Abstract
Alloys with melting points < 150 °C are required for the development of flexible consumer devices. While the eutectic In–48Sn alloy is a promising candidate for these applications, its low tensile strength and low creep resistance during solid-state aging are of concern. The addition of Ag can address this issue to some extent; however, the effect of added Ag on the properties of the alloy is not well understood. Here, we studied the effects of added xAg (x = 0.5, 1.0, 1.5 wt.%) on the fusion start temperature, microstructure, and mechanical properties of the In–Sn eutectic alloy and found that the fusion start temperature of the In–48Sn–xAg alloy was reduced to around 113 °C due to the ternary eutectic reaction of In–Sn–Ag, in which ε-AgIn2 only formed in the In–48Sn–xAg alloys. In addition, smaller β-In3Sn and γ-InSn4 phases were produced through the formation of ε-AgIn2, which affected the mechanical properties of the alloy. In–48Sn–1.5Ag, with the smallest grains, exhibited the highest tensile strength of 12.5 MPa via boundary strengthening. In–48Sn–1.5Ag fractured in transgranular mode, which is different from the intergranular fracturing of the eutectic alloy with relatively large grains. Conversely, In–48Sn–0.5Ag, with the highest soft-β-In3Sn/hard-γ-InSn4 ratio, showed the longest elongation of 64%, which is twice that of the eutectic In–48Sn alloy.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Low-temperature alloys with melting points less than 150 °C are receiving increasing attention for the development of lighter and more cost-effective portable consumer devices with polymer substrates [1,2,3]. Eutectic Sn–58Bi- and Sn–Bi-based alloys, with high tensile strength and suitable melting points, are commonly studied as low-temperature alloys [4,5,6,7,8]; however, they all exhibit brittleness after thermal aging because of the coarsened bismuth phase, which is a disadvantage [9]. In addition, their operating temperatures exceed 160 °C [6, 7]. On the other hand, common flexible organic substrates, such as heat-stabilized polyethylene terephthalate (HS PET) and polyethylene naphthalate (PEN), require operating temperatures below 150 °C [10]; hence, these Sn–Bi alloys cannot be used with flexible organic substrates.
The eutectic In–48Sn alloy, with a melting point of 118 °C and good ductility and wetting behavior, is the most promising candidate for the above-mentioned low-temperature applications [11,12,13]. Nevertheless, low tensile strength and low creep resistance are critical concerns in an In–Sn-based alloy during solid-state aging [14, 15]. Some studies aimed at improving the mechanical properties of the In–Sn-based alloy by the addition of trace elements (Zn, Bi, and Ag) have been reported [16,17,18]. Among these, Sn–In alloys with added Ag not only exhibit enhanced elongation and creep resistance [15, 18, 19], but their lower electrical resistance has also been investigated [18]. However, these studies showed that amounts of Ag > 2.5% result in higher melting temperatures. Moreover, the effect of the amount of Ag in the eutectic In–Sn alloy on the tensile strength and melting point is important but is not yet well understood. To address this deficiency, in this study we investigated the fusion start temperatures, microstructures, and mechanical properties of In–48Sn–xAg (ISA) alloys in which x = 0.5, 1.0, and 1.5 wt.%.
Materials and methods
Figure 1 presents a schematic of the experimental procedure. We fabricated ISA alloys consisting of commercial In–48Sn eutectic and 0.5, 1.0, and 1.5 wt.% Ag from pure silver wire (99.99%; Nilaco Corporation, Japan). These elements were alloyed at 700 °C for 5 h in an alumina crucible inside a furnace in a nitrogen atmosphere. The composition of the new alloys was analyzed as detailed below, the results of which are summarized in Table 1. Alloy bars (50 × 10 × 5 mm) were shaped using a casting mold, and then further shaped into dumbbells by wire electrical discharge machining (Sodick AG 360L, Japan). The dimensions of the dumbbells are shown in Fig. 1. Tensile testing was conducted under a strain rate of 5 × 10−4 s−1 using a universal tensile machine (Autograph AG–X, Shimadzu, Japan). The tensile elongation and ultimate tensile strength (UTS) were determined from tensile stress–strain curves. The fracture surface of each tensile specimen was examined by scanning electron microscopy (SEM; SU-70, Hitachi, Japan).
