Abstract
Hybrid materials based on organically modified silica with immobilized boron-dipyrrins (BODIPY) dyes were obtained. Spectral characteristics of the dyes were measured during the matrix formation and in obtained materials. It was stated, that immobilization does not affect the character of the absorption and fluorescence spectra of the dye, but due to the effect of fluorescent molecular rotor observed for meso-substituted BODIPY, the fluorescent quantum yields were found to increase during the matrix sealing. Quantum yield increase is linear for all of the investigated matrices except phenyl-substituted one, where the π-π interactions of the dye molecule with matrix could be observed. This effect could be used for fine control of the matrix formation process and leads to increase of the dye emission in the final material for further practical applications. All obtained hybrid materials were found to be stable upon UV light irradiation, hence immobilization enhance the stability of the dye in comparison with the BODIPY in organic solvents.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Boron-dipyrrin (BODIPY, 4,4-difluoro-4-bora-3a,4a-diazas-indacene) based compounds are one of the most commonly used fluorescent sensors and labels due to their high photostability and fluorescence quantum yields. BODIPY-based fluorescent sensors may be used for selective detection of cations [1, 2], anions [3, 4], molecules [5] and for accurate measurement of the solvent characteristics (polarity, pH, viscosity etc.) [6,7,8]. These sensors also effectively serve the purposes of biochemistry and molecular biology [9,10,11]. Another BODIPY application is covalent binding of luminophores to various biomolecules as labels [12].
The obtainment of fluorescent molecular rotors is one of the newest directions in BODIPY chemistry [13, 14]. While being exited one part of this molecule is able to rotate towards the other, causing the intramolecular charge transfer (this effect is called twisted intramolecular charge transfer state, or TICT state) [15, 16]. The main feature of such molecular rotors is a strong dependence between quantum yield and solvent dynamic viscosity. In this case, low viscosity mixtures may cause low fluorescence quantum yields due to high probability of non-radiative relaxation [17]. Fluorescent molecular sensors gain much popularity in molecular biology, analytical and biochemical studies nowadays [18], their precise analytical feedback and high selectivity being the reason for that.
Scope of BODIPY practical applications in analytical chemistry and optical devices is limited by dye stability to aggressive factors of the medium (irradiation, chemicals, temperature). Thus studies of the materials obtained by the sol–gel process with the inclusion of BODIPY is still of current interest, since properties of luminophores are retained in the hybrid materials and, at the same time, active component becomes more resistant to high temperatures and intense ultraviolet irradiation.
Examples of such an improvement could be found in the literature stated: In the study of Chateau D. materials with MTEOS (methyltriethoxysilane) and VTEOS (vinyltriethoxylsilane) as a precursors with inclusion of BODIPY were synthesized for use as an optical limiter operating in the NIR (near infrared) region [19]. In study of Hong, X.,BODIPY was immobilized into VTEOS matrix for cellular imaging [20]. In research of Hungerford, G., sol-to-gel transition was monitored by dint of the changes in the fluorescence of the dye in the reaction mixture [21].
The use of sol–gel precursors of different nature allows one to initiate various interactions between the matrix and the dye from the simple intermolecular interactions, to the donor–acceptor or covalent binding.
In this article, we contribute to the variety of the optically active hybrid materials, investigate the spectral properties of the obtained hybrid materials and explore principal possibility and nature of matrix binding with the dye. The dyes used in this study are presented in Fig. 1. These dyes and precursors have been chosen due to the assumption of their ability to form various interactions between the dye and the organomodified part of the matrix, namely the hydrogen bonding (in case of BODIPY 1, BODIPY 2 and APTMOS2, ClTEOS silica precursors) and π-π stacking (via phenyl substituent in BODIPY core and PhTEOS).
Experimental
Reagents and Apparatus
Triethoxyphenylsilane 98% (PhTEOS/PhSiO2), (3-Aminopropyl)trimethoxysilane 97% (APTMOS/NH2SiO2(precursor/ in material)) and (3-Chloropropyl)triethoxysilane 95% (ClTEOS/ClSiO2) were purchased on Sigma–Aldrich.. Tetraethoxysilane (TEOS/SiO2), hydrochloric acid, ethanol were purchased on Chimmed, Russia and used as is, without further purification. BODIPY dyes were synthesized by the authors previously [22].
