Abstract
Congenital toxoplasmosis (CT) is a zoonosis resulting from the fetal transmission of the obligate intracellular parasite, T. gondii, transplacentally in pregnant women usually in the first trimester of pregnancy. This research consisted in the review of indexed articles made in the Ecuador in the period between the years 2012 and 2017 with prevalence studies of the aforementioned pathology. The purpose of this collection was to determine the current situation of this disease in the Ecuadorian territory and, in addition, to recognize the risk factors involved, affected age groups, signs and symptoms, diagnosis and prevalence of the infection. Among the risk factors were the consumption of raw or undercooked meat, contact with feces of young cats and climatic conditions that stimulate the spread of oocysts, among others. Most affected ages by this disease fluctuate between 20 and 30 years. In 90% of the cases, the disease is asymptomatic or it may present adenopathies, maculo papular erythema, hepatosplenomegaly and other general signs such as fever, malaise, headache and myalgias. According to studies conducted with specific populations, it was established that the causative agent remains latent in first-trimester pregnant women from the provinces of Pichincha (71.4% of 140 pregnant women), Guayas (73% of 5683) and El Oro (16% of 250). The results of this research evidenced missing information in Ecuador, finding few and isolated studies regarding to this pathology. Therefore, it is concluded that an updated research should be conducted in order to elucidate the true epidemiological situation of congenital toxoplasmosis in Ecuador.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Toxoplasmosis is a zoonotic parasitosis prevalent in humans [1]. Toxoplasma gondii, etiological agent of this disease, is a protozoa of global distribution [2]. The parasite shows a complex life cycle that presents three states: taquizoite, bradizote and sporozoite. Its definitive host are felines, including domestic cats [3]. It affects about one-third of people worldwide, and can be asymptomatic until severe symptoms appear in rare cases, depending on the suitability of the immune antiparasitic response [4].
Congenital toxoplasmosis (CT) is the consequence of fetal transplacental transmission of T. gondii after maternal infection [5]. When the infection is first acquired during pregnancy, it can cause serious consequences for the fetus, such as hydrocephalus, microcephaly, brain calcifications, chorioretinitis, or in the worst case, end with an abortion, depending on the date the mother became infected [6]. Children born to an infected mother in the first trimester are highly probable to be a severely affected, while 80% of children infected during the third trimester are born asymptomatic [7].
The infection caused by T. gondii is a globally distributed zoonosis, unlike other parasitosis that mainly affects tropical countries [8]. The annual incidence of CT is estimated at 190,100 cases (95% confidence interval: 179,300—206,300) [9].
Regarding Ecuador, cities such as Riobamba show a prevalence of 40–50% [10]. In Quito, the prevalence of CT is 40% [6]. In the coast region, there is a prevalence of 74%. In Guayaquil, the current prevalence is 80.1% [11]. The risk of CT in Guayaquil–Ecuador is 2, 4 for ten thousand and it is to be expected the annual birth of 147 affected children, with 95 (65%) asymptomatic, but with mixed repercussions on its subsequent development. However, the reality of the CT in the country has not yet been fully established [12].
Pregnant women in the chronic stage of T. gondii infection may experience complications of the disease. This parasite reaches the placenta by heamegran, potentially infecting the fetus resulting in a CT. This infection may cause death to the fetus, or cause changes that aggravate the health of children after surviving this pathology. Several clinical alterations such as seizures only appear after the second or third decade of life. Also, previous studies have shown that 80% of children with subclinical infection develope eye sequelae at some point in their lives. Therefore, an asymptomatic child at birth is not necessarily indicative of a better prognosis [13].
In the Ecuadorian region, research on CT prevalence data is insufficient or outdated, so further revision is needed in order to understand the current situation of the disease. However, despite significant results from studies on Congenital Toxoplasmosis (CT) in Ecuador, their current situation is not fully clarified because they remain scarce, inaccurate and obsolete [14].
