polym. Mater. Polym. Biomaterials 65(6), 315–322 (2016)
P. Song et al., Novel 3D porous biocomposite scaffolds fabricated by fused deposition modeling and gas foaming combined technology. Compos. Part B: Eng. 152, 151–159 (2018)
F.-L. He et al., A novel layer-structured scaffold with large pore sizes suitable for 3D cell culture prepared by near-field electrospinning. Mater. Sci. Engineering: C 86, 18–27 (2018)
J.E. Samorezov, E. Alsberg, Spatial regulation of controlled bioactive factor delivery for bone tissue engineering. Adv. Drug Deliv. Rev 84, 45–67 (2015)
M. Mofarrah et al., Fabricating ZSM-5 zeolite/polycaprolactone nano-fibers co-loaded with dexamethasone and ascorbic acid: potential application in osteogenic differentiation of human adipose-derived stem cells. J. Drug Deliv. Sci. Technol 79, 103999 (2023)
S. Sahabi, D. Jafari-Gharabaghlou, N. Zarghami, A new insight into cell biological and biochemical changes through aging. Acta Histochem 124(1), 151841 (2022)
H. Serati-Nouri et al., Biomedical applications of zeolite-based materials: a review. Mater. Sci. Engineering: C 116, 111225 (2020)
R. Goyal et al., Nanoparticles and nanofibers for topical drug delivery. J. Controlled Release 240, 77–92 (2016)
M.F.S. Jadid et al., Enhanced anti-cancer effect of curcumin loaded-niosomal nanoparticles in combination with heat-killed Saccharomyces cerevisiae against human colon cancer cells. Journal of Drug Delivery Science and Technology, 2023: p. 104167
E. Salmani Javan et al., Development of a magnetic nanostructure for co-delivery of metformin and silibinin on growth of lung cancer cells: possible action through leptin gene and its receptor regulation. Asian Pac. J. Cancer Prev 23(2), 519–527 (2022)
Y. Alagheband et al., Design and fabrication of a dual-drug loaded nano-platform for synergistic anticancer and cytotoxicity effects on the expression of leptin in lung cancer treatment. J. Drug Deliv. Sci. Technol 73, 103389 (2022)
N. Goonoo, A. Bhaw-Luximon, D. Jhurry, Drug loading and release from electrospun biodegradable nanofibers. J. Biomed. Nanotechnol 10(9), 2173–2199 (2014)
S.T. Yohe et al., Triggered drug release from Superhydrophobic meshes using high-intensity focused Ultrasound. Adv. Healthc. Mater. 2(9), 1204–1208 (2013)
S. Demirci et al., pH-responsive nanofibers with controlled drug release properties. Polym. Chem 5(6), 2050–2056 (2014)
J. Hrib et al., Nanofibers for drug delivery–incorporation and release of model molecules, influence of molecular weight and polymer structure. Beilstein J. Nanotechnol 6(1), 1939–1945 (2015)
M.V. Natu, H.C. de Sousa, M. Gil, Effects of drug solubility, state and loading on controlled release in bicomponent electrospun fibers. Int. J. Pharm 397(1–2), 50–58 (2010)
S.-F. Chou, D. Carson, K.A. Woodrow, Current strategies for sustaining drug release from electrospun nanofibers. J. Controlled Release 220, 584–591 (2015)
S.-F. Chou, K.A. Woodrow, Relationships between mechanical properties and drug release from electrospun fibers of PCL and PLGA blends. J. Mech. Behav. Biomed. Mater 65, 724–733 (2017)
A. Eatemadi et al., Synthesis and characterization of chrysin-loaded PCL-PEG-PCL nanoparticle and its effect on breast cancer cell line. Biomed. Pharmacother 84, 1915–1922 (2016)
J.T. Ratnayake, M. Mucalo, G.J. Dias, Substituted hydroxyapatites for bone regeneration: a review of current trends. J. Biomedical Mater. Res. Part B: Appl. Biomaterials 105(5), 1285–1299 (2017)
P. Chatakun et al., The effect of five proteins on stem cells used for osteoblast differentiation and proliferation: a current review of the literature. Cell. Mol. Life Sci 71(1), 113–142 (2014)
S. Morelli et al., Osteogenic and osteoclastogenic differentiation of co-cultured cells in polylactic acid–nanohydroxyapatite fiber scaffolds. J. Biotechnol 204, 53–62 (2015)
X. Liu, P.X. Ma, Phase separation, pore structure, and properties of nanofibrous gelatin scaffolds. Biomaterials 30(25), 4094–4103 (2009)
Q. Yao et al., Three dimensional electrospun PCL/PLA blend nanofibrous scaffolds with significantly improved stem cells osteogenic differentiation and cranial bone formation. Biomaterials 115, 115–127 (2017)