U.v.–vis. electronic absorption spectra (EAS) were recorded on an SF-104 spectrophotometer (Aquilon, Russia) controlled with a PC using the software package UVWin 5.1.0. Absorption spectra of the thin films of hybrid materials were registered using solid samples holder, providing strictly perpendicular position of the test sample to the light beam. The accuracy of the measurements was ± 0.03 on the scale of optical density; wavelength accuracy was ± 0.05 nm. The fluorescence spectra for film materials was obtained using solid samples holder, providing angle of 45 degrees of the test sample to the light beam, with Cary Eclipse fluorescence spectrometer (Agilent technologies, USA). The slit widths for excitation beam and detector were ranged 2.5 to 10 nm. The accuracy of the wavelength measurements was ± 0.3 nm in the wavelength range 500–700 nm. Excitation wavelength was 480 nm. FTIR spectra were recorded on a spectrometer Avatar 360 (“Thermo Nicolet”, USA). Spectra were recorded in the range of 400–4000 cm− 1 at room temperature.
Irradiation of the samples was carried out by the u.v.-irradiator OUFB-04 (Russia) with the integral light of 180–270 nm range (u.v. C range). Effective irradiance in the stated spectral range was 11.4 W/m2. Degradation constant was calculated by the formula \(K=\frac{\varDelta I}{\varDelta \tau }\), where K – photodegradation rate constant, ΔI – change in fluorescence intensity during irradiation, Δτ – time period.
Preparation of Materials
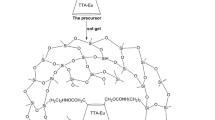
The materials were prepared by acid hydrolysis of the precursors. Molar ratios of reagents was n(precursor): n(C2H5OH) : n(H2O) : n(HCl) = 1:4:2:0.001. The reaction temperature was 25 °C. Appropriate BODIPY was added to the reaction mixture in concentration 10− 4 M. Obtained system was stirred for one hour, and then, after addition of the dye, material was deposited to a glass substrate by spin-coating. The glass substrates were rotated at 3500 rpm for 15 s. The slides had previously been treated with concentrated KOH for cleaning and degreasing purposes. After coating, the films were aged for 24 h at 25 °C.
For verification of molecular rotary properties, sol–gel process was carried out in 3.5 ml quartz cuvette with addition of the BODIPY. Cuvette was sealed, absorption and fluorescence spectra were measured with the fixed time interval.
Results and Discussion
Spectral Characteristics
Typical electronic absorption spectra of the sol of the hybrid materials are presented on the Fig. 2. Spectra of the films are identical to the sol–gel mixture in cuvette. Spectral characteristics of materials were compared with appropriate BODIPY in ethanol solution [22, 23].
Spectral characteristics of the obtained materials and dyes in ethanol are shown in Tables 1 and 1 of supplementary materials. SiO2 based hybrids are shown for comparison to demonstrate the influence of organic moiety on dye molecules and hybrid films morphology.
Spectral characteristics of all dyes differ from the ones in ethanolic solutions. 8–9 nm bathochromic shifts and decrease in quantum yields were observed in absorption spectra of SiO2 hybrids in comparison to solutions. Intense dye–dye interaction in the matrix is confirmed by the fact, that spectral characteristics of the hybrid materials are closer to the ones of the individual dyes films [24], than to ethanolic solutions. Simultaneously, taking into account negative solvatochromic effect of the BODIPY dyes [25], peak shifts could be attributed to lesser polarity of the polymeric matrix in comparison with ethanol.
Photophysical characteristics of the dyes vary depending on the matrix nature thereby confirming the specificity of the dye interaction with organic part of the polymeric matrix.
FTIR spectra of hybrid materials were taken in order to figure out specific interactions taking place between the matrix and the dyes (Fig. 3 and Fig. S1–S8 of supplementary materials). The intermolecular interactions of the BODIPY with the matrix were determined by the changes in position and intensity of the peaks in FTIR spectra of dyes (Table 2).
