Abstract
Our main objective was to describe the course of GLS during the first days of septic shock and to assess the agreement between GLS values and longitudinal strain measured in apical four chambers. A prospective observational single centre study was conducted at the Nimes University Hospital’s ICU. All patients admitted for a diagnosis of septic shock without pre-existing heart disease were eligible. Echocardiography (LVEF and GLS) was performed on the first day, and repeated once between day 3 and day 5 then once between day 6 and day 8. We enrolled 40 consecutive patients. Four patients were excluded. In overall population, GLS at T1 was impaired (− 11.0%, IQR(interquartile range) [− 15; − 10]). On T2 exams, a significant improvement of the GLS (− 11% vs − 16% p = 0.02) was observed whereas LVEF remained stable over time. A good agreement between GLS and longitudinal strain measured on a four chambers view was found. Based on the Bland and Altman method, the mean of differences for T1 exams was 0.1 (95% CI [− 0.6; 0.8]) with limits of agreement ranging from − 4 to 4. Myocardial strain is depressed at the early phase of septic shock and improves over time. A single measurement of LS4C view appears sufficient at bedside.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Septic cardiomyopathy (SCM) is observed in up to 40% of patients with septic shock [1]. This clinical entity has been firstly described by Parker in the 80’s, who suggested that left ventricle ejection fraction (LVEF) was paradoxically higher in non-survivors of septic shock [2]. Subsequent studies did not confirm these results and a meta-analysis did not show any link between LVEF and mortality in septic population [3]. Several reviews discuss the pathogenesis of cardiac impairment in sepsis. Although no improvement in patient prognosis has been shown, ultrasound remains the more suitable tool for the evaluation of SCM [4].
The main mechanism for high LVEF is a profound inflammatory-induced vasoplegia, which decreases cardiac afterload and artificially increases LVEF. Severe vasoplegia and high dose of vasopressors requirement is associated with poor outcome, leading to refractory shock [5]. Thus, LVEF was considered as too dependent on loading conditions to be a reliable marker of intrinsic myocardial contractility in septic shock [6, 7]. It could be hypothesized that other echocardiographic markers of intrinsic contractility could be interesting to detect SCM. The use of conventional echocardiography is now widely recommended at bedside for non-invasive hemodynamic assessment in intensive care unit (ICU) patients, but the clinical usefulness of myocardial strain assessment remains unclear [8, 9].
Real-time speckle-tracking imaging is now widely available on echocardiography machine at bedside. This technology allows to assessing myocardial deformation over the cardiac cycle. Myocardial deformation is quantified by measuring global longitudinal strain (GLS). This index of segmental and global myocardial function could be less dependent of loading conditions. In cardiology, this technique has been validated using comparison with gold standard techniques such as magnetic resonance imaging [10]. Myocardial longitudinal strain assessment is noteworthy due to a fast learning curve, an automatized measurement and a good inter- and intra-observer reproducibility [11,12,13]. Initial experimental and humans data suggest that initial GLS could be a better and earlier marker of SCM than LVEF [7, 14]. The impact of GLS alterations on prognosis remains debated. An altered GLS value at admission (> − 13%) has been shown to independently predict mortality in septic shock [15, 15] whereas one study did not confirm the association between GLS and prognosis [17]. Two issues remain unclear about myocardial strain in ICU. First, the evolution of GLS over time can be of clinical importance but has been poorly described [17]. Second, the possibility to simplify strain assessment in ICU patients seems necessary as previous reports have suggested a feasibility ranging from 42% to 77% [7, 18]. This variability is mainly related to difficult assessment of GLS due to limited echogenicity of anterior wall in 2 and 3 chambers views [19]. Theoretically, GLS should be assessed in 2, 3 and 4 chambers views and subsequently averaged. This is difficult and time consuming in ICU patients, even if automatic real-time measurement simplifies the procedure. We hypothesized that SCM is a global heart disease and that there would be no difference between average left ventricle GLS and single value of left ventricle longitudinal strain recorded in a single 4 chamber view (LS4C).
Our main objective was to address these issues, by describing the natural history of left ventricle GLS during septic shock and evaluating the agreement between average left ventricle GLS and a single value of left ventricle longitudinal strain recorded in a single 4 chamber view (LS4C).
