Abstract
Deaths from liver cancer are on the rise and disproportionately affect minority racial/ethnic groups. In this study, we examined associations between physicians’ recommendations for hepatitis B virus (HBV) and hepatitis C virus (HCV) screening and sociodemographic and lifestyle factors among minority populations in the areas of Greater Philadelphia and New York City. Using Poisson regression with robust variance estimation, we evaluated potential associations for 576 Hispanic American (HA), African American (AA), and Asian Pacific American (APA) adults, using blood tests as an outcome measure, with adjustment for sociodemographic factors We found that APAs (34.2%) were most likely to have a physician recommend HBV and HCV screening tests (34.2% and 27.1%, respectively), while HAs were least likely to receive an HBV recommendation (15.0%) and AAs were least likely to receive an HCV recommendation (15.3%). HAs were significantly likely to have never received a blood test for either HBV or HCV (RR = 1.25, 95% CI: 1.05, 1.49). APAs were significantly more likely to receive a screening recommendation for HBV (RR = 1.10, 95%CI: 1.01, 1.20) and to have a blood test (RR = 1.57, 95% CI: 1.06, 2.33). Our findings show that, among HAs, AAs, and APAs, physician recommendations are strongly associated with patients undergoing blood tests for HBV and HCV and that minority populations should increasingly be recommended to screen for HBV and HCV, especially given their elevated risk.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Deaths from liver cancer have increased in the United States (U.S.) [1]. The incidence of liver cancer is associated with several risk factors, including gender, age, diet, and race/ethnicity, as well as infection with hepatitis B virus (HBV) or hepatitis C virus (HCV). HBV and HCV together account for 65% of liver cancer incidence in the U.S. [2,3,4].
Among primary liver cancers, hepatocellular carcinoma (HCC) is the most prevalent form. Historically, in the U.S., the highest HCC incidence rates were observed in Asian Pacific Americans (APAs) [5], mainly due to the high prevalence of HBV infection among immigrants from HBV-endemic areas. Although APAs make up 6% of the U.S. population, they account for more than 50% of Americans with chronic hepatitis B [6]. As a result, APAs are 8–13 times more likely than non-Hispanic whites (NHWs) to develop liver cancer, with a 60% higher death rate.
Hispanic American (HA) men and women have the fastest-rising incidence rates of HCC among all racial/ethnic groups [7,8,9]. Previous research has shown that mortality from HCC is lower among immigrant HAs than among HA men born in the U.S. [8]. While the reasons for this disparity are unclear, native HAs may be affected by a higher prevalence of HCC risk factors, including HCV infection and lifestyle factors related to obesity and diabetes mellitus [8].
Liver cancer incidence rates are also higher among African Americans (AAs) (10.2 per 100,000) compared to NHWs (6.3 per 100,000) [10]. Evidence suggests that among AAs, HCV is the population-attributable fraction (PAF) most likely influencing HCC, contributing to more than 36% of cases [4]. Additionally, aging of the baby boomer birth cohort (individuals born between 1945 and 1965) contributes to elevated risk among AA adults [9, 10]. In the U.S., about three-quarters of chronic HCV infections occur among baby boomers [11].
The Centers for Disease Control and Prevention (CDC) recommends HCV screening at least once in a lifetime for all adults aged > 18 years and HBV screening for adults aged 19–59 years and adults ≥ 60 years with risk factors for hepatitis [12, 13]. Nevertheless, evident disparities exist in HBV and HCV screening rates for minority groups [14]. AAs are the least likely to be screened for HBV, followed by Asian and Hispanic adults, despite being populations with known cancer disparities [15]. Analysis of the 2009–2010 Racial and Ethnic Approaches to Community Health Across the U.S. Risk Factor Survey, involving 53,896 minority adults, showed that only 19% of participants were ever tested for HCV [16]. Hispanics and APAs were the least likely to be screened, followed by AAs and indigenous individuals. Research shows that a strong provider recommendation is an essential factor in the uptake of preventive services, including the HBV and HCV screening [17, 18]. However, In the U.S., physicians do not usually test for HBV and HCV unless it is requested by the patient or the patient is a pregnant woman, a high-risk individual with a current or past history of illicit injection drug use, or is a donor for blood transfusions [19].
The objective of this paper is to assess minority community sociodemographic factors that are associated with physicians’ recommendations for HBV and HCV screening. Knowledge of such associations can potentially help physicians and public health professionals develop interventions for increasing HBV and HCV screening among minority populations.
Methods
Study Design and Participants
This project is part of a 5-Year Temple University/Fox Chase Cancer Center and Hunter College (TUFCCC/HC), U54 Regional Cancer Health Disparity Partnership funded by the National Cancer Institute. This Partnership aims to reduce cancer health disparities among underserved minority populations in the Pennsylvania-New Jersey-New York City (PNN) region through cancer disparities research, community outreach, and career development for underrepresented early-stage investigators and students. A strength of the Partnership is its Community Outreach Core (COC). One of the aims of the COC is to engage individuals in the community in the process of implementing liver, colorectal, and lung cancer education initiatives in underserved African, Asian, and Hispanic American disparity populations. In the first three years of the TUFCCC/HC Partnership program, we focused on HBV/HCV and liver cancer education prevention; we engaged 50 community-based organizations (CBOs) serving the AA, APA, and HA communities in the PNN region. The CBO leaders and staff were actively engaged in developing participant recruitment strategies, conducting outreach, co-delivering educational workshops, and collecting survey data at collaborating CBO sites. This study used baseline data from the initiative of HBV/HCV and liver cancer education prevention. Eligibility criteria included that participant be at least 18 years of age; self-identify as AA, APA, or HA; and reside in New York City and the greater Philadelphia metropolitan area during the enrollment period.
