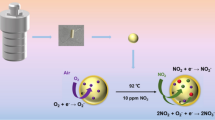
Abstract
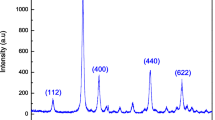
In this work we are presenting the hydrothermal method to synthesize NiTiO3 perovskite nanoparticles (NPs). The effect of variation in reaction temperature on the structural, optical, electrical and gas sensing properties of NiTiO3 nanoparticles was investigated. The nanoparticles synthesized at different reaction temperatures were characterized by various characterization methods like XRD, FTIR, UV–Visible Spectroscopy, FESEM, TEM, HRTEM and SAED. The results of UV–Visible analysis revealed that band gap of NiTiO3 decreased from 2.90 to 2.56 eV on increase in reaction temperature from 140 to 200 °C. The XRD analysis showed that crystallite size decreased in the range of 21 to 12 nm on increase in reaction temperature. The various parameters of the material like dislocation density, microstrain and crystallinity were also calculated from XRD data. The average particle size was estimated by FESEM analysis and found to be decreased on increase in reaction temperature. FTIR analysis confirmed the formation of NiTiO3. Study of electrical properties proved the semiconducting behaviour of NiTiO3. The detail analysis of NiTiO3 sensor characteristics in terms of sensitivity, selectivity, response and recovery time was carried out. The study of gas sensing performance of NiTiO3 revealed that NiTiO3 synthesized at 140 °C showed maximum sensitivity to CO2 gas at room temperature, whereas NiTiO3 synthesized at 200 °C showed maximum sensitivity to H2S gas at 250 °C.














Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
M.A. Ruiz-Preciado, A. Kassiba, A. Gibaud, A. Morales-Acevedo, Comparison of nickel titanate (NiTiO3) powders synthesized by sol-gel and solid state reaction. Mater. Sci. Semicond. Process. 37, 171–178 (2015). https://doi.org/10.1016/j.mssp.2015.02.063
T. Li, C.C. Wang, C.M. Lei, X.H. Sun, G.J. Wang, L.N. Liu, Conductivity relaxation in NiTiO3 at high temperatures. Curr. Appl. Phys. 13(8), 1728–1731 (2013). https://doi.org/10.1016/j.cap.2013.07.002
N. Dharmaraj, H.C. Park, C.K. Kim, H.Y. Kim, D.R. Lee, Nickel titanate nanofibers by electrospinning. Mater. Chem. Phys. 87(1), 5–9 (2004). https://doi.org/10.1016/j.matchemphys.2004.05.005
O. Yamamoto, Y. Takeda, R. Kanno, M. Noda, perovskite-type oxides as oxygen electrodes for high temperature oxide fuel cells. Solid State Ionics 22, 241–246 (1986)
D.D. Kajale, V.B. Gaikwad, S.D. Shinde, D.N. Chavan, G.E. Patil, Effect of surface modification on SrTiO3 thick films: room temperature H2S Gas sensor. Sens. Transducers 137(2), 10–21 (2012)
J.W. Fergus, Perovskite oxides for semiconductor-based gas sensors. Sens. Actuators, B Chem. 123(2), 1169–1179 (2007). https://doi.org/10.1016/j.snb.2006.10.051
M. Singh, B.C. Yadav, A. Ranjan, M. Kaur, S.K. Gupta, Synthesis and characterization of perovskite barium titanate thin film and its application as LPG sensor. Sens. Actuators, B Chem. 241, 1170–1178 (2017). https://doi.org/10.1016/j.snb.2016.10.018