2 Methods
2.1 Study design
A prospective, observational cohort study was conducted at a single medico-surgical ICU of the Nimes University Hospital from February 2016 to march 2018 [20].
The study protocol was approved by the Local Ethics Committee (IRB/16.02.07) that waived written inform consent. Written information was given to the patient or his/her relatives who could refuse to participate.
All patients aged ≥ 18 years admitted for a diagnosis of septic shock were enrolled in the present study. Septic shock was defined by clinical evidence of sepsis with persisting hypotension requiring vasopressors to maintain MAP ≥ 65 mm Hg and a serum lactate level > 2 mmol/L (18 mg/dL) despite adequate volume resuscitation, according to current guidelines [21].
Exclusion criteria were age < 18 years, pregnancy, severe heart valvular disease, ischemic heart disease with known past history of ischemic sequelae, others cardiomyopathies (dilated cardiomyopathy, hypertrophic heart disease), atrial fibrillation or tachycardia > 120/min, patients in whom LV walls could not be evaluated through standard apical views. One untracked segment by view was considered as an exclusion criteria.
The following variables were recorded: age, gender, body mass index, past medical history, septic origin, vasopressor doses, mechanical ventilation use, ICU length of stay and 30 days mortality [22]. The severity of organ failure was assessed with the simplified acute physiology score (SAPS) II at admission [23].
All patients were treated according to international guidelines septic shock management [24].
2.2 Ultrasound protocol
The echocardiographic examination was firstly performed during the first 24 h after ICU admission (T1) and repeated once between day 3 and day 5 (T2) then once between day 6 and day 8 (T3).
All echocardiographic exams were performed by an expert in echocardiography using a Vivid S70 ultrasound device (GE Healthcare) [25] In addition, they performed an additional specialized training. Two-dimensional apical two-chamber, four-chamber and long-axis views (2C, 4C and ALAX) were recorded. Speckle tracking analysis was performed during a post treatment analysis after recording the echocardiographic loops. Briefly, analysis of LV myocardial deformation is performed from 2-dimensional gray-scale loops by automatic tracking of myocardial speckles after manual selection of landmark points using apical views of the left ventricle. GLS was calculated as the average of negative peak of longitudinal strain from each of the 18 LV segments from the 2C, 4C and ALAX views. Formula: GLS = (Ls − Ld)/Ld) (where Ls = systolic length and Ld = diastolic Length).
The main objective of the present study was to report the course of GLS throughout sepsis. We also compared GLS with LVEF in the overall population and in two subgroups: LVEF ≥ 45% and LVEF < 45%.
Left ventricle volumes and LVEF were calculated using the Simpson’s method [26]. Diastolic function was assessed according to international guidelines [27] The E and A waves velocities and E wave deceleration time were measured using pulsed wave (PW) Doppler in the mitral inflow at the tip of the valve visualized by an apical 4-chambers view. Early diastolic tissue wave velocity of the lateral mitral annulus (E’ wave velocity) was measured in the apical 4C view using PW tissue Doppler. Then we calculated the E/E’ ratio. Velocity Time Integral (VTI) of the left ventricular outflow tract we measured on an apical five-chambers view [28]. For the right ventricular (RV) evaluation the dilatation is assessed in four chambers with focus on the right ventricle [29]. The longitudinal contractility was assessed by the tricuspid annular plane systolic excursion (TAPSE) by using M mode on a 4-chambers view. For calculating RV strain, only the free wall right ventricle from a 4 chamber view was considered. We performed automatic tracking of myocardial speckles after manual selection of landmark points (lateral tricuspid annular, medial tricuspid annular and apex). Applying the LV speckle tracking method to RV walls, GLS of RV was calculated from values of the three segments of the free wall right ventricle. The three septal segments were excluded for the calculation of RV GLS.
At the time of echocardiographic examination, were also recorded clinical parameters: heart rate (HR), systolic (SBP), diastolic (DBP), and mean arterial (MAP) blood pressure, as well as biological parameters: pH, pO2, pCO2, base excess, bilirubin, lactates, troponins, platelets and serum creatinine.