The baseline surveys were provided in multiple formats (paper or online via Qualtrics software; copyright © 2020 Qualtrics, Provo, UT) to accommodate the needs of the participants. In addition, trained multilingual community educators helped participants who needed assistance for understanding survey questions. In this analysis, we assessed the likelihood of a participant’s physician providing a recommendation for HBV or HCV screening and the likelihood of the individual undergoing a blood test based on demographic, lifestyle, and behavioral factors. A detailed study protocol has been published previously [20].
Measurements
Participants self-reported whether they were ever recommended to receive an HBV or HCV screening blood test. If they reported that they did not receive a recommendation for either HBV or HCV, we coded the response as not receiving a recommendation for either. Similarly, we asked participants to report whether they ever received a screening blood test for either HBV or HCV or neither.
Lifestyle and behavioral factors were self-reported by the study participant. To assess alcohol consumption, we asked participants how many servings of alcohol they usually have per week by alcohol type: beer, wine, or liquor. If a male consumed more than two servings of alcohol per day, we classified the individual as being an excessive drinker; for females, the cutoff was more than one drink per day [21]. To find other risk factors for liver cancer, we asked participants if a physician had previously diagnosed them with diabetes, high blood pressure, heart disease, or liver disease. We also asked participants whether any of their family members had liver disease. These questions helped us determine whether participants had any risk factors for liver cancer or whether their physicians would be concerned that they may be at high risk of developing liver cancer.
Statistical Analysis
We characterized the distributions of all covariates and clinical factors. We used chi-square tests to compare the categorical demographic characteristics and lifestyle and behavioral factors across AA, APA, and HAs. We used ANOVA to assess differences in the likelihood of physicians recommending testing for HBV and HCV and differences in patients receiving blood tests for the viruses across the ethnic and racial groups. We used Poisson regression with robust variance estimation [22] to evaluate associations between physicians’ recommendations for testing and patients receiving blood tests. Race and ethnicity were used as predictors adjusted for demographics, and lifestyle factors were used as covariates selected a prior based on literature. We adjusted for age [23], sex [24,25,26], quality of health [27], smoking status [28], physical activity [29], excessive drinking [30], number of preexisting health conditions (0–3) [31], and family history of liver cancer. Finally, we examined model residuals to assess the adequacy of models.
We set significance as α = 0.05 for a 2-tailed test. We did not correct for multiple testing to increase sensitivity for detecting associations to be confirmed by a future investigation. We used R studio (version 4.0.3; R Development Core Team) for the statistical analysis.
Results
Demographic Distribution of Participants
Table 1 describes the demographic factors of 181 males and 395 females participating in this study. We had 167 individuals of Hispanic, Latino, or Spanish origin; 25 individuals identifying as white; 137 AA or Black participants; 266 APA; and 15 adults identifying as other. Our HA (73.7%) and AA (62.0%) participants were primarily born in the U.S., but most APA (95.1%) participants were foreign-born. A majority of our participants earned less than $19,999 annually (n = 296, 51.4%).
Lifestyle and Behavioral Factors
The lifestyle and behavioral factors of our study participants are in Table 2. Most study participants said their overall health quality was good or very good. However, most APA adults said their health was only fair (38.3%). Most of our participants did not consume excessive alcohol, with 2 drinks per day for males and one drink per day for females [21]. Many HA and APA adults had either pre-existing high blood pressure (27.5% and 27.4%, respectively) or diabetes (26.3% and 16.2%, respectively). AA (51.1%) and white HA (35.4%) participants mostly had pre-existing high blood pressure. While many study participants did not have a family history of liver disease, 21.1% of APA participants reported having a family member with liver disease.
Physician-Recommendation for HBV/HCV Screening
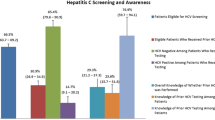
Of the 167 HA participants, 35 (15.0%) reported a physician-recommended HBV screening test and 15.6% (n = 26) received an HCV screening recommendation. Of 137 AA participants, 31 (22.6%) received screening recommendations for HBV and 21 (15.3%) for HCV screening tests. We had 265 APAs participants, 91 of whom (34.2%) reported physician-recommended HBV screening, with 72 (27.1%) having received recommendations for HCV testing.
Self-reported Screening for HBV/HCV
Of the 35 HA individuals who received a physician HBV screening recommendation, 26 (74.3%) followed through and underwent a blood test for HBV. Of the HA participants with recommendations for HCV screening, 88.5% (n = 23) received a blood test. Of the AA participants with a physician screening recommendation, 90.3% received a blood test for HBV and 123.8% received an HCV screening test. Five AA participants proactively asked their physicians for an HCV screening test. Eighty-one (89.0%) of the 91 APAs with a doctor’s recommendation for an HBV test followed through and had the test completed. One-third (33.3%, n = 24) of APAs with an HCV recommendation received the blood test.
