Abstract
Purpose
The Primary Immune Deficiency Treatment Consortium (PIDTC) enrolled children with severe combined immunodeficiency (SCID) in a prospective natural history study of hematopoietic stem cell transplant (HSCT) outcomes over the last decade. Despite newborn screening (NBS) for SCID, infections occurred prior to HSCT. This study’s objectives were to define the types and timing of infection prior to HSCT in patients diagnosed via NBS or by family history (FH) and to understand the breadth of strategies employed at PIDTC centers for infection prevention.
Methods
We analyzed retrospective data on infections and pre-transplant management in patients with SCID diagnosed by NBS and/or FH and treated with HSCT between 2010 and 2014. PIDTC centers were surveyed in 2018 to understand their practices and protocols for pre-HSCT management.
Results
Infections were more common in patients diagnosed via NBS (55%) versus those diagnosed via FH (19%) (p = 0.012). Outpatient versus inpatient management did not impact infections (47% vs 35%, respectively; p = 0.423). There was no consensus among PIDTC survey respondents as to the best setting (inpatient vs outpatient) for pre-HSCT management. While isolation practices varied, immunoglobulin replacement and antimicrobial prophylaxis were more uniformly implemented.
Conclusion
Infants with SCID diagnosed due to FH had lower rates of infection and proceeded to HSCT more quickly than did those diagnosed via NBS. Pre-HSCT management practices were highly variable between centers, although uses of prophylaxis and immunoglobulin support were more consistent. This study demonstrates a critical need for development of evidence-based guidelines for the pre-HSCT management of infants with SCID following an abnormal NBS.
Trial Registration
NCT01186913
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Severe combined immunodeficiency (SCID), characterized by profound cellular and humoral immune defects, leads to lethal infections unless treated by hematopoietic cell transplant (HSCT), gene therapy (GT), or enzyme replacement therapy (ERT) [1,2,3,4]. Newborn screening (NBS) for SCID offers the potential to identify affected infants early in life, permitting prompt definitive diagnosis and treatment [4, 5]. The Primary Immune Deficiency Treatment Consortium (PIDTC) has studied SCID since 2010, reporting outcomes for 100 SCID patients receiving HSCT between 2010 and 2014, and demonstrating in larger cohorts that infection at the time of HSCT for SCID was associated with increased mortality [6,7,8,9].
While only a minority of infants with SCID were recognized due to a positive family history (FH), the rate of diagnosis via NBS increased during the period of PIDTC study enrollment. Children with SCID diagnosed via NBS or FH, perhaps related to fewer pre-HSCT infections and medical comorbidities, had better neurologic outcomes than did children diagnosed following clinical illness [10].
However, despite lower infection rates in infants with SCID diagnosed via NBS or FH, compared with those presenting with clinical symptoms, 42% of these infants still experienced at least one infection prior to HSCT, and 27% had active infection at the time of HSCT [7]. Moreover, 76% of these infections were identified after the SCID diagnosis was confirmed [7]. We asked whether infection rates were the same in infants diagnosed by NBS versus FH and whether time to HSCT was similar between these groups. Our objective was to retrospectively analyze the types and timing of infections in our existing dataset of patients who received HSCT at PIDTC centers to better define strategies for infection prevention in a future trial. Infection data alone were insufficient to determine sources of exposure or potential approaches to prevention of infections prior to HSCT; we therefore surveyed PIDTC centers to establish the spectrum of current management practices.
