Abstract
Measuring cardiac output (CO) is an integral part of the diagnostic and therapeutic strategy in critically ill patients. During the last decade, the single transpulmonary thermodilution (TPTD) technique was implemented in clinical practice. The purpose of this paper was to systematically review and critically assess the existing data concerning the reproducibility of CO measured using TPTD (COTPTD). A total of 16 studies were identified to potentially be included in our study because these studies had the required information that allowed for calculating the reproducibility of COTPTD measurements. 14 adult studies and 2 pediatric studies were analyzed. In total, 3432 averaged CO values in the adult population and 78 averaged CO values in the pediatric population were analyzed. The overall reproducibility of COTPTD measurements was 6.1 ± 2.0 % in the adult studies and 3.9 ± 2.9 % in the pediatric studies. An average of 3 boluses was necessary for obtaining a mean CO value. Achieving more than 3 boluses did not improve reproducibility; however, achieving less than 3 boluses significantly affects the reproducibility of this technique. The present results emphasize that TPTD is a highly reproducible technique for monitoring CO in critically ill patients, especially in the pediatric population. Our findings suggest that obtaining a mean of 3 measurements for determining CO values is recommended.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Cardiac output (CO) measurement constitutes an integral part of the diagnostic and therapeutic strategy in critically ill patients. Measuring CO allows physicians to characterize shock states in patients and to guide therapy at the bedside, especially during fluid resuscitation and adaptation of vasoactive or inotropic support.
During the last decade, transpulmonary thermodilution (TPTD) using the single indicator technique [1] was implemented in clinical practice and gradually replaced the pulmonary thermodilution. The development and the implementation of this new technique (TPTD) in clinical practice are significantly reflected by the number of publications in Index Medicus between 1982 and 2014. A total of 349 citations appear when the keyword “transpulmonary thermodilution” is typed.
The TPTD curve allows the measurement of CO (COTPTD) and the estimate of derived indices, thereby permitting assessment of cardiac preload [2, 3] and extravascular lung water [4–6]. COTPTD is determined using the Stewart-Hamilton equation applied to a thermodilution curve [7], which is similar when using a pulmonary artery catheter [8]. Compared to the right heart thermodilution curve, a notable exception lies in the injection site of the thermal indicator and the recording site of the thermodilution curve: the TPTD curve is obtained by injecting the thermal indicator into a central intrathoracic vein instead of the right atrium, and the thermal shift is collected via an arterial catheter inserted in a large systemic arterial trunk (femoral, axillary or brachial artery), instead of the pulmonary artery, after a transpulmonary transit of the cold indicator.
At its output, the TPTD technique was the subject of debate concerning whether this new technique could be a surrogate of CO measurement by pulmonary artery catheter. In this regard, the evaluation of the reproducibility of the present technique is essential for recognizing the difference between changes in the patient’s physiological status and errors associated with the technique. However, only one study [9] has specifically evaluated the reproducibility of COTPTD and the assessment of how closely individual measurements agree with each other when all efforts attempt to keep hemodynamic conditions constant.
The purpose of this paper is to systematically review and critically assess the existing data concerning the reproducibility of COTPTD. The present analysis may, in large part, impact clinical practice and the definition of a positive response to fluid infusion. The ‘magic threshold value’ of a 15 % increase in CO, which is adapted to assess fluid responsiveness, is primarily related to the precision of the pulmonary artery catheter [10].
2 Methods
The statistical definition of precision is the variability of values attributed to random measurement errors. Precision means repeatability when the technique of measurements is performed by the same operator, during constant conditions, and repeated within a short time period. Precision equals reproducibility when the process of measurements is performed among different operators (laboratories) and over longer time periods. Thus, when performing a systematic analysis and adding repeatability of COTPTD values obtained from distinctive studies, the computation equals the measurement of reproducibility.
2.1 Search strategy
Combinations of key words related to “Transpulmonary thermodilution AND precision”; “Transpulmonary thermodilution AND reproducibility”; “Transpulmonary thermodilution AND CO” were used to search reviews and reports on the MEDLINE and Embase databases and the Cochrane Group trial register without any language restriction. The number of articles published in each category was 25, 52, and 234, respectively. The last search was performed in December 2014. We checked the bibliographies of the retrieved reports and reviews. For conducting and reporting the present systematic review, the authors followed the MOOSE Guidelines for observational studies [11], when indicated. As there is no gold-standard validated method for assessing bias and the quality of observational studies (10, [12] but there is consensus that the three most fundamental domains are (1) appropriate selection of studies, (2) appropriate measurement of variables, and (3) appropriate control of confounding, as well as considering design-specific biases, we therefore discuss these quality criteria for each study in a narrative fashion. Also, we did not consider data from abstracts, letters and animal studies. Because this study did not involve human participants or medical records, institutional review board approval was neither required nor obtained.