In case of the hybrids obtained from the PhTEOS, formation of a π-π stacking between phenyl ring of the matrix and phenyl moiety of the dye could be noticed for all of the studied BODIPYs.
Hydrogen bond formation between the lone pair of the nitrogen of the dye and hydroxyl groups hydrogens, situated on the surface of the matrix, was shown to occur in materials with BODIPY 2. A similar interaction was noticed for the BODIPY1@NH2SiO2 hybrid, in this case the hydrogen bond is formed between the nitrogen of the amino group of the matrix and the hydrogen in the carboxyl group of the dye.
Rotary Effect
Sensory properties of BODIPY 3 for dynamic viscosity of the sol–gel systems (molecular rotary properties) were studied. Several systems with different silica precursors were marked with rotor dye. Absorption and fluorescence spectra were measured at regular time intervals during 14 days at room temperature (Fig. 4). Increase in both absorbance and fluorescence intensity was stated for all of the systems. This fact could be explained by dye concentration increase caused by gel compression and solvent evaporation within system aging. Decline in the fluorescence and 7 nm batochromic shift in EAS noticed after 480 h of PhSiO2 system aging could be explained by specific matrix-dye interactions and rotary response of the BODIPY. The ability of the dye to act as rotor was determined from the changes in the fluorescence quantum yield, the calculation of which takes into account changes in absorption and fluorescence.
Gelation time for the pure SiO2 is 312 h while for PhSiO2 and NH2SiO2 it was measured to be 576 and 504 h respectively.
Based on the data we have plot the dependence between the fluorescence quantum yield of the gel aging time, data obtained was processed to establish the dependence between fluorescence quantum yield and gel aging time (Fig. 5). For SiO2 dependence between fluorescence quantum yield and aging time of the gel has a linear shape, confirming the hypothesis of the dye rotary properties. This result coincides with the work of Graham Hungerford [21].
Contrariwise, in case of the PhSiO2 system, dependence has a complex nature: intense fluorescence increase could be observed in the first time interval with further reaching a plateau and even intensity decrease during gel aging. Fluorescence decrease could be caused by the phenyl substituent of dye reaction with the phenyl from matrix causing energy transfer from the dye molecule to the matrix. Dependence found for NH2SiO2 is also close to a linear shape, but using of the BODIPY 3 for the dynamic viscosity control in this matrix is difficult due to specific interactions of the dye molecule with the matrix. From the abovementioned it could be stated that the BODIPY 3 can be used as the molecular rotary sensing probe in systems obtained from TEOS and PhTEOS.
Photochemical Stability of Materials
Irradiation with intense UV light was carried out for 30 min with fluorescence intensity control in 3 min time intervals, to test the photochemical stability of the obtained materials. An example of the obtained spectra is shown on the Fig. 6. Strong decrease of fluorescence in the initial moments of irradiation could be caused by the photochemical destruction of the BODIPY dyes situated on the surface of the hybrid material.
Degradation constants for the materials were calculated. Results are shown in Table 3.
BODIPY1@PhSiO2 was found to be the most photochemically resistant material and BODIPY3@PhSiO2 is, vice versa, the most labile one. Probably hydrogen bonding and π-π stacking between dye and matrix increases the stability of the dye molecules. Advanced resistance of the BODIPY 1 to UV irradiation may be caused by delocalization of the electron density at the carboxyl group.
Sensory Activity
Sensory activity of BODIPY1/2@NH2SiO2 and BODIPY1/2@SiO2 hybrids towards the pH of the solution was investigated (Fig. 7).
The dependence obtained from the material derived from SiO2 has a linear shape: fluorescence intensity grows with an increase in the alkalinity of the medium. For the materials obtained from APTMOS dependence is reversed. These changes in the spectra are reversible at pH decrease. Dyes were found to destruct at a pH of less than 2 with a non-fluorescent products formation. From the abovementioned it could be stated that obtained materials could be used for effective medum pH measurement in the range of 2 to 12.
Conclusions
In all of the obtained materials dyes retain their photophysical characteristics. The highest quantum yields were observed for the BODIPY1@ClSiO2 and BODIPY3@ClSiO2 hybrids. Formation of the hydrogen bonds and π-π stacking between the matrix and the dye was observed and proved using FTIR method, however a covalent bond formation was not stated.