Methods
Analysis of Infections in SCID Patients Diagnosed via NBS or FH
The PIDTC, a collaboration of immunology and HSCT centers in the USA and Canada, conducts research to improve the diagnosis and treatment of primary immunodeficiencies [1,2,3]. PIDTC Protocol 6901 (NCT01186913, www.clinicaltrials.gov) opened in August 2010 as a prospective, natural history study of patients diagnosed and treated for SCID, performed in accordance with the 1964 Declaration of Helsinki and its later amendments and approved by the Institutional Review Boards of individual centers and the UCSF central IRB. Of the 32 participating centers at the time of this analysis, 25 contributed SCID patients, forming the largest North American SCID cohort. Eligibility criteria included diagnosis of typical or leaky SCID based on published definitions [3], and survival to treatment with HSCT. Patients were assigned to stratum A if they were diagnosed with typical SCID and stratum B if they had leaky SCID or Omenn syndrome. Data for the 59 patients diagnosed via NBS or FH within the first 100 patients enrolled and treated with HSCT were reexamined for age at SCID diagnosis, age at infection onset, type of infection, location of pre-HSCT care (inpatient or outpatient) once SCID diagnosis was confirmed, and age at HSCT [5]. Diagnosis by family history is defined as a patient who had a relative with a diagnosis of SCID that was known prior to the patient’s delivery; testing for SCID may have been done prenatally or following delivery. Seven patients treated who received GT and/or ERT were not included, as this study focused on rates of infection prior to HSCT. Statistical analyses were primarily descriptive or univariate, with comparisons of categorical outcomes between NBS and FH groups done using Fisher’s exact test, and comparisons of quantitative variables between NBS and FH groups done using the Wilcoxon rank-sum test. Infection density, computed as infections per month, was compared between NBS and FH groups using Poisson’s regression with a log link and offset equal to the log age at HSCT. Survival was estimated using the Kaplan–Meier method and compared between groups using the log-rank test. Multivariable analyses were not done due to limited sample size.
Survey of Current Pre-HSCT Management of SCID Infants at PIDTC Centers
A 29-question survey was initially developed by 18 PIDTC clinical immunologists and transplant specialists. It was then further refined by the larger 6901 protocol working group (Data Supplement 1). The survey aimed to assess the following: patient age at time of SCID diagnosis, timing of HSCT, inpatient versus outpatient pre-HSCT management, inpatient protective environments, outpatient monitoring practices, antimicrobial prophylaxis and immunoglobulin therapies, and practices for monitoring and prevention of viral infections. The survey was completed between October 2017 and January 2018 by the 42 PIDTC centers active at that time. Centers were also asked to share any written protocols for SCID management.
Results
Infection and Survival Outcomes of the Cohort (Birth Years 2010–2014)
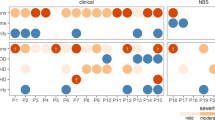
Table 1 and Fig. 1 show infections in infants diagnosed with SCID via NBS or FH. Twenty-one of the 38 infants diagnosed via NBS (55%) experienced at least one pre-HSCT infection, compared with 4 of the 21 infants diagnosed via FH (19%) (p = 0.012). The median age of diagnosis in patients diagnosed via NBS was 25 days (0–85), versus 6 days (0–32) for those diagnosed via FH (p < 0.001). Thirteen infections occurred before a diagnosis of SCID was confirmed; all but one of these occurred in patients diagnosed by NBS. To account for potential differential time to HSCT, we analyzed infection density, which was 0.3 per month in infants diagnosed via NBS compared with 0.1 per month in infants diagnosed via FH (p = 0.029). Active infection at the time of HSCT was present in 15 of 38 diagnosed via NBS (39%), but only 1 of the 21 diagnosed via FH (5%) (p = 0.005). Several infectious pathogens were seen; some infants had multiple infections (Table 1). The most common were cytomegalovirus (CMV) (n = 5), Epstein–Barr virus (EBV) (n = 2), and respiratory viruses (n = 9). The median time to HSCT was 96.5 days (range 45–251) for those diagnosed via NBS, but only 49 days (range 16–167) for those diagnosed via FH (p < 0.001). Two-year post-HSCT survival was 89% in both NBS and FH groups.
Centers were queried to determine if the 59 infants diagnosed by NBS or FH were managed as inpatients or outpatients once the SCID diagnosis was confirmed. Infants initially managed as outpatients, but later hospitalized were considered outpatients for this study. Thirty-six of the 59 infants (61%) were hospitalized continuously once the SCID diagnosis was confirmed, while 23 (39%) were managed as outpatients. The incidence of infection between inpatients (47%) compared with outpatients (35%) was not significantly different (p = 0.423; Table 1).