2.1.1 Inclusion criteria
To assess the reproducibility of TPTD technique in all of the studies, we assessed the precision reported in each study (repeatability). The precision was often referred to the coefficient of error of the estimate, which is a statistical expression for the size of the standard error of the mean of repeated estimates, relative to the mean of the estimates. In cases where we were unable to find the COTPTD precision value, we extracted either the coefficient of variation (CV), or the coefficient of error (CE), or the standard deviation of the mean (SEM), or the least significance change (LSC). CE and CV were obtained using the formula: CE = CV/√n or CV = CE × √n, respectively, where n is the number of replicates of measurements in each patient. The precision is calculated as two CEs. A measurement precision level of ≤10 % is desirable [13]. The LSC is the minimum change that must be measured by a device to recognize a real change of measurement [14]. LSC was calculated using the following equation: LSC = CE × 1.96 × √2 [9].
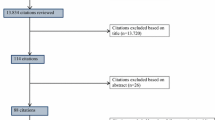
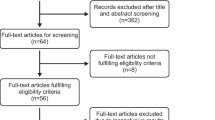
After reading and analyzing these articles, 16 studies (n = 14 adult studies and 2 pediatric studies) that met the inclusion criteria were identified (Fig. 1). An additional computerized search, “ISI Web of Knowledge”, which analyzed citing and cited articles as well as a manual search in the references of relevant articles, did not uncover any more articles that could be included in our systematic analysis. The characteristics of all of the studies were independently rated by R.G. And K.B., and the discrepancies were discussed.
We found 14 studies in which TPTD was used to measure CO, without including reproducibility results in their articles. We attempted to contact these eight research teams to obtain additional data. Unfortunately, only one response was sent to us with unusable data in the context of our systematic review. In selected studies for this systematic review, we wanted to obtain additional data, such as the standard deviation (SD) of CO measurements, to perform a meta-analysis with complete results. We contacted the authors of these studies (14 e-mails were sent twice a month apart); however, we received only two responses.
In each case, the number of measurements for determining a CO value, number of CO averaged measurements, temperature, nature, volume and injection site of the indicator and insertion site of the arterial catheter were collected. Additionally, the number of cold boluses to obtain a CO averaged value and the total numbers of thermodilution curves were required for calculating reproducibility.
2.2 Statistical analysis
The results are expressed as absolute values for the number of averaged CO measurements and for the total thermodilution curves achieved, as the mean ± SD and the median for CO measurements or cardiac index and as percentages for precision, CV and LSC. Graphs were constructed using GraphPad Prism 5.0 (GraphPad Software, San Diego, CA, USA) and Microsoft office Excel 2007.
3 Results
A flow diagram is shown in Fig. 1, and demographics data are presented in Table 1 for adult studies and in Table 2 for pediatric studies. In these tables, we wanted to show CV, LCS and precision and to clarify which values were extracted from specific articles to achieve the present results.
The primary results are shown in Tables 1 and 2. A total of 3432 averaged CO values (9775 TPTD boluses) were reviewed from 14 adult studies and 78 averaged CO values (210 TPTD boluses) were reviewed from 2 pediatric studies. In all studies CVC were positioned in the superior vena cava. The average number of bolus made by TPTD to obtain a CO value was 3.3 ± 0.6 in the adult studies and 2.7 ± 0.6 in the pediatric studies. In 12 of 14 adult studies and in 2 pediatric studies, the authors conducted three thermodilution boluses; in 2 adult studies, they performed 4 boluses; in 1 adult study, they performed 5 boluses; and in 1 adult study, they performed 2 boluses.
Concerning the study conducted by Kiefer et al., the same 72 patients received 3 boluses to assess a CO value. The TPTD curves were analyzed twice: on the one hand, on-line using the EV1000TM algorithm; and on the other hand, off-line, using the PiCCO TM2 algorithm (this study appears twice and is counted twice, as shown in Table 1).