The melting behavior of each ISA alloy was examined by differential scanning calorimetry (DSC; 7020 clinical analyzer, Hitachi, Japan) under a constant flow of N2 in the range of 50–250 °C at a rate of approximately 10 °C/min. The fusion start temperature (Tf) was determined as required by the JIS Z 3198-1 standard. The microstructure of the ISAs was examined after grinding with SiC grit paper in the order #800, #1200, and #2000, and then successive polishing with 1.0 and 0.3 µm alumina suspension, respectively (Buehler, USA). The crystal structure of the ISA alloys was determined by X-ray diffraction (XRD; Ultima IV, Rigaku, Japan) in a range of 30°–80° 2θ at a scan rate of 1°/min. The microstructure and the elemental distribution of the ISAs was determined by SEM and field-emission electron-probe microanalysis (FE-EPMA; JXA-8530F JEOL). ImageJ software was used to calculate phase size and number.
Results and discussion
Melting behavior in relation to alloy phase constitution and microstructure
The DSC curves for all alloys are shown in Fig. 2a, from which the fusion start temperatures for the In–48Sn–0.5Ag (ISA4805), In–48Sn–1.0Ag (ISA4810), and In–48Sn–1.5Ag (ISA 4815) alloys were determined to be 112.6, 112.7, and 113.2 °C, respectively, which are all lower than that of the eutectic In–48Sn alloy (117.2 °C). The addition of Ag slightly decreases the fusion start temperature of eutectic In–48Sn, which is ascribable to the ternary eutectic reaction, and was calculated by Ohnuma et al. to be 114 °C for Sn–52.2In–0.9Ag (Eq. (1)). The melting temperature of Sn-51.7In with 1.0–5.0 wt.% added Ag was predicted to increase with increasing silver content [20], in agreement with the results in this study.
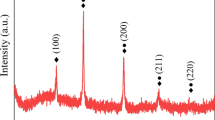
The XRD patterns for all alloys are displayed in Fig. 2b, which reveals the presence of ε-AgIn2 in the new ISA alloys, as observed in other In–Sn–Ag alloys [19]. Although few studies have reported the presence of the Ag2In intermetallic compound (IMC) in ISA alloys [19, 21], Chuang et al. [22] reported that Ag2In and AgIn2 compete in In–49Sn/Ag alloyed specimens during aging. At temperatures under 75 °C, the dominant reaction is:
Hence, the Ag-In IMC is transformed from Ag2In to AgIn2, according to Eq. (2), in ISA alloys during slow cooling. In addition, EPMA maps of the alloys are shown in Fig. 3, with EPMA point analyses providing the elemental content of all phases, as summarized in Table 2. AgIn2, which is circular in shape, exists in the In3Sn phase in the ISA alloys because the In-rich phase is required for transformation to occur, according to Eq. (2). The diameter of the AgIn2 phase increases with the addition of more Ag, from 0.5–2.0 μm in the ISA 4805 and 4810 alloys to 2.0–4.0 μm in the ISA 4815 alloy, leading to a significant reduction in the γ-InSn4 size. The microstructures of the eutectic In–48Sn and ISA alloys are displayed in Fig. 4a–d, which shows that the In-rich β-In3Sn appeared as dark regions, while the light areas correspond to γ-InSn4 [14, 19]. We measured the area and number of phases, as summarized in Fig. 4e–g, which shows that the β-In3Sn + ε-AgIn2 phases in ISA4815 (135 μm2) are smaller than β-In3Sn in eutectic In–48Sn (270 μm2). Moreover, the γ-InSn4 phase in eutectic In–48Sn is, on average, 215 μm2 in size; ISA4805 and ISA4810 show similar γ-phases that are approximately 120 μm2 in size, while the average γ-InSn4 phase in ISA4815 was found to be 300% smaller (65 μm2), as shown in Fig. 4f. The β-In3Sn/γ–InSn4 ratio and the total number of phases in each alloy are displayed in Fig. 4g. The size of grains or phases in alloys is usually determined by the number of nucleation sites during solidification. When Ag was added to the eutectic In–48Sn alloy, the ε-AgIn2 formed along the phase boundaries created additional sites for nucleation during cooling. Therefore, the addition of Ag affects the size of the phases, and its effects on the mechanical properties of the ISA alloys need to be considered.