While none of the 21 infants diagnosed by FH was infected with CMV, 5 of the 38 (13%) diagnosed by NBS had CMV (p = 0.150), 4 inpatient and 1 initially outpatient. In 2 of these infants, CMV was detected within a month of diagnosis, suggesting that the initial exposure may have occurred prior to confirmation of the SCID diagnosis. Of the 5 patients with CMV, 3 had been breastfed, 1 had not been breastfed, and breastfeeding status was unknown for 1. Both patients with EBV viremia were managed as inpatients from the time of SCID diagnosed by NBS. Eight out of 38 infants (21%) diagnosed by NBS had a respiratory viral infections (4 inpatient and 1 initially outpatient), while 1 of 21 (5%) diagnosed by FH had a respiratory viral infection (p = 0.138). Of the respiratory viral infections, 2 were diagnosed prior to confirmation of the SCID diagnosis.
Four of the 38 infants diagnosed via NBS (11%) died after HSCT, all of whom had an active infection at the time of HSCT (Table 1). One of these infants had received no pre-HSCT conditioning, while 3 had received myeloablative conditioning (MAC). Three were initially managed as inpatients, and their causes of death included one or more infections (CMV, EBV, adenovirus, aspergillus, staphylococcal bacteremia, and rhinovirus) and hepatic veno-occlusive disease (VOD), a complication of chemotherapy, in one infant. One infant initially managed as an outpatient died of VOD.
Two of the 21 infants diagnosed via FH died post HSCT (9.5%), neither of whom experienced infection prior to HSCT. One, who did not receive pre-HSCT conditioning, had respiratory failure, while the other, who received MAC, had EBV with post-transplant lymphoproliferative disease.
Survey Results
Because the survey focused on general practice patterns, it was not correlated to patient-level data. The response rate was 100%, with 50 evaluable responses from physicians at 42 PIDTC centers, some centers’ responses from both a HSCT specialist and an immunologist. The respondents included 25 HSCT specialists, 16 immunologists, and 9 with dual expertise. Responses according to subspecialty are in Supplemental Table 1. Consensus responses for the 7 centers with > 1 respondent were solicited. Twenty-one of the 27 centers with formal supportive care protocols for SCID management submitted their protocol for analysis.
Median Age at the Time SCID Management Commenced
At the time of the survey, 91% of PIDTC centers were in states with SCID NBS programs. Based on survey responses, abnormal NBS results were reported to the specialist at a median age of 16 days from birth (range 1–28 days). Delays in reporting included local NBS program process issues and delays where only the primary care provider was notified of abnormal results and took a variable amount of time to notify specialists. Initiation of management as a SCID patient (when protective actions and prophylaxis were started) was at a median age of 21 days (range 2–30) and was based on lymphocyte subset values in most centers (38 centers, 90%). Additional testing used to determine when an infant should be managed as a SCID patient included as follows: naïve T cells, 25 centers (60%); T cell proliferation, 16 centers (38%); genetic testing, 6 centers (14%); maternal engraftment, 7 centers (17%); repeat TREC, 2 centers (5%); and negative results from testing for DiGeorge syndrome, 1 center (2%).
Timing of Transplant for all Infants with SCID
When asked for criteria regarding timing of an HSCT with no chemotherapy (“unconditioned”), 69% of centers proceeded as soon as possible. Others cited these criteria to determine timing of an unconditioned HSCT: resolution of infection (29%), return of genetic test results (24%), return of an unrelated donor search (19%), and development of infection (7%). Timing for conditioned HSCT was not assessed in this study as this may have been influenced by multiple factors including time required to find a donor and type of donor selected, time to rule out radiosensitivity, and controversy regarding how soon it is safe to use specific conditioning agents in an infant.
Outpatient Versus Inpatient Management Pre-HSCT
For 52% of centers, the pre-HSCT management location for infants with SCID varied, while 31% routinely planned for inpatient and 17% for outpatient care. Criteria to determine an appropriate home environment varied greatly between centers (Fig. 2). Centers having multiple provider responses cited their criteria inconsistently prior to providing center consensus responses. Of 11 centers that addressed disposition in their written protocol, 64% of these were consistent with their survey responses.