The overall reproducibility of COTPTD was 6.1 ± 2.0 % and LSC was 8.5 ± 2.7 % for CO measurements from the 14 selected adult studies. The overall reproducibility of COTPTD was 3.9 ± 2.9 % and LSC was 5.4 ± 4.0 % for CO measurements from the two selected pediatric studies (Table 2) (Fig. 2a, b).
In the present study, we performed a subgroup analysis after classifying the data into three categories based on the number of boluses injected to determine an average CO value: less than 3 boluses, 3 boluses and more than 3 boluses. Reproducibility was 6.6 ± 1.7, 5.4 ± 1.7 and 10.2 ± 3.7 %, depending on whether 3, more than 3 or less than 3 boluses were performed, respectively. LSC was 9.1 ± 2.3, 7.5 ± 2.3 and 14.2 ± 5.1 %, depending on whether 3, more than 3 or less than 3 boluses were performed, respectively (Fig. 3a, b).
The cold boluses were injected through the proximal port of the pulmonary artery catheter (PAC) in 5 studies or through the central venous catheter (CVC) in 9 studies. In one study, it was not apparent in which site the cold boluses were injected. The reproducibility and the LSC of the COTPTD measurements were not affected by the injection site (Supplemental Digital Content 1 a and b). In 12 studies, the PiCCO arterial catheter was inserted in the femoral artery, whereas in 2 studies, the arterial catheter was inserted in the axillary artery (Supplemental Digital Content 2 a and b).
The amount of cold injected liquid was variable according to the studies. The injected liquid ranged from 10 to 20 mL, knowing that in 10 studies, the cold bolus was at least 15 mL. The injection site did not seem to influence the reproducibility nor the LSC of the CO measurement by TPTD (Supplemental Digital Content 1. a and b and 2. a and b). However, when the amount of fluid injected and the site of injection were analyzed together (CVC 15 mL vs. CVC 20 mL and PAC 10 mL vs. PAC 15 mL), the precision and the LSC were better for both the injection of 20 versus 15 mL cold liquid through the CVC and the injection of 15 vs. 10 mL via the PAC (Fig. 4a, b). Moreover, in all of the studies, the cold bolus was a liquid with a temperature below 8 °C.
Finally, we constructed a bubble graph, which illustrates that the precision of TPTD depends on the number of boluses to calculate a CO value and the number of boluses achieved in each study. The bubble graph showed that reproducibility improves with an increased number of boluses (Fig. 5).
Bubble graph illustrating the precision of TPTD assessed in each study depending on the number of boluses achieved to calculate a CO value. The surface of each bubble represents the magnitude of boluses made in each study and considered in this review. Pink bubbles adult Studies, Blue bubbles pediatric studies
4 Discussion
The primary finding of the present systematic review is that the TPTD technique has a higher reproducibility value compared to the pulmonary artery catheter [1, 10]. This result is likely related to the longer transit time of the thermal bolus, which finally is not influenced by respiration and arrhythmias.
To detect 15 % change in CO, a monitor must have a level of precision that can detect this change with 95 % certainty. However, measuring a change does not necessarily mean that the patient’s physiological status has changed [15]. The reproducibility of a technique is its ability to provide CO values that are close to each other. This factor is especially important for techniques measuring CO [16] because we are more interested in variations of CO values over time than in a given CO value. In this regard, reproducibility of a technique capable of measuring CO is important, particularly when the task is to assess fluid responsiveness. For example, when a patient is considered to be fluid responsive because his CO increases above 15 %, it is essential to ensure that the difference in CO measured before and after volume expansion is actually the patient’s physiological reaction to the volume administration, rather than related to the error associated to the measurement technique. As the magic threshold value of 15 % CO increase is primarily related to the clinical significance and reproducibility of the pulmonary artery catheter [10, 17], we could argue that when CO is measured by TPTD, a technique that has a reproducibility of 6.1 ± 2.0 %, a 10 % CO increase could define a patient to be fluid responsive.
The good reproducibility of TPTD can be explained by the fact that using this technique, the cold indicator takes approximately twenty-seconds to be captured by the femoral temperature sensor, whereas using the right heart thermodilution, the temperature variation is detected after approximately 3–4 s. The present longer transit time reduces artefacts produced by airway pressure and arrhythmia on venous return-CO steady state, which have more impact on the results of right heart thermodilution [18, 19]. Also, the relative very good reproducibility observed in the pediatric population [20, 21] can be explained by several mechanisms: first, the small size of children allows arterial and venous catheters to be inserted specifically in the thoracic compartment. Second, the heart rate and respiratory rate are higher for children than for adults, thus inducing a lower influence on the TPTD curve. Finally, these results support the use of TPTD for measuring CO in pediatric patients because this technique can be easily performed in children, unlike the pulmonary artery catheter [22].