In this regard, adolescent pregnancy is a public health priority in Latin America and the Caribbean, ranking second globally, after Africa. According to the Statistical Center for Latin America and the Caribbean (ECLAC), Ecuador is the third country in the region with the highest rate of teenage pregnancy (10–19 years), after Nicaragua and the Dominican Republic [15]. As of 2013, the CT was removed from the epidemiological gazzette as a mandatory reporting disease. From there until now, congenital toxoplasmosis has been excluded as a zoonotic disease from which data is periodically collected.
This bibliographic review aims to provide a search tool for information related to the status of epidemiological, symptomatalogical and prevalence data of Congenital Toxoplasmosis infection in Ecuador, with the purpose to deliver reliable and up-to-date information. It also specifies the risk factors that favor transmission, compared with other countries of Latin American and the locations (provinces) of Ecuador, where there is a higher prevalence of CT in 2017. After this compilation it is expected to satisfy doubts of students and health professionals.
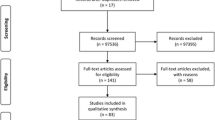
Materials and Methods
Studio Design
This was a descriptive, documentary, retrospective and cross-sectional research. References of research work carried out in Ecuador were reviewed, related to pregnant women residing in the country by 2017, in various health centers belonging to the Provinces of Pichincha, Guayas and El Oro which were registered as confirmed cases by serological examinations of Anti Toxoplasma gondii IgM and IgG antibodies. More than 41 revised articles (13 of them of Ecuador) were considered, studies conducted by the World Health Organization (WHO), The Panamerican Health Organization (PAHO), Ecuador’s Ministry of Public Health (MSP), indexed scientific articles in the region and other indexed bibliographic compilations, which allowed such data to be grouped into an instrument and establish the risk factors associated with the transmission of the disease, age groups, most common signs and symptoms as well as the prevalence of CT in Ecuador. For the collection of information was established as a single collection period from May of 2018 to september of the same year.
Risk Factors
Based on the compilation of ecuadorian articles found, the intake of undercooked foods was accepted as the first risk factor, especially pork, lamb, beef and hunting animals that have come into contact with the causative agent [16]. The transmission of T. gondii through the consumption of tissue cysts in the tissues of animals for human consumption or by the presence of oocysts in fruits and vegetables, is also considered a way in which pregnant women acquire the infection, although it is less frequent than by ingestion of undercooked meats [6].
The second risk factor that most affects women in pregnancy is contact with cats faeces, which due to a low socioeconomic situation and the lack of frequent veterinary control, these are more likely to acquire this parasite and use it as a definitive guest. It is understood that the method by which the parasite reaches fruits and vegetables is through the faeces of these felines. In addition, it should be emphasized those younger felines (under 6 months) are the most exposed to acquire the causal agent [17].
On the other hand, there is considered to be a link between climate and T. gondii infection in pregnancy [18]. The oocyst form of the parasite has high resistance to environmental factors and can survive for long periods of time. Rain is essential for the spread of oocysts, because they are released from faeces and distributed in large wet areas, while the drying and direct action of sunlight destroy them [11]. Therefore, the parasite tends to be more endemic in wet areas; however, this difference between zones is minimal as, once sporulated and in conditions of 4 °C can remain infective for 4.5 years and in conditions of 10–25 °C are infective for 6 months, but then lose their infective capacity by 1 min at 60 °C. Due to these reasons this factor is considered in reviews in a third place [19].
There are other factors that may set pregnant women at risk. It has been noted: distribution of the parasite by other mechanical vectors such as flies, cockroaches [19], and also by the human being himself, who is able to transport the agent with the hands to his organism (self-inoculation) or to others, through contact with food and drinks. Similarly, there is parenteral transmission caused by the use of syringes or blood transfusions in pregnant women (point cases), among others [20].
Age Groups
According to the compilation of results obtained in the 41 reviewed articles, the most affected ages in pregnant women range from 20 to 30 years. Based on the abovementioned, it can be observed that there is a similarity within this section in respect to other Latin American countries. A study in Valledupar—Colombia found, in relation to age, a higher prevalence (54.6%) of positive antibodies for ages 20 to 29 [21]. For studies belonging to Iquitos—Peru, a higher prevalence was shown in pregnant women aged 15–30 years [22]. While, in the Palavecino Municipality, Lara—Venezuela, one study showed the HIV-positive presence of T. gondii among age groups of 20–41 years [23].