Inpatient Protective Environments
In addition to universal hand hygiene, 20% of centers required a 2-min scrub. Most centers required staff to wear some form of personal protective equipment (PPE) in SCID patient rooms. This included reverse isolation (gown, glove, mask) either with (n = 25) or without (n = 10) positive pressure ventilation (PPV); gown and gloves, but no mask (n = 1); and mask only (n = 2). Six centers did not indicate a requirement for staff to wear any PPE. Of 16 centers that addressed protective isolation components in their written protocol, 92% were consistent with their survey responses.
Most centers (79%) restricted non-staff caregivers to a maximum of 2 individuals; 7% reported no restrictions. There was significant variability with regard to the allowed number of non-care-providing visitors, including siblings (Fig. 3a and b).
Thirty-five (83%) centers required non-staff caregivers and visitors to perform hand hygiene prior to entering a SCID patient room. Thirteen (31%) required these individuals to wear gowns, gloves and masks.
Outpatient Monitoring
Infants managed as outpatients resided in private homes (n = 22), accommodations near the hospital (n = 1), or a communal hospital-associated facility (n = 2). When queried regarding recommended frequency for outpatient clinic follow-up of these patients, 8 indicated no formal policy, 8 recommended weekly visits, 5 twice a week, 4 every other week, and 1 monthly.
Prophylaxis and Immunoglobulin Replacement
Prophylaxis against Pneumocystis jiroveci (PJP) infection was started universally, although age at initiating trimethoprim/sulfamethoxazole (TMP-SMX) varied (Fig. 4a and b). Fungal prophylaxis was routinely started by 79% of centers with various agents (Fig. 4c). Most centers (98%) administered immunoglobulin to all patients with varied routes (Fig. 4d). Of 17 centers that addressed use of immunoglobulin and antimicrobial prophylaxis in a written protocol, 70% were consistent with their survey responses.
a PJP prophylaxis medications. *Dose varied, but generally ranged between 2.5 and 5 mg/kg/day of the trimethoprim component divided to twice a day administration and given 2–3 times a week. b Age at initiation of TMP-SMX. c Fungal prophylaxis medications. *Dose varied, but generally ranged between 3 and 6 mg/kg/day. d Route of immunoglobulin replacement
Viral Infection Testing, Surveillance, Prophylaxis, and Prevention
Viral Testing
Routine viral monitoring for CMV was performed at 93% of centers, with blood PCR the most common method. Routine monitoring for EBV and adenovirus via blood PCR was performed by 69% and 57% of centers, respectively. Respiratory virus monitoring was routine at 7% of centers. Of the 13 centers that addressed CMV screening in a written protocol, 40% were consistent with their survey responses.
CMV and Breastfeeding Recommendations
Of the 36 centers that made recommendations regarding breastfeeding, 81% recommended against breastfeeding if the mother was CMV seropositive. Of these, 45% based their recommendations on anti-CMV IgG alone, 45% considered anti-CMV IgG and/or IgM, and 3% considered anti-CMV IgM alone. Twenty-eight percent of centers recommended against breastfeeding if mothers had infectious symptoms.
Viral Prophylaxis
A minority of centers (45%) routinely started all patients on prophylaxis against herpes viruses with acyclovir and continued through HSCT. If used, 94% started either at diagnosis or within the first month of life.
Discussion
Despite diagnosis following NBS, some SCID patients enrolled in PIDTC Protocol 6901 developed respiratory viral (21%) and/or CMV (13%) infections prior to HSCT. These infections may have contributed to the surprising lack of difference in overall survival between patients diagnosed clinically compared with those diagnosed via NBS or FH noted in our recent PIDTC analysis [7]. Prior to the era of NBS, improved outcomes in patients diagnosed via FH versus as sporadic cases have been attributed to earlier institution of protective measures resulting in reduced infections, so our findings were unexpected [11]. To address this issue, we reassessed the patient data and surveyed the clinical practices of PIDTC providers regarding diagnosis and management of newborn SCID prior to HSCT.