A mean of 3 measurements is recommended by the manufacturer for determining CO [23]. However, in practice, some teams realize 2 or even only one measurement for determining CO. Our results suggest that injecting more than 2 boluses may improve the reproducibility of TPTD measurement. However, when the amount of fluid injected and the site of injection were analyzed together, the reproducibility and LSC were improved when a higher volume was injected. The injection of 15 mL of iced-cold saline is recommended by the manufacturer for each TPTD bolus. Our results showed that a higher volume of cold indicator injected provides a better precision with an improvement in the reproducibility. Indeed, a potential loss of the thermal indicator is a well-known weakness of any thermodilution technique due to thermic exchange between the indicator and the surrounding anatomic structures (vessels, valves, heart chambers, etc.). [24]. Compared with pulmonary arterial thermodilution, the thermodilution bolus in TPTD is more prone to indicator loss with regard to the longer transit time of the thermal bolus and to an increased contact surfaces in the pulmonary circulation. Increasing the amount of cold indicator volume (20 mL) are likely to better compensate for indicator loss.
The present review has a number of limitations. First, the number of studies from which we could extract the data on TPTD reproducibility remained low compared with all of the studies published using this technique. Second, patients included in different studies are not homogeneous in terms of medical and technical conditions. Third, this systematic review included studies published over the last 12 years. During this period, the PiCCO™ software has been edited twice: PiCCO™, PiCCOPlus™ and PiCCO2™ could impact the reliability of our results. Fourth, we had not included the lithium dilution technique (LiDCO Ltd, Cambridge, UK) in the present systematic review as the aim was to measure the precision of transpulmonary thermodilution method and not the precision of transpulmonary lithium-dilution technique. Fifth, the use of TPTD in pediatric patients could be tempered. Only two small studies (30 patients in all) have been included in this systematic review. Finally because our study is a systematic review without meta-analysis, we only conducted a graphical representation of results.
5 Conclusion
Our results showed that TPTD has a very good reproducibility compared to the right heart thermodilution. Moreover, the present study may suggest that the definition of a positive response to fluid infusion fixed at 15 % due to the lack of reproducibility of PAC could be redefined at only 10 % when TPTD is used. We believe that this 10 % change in CO could be statistically and clinically relevant.
Abbreviations
- CE:
-
Coefficient of error
- CO:
-
Cardiac output
- CV:
-
Coefficient of variation
- CVC:
-
Central venous catheter
- LSC:
-
Least significance change
- PAC:
-
Pulmonary arterial catheter
- SEM:
-
Standard deviation of the mean
- SD:
-
Standard deviation
- TPTD:
-
Transpulmonary thermodilution
References
Sakka SG, Reinhart K, Meier-Hellmann A. Comparison of pulmonary artery and arterial thermodilution cardiac output in critically ill patients. Intensive Care Med. 1999;25(8):843–6.
Sakka SG, Ruhl CC, Pfeiffer UJ, Beale R, McLuckie A, Reinhart K, Meier-Hellmann A. Assessment of cardiac preload and extravascular lung water by single transpulmonary thermodilution. Intensive Care Med. 2000;26(2):180–7.
Michard F, Alaya S, Zarka V, Bahloul M, Richard C, Teboul JL. Global end-diastolic volume as an indicator of cardiac preload in patients with septic shock. Chest. 2003;124(5):1900–8.
Bendjelid K, Giraud R, Siegenthaler N, Michard F. Validation of a new transpulmonary thermodilution system to assess global end-diastolic volume and extravascular lung water. Crit Care. 2010;14(6):R209. doi:10.1186/cc9332.
Combes A, Berneau JB, Luyt CE, Trouillet JL. Estimation of left ventricular systolic function by single transpulmonary thermodilution. Intensive Care Med. 2004;30(7):1377–83. doi:10.1007/s00134-004-2289-2.
Ritter S, Rudiger A, Maggiorini M. Transpulmonary thermodilution-derived cardiac function index identifies cardiac dysfunction in acute heart failure and septic patients: an observational study. Crit Care. 2009;13(4):R133. doi:10.1186/cc7994.