The small presence of articles in Ecuador, although articulating much information, is not sufficient to make an adequate conclusion at the national level [12, 14] However, thanks to this data collection, the presence of T. gondii in Ecuador is undeniable, existing possible endemic areas in the provinces studied (Quito, Guayaquil and El Oro) and establishing as possible a relationship between susceptibility of infection and groups of pregnant women between 20 and 35 years in the aforementioned locations.
Signs and Symptoms
The affection of T. gondii is a zoonosis, capable of inhabiting humans and several species of mammals and birds [19]. Its transmission between hosts occurs in a congenital or acquired manner. In relation to congenital toxoplasmosis, 90% of children born with congenital toxoplasmosis have a normal appearance and remain asymptomatic for the first few years of life especially if the mother became infected for the first time during the second or third trimester of gestation, but later had various neurological problems. The symptoms that appear in the newborn depend on the moment in which the fetus becomes infected and are: generalized infection, acute encephalitis and irreversible sequelae [24]. However, if the infection occurs during the first trimester, the fetus is contaminated by the taquizoates that cross the placental barrier. Also, cysts with bradyzoites can occur and result in an active multiplication of tachyzoites, with widespread dissemination, usually fatal (intrauterine death where the embryo or fetus is removed) or result in lesions of the central nervous system such as toxoplasma chorioretinitis [25]. This is the main ocular manifestation of congenital toxoplasmosis. In relation to this chorioretinitis it is usually the late sequela of an acquired infection in the uterus, although it also occurs due to an acute infection acquired in immunocompetent individuals and as a result of an acute acquired infection or the reactivation of a latent infection in immuno-compromised individuals [26].
In addition, Sabin syndrome can be observed in humans [25]. This is characterized by hydrocephalus with microcephaly, brain calcifications, chorioretinitis and mental impairment [27, 28]. In addition to these, T. gondii infection has a wide variety of ultrasound signs, with a rare presence of fetal hydrops [29]. In South America, this acute phase parasitosis is infrequent in the general population, reporting incidences of seroconversion in susceptible pregnant women, 1 in 1000, therefore; most institutions recommend screening only at risk population [30].
Prevalence
In Ecuador, the risk of congenital toxoplasmosis in Guayaquil is 2.4 per 10,000 and 147 affected children are expected to be born annually, with 95 (65%) asymptomatic, but with mixed repercussions on their subsequent development, incorporating children with visual impairments or motor or intellectual disorders into the community, which, on the other hand, its impact could be prevented or diminished with timely treatment at birth and permanent evaluation. However, the reality of congenital toxoplasmosis in the country has not yet been established [12].
The latest records of government-level surveys on congenital toxoplasmosis can be found in the Weekly Epidemiological Gazette Number 16 of the year 2013 [14]. It presents an overview of toxoplasmosis, finally reporting 51 cases, of which the most affected provinces in descending order are: Pichincha, Guayas, Manabí and El Oro. From there until today, toxoplasmosis has been excluded as a zoonotic disease from which data are collected periodically and as a pathology to consider in pregnancy. Proof of the above is the lack of reliable epidemiological data generated by the public health system, focused on women in gestation. The lack of this information makes it impossible to assert a figure that is true for the whole country. However, data regarding the disease within the Ecuadorian territory have been found thanks to non-governmental studies in specific populations within the aforementioned provinces. The following information was collected:
In the city of Quito, a study conducted in October 2008, in 140 pregnant women (first trimester of gestation) at the Gyneco-Obstetric Hospital Isidro Ayora found a prevalence relative to 71.4%, with an increased risk in adolescents and mothers over 34 years old [31].