2.3 Statistical analysis
Descriptive statistics were reported using median and interquartile range (25–75th percentile) for continuous variables. For categorical variables, frequencies and proportions were given. To assess the change between T1 and T2 for the average GLS and LVEF, we used a Wilcoxon signed-rank test with continuity correction. We also studied the course of GLS in the subgroup defined by an initial impairment of LVEF below 45%. Finally, to assess the agreement between average GLS and the four-chamber view, we used the Bland and Altman method by computing the mean difference and its 95% confidence interval and limits of agreement defined as mean difference ± 1.96 SD. A p value of 0.05 was considered as significant. All statistical analyses were performed using R software (version 3.3.2).
3 Results
3.1 Patients’ characteristics
Among the 40 consecutive patients enrolled, 4 patients were excluded: two patients had previous ischemic cardiomyopathy with anterior sequelae, one patient had an atrial fibrillation and one had missing data for GLS. Table 1 summarizes the main clinical characteristics of the 36 included patients. All patients reached the targeted mean arterial blood pressure > 65 mmHg as usually recommended [9]. No patient was lost for follow up. No patient died during study protocol. Eight patients (22%) died during the month after ICU admission. The site of infection is shown in Table 1.
3.2 Hemodynamic and biological parameters (Tables 2 and 3)
Between T1 and T2 exams, a decrease in HR over time was observed, and increases in SBP and pulse pressure.
Echography was performed respectively 9.65 (IQR [3.07; 16.88]), 74.88 (IQR[62; 90.75]) and 182 (IQR [164.51; 199.33]) hours after admission, respectively for T1, T2 and T3 exams.
Considering the echocardiography parameters, VTI increased between T1 and T2, LVEF was not statistically different between T1 and T2 (p = 0.2). Others parameters remained stable (Table 2).
Table 3 shows the biological parameters with a decrease in arterial lactate and an increase in base excess between T1 and T2.
3.3 Global longitudinal strain
GLS could be calculated in 36, 22 and 9 patients at T1, T2 and T3, respectively. Patients discharged from the ICU before T2 and/or T3 assessments occurred together with experts unavailability at T2 and/or T3 were the main reasons for missing GLS data over time.
Baseline GLS was impaired (> − 13%) in 23 patients (65.7%). In overall population GLS, improved throughout the study whereas LVEF did not change (Fig. 1).
Evolution of longitudinal strain and left ventricle ejection fraction values over time during septic shock (median, interquartile). T1 = day 1, T2 = day 3–5, T3 = day 8. lower whisker = smallest observation greater than or equal to lower hinge−1.5 * IQR, upper whisker = largest observation less than or equal to upper hinge + 1.5 * IQR)
For the 22 patients for whom GLS was available at T1 and T2, GLS appeared significantly improved at T2 (− 11% vs − 16%) (p = 0.02) (Fig. 1). This significant improvement was also found when considering LS4C (− 11% vs − 15%, p = 0.004).
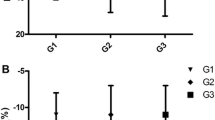
In the subgroup of patients with SCM defined by LVEF < 45% (7 patients), LVEF and GLS evolution over time seemed to be similar (Fig. 2a, b). The initial GLS was impaired in low LVEF Group compared to LVEF ≥ 45% group (− 9.6 vs − 11%). In this sub group, both GLS and LVEF improved at T2 (GLS: − 9.6 vs − 15.5%) (LVEF: 29% vs 50%) (Fig. 2a, b).
The comparison between mean longitudinal strain values in 2, 3 and 4 chambers view and the averaged GLS are reported in Fig. 3. The Bland and Altman method showed a good agreement between GLS and LS4C: For T1 exams (n = 36) mean of differences 0.1 (95% CI [− 0.6; 0.8]); For T2 exams (n = 23) mean of differences 0.1 (95% CI [− 0.96; 1.16]) (Fig. 4a, b).
Bland and Altman diagram to assess agreement between average GLS (average of GLS value obtained for two chambers, four chambers and APLAX views) and four-chamber GLS as a simplified method for strain assessment (single Longitudinal strain: SLS method) in T1 (a) and T2 (b). Computing the mean difference and its 95% confidence interval and limits of agreement defined as mean difference ± 1.96 SD
The course of GLS in survivors and non-survivors is shown in Table 4.