Kiefer N, Hofer CK, Marx G, Geisen M, Giraud R, Siegenthaler N, Hoeft A, Bendjelid K, Rex S. Clinical validation of a new thermodilution system for the assessment of cardiac output and volumetric parameters. Crit Care. 2012;16(3):R98. doi:10.1186/cc11366.
Bendjelid K, Schutz N, Suter PM, Romand JA. Continuous cardiac output monitoring after cardiopulmonary bypass: a comparison with bolus thermodilution measurement. Intensive Care Med. 2006;32(6):919–22. doi:10.1007/s00134-006-0161-2.
Monnet X, Persichini R, Ktari M, Jozwiak M, Richard C, Teboul JL. Precision of the transpulmonary thermodilution measurements. Crit Care. 2011;15(4):R204. doi:10.1186/cc10421.
Stetz CW, Miller RG, Kelly GE, Raffin TA. Reliability of the thermodilution method in the determination of cardiac output in clinical practice. Am Rev Respir Dis. 1982;126(6):1001–4.
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, Moher D, Becker BJ, Sipe TA, Thacker SB. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis of observational studies in epidemiology (MOOSE) group. JAMA. 2000;283(15):2008–12.
Sanderson S, Tatt ID, Higgins JP. Tools for assessing quality and susceptibility to bias in observational studies in epidemiology: a systematic review and annotated bibliography. Int J Epidemiol. 2007;36(3):666–76. doi:10.1093/ije/dym018.
Squara P, Cecconi M, Rhodes A, Singer M, Chiche JD. Tracking changes in cardiac output: methodological considerations for the validation of monitoring devices. Intensive Care Med. 2009;35(10):1801–8. doi:10.1007/s00134-009-1570-9.
Cecconi M, Rhodes A, Poloniecki J, Della Rocca G, Grounds RM. Bench-to-bedside review: the importance of the precision of the reference technique in method comparison studies–with specific reference to the measurement of cardiac output. Crit Care. 2009;13(1):201. doi:10.1186/cc7129.
Wood A, Bendjelid SM, Bendjelid K. Primary aortoenteric fistula: should enhanced computed tomography be considered in the diagnostic work-up? Anesth Analg. 2005;101(4):1157–9. doi:10.1213/01.ANE.0000175769.11560.FE.
Critchley LA, Lee A, Ho AM. A critical review of the ability of continuous cardiac output monitors to measure trends in cardiac output. Anesth Analg. 2010;111(5):1180–92. doi:10.1213/ANE.0b013e3181f08a5b.
Giraud R, Siegenthaler N, Bendjelid K. Transpulmonary thermodilution assessments: precise measurements require a precise procedure. Crit Care. 2011;15(5):195. doi:10.1186/cc10459.
Groeneveld AB, Berendsen RR, Schneider AJ, Pneumatikos IA, Stokkel LA, Thijs LG. Effect of the mechanical ventilatory cycle on thermodilution right ventricular volumes and cardiac output. J Appl Physiol. 2000;89(1):89–96.
Sasse SA, Chen PA, Berry RB, Sassoon CS, Mahutte CK. Variability of cardiac output over time in medical intensive care unit patients. Crit Care Med. 1994;22(2):225–32.
Lemson J, Backx AP, van Oort AM, Bouw TP, van der Hoeven JG. Extravascular lung water measurement using transpulmonary thermodilution in children. Pediatr Crit Care Med. 2009;10(2):227–33. doi:10.1097/PCC.0b013e3181937227.
Pauli C, Fakler U, Genz T, Hennig M, Lorenz HP, Hess J. Cardiac output determination in children: equivalence of the transpulmonary thermodilution method to the direct Fick principle. Intensive Care Med. 2002;28(7):947–52. doi:10.1007/s00134-002-1334-2.
Tibby S. Transpulmonary thermodilution: finally, a gold standard for pediatric cardiac output measurement. Pediatr Crit Care Med. 2008;9(3):341–2. doi:10.1097/PCC.0b013e318172ea56.
Bendjelid K, Marx G, Kiefer N, Simon TP, Geisen M, Hoeft A, Siegenthaler N, Hofer CK. Performance of a new pulse contour method for continuous cardiac output monitoring: validation in critically ill patients. Br J Anaesth. 2013;111(4):573–9. doi:10.1093/bja/aet116.