F. Zaza, G. Orio, E. Serra, F. Caprioli, M. Pasquali, Low-Temperature capacitive sensor based on perovskite oxides. AIP Conference Proceedings 1667, (2015). https://doi.org/10.1063/1.4922560
P.M. Bulemo, I.D. Kim, Recent advances in ABO3 perovskites: their gas-sensing performance as resistive-type gas sensors. J. Korean Ceram. Soc. 57(1), 24–39 (2020). https://doi.org/10.1007/s43207-019-00003-1
B. Szafraniak, Ł Fuśnik, J. Xu, F. Gao, A. Brudnik, A. Rydosz, Semiconducting metal oxides: SrTiO3, BaTiO3 and BaSrTiO3 in gas-sensing applications: a review. Coatings 11(2), 1–23 (2021). https://doi.org/10.3390/coatings11020185
K.S. Beenakumari, Synthesis and characterisation of nano-sized nickel titanate photocatalyst. J. Exp. Nanosci. 8(2), 203–209 (2013). https://doi.org/10.1080/17458080.2011.566631
H. Khojasteh, M. Salavati-Niasari, S. Mortazavi-Derazkola, Synthesis, characterization and photocatalytic properties of nickel-doped TiO2 and nickel titanate nanoparticles. J. Mater. Sci. Mater. Electron. 27(4), 3599–3607 (2016). https://doi.org/10.1007/s10854-015-4197-3
D.J. Taylor, P.F. Fleig, R.A. Page, Characterization of nickel titanate synthesized by sol-gel processing. Thin Solid Films 408(1–2), 104–110 (2002). https://doi.org/10.1016/S0040-6090(02)00143-8
A.R. Phani, S. Santucci, Structural characterization of nickel titanium oxide synthesized by sol-gel spin coating technique. Thin Solid Films 396(1–2), 1–4 (2001). https://doi.org/10.1016/S0040-6090(01)01131-2
S. Chuang, M. Hsieha, D. Wang, Structure and dielectric properties of NiTiO3 powders synthesized by the modified sol–gel method. J. Chin. Chem. Soc. 59(5), 628–632 (2012). https://doi.org/10.1002/jccs.201100496
V. Gupta, K.K. Bamzai, P.N. Kotru, B.M. Wanklyn, Mechanical characteristics of flux-grown calcium titanate and nickel titanate crystals. Mater. Chem. Phys. 89(1), 64–71 (2005). https://doi.org/10.1016/j.matchemphys.2004.08.027
A.V. Murugan, V. Samuel, S.C. Navale, V. Ravi, Phase evolution of NiTiO3 prepared by coprecipitation method. Mater. Lett. 60(15), 1791–1792 (2006). https://doi.org/10.1016/j.matlet.2005.12.023
T.T. Pham, S.G. Kang, E.W. Shin, Optical and structural properties of Mo-doped NiTiO3 materials synthesized via modified Pechini methods. Appl. Surf. Sci. 411, 18–26 (2017). https://doi.org/10.1016/j.apsusc.2017.03.123
P. Yuan, C. Fan, G. Ding, Y. Wang, X. Zhang, Preparation and photocatalytic properties of ilmenite NiTiO3 powders for degradation of humic acid in water. Int. J. Miner. Metall. Mater. 19(4), 372–376 (2012). https://doi.org/10.1007/s12613-012-0566-6
N. Kaur, M. Singh, A. Moumen, G. Duina, E. Comini, 1D titanium dioxide: achievements in chemical sensing. Materials 13(13), 1–21 (2020). https://doi.org/10.3390/ma13132974
B. Zhao, L. Lin, D. He, Phase and morphological transitions of titania/titanate nanostructures from an acid to an alkali hydrothermal environment. J. Mater. Chem. A 1(5), 1659–1668 (2013). https://doi.org/10.1039/c2ta00755j
J. Casanova-Chafer, R. Garcia-Aboal, P. Atienzar, E. Llobet, Unraveling the gas-sensing mechanisms of lead-free perovskites supported on graphene. ACS Sens. 7(12), 3753–3763 (2022). https://doi.org/10.1021/acssensors.2c01581