The association between race/ethnicity and physicians’ recommendation for testing, along with completion of blood tests completed, is shown in Supplemental Table 1. Supplemental Table 2 shows demographic, lifestyle, and behavioral factors and their association with physicians’ recommendations for testing for HBV and HCV and the respective blood tests. Supplemental Table 3 displays associations of demographic, lifestyle, and behavioral factors with race and ethnicity.
Table 3 shows the results of the Poisson regression analysis for race/ethnicity, physician’s recommendations for HBV and HCV testing, and actual blood tests for 572 adults, adjusted for demographic, lifestyle, and behavioral factors. We found that physicians were less likely to recommend HA adults for receiving a blood test for HBV (relative risk (RR) = 0.95, 95% confidence interval (CI): 0.87, 1.03)) or HCV (RR = 0.93, 95% CI: 0.85, 1.01), albeit without any statistical significance. Hence HA adults were less likely to have received a blood test for HBV (RR = 0.75, 95% CI: 0.53, 1.08) or HCV (RR = 0.77, 95% CI: 0.45, 1.32). As expected, HA individuals were significantly likely to have never undergone a blood test for either HBV or HCV (RR = 1.25, 95% CI: 1.05, 1.49). Similar to the larger HA group, the HA white adults were less likely to have a physician recommend HBV or HCV (RR = 0.90, 95% CI: 0.78, 1.05) testing and were less likely to receive a blood test for either HBV or HCV (RR = 1.18, 95% CI: 0.92, 1.52).
While AA adults were less likely to have a physician recommend undergoing a blood test for HBV (RR = 0.95, 95% CI: 0.87, 1.04) or HCV (RR = 0.91, 95% CI: 0.83, 1.00), they were more likely to have a blood test done for HCV (RR = 1.42, 95% CI: 0.83, 2.44), although this was not a statistically significant finding. Unlike the other ethnic and racial groups in our study, APA adults were significantly more likely to have a physician recommend both HBV (RR = 1.10, 95% CI: 1.01, 1.20) and HCV (RR = 1.10, 95% CI: 1.00, 1.20) testing and had higher likelihoods of following through and receiving a blood test for HBV (RR = 1.57, 95% CI: 1.06, 2.33). Supplemental Table 4 shows the unadjusted association between race/ethnicity, physician’s recommendations for HBV and HCV testing, and blood tests conducted. The findings of both the unadjusted and the adjusted results were similar, though the relative risks were closer to the null finding in the covariate-adjusted models.
Discussion
Despite medical advancements, liver cancer-related deaths have increased in the U.S., with primary liver cancer being the 7th most significant contributor to cancer mortality [1]. In this study, we aimed to examine differences in sociodemographic factors and lifestyle behaviors among minority populations in the U.S. in the context of physician recommendations for HBV and HCV screening. We found that HA and AA adults were less likely to have a physician recommend HBV or HCV screening, despite having the highest prevalence of HCC. Conversely, APAs were most likely to have a physician recommend screening tests for both HBV and HCV. Given the endemic nature of chronic hepatitis B in Asian countries, a baseline HBV recommendation rate of approximately 34 out of every 100 individuals is relatively low. Specifically for HCV, APAs had the lowest baseline rate of receiving a blood test for HCV and the lowest uptake of physician screening recommendations for HCV tests. For the HBV screening tests, APAs had the highest follow-through on physician’s recommendations, followed by AAs and HAs. We found that follow-through on physician screening recommendations ranged from 33.3 to 123.8% across all our communities. Uptake of HCV screening was the highest among AAs, followed by HAs and APAs.
HBV and HCV Infection among APAs
APAs were most likely to have a physician recommend HBV screening, and a majority of participants followed through (89.0%). Analysis of NHANES 1999–2016 data found no statistically significant differences in the liver disease awareness rate between U.S.-born and foreign-born persons with HBV infection [32]. However, evidence shows that HBV infection is more prevalent among foreign-born individuals. Chinese Americans have a three-fold higher risk of dying from liver cancer compared to NHWs, Koreans have an eight-fold higher risk, and Vietnamese Americans have a 13-fold higher risk [33, 34]. The prevalence of HBV screening and vaccination among APAs and ethnic differences for factors associated with screening and vaccination behaviors have been investigated previously [35]. One study found that 47% percent of participants reported having received HBV screening [36]–a higher rate of screening recommendations by physicians than our study (34.2%).
We also found that fewer than 30% of APA participants received a physician recommendation for HCV screening, and only one-third of those with a screening recommendation followed through on the recommendation. Screening for HCV was poorer than HBV in our study population, suggesting that intervention/prevention needs to be stronger for HCV.
HBV and HCV Infection among AAs/ Blacks
In this study, we found that fewer than one out of every four AA participants received a recommendation for HBV screening; fewer than one out of every five AAs received an HCV screening recommendation. About 13% of the U.S. population is represented by AAs; of the estimated 3.2 million persons with chronic HCV infection, however, AAs make up roughly 23% [37,38,39]. HCV infection rates are high particularly among Black men, with the highest prevalence rate, 9.8%, occurring among Black men ages 40 to 49 years [38]. Moreover, among AAs ages 45–64, chronic liver disease, often chronic hepatitis C-related, is a major cause of death [38, 39].