In 2014, in the province of El Oro, a screening study of IgG–IgM antibodies was conducted on 250 pregnant women (15–40 years) who were in the first trimester of pregnancy in which a prevalence of 18.8% was determined (Table 1). It was found 203 negative cases, 40 of them were positive for T. gondii, 5 IgG positives and IgM negatives, and 2 negative IgG and IgM positive cases were determined to be considered as acute infections. 50% of cases in which antibodies were presented for T. gondii belong to the group between 26 and 30 years, being this the age group in which the majority of cases were observed, followed by 28% with pregnant women aged between 21 and 26 years [32].
As a result of the investigations, 74% is indicated for the entire coastal region or coast [12, 33], so a decrease of 18% compared to 1989 is evident. This decline in prevalence can be inferred from the evident changes, since 1989, in basic services in the city: provision of drinking water and sewerage and garbage collection, as well as education for health currently being taught in schools [11].
In the period from August 2016 to July 2017, a seroprevalence study conducted in Guayaquil at the Hospital de la Mujer “Alfredo G. Paulson” showed a prevalence of 73%. Results from serological analyses of the 5683 patients under prenatal control indicated that 4154 patients had low IgG titrations corresponding to serological scarring, indicating that they were infected prior to pregnancy. The remaining 1529 patients were seronegative in the initial study, further analysis showed that 87 patients (2%) of these had IgM > 3 being indicative of first infection by toxoplasmosis during pregnancy. Of this group of patients, 1442 remained seronegative until the end of pregnancy. In the group of patients over 30 years of age, 64% of patients had positive (> 3) anti-toxoplasma IgG titrations [34] (Table 1).
Discussion
From data collected in ecuadorian articles, the intake of poorly cooked meats such as: beef, pork, among others, that have been in contact with the causal agent was evident, and this is the main risk factor for CT infections, as referred by Bahia et al. [35] (Brazil), Carrada [36] (Mexico) and Romero et al. [37] (Paraguay). In addition, other aspects associated with the disease such as age, level of studies, occupation, hygiene, poorly washed fruits and vegetables, high density of stray cats population, place of residence, atmospheric variables (temperature–humidity) and socioeconomic factors [37].
However, this information does not seem to correspond to what Fernandez et al. [12] pointed out in the study in Guayaquil, who indicate that there were people who eat red meat, a few times a week and mostly well cooked.
The 5-year risk shown has been mainly in women’s groups between the ages of 20–30, due to the shortage of articles in Ecuador and, based on the data collected, it can be determined that the most affected groups are in the Coast region [12]. This information could be consistent with a study conducted in El Oro province in which the evidence of IgG–IgM antibodies against T. gondii was found in women between 26 and 30 years of age in 50% of cases studied [32]. However, in a study conducted in Divinópolis, a municipality in the southern Brazilian state of Minas Gerais in Brazil by Fonseca et al. [38] was found that the age group with the highest prevalence was > 30 years.
Although during the first years of life the infection has usually been asymptomatic, among the main symptoms: generalized infection, acute encephalitis, irreversible sequelae present in infections during the second or third trimester and fatal problems or neurological involvements during the first trimester of gestation [24]. In the same way, Guerra et al. refers the presence of Sabin syndrome [25], while in Ecuador; a study by Toapanta [39] has identified asymptomatic children with subsequent developmental impacts such as visual problems, motor or intellectual disorders.
In the study conducted by Fernández et al. [40] indicate that in Ecuador there is a prevalence of antibodies that is located around 74% in the coast region where infection rates in the age group between 15 and 20 years was 79%. Comparing this research with Fernandez et al. [40] study showed a prevalence for the country of 78.3% and a rate of 60.2% for the same age group denoting a percentage difference of 18% [40]. The presence of CT occurs mainly in populations of susceptible or seronegative pregnant women (without prior infection), which may suggest patients have a risk of infection during pregnancy, however; it is not entirely true, as it also depends on the circulation of the parasite in the environment [12]. It was further evident as a result of the review that from week 30 to childbirth a higher probability of congenital infection is shown, whose percentage ranges from 60 to 80% risk, generating possible irreversible sequelae, including classic clinical signs (intracranial calcifications, chorioretinitis and/or hydrocephalus before 3 years of age) [41].