G.J. Mogal, D.V. Ahire, G.E. Patil, G.H. Jain Jain, V.B. Gaikwad, Hydrothermal synthesis of TiO2 nanorods using TiCl3 and its gas sensing properties. Sci. Technol. Eng. 3(11), 123–130 (2014)
R.P. Patil, P.V. More, G.H. Jain, P.K. Khanna, V.B. Gaikwad, BaTiO3 nanostructures for H2S gas sensor: Influence of band-gap, size and shape on sensing mechanism. Vacuum 146, 455–461 (2017). https://doi.org/10.1016/j.vacuum.2017.08.008
S.D. Shinde, V.B. Gaikwad, G.E. Patil, D.D. Kajale, G.H. Jain, Gas sensing performance of nanostructured ZnO thick film resistors. Int. J. Nanoparticles 5(2), 126–135 (2012). https://doi.org/10.1504/IJNP.2012.046239
M.K. Deore, V.B. Gaikwad, R.M. Chaudhari, N.U. Patil, P.D. Hire, S.B. Deshmukh, G.E. Patil, V.G. Wagh, G.H. Jain, Formulation, Characterization and LPG-sensing properties of CuO-doped ZnO thick film resistor. Adv. Sens. Technol. (2013). https://doi.org/10.1007/978-3-642-32180-1_16
M. Shellaiah, K.W. Sun, Review on sensing applications of perovskite nanomaterials. Chemosensors (2020). https://doi.org/10.3390/chemosensors8030055
G.H. Jain, V.B. Gaikwad, D.D. Kajale, R.M. Chaudhari, R.L. Patil, N.K. Pawar, M.K. Deore, S.D. Shinde, L.A. Patil, Gas sensing performance of pure and modified BST thick film resistor. Sens. Transducers 90, 160–173 (2008)
D.N. Chavan, G.E. Patil, D.D. Kajale, V.B. Gaikwad, P.K. Khanna, G.H. Jain, Nano Ag-doped In2O3 thick film: a low-temperature H2S gas sensor. J. Sens. (2011). https://doi.org/10.1155/2011/824215
A. Habib, R. Haubner, N. Stelzer, Effect of temperature, time and particle size of Ti precursor on hydrothermal synthesis of barium titanate. Mater. Sci. Eng. B 152(1–3), 60–65 (2008). https://doi.org/10.1016/j.mseb.2008.06.018
S. García-García, S. Wold, M. Jonsson, Effects of temperature on the stability of colloidal montmorillonite particles at different pH and ionic strength. Appl. Clay Sci. 43(1), 21–26 (2009). https://doi.org/10.1016/j.clay.2008.07.011
A. Noviyanto, R. Amalia, P.Y.D. Maulida, M. Dioktyanto, B.H. Arrosyid, D. Aryanto, L. Zhang, A.T.S. Wee, N. Arramel, Anomalous temperature-induced particle size reduction in manganese oxide nanoparticles. ACS Omega 8(47), 45152–45162 (2023). https://doi.org/10.1021/acsomega.3c08012
N.R. Yogamalar, A.C. Bose, Burstein-Moss shift and room temperature near-band-edge luminescence in lithium-doped zinc oxide. Appl. Phys. A Mater. Sci. Process. 103(1), 33–42 (2011). https://doi.org/10.1007/s00339-011-6304-5
Y.G. Wang, S.P. Lau, H.W. Lee, S.F. Yu, B.K. Tay, X.H. Zhang, H.H. Hng, Photoluminescence study of ZnO films prepared by thermal oxidation of Zn metallic films in air. J. Appl. Phys. 94(1), 354–358 (2003). https://doi.org/10.1063/1.1577819
S. Dutta, S. Chattopadhyay, M. Sutradhar, A. Sarkar, M. Chakrabarti, D. Sanyal, D. Jana, Defects and the optical absorption in nanocrystalline ZnO. J. Phys. Condens. Matter 19(23), 236218 (2007). https://doi.org/10.1088/0953-8984/19/23/236218
Q. Wang, M. Brier, S. Joshi, A. Puntambekar, V. Chakrapani, Defect-induced Burstein-Moss shift in reduced V2O5 nanostructures. Phys. Rev. B (2016). https://doi.org/10.1103/PhysRevB.94.245305