Possibility of the BODIPY usage as the molecular rotary probe in the systems obtained from TEOS and PhTEOS was shown. In case of the NH2SiO2 matrix, the viscosity control is hindered by possible side matrix-dye interactions.
Obtained materials have shown good resistance to UV irradiation compared with films made from PMMA [24] and can be used as optical conversion devices, moreover, the material BODIPY1@PhSiO2 was found to be the most resistant one due to the formation of the hydrogen bonds and intense π-π stacking between the dye and the matrix.
Ability for evaluation of the pH of the environment in the range of 2 to 12 units was also shown for the obtained materials.
References
Wang LY, Fang GP, Cao DR (2015) A novel phenol-based BODIPY chemosensor for selective detection Fe3+ with colorimetric and fluorometric dual-mode. Sensors Actuators B Chem 207:849–857. https://doi.org/10.1016/j.snb.2014.10.110
He Y, Feng RK, Yi YR, Liu ZX (2014) Recent progress in the research of borondipyrromethene-based fluorescent ion chemosensor. Chinese J Org Chem 34:2236–2248. https://doi.org/10.6023/cjoc201403066
Madhu S, Kalaiyarasi R, Basu SK, Jadhavb S, Ravikanth M (2014) A boron-dipyrrin–mercury (II) complex as a fluorescence turn-on sensor for chloride and applications towards logic gates. J Mater Chem C 2:2534–2544. https://doi.org/10.1039/C3TC32188F
Liu J, He XX, Zhang J, He T, Huang LQ, Shen JQ, Li D, Qiu HY, Yin SC (2015) A BODIPY derivative for colorimetric and fluorometric sensing of fluoride ion and its logic gates behavior. Sensors Actuators B Chem 208:538–545. https://doi.org/10.1016/j.snb.2014.11.094
Niu Li-Ya et al (2012) BODIPY-based ratiometric fluorescent sensor for highly selective detection of glutathione over cysteine and homocysteine. J Am Chem Soc 134(46): 18928–18931. https://doi.org/10.1021/ja309079f
Gareis T, Huber C, Wolfbeis OS, Daub J (1997) Phenol/phenolatedependent on/off switching of the luminescence of 4,4-difluoro-4- Bora-3a,4a-diaza-S-indacenes. Chem Commun 18:1717–1718. https://doi.org/10.1039/A703536E
Werner T, Huber C, Heinl S, Kollmannsberger M, Daub J, Wolfbeis OS (1997) Novel optical Ph-sensor based on a boradiaza-indacene derivative. Fresenius J Anal Chem 359:150–154. https://doi.org/10.1007/s002160050552
Wu Y, Štefl M, Olzyńska (2013) Molecular rheometry: direct determination of viscosity in L o and L d lipid phases via fluorescence lifetime imaging. Phys Chem Chem Phys 15(36):14986–14993. https://doi.org/10.1039/c3cp51953h
Sui B, Tang S, Liu T (2014) Novel BODIPY-based fluorescence turn-on sensor for Fe3+ and its bioimaging application in living cells. ACS Appl Mater Interfaces 21:18408–18412. https://doi.org/10.1021/am506262u
Andrea B, Costero M, Salvador AG (2014) Chromo-fluorogenic BODIPY-complexes for selective detection of V-type nerve agent surrogates. Chem Commun 50:13289–13291. https://doi.org/10.1039/C4CC05945J
Fengling S, Yingying X, Xu W, Jingyun W, Xiaoqing X, Xiaojun P (2014) Ratiometric fluorescent probe based on novel red-emission BODIPY for determination of bovine serum albumin. Chem Res Chin Univ 30:738–742. https://doi.org/10.1007/s40242-014-4029-7
Ma D, Kim D, Seo E, Lee S, Ahn KH (2015) Ratiometric fluorescence detection of cysteine and homocysteine with a BODIPY dye by mimicking the native chemical ligation. Analyst 140:422–427. https://doi.org/10.1039/c4an01791a