Conclusions
According to the data consulted, the main risk factor for contracting CT is the consumption of undercooked foods, especially pork. As a second and third factor is the contact with faeces of cats and to weather conditions, respectively. The mostly affected age group corresponds to the range between 20 and 30 years of age. Signs and symptoms of CT are variable according to the mother’s period of infection, which are much more serious if transmission occurs during the first trimester of pregnancy. The most common symptoms include generalized infection, acute encephalitis, Sabin syndrome or even irreversible sequelae, which include classic clinical signs (intracranial calcifications, chorioretinitis and/or hydrocephalus before 3 years old).
According to the review of studies conducted in Ecuador, the prevalence of the disease ranges from 71 to 74%; however, prevalence data at the province level is scarce.
Recommendations
We invite all students and health professionals to contribute with new research that allows us to have a much clearer notion of the situation in the Ecuadorian territory.
In the same way, it should be considered that public health institutions implement an adequate epidemiological surveillance system and disseminate prevention measures in accordance with the community, mainly in the educational field in order to obtain changes in habits and lifestyles that act as major risk factors.
References
Mimica, F., Muñoz, C., Torres, M., & Padilla, O. (2015). Toxoplasmosis, zoonosis parasitaria prevalente en Chile: Recuento y desafíos. Revista Chilena de Infectología.,32(5), 541–549.
Álvarez, J., Martínez, M., Moreno, L., Lorente, S., & Crespo, M. (2008). Prevalencia e incidencia de la infección por Toxoplasma gondii en mujeres en edad fértil en Albacete (2001-2007). Revista Española de Salud Pública.,82(3), 333–342.
Beltrán Flores, S., Flores Arriaga, J., & Lema, Correa M. (2014). Toxoplasmosis congénita. Boletín Médico del Hospital Infantil de México,71(6), 373–376.
Pedersen, M., Mortensen, P., Norgaard, B., & Postolache, T. (2012). Toxoplasma gondii infection and self-directed violence in mothers. Archives of General Psychiatry,69(11), 1123–1130.
Baquero Artigao, F., Del Castillo, M., Fuentes, I., Goncé, A., Fortuny, C., De La Calle, M., et al. (2013). Guía de la Sociedad Española de Infectología Pediátrica para el diagnóstico y tratamiento de la toxoplasmosis congénita. Anales de Pediatría,79(2), 116p.
Peña, J. (2015). Determinación de Ac. IgM contra toxoplasmosis, mediante la técnica de quimioluminiscencia y sus posibles complicaciones en los neonatos, en mujeres embarazadas que acudieron al laboratorio clínico del hospital municipal materno infantil San José Sur a realizarse su control prenatal, en el período enero - diciembre 2014 (p. 82). Universidad Central del Ecuador.
Carral, L., Kaufer, F., Pardini, L., Durlach, R., Moré, G., & Venturini, M. (2018). Toxoplasmosis congénita: Diagnóstico serológico, RPC, aislamiento y caracterización molecular de Toxoplasma gondii. Revista Chilena de Infectología,35(1), 36–40.
Agudelo, A., Montoya, J., Isaza, A., & Rosso, F. (2007). Toxoplasmosis congénita: Aspectos clínicos y epidemiológicos de la infección durante el embarazo. Colombia Médica,38(3), 316–337.
Torgerson, P. R., & Mastroiacovo, P. (2013). The global burden of congenital toxoplasmosis: A systematic review. Bulletin of the World Health Organization,91(7), 501–508.
Cano, L., Quinchuela, J. (2017). Prevalencia de toxoplasma gondii en alumnas de la carrera de psicología clínica de la universidad nacional de Chimborazo (Tesis doctoral). Universidad Nacional de Chimborazo, Riobamba (p. 48). Ecuador.
Ponce, J. (2014). Evaluación de hemaglutinación indirecta y enzimoinmunoensayo para diagnóstico de toxoplasmosis en Guayaquil (Tesis para Magister) (p. 62). Universidad de Guayaquil. Ecuador.