S. Mohan, M. Vellakkat, A. Aravind, U. Reka, Hydrothermal synthesis and characterization of zinc oxide nanoparticles of various shapes under different reaction conditions. Nano Express (2020). https://doi.org/10.1088/2632-959X/abc813
R.S. Singh, T.H. Ansari, R.A. Singh, B.M. Wanklyn, Electrical conduction in NiTiO3 single crystals. Mater. Chem. Phys. 40(3), 173–177 (1995). https://doi.org/10.1016/0254-0584(95)01478-0
L.G.J. de Haart, A.J. de Vries, G. Blasse, Photoelectrochemical properties of MgTiO3 and other Titanates with the ilmenite structure. Mat. Res. Bull. 19(7), 817–824 (1984). https://doi.org/10.1016/0025-5408(84)90042-4
Y.J. Lin, Y.H. Chang, W.D. Yang, B.S. Tsai, Synthesis and characterization of ilmenite NiTiO3 and CoTiO3 prepared by a modified Pechini method. J. Non-Cryst. Solids 352(8), 789–794 (2006). https://doi.org/10.1016/j.jnoncrysol.2006.02.001
M. Rahimi-Nasrabadi, F. Ahmadi, M. Eghbali-Arani, Novel route to synthesize nanocrystalline nickel titanate in the presence of amino acids as a capping agent. J. Mater. Sci. Mater. Electron. 27(11), 11873–11878 (2016). https://doi.org/10.1007/s10854-016-5331-6
T. Acharya, R.N.P. Choudhary, Structural, ferroelectric and electrical properties of NiTiO3 ceramic. J. Electron. Mater. 44(1), 271–280 (2015). https://doi.org/10.1007/s11664-014-3426-5
M.W. Li, J.P. Yuan, X.M. Gao, E.Q. Liang, C.Y. Wang, Structure and optical absorption properties of NiTiO3 nanocrystallites. Appl. Phys. A Mater. Sci. Process. (2016). https://doi.org/10.1007/s00339-016-0259-5
S. Karmakar, A.K. Manna, S. Varma, D. Behera, Investigation of optical, electrical and magnetic properties of hexagonal NiTiO3 nanoparticles prepared via ultrasonic dispersion techniques for high power applications. Mater. Res. Express (2018). https://doi.org/10.1088/2053-1591/aac404
G. Nagaraju, Udayabhanu, Shivaraj, S.A. Prashanth, M. Shastri, K.V. Yathish, C. Anupama, D. Rangappa, Electrochemical heavy metal detection, photocatalytic, photoluminescence, biodiesel production and antibacterial activities of Ag–ZnO nanomaterial. Mater. Res. Bull. 94, 54–63 (2017). https://doi.org/10.1016/j.materresbull.2017.05.043
M. El-Kemary, N. Nagy, I. El-Mehasseb, Nickel oxide nanoparticles: synthesis and spectral studies of interactions with glucose. Mater. Sci. Semicond. Process. 16(6), 1747–1752 (2013). https://doi.org/10.1016/j.mssp.2013.05.018
E.A. Al-Oubidy, F.J. Kadhim, Photocatalytic activity of anatase titanium dioxide nanostructures prepared by reactive magnetron sputtering technique. Opt. Quant. Electron. (2019). https://doi.org/10.1007/s11082-018-1738-z
S. Yuvaraj, V.D. Nithya, K.S. Fathima, C. Sanjeeviraja, G.K. Selvan, S. Arumugam, R.K. Selvan, Investigations on the temperature dependent electrical and magnetic properties of NiTiO3 by molten salt synthesis. Mater. Res. Bull. 48(3), 1110–1116 (2013). https://doi.org/10.1016/j.materresbull.2012.12.001
R. Vijayalakshmi, V. Rajendran, Effect of reaction temperature on size and optical properties of NiTiO3 nanoparticles. E-J. Chem. 9(1), 282–288 (2012). https://doi.org/10.1155/2012/607289