Mechanical properties of alloys
The tensile strength and elongation of the alloys are presented in Fig. 5, which reveals that eutectic In–48Sn and ISA4810 exhibit the same tensile strength of about 10.5 MPa, while ISA4805 exhibits a lower tensile strength of 9.3 MPa, and the ISA4815 alloy exhibits the highest tensile strength of 12.5 MPa, as shown in Fig. 5a. In addition, the elongation of the new alloys was improved due to the addition of Ag, as is clearly evident in Fig. 5b, which shows that the ISA4805 alloy presents the greatest elongation of 64%, which is twice that of the eutectic In–48Sn alloy (32%), followed by ISA4810 (52%) and ISA4815 (46%). The new alloys exhibit higher elongation but lower tensile strength compared with the eutectic Sn-58Bi alloy, namely 37.5% and 48.0 MPa, respectively [5].
The microstructure of a metal has an essential effect on its mechanical properties. As the microstructure was observed to change in this study, we were concerned about the impact of any change on the mechanical properties of the resultant alloys. Figure 5 shows typical stress–strain curves and a fracture image for each alloy; the maximum stress point and maximum strain in each curve determine the tensile strength and elongation of the alloy, respectively. The stress–strain curve of In–48Sn is typical of the ductile mode, with little plastic deformation; the curve tends to bulge before significantly dropping, while the specimen shows a typical brittle mode with planar stress fractures on inclined planes. We note that the eutectic In–48Sn alloy exhibits moderately ductile fracturing [19] or semi-ductile behavior. Conversely, the ISA samples exhibit ductile behavior with superior strain; the curves transit smoothly and fall slightly with the extensional tailing, in agreement with their macroscopic fracture morphologies, in which ISA4805 shows extreme elongation, with the longest necking region, followed by the ISA4810 and ISA4815 alloys. This alloy behavior is consistent with the elongation values, as shown in Fig. 5b.
The tensile strength was improved by the addition of more than 1.0 wt.% Ag. In particular, the ISA4815 alloy exhibited the highest tensile strength of 12.5 MPa. Mechanical strength depends on the dislocation capacity during plastic deformation [23], and the dislocations that accumulate at grain or phase boundaries induce grain-boundary strengthening in metals [24]. Therefore, the tensile strength of ISA4815 may be enhanced by its significantly smaller phases, as shown in Fig. 4e, f.
Figure 6a, b present dislocation-density gradients in large and small grains. The grain boundary is weakened in a large grain when a considerable dislocation-density gradient exists between the grain interior and the grain boundary, as shown in Fig. 6a [23]. Intergranular fracturing usually occurs in crystals with larger grains, because dislocation accumulations propagate cracks at grain boundaries during tensile testing [24]. Conversely, as shown in Fig. 6b, the grain boundaries are not the weakest regions during tensile testing in smaller grains with more uniform dislocation distributions, and transgranular fracturing, as opposed to intergranular fracturing, is observed.
Fracture images following tensile testing are shown in Fig. 6c–f, with phases determined by energy-dispersive spectroscopy (EDS). Figure 6c reveals that the eutectic In–48Sn, with the largest phases, exhibits intergranular fracturing through cleavage along the grain boundaries. Each cracked grain is differently oriented to give a crystalline structure that resembles fractured rock candy [25]. The SEM images in Fig. 6d and e reveal that the phases in ISA4805 and ISA4810 are elongated through grain boundary sliding (GBS) motions along their tensile axes as well as cleavage that leads to grain cracking inside the phase [23]. On the other hand, the ISA4815 alloy, with the largest number of small phases (Fig. 4), shows the most transgranular fractures, with collision areas (Fig. 6f) and the highest tensile strength, as shown in Fig. 5a.