Fernández, T., Acosta, Y., & Montaño, M. (2010). Toxoplasmosis congénita: Reporte de casos. Revista de la Facultad de Ciencias Médicas de la Universidad de Guayaquil,17(3), 192–197.
Avelar, J., Da Silva, M., Alves, H., Ribeiro, H., Do Amaral, W., Rodriguez, I., et al. (2018). Epidemiological factors associated with Toxoplasma gondii infection in postpartum women treated in the public healthcare system of goiânia, state of Goiás, Brazil. Revista da Sociedade Brasileira de Medicina Tropical,51(1), 57–62.
Salud Ecuador. SIVE – ALERTA. [Internet]. MSP. Ecuador. [updated 2013 Jun 7; cited 2018]. Retrieved from https://issuu.com/saludecuador/docs/gaceta13/19.
Ministerio de Salud Pública del Ecuador, Plan Nacional de Salud Sexual y Salud Reproductiva: Viceministerio de Gobernanza de la Salud Pública, Marzo, 2017, Quito, Ecuador.
Zambrano Demera, I. E. (2016) Prevalencia de toxoplasmosis en gestantes primer trimestre que acudieron al laboratorio clínico APROFE Central, Guayaquil 2012 a 2014. (Trabajo de Titulación) (p. 80). Universidad de Guayaquil. Ecuador.
Rivera Fernández, N., & García Dávila, P. (2017). El papel de los gatos en la toxoplasmosis. Realidades y responsabilidades. Revista de la Facultad de Medicina de la UNAM,60(6), 7–18.
Sanchez, R., Couret, M., Ginorio, D., Nodarse, A., Sanchez, N., Soler, I., et al. (2012). Toxoplasmosis y embarazo. Revista Cubana de Obstetricia y Ginecología,38(1), 99–106.
Raiden Grandía, G., Angel Entrena, G., & Jeddú, Cruz H. (2013). Toxoplasmosis en Felis catus: Etiología, epidemiología y enfermedad. Revista de Investigaciones Veterinarias del Peru,24(2), 131–149.
Almirall, P., Escobedo, A., Nuñez, F., & Ginorio, E. (2002). Toxoplasmosis: Aspectos de interés sobre el manejo de la toxoplasmosis. Colombia Médica,7(1), 1–9.
Jácome, Torres J. (2013). Prevalencia de infección por Toxoplasma gondii en mujeres embarazadas, en Valledupar, César. Revista Colombiana de Microbiología,3(1), 31–44.
Reátegui, B., & Vela, G. (2011). Factores socioeconómicos - epidemiológicos y su relación con la seroprevalencia de toxoplasmosis en gestantes atendidas en los hospitales “Felipe Arriola” y “Cesar Garayar”, Iquitos, Perú, 2009. Neotropical Helminthology,5(1), 31–40.
Triolo Mieses, M., & Traviezo, Valles L. (2004). Seroprevalencia de anticuerpos contra toxoplasma gondii en gestantes del municipio Palavecino, Estado Lara, Venezuela. Revista BIOMED,15(1), 181–190.
Ocampo, L., & Duarte, I. (2010). Modelo para la dinámica de transmisión de la toxoplasmosis congénita. Revista de Salud Pública.,12(2), 317–326.
Guerra, F., Norberg, A., Covarrubias, E., Aguillar, M., Madeira, J., & Serra, N. (2014). Toxoplasmosis aguda en embarazadas asintomáticas de Rio de Janeiro, Brasil. Revista Médica Herediana,25(4), 204–207.
González Núñez, I., Díaz Jidy, M., & Pérez, Avila J. (1999). Coriorretinitis por toxoplasma en niños. Revista Cubana de Medicina Tropical,51(2), 138–142.
Solis, Duran M. (2010). Toxoplasmosis Congénita. Revista Médica de Costa Rica y Centroamérica,67(592), 127–130.
Díaz, L., Zambrano, B., Chacón, G., Rocha, A., & Diaz, S. (2010). Toxoplasmosis y embarazo. Revista de Obstetricia y Ginecología de Venezuela,70(3), 190–205.