Renner LE, Morton MJ, Sakuma GY. Indicator amount, temperature, and intrinsic cardiac output affect thermodilution cardiac output accuracy and reproducibility. Crit Care Med. 1993;21(4):586–97.
Muller L, Candela D, Nyonzyma L, Mattatia L, Suehs C, Fabbro-Peray P, Louart G, de La Coussaye JE, Jaber S, Leone M, Lefrant JY. Disagreement between pulse contour analysis and transpulmonary thermodilution for cardiac output monitoring after routine therapeutic interventions in ICU patients with acute circulatory failure. Eur J Anaesthesiol. 2011;28(9):664–9. doi:10.1097/EJA.0b013e328346adda.
Goedje O, Hoeke K, Lichtwarck-Aschoff M, Faltchauser A, Lamm P, Reichart B. Continuous cardiac output by femoral arterial thermodilution calibrated pulse contour analysis: comparison with pulmonary arterial thermodilution. Crit Care Med. 1999;27(11):2407–12.
Wiesenack C, Prasser C, Keyl C, Rodig G. Assessment of intrathoracic blood volume as an indicator of cardiac preload: single transpulmonary thermodilution technique versus assessment of pressure preload parameters derived from a pulmonary artery catheter. J Cardiothorac Vasc Anesth. 2001;15(5):584–8.
Segal E, Katzenelson R, Berkenstadt H, Perel A. Transpulmonary thermodilution cardiac output measurement using the axillary artery in critically ill patients. J Clin Anesth. 2002;14(3):210–3.
Wouters PF, Quaghebeur B, Sergeant P, Van Hemelrijck J, Vandermeersch E. Cardiac output monitoring using a brachial arterial catheter during off-pump coronary artery bypass grafting. J Cardiothorac Vasc Anesth. 2005;19(2):160–4.
Ostergaard M, Nielsen J, Rasmussen JP, Berthelsen PG. Cardiac output–pulse contour analysis vs. pulmonary artery thermodilution. Acta Anaesthesiol Scand. 2006;50(9):1044–9. doi:10.1111/j.1399-6576.2006.01080.x.
Sakka SG, Kozieras J, Thuemer O, van Hout N. Measurement of cardiac output: a comparison between transpulmonary thermodilution and uncalibrated pulse contour analysis. Br J Anaesth. 2007;99(3):337–42. doi:10.1093/bja/aem177.
Hamzaoui O, Monnet X, Richard C, Osman D, Chemla D, Teboul JL. Effects of changes in vascular tone on the agreement between pulse contour and transpulmonary thermodilution cardiac output measurements within an up to 6-hour calibration-free period. Crit Care Med. 2008;36(2):434–40. doi:10.1097/01.CCM.OB013E318161FEC4.
Monnet X, Anguel N, Naudin B, Jabot J, Richard C, Teboul JL. Arterial pressure-based cardiac output in septic patients: different accuracy of pulse contour and uncalibrated pressure waveform devices. Crit Care. 2010;14(3):R109. doi:10.1186/cc9058.
Metzelder S, Coburn M, Fries M, Reinges M, Reich S, Rossaint R, Marx G, Rex S. Performance of cardiac output measurement derived from arterial pressure waveform analysis in patients requiring high-dose vasopressor therapy. Br J Anaesth. 2011;106(6):776–84. doi:10.1093/bja/aer066.
Gruenewald M, Meybohm P, Renner J, Broch O, Caliebe A, Weiler N, Steinfath M, Scholz J, Bein B. Effect of norepinephrine dosage and calibration frequency on accuracy of pulse contour-derived cardiac output. Crit Care. 2011;15(1):R22. doi:10.1186/cc9967.
Authors’ Contributions
RG participated in the design of the study, collected and analyzed the data, performed statistical analysis and drafted the manuscript. NS participated in the collection of data and helped to draft the manuscript. PM performed statistical and data analysis and helped to draft the manuscript. KB designed the study, collected the data and drafted the manuscript. All of the authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
In 2010, KB received consultant fees from Edwards Lifesciences. The other authors do not have any conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Giraud, R., Siegenthaler, N., Merlani, P. et al. Reproducibility of transpulmonary thermodilution cardiac output measurements in clinical practice: a systematic review. J Clin Monit Comput 31, 43–51 (2017). https://doi.org/10.1007/s10877-016-9823-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-016-9823-y