J. Menashi, R. C. Reid, L.Wagner, Doped barium titanate based compositions. U.S. Patent 4863883 (1989).
K. Suematsu, M. Yuasa, T. Kida, N. Yamazoe, K. Shimanoe, Determination of oxygen adsorption species on SnO2: exact analysis of gas sensing properties using a sample gas pretreatment system. J. Electrochem. Soc. 161(6), 123–128 (2014). https://doi.org/10.1149/2.004406jes
R. Bhusari, J.S. Thomann, J. Guillot, R. Leturcq, Oxygen adsorption and desorption kinetics in CuO nanowire bundle networks: implications for MOx-based gas sensors. ACS Appl. Nano Mater. 5, 10248–10257 (2022). https://doi.org/10.1021/acsanm.2c01245
F.J. Meng, R.F. Xin, S.X. Li, Metal oxide heterostructures for improving gas sensing properties: a review. Materials (2022). https://doi.org/10.3390/ma16010263
C. Balamurugan, D.-W. Lee, Perovskite hexagonal YMnO3 nanopowder as p-type semiconductor gas sensor for H2S detection. Sens. Actuators B Chem. 221, 857–866 (2015). https://doi.org/10.1016/j.snb.2015.07.018
X. Li, L. Zhang, N. Luo, J. Chen, J. Cheng, W. Ren, J. Xu, Enhanced H2S sensing performance of BiFeO3 based MEMS gas sensor with corona poling. Sens. Actuators B: Chem. (2022). https://doi.org/10.1016/j.snb.2022.131477
A. Marikutsa, A.A. Dobrovolskii, M.N. Rumyantseva, A.A. Mikhaylov, A.G. Medvedev, O. Lev, P.V. Prichodchenko, Improved H2S sensitivity of nanosized BaSnO3 obtained by hydrogen peroxide assisted sol-gel processing. J. Alloys Compd. (2023). https://doi.org/10.1016/j.jallcom.2023.169141
H. Shan, W. Xuan, Z. Li, D. Hu, X. Gu, S. Huang, Room-temperature hydrogen sulfide sensor based on tributyltin oxide functionalized perovskite CsPbBr3 quantum dots. ACS Appl. Nano Mater. 5(5), 6801–6809 (2022). https://doi.org/10.1021/acsanm.2c00791
D.D. Kajale, G.E. Patil, V.B. Gaikwad, S.D. Shinde, D.N. Chavan, N.K. Pawar, S.R. Shirsath, G.H. Jain, Synthesis of SrTiO3 nanopowder by sol-gel-hydrothemal method for gas sensing application. Int. J. Smart Sens. Intell. Syst. 5(2), 382–400 (2012). https://doi.org/10.21307/ijssis-2017-487
G.H. Jain, L.A. Patil, P.P. Patil, U.P. Mulik, K.R. Patil, Studies on gas sensing performance of pure and modified barium strontium titanate thick film resistors. Bull. Mater. Sci. 30(1), 9–17 (2007). https://doi.org/10.1007/s12034-007-0003-z
W. Zhang, C. Xie, G. Zhang, J. Zhang, S. Zhang, D. Zeng, Porous LaFeO3/SnO2 nanocomposite film for CO2 detection with high sensitivity. Mater. Chem. Phys. 186, 228–236 (2017). https://doi.org/10.1016/j.matchemphys.2016.10.048
S.F.H. Karouei, H.M. Moghaddam, P-p heterojunction of polymer/hierarchical mesoporous LaFeO3 microsphere as CO2 gas sensing under high humidity. Appl. Surf. Sci. 479, 1029–1038 (2019). https://doi.org/10.1016/j.apsusc.2019.02.099
S. Joshi, S.J. Ippolito, S.R. Periasamy, Y.M. Sabri, M.V. Sunkara, Efficient heterostructures of Ag@CuO/BaTiO3 for low-temperature CO2 gas detection: assessing the role of nanointerfaces during sensing by operando DRIFTS technique. ACS Appl. Mater. Interfaces 9(32), 27014–27026 (2017). https://doi.org/10.1021/acsami.7b07051