4 Discussion
4.1 Key findings
GLS was altered at day 1 of septic shock and significantly improved over time, until normalization. On the opposite, conventional methods for LV systolic function assessment as LVEF and S wave velocity were not affected by the natural course of sepsis. As a second result, GLS values were comparable whatever the echocardiographic view (2, 4 chambers and APLAX views) considered, suggesting that a single measurement in 4 chambers view (SL4C)) is sufficient at bedside.
4.2 Relationship with previous literature
SCM has been well reported during septic shock [30]. Recent data suggest that diastolic dysfunction is more frequent and more associated with prognosis than systolic dysfunction [31, 32]. Moreover, supranormal systolic function (hyperkinetic state, defined as an LVEF > 60%) is associated with worse prognosis [7]. Systolic function is usually assessed by echocardiography, by measuring conventional LVEF. Diastolic function is assessed by E’ wave velocity recording [17]. GLS is correlated to both systolic and diastolic function during sepsis [17]. GLS can be altered while LVEF remains preserved. Therefore, GLS could be a better and earlier indicator of systolic dysfunction than LVEF. In an experimental model of E coli-induced sepsis in anesthetized and ventilated piglets, Hestenes et al. [14] reported a significant linear alteration of LV GLS after E coli infusion (from − 17.2% ± 2.8% to − 12.3% ± 3.2%, p = 0.04) on a study period of 240 min. Right ventricle GLS was also significantly altered (from − 24.2% ± 4.1% to − 16.9% ± 5.7%, p = 0.02). In this model, LVEF remained unaltered (from 48% ± 7% to 49% ± 5% over the study period, p = 0.4). The conclusion was that GLS helps at assessing myocardial dysfunction during sepsis while LVEF assessment may underestimate this diagnosis. Analogous results have been obtained in humans. The evolution of strain has been studied in the first day of sepsis and septic shock [33]. At baseline, GLS was abnormal both in sepsis and septic shock patients (− 15%). At 24 h, GLS was significantly impaired only in septic shock patients (− 13% at H24) but unchanged in sepsis group (− 17% at H24), suggesting a more profound myocardial impairment during septic shock than during sepsis and a link between severity and GLS. In this study, in contrast to GLS, LVEF remained normal in both groups. This study showed that rapid and profound change in GLS value can occur during the first 24 h of septic shock, without concomitant alterations of LVEF. In our study, patients were mainly analyzed at the end of the first 24 h of septic shock, explaining why a very poor value of GLS (− 11.7%) was reported at day 1, close to value reported by Shahul et al. [33]. In a large study involving 132 septic patients, GLS was altered in 70% of patients, whatever the value of LVEF [7]. Similarly, our results confirm that GLS is altered during sepsis whereas LVEF remains normal as well as S wave velocity, a classical marker of systolic function, that is not altered over time in the present study (Table 2). Additionally, the present results show that, in the whole population, GLS improves over time while LVEF and S wave velocity remain stable in the natural course of sepsis. In the sub group of patients with altered LVEF at baseline, both LVEF and strain improved from T1 to T2. In the sub group of patients with altered LVEF, the clinical interest of strain to characterize SCM is less obvious than for patients with normal LVEF.
Because both preload and afterload are decreased at the early phase of sepsis, LVEF can be artificially high and usually normal in both survivors and non-survivors [31]. Theoretically, GLS should be less affected by loading conditions than LVEF [34] because it does not depend on ventricular volume [17, 35]. However, recent studies have challenged the concept that myocardial strain is independent from loading conditions. Nafati et al. recently reported that, in preload dependent patients, strain can move from pathological to normal value after fluid challenge (− 13.3 ± 3.5 to − 18.4% ± 4.5 (p < 0.01) [36]. This means that the improvement of GLS over time during sepsis observed in the present report could be partially due to improvement of preload conditions during fluid resuscitation. Although we cannot formally exclude a correction improvement of preload conditions, static echocardiographic preload indicators (E wave velocity, E/E’ ratio, E/A ratio) [7] suggest that GLS improvement over time is rather due to favorable myocardial function evolution during sepsis rather than to preload increase. Our results slightly differ from those obtained by De Geer et al. in the timing of improvement [17]. This group reported a significant impairment of GLS from day 1 to day 3–4 and a return to baseline value at the end of sepsis. Both studies suggest that GLS is affected by sepsis and subsequently improves.