An analysis calculating PAFs for each HCC risk factor found that for AAs, HCV had the greatest PAF (36.1%, 95%CI = 31.8–40.4) [4]. Metabolic disorders had a PAF of 14.4% (95%CI = 6.4–22.3), and the PAF of smoking was 10.6% (95%CI = 2.8–18.5). Among AAs, the high burden of HCV-related HCC is consistent with race-specific HCV prevalence estimates from U.S. surveys [40]. However, data on the natural history and treatment of hepatitis C in the AA population is relatively scarce. Compared with NHWs, AAs have lower viral clearance rates, resulting in higher incidence of chronic hepatitis C [41]. AAs, however, may also have a lower rate of fibrosis progression than NHWs. In the case of hepatitis C-related cirrhosis, HCC- and liver cancer-related mortality among AAs is higher than NHWs.
Most AAs in the U.S. are native-born. However, the population of Black immigrants is increasing; in 2019, 4.6 million Blacks living in the U.S. had been born outside of the country [42]. Among foreign-born AAs, sexual and vertical transmission of hepatitis are the most common sources of infection. The adult cohort study of the Hepatitis B Research Network found that in AAs, transmission, clinical presentation, and viral characteristics of HBV vary extensively [43]. This suggests that while efforts are underway to improve awareness of HBV and the need for screening, more research is needed to determine why foreign-born AAs are at higher risk of contracting HBV to identify impactful intervention points. Sociodemographic variables, such as those identified in this study, are useful for targeted intervention.
HBV and HCV Infection among HAs
We found that HAs were the least likely group to receive a physician recommendation for HBV, and fewer than one-in-five were likely to receive an HCV screening recommendation. This is alarming, since Hispanic individuals have the highest age-adjusted incidence rates of HCC, surpassing that of Asians, the previously most-affected ethnic group [8]. The yearly age-adjusted incidence rates of HCC for HA individuals are at least 2.5 times higher than their NHW counterparts. The HA population is currently the second largest ethnic population in the U.S.; rising HCC mortality among HAs may be driven by increasing growth in the population of this group [44].
Hispanic individuals tend to be diagnosed with HCC at later stages than other racial/ethnic groups. HAs who are diagnosed with early-stage HCC have significantly decreased rates of receiving curative therapy [44, 45]. The reasons behind these disparities were investigated in a large, multicenter, cohort study in the U.S., [44] the findings of which suggest that social determinants and access to health care play a vital role. In particular, reduced rates of screening among HAs potentially contributes to delays in diagnosis, resulting in more advanced disease. While the Advisory Committee on Immunization Practices (ACIP) recommendation for universal infant hepatitis B vaccination has successfully reduced HCV prevalence in the U.S., current practices should be updated to include regular screening of all Americans for HBV and HCV as part of their primary care.
Other factors that potentially contribute to differences in HCC between HA and NHW patients are variations in rates of metabolic syndrome and nonalcoholic fatty liver disease, both of which are independently associated with increased risk of HCC. A study from 2013 found that, among individuals aged 68 or older, metabolic disorders were the most significant PAF contributing to HCC among whites and Hispanics, with approximately a 39% contribution to HCC cases diagnosed between 1994 and 2007 [4]. Given the prevalence of metabolic syndrome and its components in HA patients, and the increased risk that these risk factors pose for the development of HCC, both general practitioners and researchers should work collectively to advise patients to prevent the development of metabolic syndrome [44].
Physician’s Recommendation for HBV and HCV Screening Tests
Minority populations in the U.S. have low HBV and HCV screening rates. A national survey of 53,896 minority individuals found that overall, 39.2% of respondents reported having undergone testing for hepatitis B, with percentages varying from 42.5% (95% CI: 41.4–43.6) among APAs to 35.6% (95% CI: 34.7–36.5) among HAs [46]. The same survey found that overall, 19.5% of individuals had received testing for hepatitis C; testing rate varied between groups (AAs, 20.7%; APAs, 17.7%; HAs, 17.3%) [16]. Although access to health insurance, along with factors such as age and education status, influence hepatitis B testing, whether an individual is currently seeing a physician and having regular primary care has greater influence over testing [16, 36, 46]. Our study found that, in most cases, participants who received a physician recommendation followed through with screening blood tests. Compliance with HBV screening tests ranged from 74.3 to 89.0% [47].
Despite the high prevalence of hepatitis B among racial/ethnic minorities, a lack of awareness about HBV risk factors among some providers potentially hinders testing. A survey showed that fewer than 70% of family physicians would recommend screening for immigrants from HBV-endemic regions; less than half would recommend testing for household contacts of HBV-infected persons [48]. Overall, providers were found to be only slightly more knowledgeable about HBV than HCV screening. For patients who test positive for HBV, 21% of physicians were uncertain of next steps and would refer patients to a specialist for follow-up. In the case of HCV, 31% of physicians were unaware of additional steps. Screening rates were highest for physicians who had been in practice for 10–20 years (p-value = 0.04), and screening for HCV was more likely to be recommended or carried out by physicians in academic settings.
Research suggests that medical school and residency training do not adequately prepare physicians for screening and management of chronic hepatitis [48,49,50,51,52,53]. Whether patients undergo testing is associated with physician recommendation and patient request. If a physician recommends screening, most patients are likely to follow though [36, 46, 54]. However, most of the time physicians are unable to identify the appropriate serologic test to screen for chronic hepatitis. Lack of understanding about HBV and HCV prevents physicians from adequately advising patients on next steps.