Lacunza, R., & Boza, M. (2012). Hidropesía fetal como signo ecográfico de toxoplasmosis congénita. Revista Peruana de Ginecología y Obstetricia,58(1), 55–58.
Cárdenas, D., Lozano, C., Castillo, Z., Cedeño, J., Galvis, V., Rios, J., et al. (2015). Frecuencia de anticuerpo anti Toxoplasma gondii en gestantes de Cúcuta, Colombia. Revista Medica Herediana,26(4), 230–237.
Mayorga, B. (2008). Serodiagnostico mediante IgG, IgM, IgA ELISA de toxoplasmosis en mujeres en el primer trimestre de embarazo del Hospital Gineco Obstetrico Isidro Ayora de la ciudad de Quito en octubre del 2008 (tesis de fin de grado de la carrera de medicina) (p. 62). Universidad San Francisco de Quito. Ecuador.
Lam, A., Segura, M., Santos, J., Sanmartín, D., & López, M. (2016). Toxoplasma gondii en mujeres embarazadas en la provincia de El Oro, 2014. Revista Ciencia UNEMI,9(21), 135–141.
Frenkel, J. K., Lazo, Y., & Lazo, J. (1984). Encuesta sobre infección toxoplásmica en un grupo de alumnos del tercer año de medicina y en un número igual de gatos, de la ciudad de Guayaquil. Revista de Medicina Tropical y Parasitología,1(119), 81–88.
Aguirre, A. (2018). Seroprevalencia anti-toxoplasma gondii en embarazadas que acuden al Hospital Alfredo G. Paulson. Guayaquil, agosto 2016 – julio 2017 (p. 69). Universidad de Guayaquil.
Bahia Oliveira, L. M., Jones, J. L., & Azevedo, Silva J. (2003). Highly endemic, waterborne toxoplasmosis in North Rio de Janeiro State, Brazil. Emerging Infectious Diseases,9(1), 55–62.
Carrada, T. (2005). Toxoplasmosis: Parasitosis reemergente del nuevo milenio. Revista Mexicana de Patología Clínica,52(3), 151–162.
Romero, D., Gonzalez, C., Guillen, I., Aria, L., Meza, T., Rojas, A., et al. (2017). Seroprevalencia y factores de riesgo asociados a la toxoplasmosis en mujeres en edad reproductiva que acudieron al hospital distrital de Lambaré, Paraguay. Inicio Acerca de Iniciar sesión Registrarse Buscar Actual Archivos Avisos,15(3), 83–88.
Fonseca, A. L., Andrade Silva, R., Blima, F., Madureira, A. P., de Sousa, F. F., & Margonari, C. (2012). Epidemiologic aspects of toxoplasmosis and evaluation of its seroprevalence in pregnant women. Revista da Sociedade Brasileira de Medicina Tropical,45(3), 357–364.
Toapanta Cepa, J. (2016). Determinación de ac de toxoplasma (IgG – IgM) por el método de electroquimioluminiscencia y su relación con los factores de riesgo en mujeres embarazadas que acuden al hospital general docente Ambato (p. 90). Universidad Técnica de Ambato.
Fernández, T., Montaño, M., Basantes, S., & Ponce, J. (2014). Estudio seroepidemiológico para estimar el riesgo de infección congénita por Toxoplasma gondii. Revista de Patologia Tropical,43(2), 182–194.
Rosso, F., Agudelo, A., Isaza, A., & Montoya, J. (2007). Toxoplasmosis congénita: aspectos clínicos y epidemiológicos de la infección durante el embarazo. Colombia Médica,38(3), 316–337.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no conflicts of interest associated with the material presented in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Velásquez Serra, G.C., Piloso Urgiles, L.I., Guerrero Cabredo, B.P. et al. Current Situation of Congenital Toxoplasmosis in Ecuador. J Community Health 45, 170–175 (2020). https://doi.org/10.1007/s10900-019-00729-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10900-019-00729-3