The rate of pre-HSCT infections in patients diagnosed via NBS (55%) was nearly 3-fold higher than that of patients diagnosed based on family history (19%). Furthermore, patients diagnosed by FH received HSCT at a younger age than those diagnosed via NBS (median 49 vs 96 days). We hypothesize that awareness of genetic risk and early testing (including prenatal testing) in the setting of recognized FH prompted planning for birth to occur in a controlled environment with immediate protection from infectious exposures and rapid progression to HSCT. In addition, psychosocial factors and awareness of the importance of infection prevention may have contributed to the advantages of infants with positive FH; families who have had a prior child with SCID likely require less time to adjust to the diagnosis, are experienced in instituting isolation protocols, and may have organized the support required to manage a SCID baby prior to the child’s delivery. Notably, no CMV infections occurred in the FH cohort, possibly reflecting differences in prospective monitoring, prophylaxis, or adherence to prevention counseling from the time of birth (i.e., avoiding breastfeeding from CMV seropositive mothers, isolation from potentially infectious contacts).
In contrast, infants diagnosed via NBS in the absence of FH were not diagnosed immediately at birth; as noted in our survey, NBS results took as long as 28 days to return. Time required to obtain results of confirmatory testing resulted in further delays; some centers reported not initiating SCID management until results of T cell proliferation studies, genetics, and maternal engraftment studies have returned. These investigations can take 2 or more weeks to be reported, which may result in significant delays in initiating protective measures and commencing work-up for HSCT in infants diagnosed with NBS. Thirteen potentially preventable infections were seen in the period prior to confirmation of SCID diagnosis in our cohort. Thus, delays between birth, reporting of screening results, and referral for diagnosis and definitive therapy allowed for exposures to infections. Additionally, as we learned from the survey, some centers always awaited genetic testing results before HSCT, lengthening the window for infections to develop. Families who were not aware of SCID may have needed more counseling and time to comprehend the impact of their child’s condition, including the importance of infection prevention through isolation. Although NBS afforded an opportunity for sporadic SCID cases to have earlier diagnosis, further improvements in isolation, preventative care, and infectious screening practices, as well as shorter time to definitive therapy, should be sought so that infection risk can be further decreased.
Analysis of the early life data did not demonstrate a clear advantage for inpatient versus outpatient management prior to HSCT in terms of survival or risk of development of infections. Based on our survey of PIDTC centers, there was no consensus regarding inpatient versus outpatient management prior to transplant. Practices for inpatients commonly included hand hygiene and PPE, as well as a protective PPV environment. The clinical status of the patient and appropriateness of the home environment were important factors contributing to the decision to allow outpatient management. However, what comprised an appropriate home environment was multifactorial. The recommended frequency of follow-up while awaiting HSCT varied. Notably, there were also discrepancies between respondents from a single center, the majority of which concerned isolation of patients prior to HSCT. Standard practice documents existed at many centers, but were not always consistently followed. Further study is needed to determine which environmental factors provide best protection from infection for SCID patients diagnosed via NBS.
An area of unanimity in the SCID provider community was the routine use of antimicrobial prophylaxis and immunoglobulin for SCID patients prior to definitive therapy, although timing of initiation and dosing were not uniform.
Viral infections led to significant morbidity and mortality, even in this cohort of SCID patients diagnosed as newborns. While screening for CMV, EBV, and adenovirus was widespread, respiratory virus screening was less common in the absence of symptoms. With regard to CMV, blood-based PCR screening was routinely performed, and a majority of providers recommended against breastfeeding by CMV-seropositive mothers. A prior retrospective analysis of patients in the UK suggested that CMV disease disproportionately occurred in those who were breastfed [12]. A recent single-center study had equal cases in breastfed and non-breastfed babies, but had very low overall rates of CMV infection [13]. Further study is needed to determine what screening should be performed and at what interval, in order to best detect and treat CMV. Similarly, there is a need to determine whether interventions will be effective in preventing CMV in the pre-HSCT SCID population, and whether newer anti-CMV antivirals and cellular therapies will make treatment more successful. Based on current understanding of CMV transmission in neonates, we propose the following management strategy [14,15,16]:
-
Infants suspected of having SCID should discontinue breastfeeding and start ready-to-feed infant formula until maternal CMV status is known.
-
Infant should undergo work-up for CMV infection with blood CMV DNA PCR and urine CMV DNA PCR; and mother should undergo CMV IgG testing. While awaiting maternal CMV results consider valganciclovir for baby and advise mother to hold breastfeeding (provide breast pump and lactation consult) [17, 18].