Awareness of HBV and HCV tends to be especially low among younger providers [51, 52]. Thus, efforts to improve patient adherence to testing should target not only patients but also medical trainees, with emphasis on the importance of obtaining relevant patient histories and with instruction on identifying HBV and HCV risk factors in patients, including sociodemographic factors. Providers should be encouraged to periodically review peer-reviewed investigations on the etiology and risk factors of liver diseases to better understand and provide optimal preventative care.
Strengths and Limitations
This study has several strengths and limitations. This study overlapped with the onset of the coronavirus disease 2019 (COVID-19) pandemic. We were able to continue with our research through the use of digital platforms, such as Zoom and teleservices. However, we cannot disentangle our analysis from the impact of the COVID-19 pandemic. Strict stay-at-home orders resulted in significant decreasing trends in the total number of liver clinic visits overall (p-trend = 0.038) [55]. Hepatoma surveillance and diagnostic abdominal imaging fell dramatically. These decreases could have impacted our data. The impacts likely would have been small, however, given that our outcomes are consistent with similar studies conducted before COVID-19. Our study may suffer from sampling/response bias, as we do not have a random sample and our participants are slightly biased by gender, as is frequent in other studies [56]. Additionally, our participants were concentrated in the greater Philadelphia metropolitan area and the New York City area, limiting our ability to generalize and extrapolate our findings to racial/ethnic groups across the U.S. Nonetheless, the findings of similar studies support our results [15, 16, 41, 46, 54]. We cannot make causal interpretations and only report associations, as we used a baseline survey.
Although some findings in our study may have been due to chance, we have a moderate sample size, giving us power to find statistically significant results. Additionally, we recruited more than 550 participants, despite the COVID-19 pandemic, due to our strong COC and historic partnership with local CBOs. Nevertheless, our study population is unlikely to represent all HAs, HA whites, AAs, and APAs and their probability of HBV and HCV screening.
Conclusion
The burden of liver cancer in the U.S. is unevenly distributed by gender, age, and race/ethnicity. We found that HA and AA adults were less likely to have a physician recommend HBV or HCV screening tests and were less likely to undergo testing for HBV and HCV, despite having the highest prevalence of HCC and HCV, respectively. APAs were most likely to have a physician recommend screening tests for HBV and had the highest follow-through. This shows that APAs have some awareness of the severity of HBV and the associated risk of liver cancer. However, there remains a significant need for targeted, culturally sensitive education on liver cancer risks and preventative measures, including screening blood tests and antiviral treatment for chronic HBV infection.
References
Siegel, R. L., Miller, K. D., & Jemal, A. (2019). Cancer statistics, 2019. CA: A Cancer Journal for Clinicians, 69(1), 7–34. https://doi.org/10.3322/CAAC.21551
Viral Hepatitis and Liver Cancer (2016, March 1). Centers for Disease Control and Prevention.
Ringehan, M., McKeating, J. A., & Protzer, U. (2017). Viral hepatitis and liver cancer. Philosophical Transactions of the Royal Society B: Biological Sciences, 372(1732), 20160274. https://doi.org/10.1098/RSTB.2016.0274
Makarova-Rusher, O. V., Altekruse, S. F., McNeel, T. S., Ulahannan, S., Duffy, A. G., Graubard, B. I., & McGlynn, K. A. (2016). Population attributable fractions of risk factors for hepatocellular carcinoma in the United States. Cancer, 122(11), 1757–1765. https://doi.org/10.1002/CNCR.29971
Chang, E. T., Keegan, T. H. M., Gomez, S. L., Le, G. M., Clarke, C. A., So, S. K. S., & Glaser, S. L. (2007). The burden of Liver cancer in asians and Pacific islanders in the Greater San Francisco Bay area, 1990 through 2004. Cancer, 109(10), 2100–2108. https://doi.org/10.1002/CNCR.22642
Chen, M. S., & Dang, J. (2015). Hepatitis B among Asian Americans: Prevalence, progress, and prospects for control. World Journal of Gastroenterology, 21(42), 11924. https://doi.org/10.3748/WJG.V21.I42.11924
El-Serag, H. B. (2012). Epidemiology of viral hepatitis and hepatocellular carcinoma. Gastroenterology, 142(6), 1264–1273.
El-Serag, H. B., Lau, M., Eschbach, K., Davila, J., & Goodwin, J. (2007). Epidemiology of hepatocellular carcinoma in hispanics in the United States. Archives of Internal Medicine, 167(18), 1983–1989. https://doi.org/10.1001/ARCHINTE.167.18.1983
Ajayi, F., Jan, J., Singal, A. G., & Rich, N. E. (2020). Racial and sex disparities in Hepatocellular Carcinoma in the USA. Current Hepatology Reports, 19(4), 462–469. https://doi.org/10.1007/S11901-020-00554-6
Islami, F., Miller, K. D., Siegel, R. L., Fedewa, S. A., Ward, E. M., & Jemal, A. (2017). Disparities in liver cancer occurrence in the United States by race/ethnicity and state. CA: A Cancer Journal for Clinicians, 67(4), 273–289. https://doi.org/10.3322/CAAC.21402
Litaker, J. R., Tamez, N., Lopez Bray, C., Allison, R. D., Durkalski, W., & Taylor, R. (2021). Hepatitis C antibody screening among baby boomers by a community-based health insurance company. Population Health Management, 24(4), 492. https://doi.org/10.1089/POP.2020.0214
Testing Recommendations for Hepatitis C Virus Infection | CDC. (n.d.). Retrieved January 22 (2023). from https://www.cdc.gov/hepatitis/hcv/guidelinesc.htm.