While a new assessment tool becomes available at bedside, it seems crucial to evaluate both its reliability and its feasibility in order to implement this tool in clinical practice. Thus, the ability for an ICU physician to rapidly and easily perform GLS in ICU patients at bedside is of importance. Boissier et al. [7] recently reported a poor feasibility (< 50%) of GLS during septic shock. Difficult assessment of GLS is classically due limited echogenicity for anterior wall in 2 and 3 chambers views [19]. Moreover, GLS assessment in 2 and 4 chambers views and APLAX incidence is time consuming, even with machines allowing automatized real time analysis. Interestingly, our results suggest that GLS values are not statistically different in these three incidences. These findings support the hypothesis that SCM is a global disease, which equally affects all LV myocardial segments. Therefore, GLS analysis from a single incidence (4 chambers view) appears to be good enough at bedside to detect SCM.
The single evaluation of LS4C is an interesting and less time-consuming approach than a complete evaluation of GLS in the ICU setting. In addition of being easier and more rapid to assess SCM, LS4C could be more precise as highlighted by the narrower CI of SL4C compared to GLS. LS2C is frequently difficult to access and a complete evaluation with difficult incidence might distort the result.
The present study was not designed for assessing the association between SL4C and mortality. The small sample size and the small number of events does not allow performing multivariate analysis for survival independent risk factors. Nevertheless, in our cohort, the strain values are more altered in non-survivors. This is consistent with literature. Previous reports suggest an association between initial GLS and mortality [15, 16]. In a recent metanalysis, Sanfilippo et al. demonstrated that altered GLS values are associated with higher mortality in patients with severe sepsis or septic shock [37]. These deserve further investigation [38] which will be facilitated by a unique LS4C assessment.
4.3 Clinical implications
The present report shows that GLS is profoundly depressed at the early phase of septic shock and subsequently improves. This early value of GLS better reflect SCM than LVEF. The second implication is that a single longitudinal strain measurement on 4 chambers view indirectly reflects the whole myocardial strain. This can be a simplest method to evaluate GLS at bedside. This can help to detect a more severe population, even when LVEF is normal.
4.4 Study limitations
This study has some limitations to acknowledge. Firstly, this is a single centre study that may prevent from extrapolating these results to other centres. Secondly, this study included a small cohort of subjects limiting the statistical power of the study. However, this is the first study describing the course of GLS during septic shock. Thirdly, several patients were not available for the third GLS assessment at day 8 due to ICU discharge or unavailability of the operators. Finally some confounders such as vasopressor were not considered in this study.
5 Conclusion
Myocardial strain is depressed at the early phase of septic shock and improves over time. Despite a profound alteration of strain, LVEF remains normal and does not reflect severity of SCM and its subsequent evolution. A single measurement of GLS in a 4 chambers view appears sufficient at bedside as compared to complete myocardial evaluation (2, 4 chambers and APLX views). Compliance with ethical standards: No conflict of interest for all authors to declare. No funding was obtaining for this works. Due to the observational design of the study, the Ethics committee (IRB/16.02.07) waived the need for patient consent.
References
Ehrman RR, Sullivan AN, Favot MJ, Sherwin RL, Reynolds CA, Abidov A, et al. Pathophysiology, echocardiographic evaluation, biomarker findings, and prognostic implications of septic cardiomyopathy: A review of the literature. Crit Care. 2018;22:112.
Parker MM, Shelhamer JH, Bacharach SL, Green MV, Natanson C, Frederick TM, et al. Profound but reversible myocardial depression in patients with septic shock. Ann Intern Med. 1984;100:483–90.
Huang SJ, Nalos M, McLean AS. Is early ventricular dysfunction or dilatation associated with lower mortality rate in adult severe sepsis and septic shock? A Meta-Analysis Crit Care. 2013;17:R96.