-
If mother is CMV IgG negative, encourage resumption of breastfeeding and stop infant valganciclovir; if mother is CMV IgG positive, CMV may be shed in breastmilk, so nursing should not continue.
-
Infant CMV DNA PCR studies should be conducted weekly for a total of 4 tests; if all are negative, recommend testing every 3 weeks. If infant has any positive CMV PCR, give provide treatment with ganciclovir and/or other effective anti-CMV therapy and work with local transplant team to plan for urgent HCT.
More complete recommendations for prophylactic medications in the pre-transplant period are available in Table 2. The PIDTC 6901 study prospectively collected patient data via standardized case report forms that assessed infections at a baseline visit prior to HSCT, but did not collect details of each patient’s state SCID NBS program nor pre-HSCT management. The wide variation in management pre- and post-HSCT limited our ability to assess the impacts of these circumstances, the incidence of infections, and ultimate survival following HSCT. A planned prospective protocol tracking pre-HSCT management will assess the impact of pre-HSCT interventions on patient outcomes. Additionally, while all PIDTC centers participated in our survey of management practices, some questions were left unanswered by the participants, decreasing our statistical power. Finally, the survey did not assess factors contributing to delay of conditioned versus unconditioned HSCT specifically. For example, some centers using conditioning delayed HSCT until an infant reached a certain age, while presence of infection could have required an urgent, unconditioned HSCT.
Ideally, consistent care guidelines should be established for management of SCID prior to definitive therapy. These guidelines should address outpatient versus inpatient pre-HSCT management, use of PPE and isolation, clinical monitoring, virologic screening, and administration of immunoglobulin and antimicrobial prophylaxis. Pre- and post-implementation assessment of outcomes may help centers to fine-tune their local practices to improve patient outcomes. While suggested practices for management of SCID have been published based on single-center experiences [19,20,21,22], evidence-based practice parameters are needed to assist in establishing universal standards of care for these infants, ultimately maximizing the potential of NBS to bring all babies with SCID to definitive treatment infection-free.
Data Availability
Deidentified individual participant data will not be made available.
Change history
03 December 2020
A Correction to this paper has been published: https://doi.org/10.1007/s10875-020-00917-0
Abbreviations
- CMV:
-
Cytomegalovirus
- EBV:
-
Epstein–Barr virus
- ERT:
-
Enzyme replacement therapy
- FH:
-
Family history
- GT:
-
Gene therapy
- HSCT:
-
Hematopoietic stem cell transplant
- MAC:
-
Myeloablative conditioning
- NBS:
-
Newborn screening
- PIDTC:
-
Primary Immunodeficiency Treatment Consortium
- PPE:
-
Personal protective equipment
- PPV:
-
Positive pressure ventilation
- SCID:
-
Severe combined immunodeficiency
- VOD:
-
Veno-occlusive disease
References
Griffith LM, Cowan MJ, Kohn DB, Notarangelo LD, Puck JM, Schultz KR, et al. Allogeneic hematopoietic cell transplantation for primary immune deficiency diseases: current status and critical needs. J Allergy Clin Immunol. 2008;122(6):1087–96.
Griffith LM, Cowan MJ, Notarangelo LD, Puck JM, Buckley RH, Candotti F, et al. Improving cellular therapy for primary immune deficiency diseases: recognition, diagnosis, and management. J Allergy Clin Immunol. 2009;124(6):1152–60.
Shearer WT, Dunn E, Notarangelo LD, Dvorak CC, Puck JM, Logan BR, et al. Establishing diagnostic criteria for severe combined immunodeficiency disease (SCID), leaky SCID, and Omenn syndrome: the Primary Immune Deficiency Treatment Consortium experience. J Allergy Clin Immunol. 2014;133(4):1092–8.
Myers LA, Patel DD, Puck JM, Buckley RH. Hematopoietic stem cell transplantation for severe combined immunodeficiency in the neonatal period leads to superior thymic output and improved survival. Blood. 2002;99(3):872–8.
Kwan A, Abraham RS, Currier R, Brower A, Andruszewski K, Abbott JK, et al. Newborn screening for severe combined immunodeficiency in 11 screening programs in the United States. JAMA. 2014;312(7):729–38.