Weng, M. K., Doshani, M., Khan, M. A., Frey, S., Ault, K., Moore, K. L., & Nelson, N. P. (2022). Universal Hepatitis B vaccination in adults aged 19–59 Years: Updated recommendations of the advisory committee on immunization practices — United States, 2022. Morbidity and Mortality Weekly Report, 71(13), 477–483. https://doi.org/10.15585/MMWR.MM7113A1
Kardashian, A., Serper, M., Terrault, N., & Nephew, L. D. (2022). Health disparities in chronic Liver Disease. Hepatology, 00, 1–22. https://doi.org/10.1002/HEP.32743
Chu, J. N., Nguyen, T. T., Rivadeneira, N. A., Hiatt, R. A., & Sarkar, U. (2022). Exploring factors associated with Hepatitis B screening in a multilingual and diverse population. BMC Health Services Research, 22(1), 479. https://doi.org/10.1186/S12913-022-07813-W
Tohme, R. A., Xing, J., Liao, Y., & Holmberg, S. D. (2013). Hepatitis C testing, infection, and linkage to care among racial and ethnic minorities in the United States, 2009–2010. American Journal of Public Health, 103(1), 112–119. https://doi.org/10.2105/AJPH.2012.300858
Caskey, R., Lindau, S. T., & Alexander, G. C. (2009). Knowledge and early adoption of the HPV Vaccine among girls and young women: Results of a National survey. Journal of Adolescent Health, 45(5), 453–462. https://doi.org/10.1016/J.JADOHEALTH.2009.04.021
Finney Rutten, L. J., Nelson, D. E., & Meissner, H. I. (2004). Examination of population-wide trends in barriers to cancer screening from a diffusion of innovation perspective (1987–2000). Preventive Medicine, 38(3), 258–268. https://doi.org/10.1016/J.YPMED.2003.10.011
NCA - Screening for Hepatitis C Virus (HCV) in Adults (CAG-00436 N) - Decision Memo. (n.d.). Retrieved November 6 (2022). from https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&NCAId=272
Zhu, L., Kim, E. J., González, E., Fraser, M. A., Zhu, S., Rubio-Torio, N., & Tan, Y. (2022). Reducing liver cancer risk through dietary change: Positive results from a community-based educational initiative in three racial/ethnic groups. Nutrients, 14(22), 4878. https://doi.org/10.3390/NU14224878
Holahan, C. J., Holahan, C. K., & Moos, R. H. (2022). Binge drinking and alcohol problems among Moderate Average-Level drinkers. American Journal of Preventive Medicine. https://doi.org/10.1016/J.AMEPRE.2022.03.021
Zou, G. (2004). A modified poisson regression approach to prospective studies with binary data. American Journal of Epidemiology. https://doi.org/10.1093/aje/kwh090
El-Serag, H. B., & Mason, A. C. (2000). Risk factors for the rising rates of primary liver cancer in the United States. Archives of Internal Medicine, 160(21), 3227–3230. https://doi.org/10.1001/ARCHINTE.160.21.3227
Bosch, F. X., Ribes, J., Díaz, M., & Cléries, R. (2004). Primary liver cancer: Worldwide incidence and trends. Gastroenterology, 127(5), S5–S16. https://doi.org/10.1053/J.GASTRO.2004.09.011
Naugler, W. E., Sakurai, T., Kim, S., Maeda, S., Kim, K. H., Elsharkawy, A. M., & Karin, M. (2007). Gender disparity in Liver cancer due to sex differences in MyD88-dependent IL-6 production. Science, 317(5834), 121–124. https://doi.org/10.1126/SCIENCE.1140485/SUPPL_FILE/NAUGLER.SOM.PDF
de Maria, N., Manno, M., & Villa, E. (2002). Sex hormones and liver cancer. Molecular and Cellular Endocrinology, 193(1–2), 59–63. https://doi.org/10.1016/S0303-7207(02)00096-5
Li, L., & Yeo, W. (2017). Value of quality of life analysis in liver cancer: A clinician’s perspective. World Journal of Hepatology, 9(20), 867. https://doi.org/10.4254/WJH.V9.I20.867
Lee, Y. C. A., Cohet, C., Yang, Y. C., Stayner, L., Hashibe, M., & Straif, K. (2009). Meta-analysis of epidemiologic studies on cigarette smoking and liver cancer. International Journal of Epidemiology, 38(6), 1497–1511. https://doi.org/10.1093/IJE/DYP280
Arem, H., Loftfield, E., Saint-Maurice, P. F., Freedman, N. D., & Matthews, C. E. (2018). Physical activity across the lifespan and liver cancer incidence in the NIH-AARP diet and health study cohort. Cancer Medicine, 7(4), 1450–1457. https://doi.org/10.1002/CAM4.1343
Turati, F., Galeone, C., Rota, M., Pelucchi, C., Negri, E., Bagnardi, V., & la Vecchia, C. (2014). Alcohol and liver cancer: A systematic review and meta-analysis ofprospective studies. Annals of Oncology, 25(8), 1526–1535. https://doi.org/10.1093/ANNONC/MDU020
Bockmann, J. H., Kohsar, M., Murray, J. M., Hamed, V., Dandri, M., Lüth, S., & Schulze-Zur-wiesch, J. (2021). High rates of liver cirrhosis and hepatocellular carcinoma in chronic Hepatitis B patients with metabolic and cardiovascular comorbidities. Microorganisms, 9(5), 968. https://doi.org/10.3390/MICROORGANISMS9050968
Le, M. H., Yeo, Y. H., Cheung, R., Henry, L., Lok, A. S., & Nguyen, M. H. (2020). Chronic Hepatitis B Prevalence Among Foreign-Born and US Born Adults in the United States, 1999–2016. Hepatology, 71(2), 431–443. https://doi.org/10.1002/HEP.30831
Fact sheet: Biden-Harris Administration Advances Equity and Opportunity for Asian American, Native Hawaiian, and Pacific Islander Communities Across the Country - The White House. (n.d.). Retrieved September 25 (2022). from https://www.whitehouse.gov/briefing-room/statements-releases/2022/01/20/fact-sheet-biden-harris-administration-advances-equity-and-opportunity-for-asian-american-native-hawaiian-and-pacific-islander-communities-across-the-country/.