J. Wang et al., Phage nanofibers induce vascularized osteogenesis in 3D printed bone scaffolds. Adv. Mater 26(29), 4961–4966 (2014)
A. Subramanian, U.M. Krishnan, S. Sethuraman, Fabrication of uniaxially aligned 3D electrospun scaffolds for neural regeneration. Biomed. Mater 6(2), 025004 (2011)
J. Wu et al., Biomimetic nanofibrous scaffolds for neural tissue engineering and drug development. Drug discovery today 22(9), 1375–1384 (2017)
V. Melissinaki et al., Direct laser writing of 3D scaffolds for neural tissue engineering applications. Biofabrication 3(4), 045005 (2011)
G. Gunay et al., Three-dimensional laminin mimetic peptide Nanofiber Gels for in vitro neural differentiation. Biotechnol. J 12(12), 1700080 (2017)
S.N. Bhatia, D.E. Ingber, Microfluidic organs-on-chips. Nat. Biotechnol 32(8), 760 (2014)
M. Serrano et al., 3D free-standing porous scaffolds made of graphene oxide as substrates for neural cell growth. J. Mater. Chem. B 2(34), 5698–5706 (2014)
M.C. Serrano et al., Chondroitin sulphate-based 3D scaffolds containing MWCNTs for nervous tissue repair. Biomaterials 35(5), 1543–1551 (2014)
S.-J. Lee et al., Application of microstereolithography in the development of three-dimensional cartilage regeneration scaffolds. Biomed. Microdevices 10(2), 233–241 (2008)
T.L. Fernandes et al., Macrophage: a potential target on cartilage regeneration. Front. Immunol 11, 111 (2020)
F.T. Moutos, F. Guilak, Functional properties of cell-seeded three-dimensionally woven poly (ε-caprolactone) scaffolds for cartilage tissue engineering. Tissue Eng. Part A 16(4), 1291–1301 (2010)
D.H. Rosenzweig et al., 3D-printed ABS and PLA scaffolds for cartilage and nucleus pulposus tissue regeneration International journal of molecular sciences, 2015. 16(7): p. 15118–15135
K.-C. Hung et al., Water-based polyurethane 3D printed scaffolds with controlled release function for customized cartilage tissue engineering. Biomaterials 83, 156–168 (2016)
F. Bray et al., Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries CA: a cancer journal for clinicians, 2018. 68(6): p. 394–424
H. Sung, J. Ferlay, R.L. Siegel, Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries 2021. 71(3): p. 209–249
R. Contreras-Cáceres et al., Electrospun nanofibers: recent applications in drug delivery and cancer therapy. Nanomaterials 9(4), 656 (2019)
N. Hassani et al., The Effect of Dual Bioactive Compounds Artemisinin and Metformin Co-loaded in PLGA-PEG Nano-particles on breast Cancer cell lines: potential apoptotic and anti-proliferative action. Applied Biochemistry and Biotechnology, 2022: p. 1–16
S. Fathi Karkan et al., Magnetic nanoparticles in cancer diagnosis and treatment: a review Artificial cells, nanomedicine, and biotechnology, 2017. 45(1): p. 1–5
Y.-J. Kim et al., Three-dimensional gastric cancer cell culture using nanofiber scaffold for chemosensitivity test. Int. J. Biol. Macromol 45(1), 65–71 (2009)
E.L.S. Fong et al., Modeling Ewing sarcoma tumors in vitro with 3D scaffolds Proceedings of the National Academy of Sciences, 2013. 110(16): p. 6500–6505
M. Santoro et al., Flow perfusion effects on three-dimensional culture and drug sensitivity of Ewing sarcoma Proceedings of the National Academy of Sciences, 2015. 112(33): p. 10304–10309
Acknowledgements
Not Applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Nazila Shabestani: Investigation; Writing; Davoud Jafari-Gharabaghlou: Investigation, Writing, Validation; Somayeh Gholami: Investigation, Writing; Nosratollah Zarghami: Supervision, Validation, Writing- Reviewing and Editing.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
Authors declare they have no financial interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shabestani, N., Jafari-Gharabaghlou, D., Gholami, S. et al. An Overview of the Various Nanofiber Scaffolds Techniques with a Focus on the 3D Nanofiber-based Scaffolds Application in Medicine. J Inorg Organomet Polym 33, 3355–3371 (2023). https://doi.org/10.1007/s10904-023-02560-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-023-02560-9