Dvorak CC, Cowan MJ, Logan BR, et al. The natural history of children with severe combined immunodeficiency: baseline features of the first fifty patients of the primary immune deficiency treatment consortium prospective study 6901. J Clin Immunol. 2013;33(7):1156–64.
Heimall J, Logan BR, Cowan MJ, Notarangelo LD, Griffith LM, Puck JM, et al. Immune reconstitution and survival of 100 SCID patients post-hematopoietic cell transplant: a PIDTC natural history study. Blood. 2017;130(25):2718–27.
Pai S-Y, Logan BR, Griffith LM, Buckley RH, Parrott RE, Dvorak CC, et al. Transplantation outcomes for severe combined immunodeficiency, 2000–2009. N Engl J Med. 2014;371(5):434–46.
Haddad E, Logan BR, Griffith LM, Buckley RH, Parrott RE, Prockop SE, et al. SCID genotype and 6-month posttransplant CD4 count predict survival and immune recovery. Blood. 2018;132(17):1737–49.
Dvorak CC, Puck JM, Wahlstrom JT, Dorsey M, Melton A, Cowan MJ. Neurologic event-free survival demonstrates a benefit for SCID patients diagnosed by newborn screening. Blood Adv. 2017;1(20):1694–8.
Brown L, Xu-Bayford J, Allwood Z, Slatter M, Cant A, Davies EG, et al. Neonatal diagnosis of severe combined immunodeficiency leads to significantly improved survival outcome: the case for newborn screening. Blood. 2011;117(11):3243–6.
Gaspar HB, Qasim W, Davies EG, Rao K, Amrolia PJ, Veys P. How I treat severe combined immunodeficiency. Blood. 2013;122(23):3749–58.
Kelty WJ, Beatty SA, Wu S, Hanson IC, Demmler-Harrison GJ, Martinez CA, et al. The role of breast-feeding in cytomegalovirus transmission and hematopoietic stem cell transplant outcomes in infants with severe combined immunodeficiency. J Allergy Clin Immunol Pract. 2019;7:2863–2865.e3.
Lanzieri TM, Dollard SC, Josephson CD, Schmid DS, Bialek SR. Breast milk–acquired cytomegalovirus infection and disease in VLBW and premature infants. Pediatrics. 2013;131(6):e1937–45.
Atkinson C, Walter S, Sharland M, Tookey P, Luck S, Peckham C, et al. Use of stored dried blood spots for retrospective diagnosis of congenital CMV. J Med Virol. 2009;81(8):1394–8.
Dergousoff BA, Vayalumkal JV, Wright NA. Survey of infection control precautions for patients with severe combined immune deficiency. J Clin Immunol. 2019;39(8):753–61.
Whitley RJ. The use of antiviral drugs during the neonatal period. Clin Perinatol. 2012;39(1):69–81.
Kimberlin DW, Jester PM, Sánchez PJ, Ahmed A, Arav-Boger R, Michaels MG, et al. Valganciclovir for symptomatic congenital cytomegalovirus disease. N Engl J Med. 2015;372(10):933–43.
Verbsky J, Thakar M, Routes J. The Wisconsin Approach to newborn screening for severe combined immunodeficiency. J Allergy Clin Immunol. 2012;129:622–7.
Kwan A, Church JA, Cowan MJ, et al. Newborn screening for SCID and T cell lymphopenia in California: results of the first 2 years. J Allergy Clin Immunol. 2013;132(1):140–50.
Dorsey MJ, Dvorak CC, Cowan MJ, Puck JM. Treatment of infants identified as having severe combined immunodeficiency by means of newborn screening. J Allergy Clin Immunol. 2017;139(3):733–42.
Amatuni GS, Currier RJ, Church JA, Bishop T, Grimbacher E, Nguyen AAC, et al. Newborn screening for severe combined immunodeficiency and T-cell lymphopenia in California, 2010-2017. Pediatrics. 2019;143(2):e20182300.
Acknowledgments
Memoriam: The authors recognize the late Dr. William T. Shearer for his contribution to this and many studies of the PIDTC. We thank Catherine Chang, Tara Bani, and Elizabeth Dunn for project management and assistance.