Philbin, M. M., Erby, L. A. H., Lee, S., & Juon, H. S. (2012). Hepatitis B and Liver cancer among three Asian American sub-groups: A focus group inquiry. Journal of Immigrant and Minority Health, 14(5), 858–868. https://doi.org/10.1007/S10903-011-9523-0/TABLES/2
Asian Americans and their origins: Key facts | Pew Research Center. (n.d.). Retrieved September 25 (2022). from https://www.pewresearch.org/fact-tank/2021/04/29/key-facts-about-asian-origin-groups-in-the-u-s/.
Strong, C., Lee, S., Tanaka, M., & Juon, H. S. (2012). Ethnic differences in prevalence and barriers of HBV screening and vaccination among Asian americans. Journal of Community Health, 37(5), 1071–1080. https://doi.org/10.1007/S10900-012-9541-4/TABLES/5
Lavanchy, D. (2009). The global burden of hepatitis C. Liver International, 29, 74–81. https://doi.org/10.1111/J.1478-3231.2008.01934.X
Alter, M. J., Kruszon-Moran, D., Nainan, O. V., McQuillan, G. M., Gao, F., Moyer, L. A., & Margolis, H. S. (1999). The prevalence of hepatitis C virus infection in the United States, 1988 through 1994. New England journal of medicine, 341(8), 556–562. https://doi.org/10.1056/NEJM199908193410802
Wilder, J., Saraswathula, A., Hasselblad, V., & Muir, A. (2016). A systematic review of race and ethnicity in Hepatitis C clinical trial enrollment. Journal of the National Medical Association, 108(1), 24–29. https://doi.org/10.1016/J.JNMA.2015.12.004
McQuillan, G. M., Kruszon-Moran, D., Kottiri, B. J., Curtin, L. R., Lucas, J. W., & Kington, R. S. (2004). Racial and ethnic differences in the seroprevalence of 6 infectious diseases in the United States: Data from NHANES III, 1988–1994. American Journal of Public Health, 94(11), 1952. https://doi.org/10.2105/AJPH.94.11.1952
Pearlman, B. L. (2006). Hepatitis C virus infection in African americans. Clinical Infectious Diseases, 42(1), 82–91. https://doi.org/10.1086/498512
One-in-Ten Black People Living in the U.S. Are Immigrants | Pew Research Center. (n.d.). Retrieved November 7 (2022). from https://www.pewresearch.org/race-ethnicity/2022/01/20/one-in-ten-black-people-living-in-the-u-s-are-immigrants/.
Hassan, M. A., Kim, W. R., Li, R., Smith, C. I., Fried, M. W., Sterling, R. K., & Lok, A. S. F. (2017). Characteristics of US-Born versus foreign-born Americans of African descent with chronic hepatitis B. American Journal of Epidemiology, 186(3), 356. https://doi.org/10.1093/AJE/KWX064
Pomenti, S., Gandle, C., Abu Sbeih, H., Phipps, M., Livanos, A., Guo, A., & Wattacheril, J. (2020). Hepatocellular Carcinoma in Hispanic Patients: Trends and Outcomes in a Large United States Cohort. Hepatology Communications, 4(11), 1708–1716. https://doi.org/10.1002/HEP4.1575
Ha, J., Yan, M., Aguilar, M., Tana, M., Liu, B., Frenette, C. T., & Wong, R. J. (2016). Race/Ethnicity-specific Disparities in Hepatocellular Carcinoma stage at diagnosis and its impact on receipt of curative therapies. Journal of Clinical Gastroenterology, 50(5), 423–430. https://doi.org/10.1097/MCG.0000000000000448
Hu, D. J., Xing, J., Tohme, R. A., Liao, Y., Pollack, H., Ward, J. W., & Holmberg, S. D. (2013). Hepatitis B testing and access to care among racial and ethnic minorities in selected communities across the United States, 2009–2010. Hepatology, 58(3), 856–862. https://doi.org/10.1002/HEP.26286
Bindman, A. B., Grumbach, K., Osmond, D., Vranizan, K., & Stewart, A. L. (1996). Primary care and receipt of preventive services. Journal of General Internal Medicine, 11(5), 269–276. https://doi.org/10.1007/BF02598266
Frrante, J. M., Winston, D. G., Chen, P. H., & de la Torre, A. N. (2008). Family physicians’ knowledge and screening of chronic hepatitis and liver cancer - PubMed. Family Medicine, 40(5), 345–351. Retrieved from https://pubmed.ncbi.nlm.nih.gov/18465284/.