Zaky A, Deem S, Bendjelid K, Treggiari MM. Characterization of cardiac dysfunction in sepsis: An ongoing challenge. Shock. 2014;41:12–24.
Martin C, Medam S, Antonini F, Alingrin J, Haddam M, Hammad E, et al. Norepinephrine: Not too much too long. Shock. 2015;44:305–9.
Vieillard-Baron A. Septic cardiomyopathy. Ann Intensive Care. 2011;1:6.
Boissier F, Razazi K, Seemann A, Bedet A, Thille AW, de Prost N, et al. Left ventricular systolic dysfunction during septic shock: The role of loading conditions. Intensive Care Med. 2017;43:633–42.
Seymour CW, Rosengart MR. Septic shock: Advances in diagnosis and treatment. JAMA. 2015;314:708–17.
Cecconi M, De Backer D, Antonelli M, Beale R, Bakker J, Hofer C, et al. Consensus on circulatory shock and hemodynamic monitoring. Task force of the European Society of Intensive Care Medicine. Intensive Care Med. 2014;40:1795–815.
Amundsen BH, Helle-Valle T, Edvardsen T, Torp H, Crosby J, Lyseggen E, et al. Noninvasive myocardial strain measurement by speckle tracking echocardiography: Validation against sonomicrometry and tagged magnetic resonance imaging. J Am Coll Cardiol. 2006;47:789–93.
Negishi T, Negishi K, Thavendiranathan P, Cho G-Y, Popescu BA, Vinereanu D, et al. Effect of experience and training on the concordance and precision of strain measurements. JACC Cardiovasc Imaging. 2017;10:518–22.
Dandel M, Lehmkuhl H, Knosalla C, Suramelashvili N, Hetzer R. Strain and strain rate imaging by echocardiography - Basic concepts and clinical applicability. Curr Cardiol Rev. 2009;5:133–48.
Mentz RJ, Khouri MG. Longitudinal strain in heart failure with preserved ejection fraction: Is there a role for prognostication? Circulation. 2015;132:368–70.
Hestenes SM, Halvorsen PS, Skulstad H, Remme EW, Espinoza A, Hyler S, et al. Advantages of strain echocardiography in assessment of myocardial function in severe sepsis: An experimental study. Crit Care Med. 2014;42:e432-440.
Chang W-T, Lee W-H, Lee W-T, Chen P-S, Su Y-R, Liu P-Y, et al. Left ventricular global longitudinal strain is independently associated with mortality in septic shock patients. Intensive Care Med. 2015;41:1791–9.
Innocenti F, Palmieri V, Guzzo A, Stefanone VT, Donnini C, Pini R. SOFA score and left ventricular systolic function as predictors of short-term outcome in patients with sepsis. Intern Emerg Med. 2018;13:51–8.
De Geer L, Engvall J, Oscarsson A. Strain echocardiography in septic shock - A comparison with systolic and diastolic function parameters, cardiac biomarkers and outcome. Crit Care. 2015;19:122.
Nafati C, Lançon V, Blasco V, Zieleskiewicz L, Harti K, Wiramus S, et al. Two-dimensional-strain echocardiography in intensive care unit patients: A prospective, observational study. J Clin Ultrasound. 2016;44:368–74.
Marwick TH, Leano RL, Brown J, Sun J-P, Hoffmann R, Lysyansky P, et al. Myocardial strain measurement with 2-dimensional speckle-tracking echocardiography: Definition of normal range. JACC Cardiovasc Imaging. 2009;2:80–4.
Leone M, Constantin J-M, Dahyot C, Duracher C, Friggeri A, Joannes Boyau O, Langeron O, Legrand M, Mahjoub Y, Mirek S, Mrozek S, Muller L, Orban J-C, Quesnel C, Virat A, Capdevila X. French intensive care unit organization. Anesthesiol Intensive Care Med. 2018;37:625–7.
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA. 2016;315:801–10.
Nouira H, Ben Abdelaziz A, Kacem M, Ben Sik Ali H, Fekih Hassen M, Ben Abdelaziz A. Which indicators used to assess quality performance in Intensive Care Units? A systematic review of medical literature. Anaesth Crit Care Pain Med. 2018;37(6):583–7
Le Gall JR, Lemeshow S, Saulnier F. A new simplified acute physiology score (SAPS II) based on a European/North American multicenter study. JAMA. 1993;270:2957–63.
Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R, et al. Surviving sepsis campaign: International guidelines for management of sepsis and septic shock: 2016. Intensive Care Med. 2017;43:304–77.
Mayo PH, Beaulieu Y, Doelken P, Feller-Kopman D, Harrod C, Kaplan A, et al. American College of Chest Physicians/La Société de Réanimation de Langue Française statement on competence in critical care ultrasonography. Chest. 2009;135:1050–60.
Lang RM, Bierig M, Devereux RB, Flachskampf FA, Foster E, Pellikka PA, et al. Recommendations for chamber quantification: A report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr. 2005;18:1440–63.
Nagueh SF, Smiseth OA, Appleton CP, Byrd BF, Dokainish H, Edvardsen T, et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2016;29:277–314.
Chengode S. Left ventricular global systolic function assessment by echocardiography. Ann Card Anaesth. 2016;19:S26-34.
Harjola V-P, Mebazaa A, Čelutkienė J, Bettex D, Bueno H, Chioncel O, et al. Contemporary management of acute right ventricular failure: A statement from the Heart Failure Association and the Working Group on Pulmonary Circulation and Right Ventricular Function of the European Society of Cardiology. Eur J Heart Fail. 2016;18:226–41.
Beesley SJ, Weber G, Sarge T, Nikravan S, Grissom CK, Lanspa MJ, et al. Septic cardiomyopathy. Crit Care Med. 2018;46:625–34.
Sanfilippo F, Corredor C, Fletcher N, Landesberg G, Benedetto U, Foex P, et al. Diastolic dysfunction and mortality in septic patients: A systematic review and meta-analysis. Intensive Care Med. 2015;41:1004–13.
Sanfilippo F, Corredor C, Arcadipane A, Landesberg G, Vieillard-Baron A, Cecconi M, et al. Tissue Doppler assessment of diastolic function and relationship with mortality in critically ill septic patients: A systematic review and meta-analysis. Br J Anaesth. 2017;119:583–94.
Shahul S, Gulati G, Hacker MR, Mahmood F, Canelli R, Nizamuddin J, et al. Detection of myocardial dysfunction in septic shock: A speckle-tracking echocardiography study. Anesth Analg. 2015;121:1547–54.
Pulido JN, Afessa B, Masaki M, Yuasa T, Gillespie S, Herasevich V, et al. Clinical spectrum, frequency, and significance of myocardial dysfunction in severe sepsis and septic shock. Mayo Clin Proc. 2012;87:620–8.
Russo C, Jin Z, Homma S, Rundek T, Elkind MSV, Sacco RL, et al. Relationship of multidirectional myocardial strain with radial thickening and ejection fraction and impact of left ventricular hypertrophy: A study in a community-based cohort. Echocardiography. 2013;30:794–802.
Nafati C, Gardette M, Leone M, Reydellet L, Blasco V, Lannelongue A, et al. Use of speckle-tracking strain in preload-dependent patients, need for cautious interpretation! Ann Intensive Care. 2018;8:29.
Sanfilippo F, Corredor C, Fletcher N, Tritapepe L, Lorini FL, Arcadipane A, et al. Left ventricular systolic function evaluated by strain echocardiography and relationship with mortality in patients with severe sepsis or septic shock: A systematic review and meta-analysis. Crit Care. 2018;22:183.
Ehrman RR, Moore SC, Favot MJ, Akers KG, Gallien JZ, Welch RD, et al. Scientific letter to the editor: Need for a definitive study of global longitudinal strain for prognostication in septic cardiomyopathy. J Am Soc Echocardiogr. 2019;32(549–552):e3.
Funding
No funding was obtaining for this works. Due to the observational design of the study, the Ethics committee (IRB/16.02.07) waived the need for patient consent.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest for all authors to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bazalgette, F., Roger, C., Louart, B. et al. Prognostic value and time course evolution left ventricular global longitudinal strain in septic shock: an exploratory prospective study. J Clin Monit Comput 35, 1501–1510 (2021). https://doi.org/10.1007/s10877-020-00620-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-020-00620-w