Authorship Contributions
• Morna Dorsey, Nicola AM Wright, Natalia S. Chaimowitz, Blachy J Dávila Saldaña, Holly Miller, Michael D. Keller, Monica S. Thakar, and Ami J. Shah, MD, designed data collection instruments, collected data, carried out the initial analyses, drafted the initial manuscript, and reviewed and revised the final manuscript.
• Morton J Cowan, Rebecca H. Buckley, Christopher C. Dvorak, Elie Haddad, Donald B. Kohn, Luigi D. Notarangelo, Sung Yun Pai, Jennifer Puck, MD, Michael Pulsipher, and Jennifer Heimall conceptualized and designed the study, designed the data collection instruments, collected data, carried out the initial analyses, drafted the initial manuscript, and reviewed and revised the manuscript.
• Linda Griffith and Brent Logan conceptualized and designed the study, designed the data collection instruments, carried out data analyses, and reviewed and revised the manuscript.
• Rolla Abu-Arja, Jeffrey Andolina, Victor Aquino, JL Barnum, Jeffrey J. Bednarski, Monica Bhatia, Francisco A. Bonilla, Manish J. Butte, Nancy J Bunin, Sharat Chandra, Sonali Chaudhury, Karin Chen, Hey Chong, Geoff Cuvelier, MD, FRCPC, Jignesh Dalal, Magee L. DeFelice, Kenneth B. DeSantes, Lisa R Forbes, Alfred Gillio, Fred Goldman, Avni Y Joshi, Neena Kapoor, MD, Alan P. Knutsen, MD, Lisa Kobrynski, Jay A Lieberman, Jennifer W Leiding, Benjamin Oshrine, Kiran P. Patel, Susan Prockop, Troy C. Quigg, Ralph Quinones, Kirk R. Schultz, Christine Seroogy, David Shyr, Subhadra Siegel, Angela R. Smith, Troy R. Torgerson, Mark T. Vander Lugt, and Lolie C Yu collected data and critically reviewed the manuscript for important intellectual content.
• All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Funding
This work was supported by the Division of Allergy, Immunology and Transplantation, National Institute of Allergy and Infectious Diseases (NIAID); and the Office of Rare Diseases Research (ORDR), National Center for Advancing Translational Sciences (NCATS), National Institutes of Health (NIH), Bethesda, MD; Public Health Service grant/cooperative agreements U54-AI082973 (PIs: MJ Cowan; September 2019 forward JM Puck and DB Kohn), U54-NS064808 and U01-TR001263 (PI: JP Krischer), R13-AI094943 (PIs: MJ Cowan; March 2018 forward JM Puck), and the Division of Intramural Research, NIAID, NIH. LD Notarangelo is supported by the Division of Intramural Research, NIAID, NIH. The PIDTC is a part of the Rare Diseases Clinical Research Network (RDCRN) of ORDR, NCATS. Collaborative work of the PIDTC with the Pediatric Blood and Marrow Transplant Consortium (PBMTC) is supported by the U54 grants above along with support of the PBMTC Operations Center by grant/cooperative agreement U10HL069254 (PI: MA Pulsipher, NHLBI/NCI) and a Johnny Cristopher Children’s Charitable Foundation St. Baldrick’s Consortium Grant. Collaborative work of the PIDTC with the Center for International Blood and Marrow Transplant Research (CIBMTR) is supported by grant/cooperative agreement U24-CA76518 (PI: MM Horowitz) from the National Cancer Institute (NCI), NHLBI, and NIAID, NIH; and grant/cooperative agreement U01HL069294 from the NHLBI and NCI; contract HHSH250201200016C and HHSH234200637015C with the Health Resources and Services Administration (HRSA/DHHS); and grants N00014-13-1-0039 and N00014-14-1-0028 from the Office of Naval Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Disclaimer
The content and opinions expressed are solely the responsibility of the authors and do not represent the official policy or position of the NIAID, ORDR, NCATS, NIH, HRSA, or any other agency of the US government.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dorsey, M.J., Wright, N.A.M., Chaimowitz, N.S. et al. Infections in Infants with SCID: Isolation, Infection Screening, and Prophylaxis in PIDTC Centers. J Clin Immunol 41, 38–50 (2021). https://doi.org/10.1007/s10875-020-00865-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-020-00865-9