Chao, S. D., Wang, B. M., Chang, E. T., Ma, L., & So, S. K. (2015). Medical training fails to prepare providers to care for patients with chronic hepatitis B Infection. World Journal of Gastroenterology: WJG, 21(22), 6914. https://doi.org/10.3748/WJG.V21.I22.6914
Hwang, J. P., Roundtree, A. K., Engebretson, J. C., & Suarez-Almazor, M. E. (2010). Medical care of Hepatitis B among Asian American populations: Perspectives from three provider groups. Journal of General Internal Medicine, 25(3), 220–227. https://doi.org/10.1007/S11606-009-1204-5/FIGURES/1
Foster, T., Hon, H., Kanwal, F., Han, S., & Spiegel, B. (2011). Screening high risk individuals for hepatitis B: Physician knowledge, attitudes, and beliefs. Digestive Diseases and Sciences, 56(12), 3471–3487. https://doi.org/10.1007/S10620-011-1928-Z/TABLES/4
Tenner, C. T., Herzog, K., Chaudhari, S., Bini, E. J., & Weinshel, E. H. (2012). Knowledge, attitudes and barriers regarding vaccination against hepatitis A and B in patients with chronic Hepatitis C virus Infection: A survey of family medicine and internal medicine physicians in the United States. International Journal of Clinical Practice, 66(10), 1009–1013. https://doi.org/10.1111/IJCP.12013
Wu, Y., Johnson, K. B., Roccaro, G., Lopez, J., Zheng, H., Muiru, A., & Chung, R. T. (2014). Poor adherence to AASLD guidelines for chronic hepatitis B management and treatment in a large academic medical center. The American Journal of Gastroenterology, 109(6), 867. https://doi.org/10.1038/AJG.2014.72
Nguyen, T. T., McPhee, S. J., Stewart, S., Gildengorin, G., Zhang, L., Wong, C., & Chen, M. S. (2010). Factors associated with hepatitis B testing among Vietnamese Americans. Journal of General Internal Medicine, 25(7), 694–700. https://doi.org/10.1007/S11606-010-1285-1/TABLES/3
Toyoda, H., Huang, D. Q., Le, M. H., & Nguyen, M. H. (2020). Liver Care and Surveillance: The Global Impact of the COVID-19 pandemic. Hepatology Communications, 4(12), 1751–1757. https://doi.org/10.1002/HEP4.1579
Cheung, K. L., ten Klooster, P. M., Smit, C., de Vries, H., & Pieterse, M. E. (2017). The impact of non-response bias due to sampling in public health studies: A comparison of voluntary versus mandatory recruitment in a Dutch national survey on adolescent health. BMC Public Health, 17(1), 1–10. https://doi.org/10.1186/S12889-017-4189-8/TABLES/2
Acknowledgements
This study was supported by TUFCCC/HC Regional Comprehensive Cancer Health Disparity Partnership, Award Number U54 CA221704(5) (Contact PIs: GX Ma, J Erblich) from the National Cancer Institute of National Institutes of Health (NCI/NIH) and by NIGMS/NIH award # 1SC3GM131949-01 (PI: MC Yeh). The author team would like to thank Community Advisory Board members, leaders, and staff members for collaborating with community/faith-based organization partners. We also acknowledge the research team staff of Planning Evaluation Core under TUFCCC/HC Regional Comprehensive Cancer Health Disparity Partnership for their support and contribution for their support and contributions to Community Outreach Core’s data management. Its contents are solely the authors’ responsibility and do not necessarily represent the official views of the NCI/NIH or the NIGMS/NIH.
Funding
This study was supported by TUFCCC/HC Regional Comprehensive Cancer Health Disparity Partnership, Award Number U54 CA221704(5) (Contact PIs: GX Ma, J Erblich) from the National Cancer Institute of National Institutes of Health (NCI/NIH) and by NIGMS/NIH award # 1SC3GM131949-01 (PI: MC Yeh).
Author information
Authors and Affiliations
Contributions
Conceptualization, MCY, GM; Methodology, MCY, GM, YT; Formal analysis, TB, VP; Investigation, MCY, GM, YT, SZ, NRT, HP, EG, MF; Resources, MCY, GM; Data curation, SZ, NRT, HP, EG, MF, YT; Writing—original draft preparation, TB; Writing—review and editing, TB, VP, LZ, YT; Supervision, MCY, GM, YT; Project administration, GM, YT, SZ, NRT, EG, MF; Funding acquisition, MCY, GM. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical Approval
The study was approved by Institutional Review Boards at Temple University and Hunter College.
Consent to Participate
All participants provided informed consent.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Begum, T.F., Patil, V.S., Zhu, L. et al. Assessing Physicians’ Recommendations for Hepatitis B Virus (HBV) and Hepatitis C Virus (HCV) Testing Among Minority Populations in Greater Philadelphia and New York City. J Community Health 49, 588–597 (2024). https://doi.org/10.1007/s10900-023-01316-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10900-023-01316-3