Bahaidarah E, Harriman A, Stachelek P, Rihn S, Heyerb E, Ziessel R (2014) Fluorescent molecular rotors based on the BODIPY motif: effect of remote substituents. Photochem Photobiol Sci 10:1397–1401. https://doi.org/10.1039/c4pp00204k
Marfin YS, Merkushev DA, Levshanov GA, Rumyantsev EV (2014) Fluorescent properties of 8-phenylBODIPY in ethanol – ethylene glycol mixed solutions. J Fluoresc 24:1613–1619. https://doi.org/10.1007/s10895-014-1447-3
Rongrong H, Lager E, Aguilar-Aguilar A, Liu J, Lam JWY, Sung HHY, Williams ID (2009) Twisted intramolecular charge transfer and aggregation-induced emission of BODIPY derivatives. J Phys Chem C 113:15845–15853. https://doi.org/10.1021/jp902962h
Dal Molin M, Verolet Q, Soleimanpour S, Matile S (2015) Mechanosensitive membrane probes. Chemistry 21:6012–6021. https://doi.org/10.1002/chem.201406320
Haidekker M, Theodorakis E (2010) Environment-sensitive behavior of fluorescent molecular rotors. J Biol Eng 4:11–25. https://doi.org/10.1186/1754-1611-4-11
Benniston AC (2014) Monitoring rheological properties in biological systems by fluorescence spectroscopy using borondipyrromethene (Bodipy) dyes: a mini review. J Anal Bioanal Tech 5:1–11. https://doi.org/10.4172/2155-9872.1000221
Château D, Bellier Q, Chaput F, Feneyrou P, Berginc G, Maury O, … & Parola S (2014) Efficient hybrid materials for optical power limiting at telecommunication wavelengths. J Mater Chem C 2(26):5105–5110. https://doi.org/10.1039/C4TC00193A
Hong X, Wang Z, Yang J, Zheng Q, Zong S, Sheng Y, … & Cui Y (2012) Silylated BODIPY dyes and their use in dye-encapsulated silica nanoparticles with switchable emitting wavelengths for cellular imaging. Analyst 137(18):4140–4149. https://doi.org/10.1039/C2AN35389J
Hungerford G, Allison A, McLoskey D, Kuimova MK, Yahioglu G, Suhling K (2009) Monitoring sol-to-gel transitions via fluorescence lifetime determination using viscosity sensitive fluorescent probes. J Phys Chem B 113(35):12067–12074. https://doi.org/10.1021/jp902727y
Marfin YS, Merkushev DA, Usoltsev SD, Shipalova MV, Rumyantsev EV (2015) Fluorescent properties of 8-substituted BODIPY dyes: influence of solvent effects. J Fluoresc 25(5):1517–1526. https://doi.org/10.1007/s10895-015-1643-9
Marfin Yu. S., Shipalova MV, Kurzin VO, Ksenofontova KV, Solomonov AV, Rumyantsev EV (2016) Fluorescent properties of BODIPY sensors based on photoinduced electron transfer. J fluoresc. https://doi.org/10.1007/s10895-016-1905-1
Marfin SY et al (2013) Spectral and photophysical properties, photo and heat resistance of dipyrrolylmethene borofluoride complex and its hybrid material with polymethylmethacrylate. Russ J Gen Chem 83(2): 381–385. https://doi.org/10.1134/S1070363213020242
Marfin YS, Vodyanova OS, Merkushev DA, Usoltsev SD, Kurzin VO, Rumyantsev EV (2016) Effect of π-extended substituents on photophysical properties of BODIPY dyes in solutions. J Fluoresc 1–11. https://doi.org/10.1007/s10895-016-1891-3
Acknowledgements
The work was partially supported by the Grant of the President of the Russian Federation (МК-8835.2016.3) and the RFBR (Grant No 16-03-01028).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bobrov, A.V., Usoltsev, S.D., Marfin, Y.S. et al. Spectral Properties and Possibilities of meso-Substituted BODIPY Usage in Sol–Gel Process and Materials. J Fluoresc 28, 277–284 (2018). https://doi.org/10.1007/s10895-017-2190-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-017-2190-3