K. Fan, H. Qin, L. Wang, L. Ju, J. Hu, CO2 gas sensors based on La1-xSrxFeO3 nanocrystalline powders. Sens. Actuators B Chem. 177, 265–269 (2013). https://doi.org/10.1016/j.snb.2012.11.004
X. Wang, H. Qin, L. Sun, J. Hu, CO2 sensing properties and mechanism of nanocrystalline LaFeO3 sensor. Sens. Actuators, B Chem. 188, 965–971 (2013). https://doi.org/10.1016/j.snb.2013.07.100
G.E. Patil, D.D. Kajale, V.B. Gaikwad, G.H. Jain, Effect of thickness on nanostructured SnO2 thin films by spray pyrolysis as highly sensitive H2S gas sensor. J. Nanosci. Nanotechnol. 12(8), 6192–6201 (2012). https://doi.org/10.1166/jnn.2012.6424
G.E. Patil, D.D. Kajale, V.B. Gaikwad, G.H. Jain, Effect of precursor concentration on gas sensing performance of nanocrystalline SnO2 thin films. J. Nanoeng. Nanomanuf. 2(3), 315–323 (2012). https://doi.org/10.1166/jnan.2012.1090
P. Shankar, J.B.B. Rayappan, Gas sensing mechanism of metal oxides: the role of ambient atmosphere, type of semiconductor and gases-A review. Sci. Lett. J. 4, 126 (2015)
G.E. Patil, D.D. Kajale, D.N. Chavan, N.K. Pawar, P.T. Ahire, S.D. Shinde, V.B. Gaikwad, G.H. Jain, Synthesis, characterization and gas sensing performance of SnO2 thin films prepared by spray pyrolysis. Bull. Mater. Sci. 34(1), 1–9 (2011). https://doi.org/10.1007/s12034-011-0045-0
Acknowledgements
Authors are very grateful to the Principal of SNJB’s KKHA Arts, SMGL Commerce and SPHJ Science College, Chandwad for providing laboratory facilities. The authors are also thankful to the C.I.F of SNJB College and C.I.F of SPPU, Pune for providing the characterization facilities. The authors are also thankful to DST, New Delhi for providing fund for research infrastructure under DST-FIST (Project No. SR/FIST-415/2018). One of the authors (MAM) is very grateful to the MAHAJYOTI Institute, Nagpur for providing Fellowship for the Doctoral Research.
Funding
The authors declare that no funds, grants or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Manoj A. More: Conceptualization, Methodology, Data curation, Validation, Experiments, Formal analysis, Investigation, Writing original draft. Swapnil A. More: Validation, Data curation. Matthew D. Femi: Validation, Data curation. Gotan H. Jain: Conceptualization, supervision. Sarika D. Shinde: Validation, Formal analysis. Dnyaneshwari Y. Patil: Validation, Formal analysis. Dnyaneshwar D. Kajale: Validation, Formal analysis. Ganesh E. Patil: Conceptualization, Supervision, Validation, Formal analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no relevant financial or non-financial conflict of interests.
Ethical approval
This paper meets the ethical standards of this journal.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
More, M.A., More, S.A., Femi, M.D. et al. Hydrothermally synthesized nanostructured NiTiO3 thick films for H2S and room temperature CO2 gas sensing. J Mater Sci: Mater Electron 35, 1706 (2024). https://doi.org/10.1007/s10854-024-13429-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10854-024-13429-z