From another point of view, the ISA samples all exhibit better elongation than the eutectic In–48Sn alloy. The γ-InSn4 in the ISA4805 alloy, which is known to be a hard phase [14], experiences a significant decrease in phase area (Fig. 4f), while the soft-β-In3Sn phase [14] is slightly reduced in size (Fig. 4e), leading to the highest β-In3Sn/γ-InSn4 ratio among the ISA alloys, as shown in Fig. 4g. On the other hand, the ISA4805 alloy, with the smallest Ag content, exhibits the lowest number of grains, while ISA4815, with the highest Ag content, shows the highest number of small grains among of the ISA alloys. In addition, as shown in the EPMA maps in Fig. 3, fewer hard γ-InSn4 regions are observed in the ISA alloys; the ε-AgIn2 is formed in the β-In3Sn phases due to the consumption of some of the In atoms, and near β-In3Sn/γ-InSn4 phase boundaries. Although the β-In3Sn regions in the ISA alloys include ε-AgIn2 formations, hardness is largely dominated by the soft-β-In3Sn phase. Accordingly, it is reasonable that ISA4805, with the highest β-In3Sn/γ-InSn4 ratio and without a large number of small grains or phases for grain-boundary strengthening, is the softest among the alloys. Therefore, this study reveals that the addition of Ag affects not only tensile strength, but also the elongation of the eutectic In–48Sn alloy.
Conclusions
In this study, eutectic In–48Sn was alloyed with 0.5–1.5 wt.% Ag to lower its fusion start temperature and enhance its mechanical properties. Due to the ternary eutectic reaction, the fusion start temperatures of the ISA alloys were all about 113 °C, which is lower than that of the eutectic In–48Sn alloy (117 °C). ε-AgIn2 formed in β-In3Sn phases and near β-In3Sn/γ-InSn4 boundaries in the ISA alloys following the addition of Ag, which reduced the average areas of both β-In3Sn phases and γ-InSn4 phases; these smaller phases enhanced tensile strength and altered the fractured mode. ISA4815, with the smallest phases, was the strongest among the alloys, with a tensile strength of 12.5 MPa; this alloy fractured in transgranular mode, as commonly seen in crystals with small grains. In addition, all ISA alloys exhibited better elongation than the eutectic In–48Sn alloy, which is ascribable to smaller hard γ-InSn4 regions. Among the alloys, ISA4805, with the highest soft-β-In3Sn/hard-γ-InSn4 ratio and devoid of very small phases, was the softest, with elongation of 64%. Therefore, the addition of Ag clearly influences the melting point, microstructures, tensile strength, and elongation of the eutectic In–48Sn alloy.
References
Kim TW, Yan M, Erlat AG et al (2005) Transparent hybrid inorganic/organic barrier coatings for plastic organic light-emitting diode substrates. J Vac Sci Technol A 23:971–977. https://doi.org/10.1116/1.1913680
Liu YF, Feng J, Bi YG et al (2019) Recent developments in flexible organic light-emitting devices. Adv Mater Technol 4:1–19. https://doi.org/10.1002/admt.201800371
Shen Y-A, Zhou S, Li J et al (2019) Sn–3.0Ag–0.5Cu/Sn–58Bi composite solder joint assembled using a low-temperature reflow process for PoP technology. Mater Des 183:108144. https://doi.org/10.1016/j.matdes.2019.108144
Shen YA, Zhou S, Li J et al (2019) Thermomigration induced microstructure and property changes in Sn–58Bi solders. Mater Des. https://doi.org/10.1016/j.matdes.2019.107619
Zhou S, Shen YA, Uresti T et al (2019) Improved mechanical properties induced by In and In & Zn double additions to eutectic Sn58Bi alloy. J Mater Sci: Mater Electron 30:7423–7434. https://doi.org/10.1007/s10854-019-01056-y
Gleason J, Schroeder V, Henshall G et al (2008) Design, materials, and assembly process of high-density packages with a low: temperature lead-free solder (SnBiAg). Solder Surf Mt Technol 20(2):11–20. https://doi.org/10.1108/09540910810871520
Mei Z, Morris JW (1992) Characterization of eutectic Sn–Bi solder joints. J Electron Mater 21:599–607. https://doi.org/10.1007/BF02655427
Zhou S, Mokhtari O, Rafique MG et al (2018) Improvement in the mechanical properties of eutectic Sn58Bi alloy by 0.5 and 1 wt.% Zn addition before and after thermal aging. J Alloys Compd 765:1243–1252. https://doi.org/10.1016/j.jallcom.2018.06.121
Wang F, Chen H, Huang Y et al (2019) Recent progress on the development of Sn–Bi based low-temperature Pb-free solders. J Mater Sci: Mater Electron 30:3222–3243
Macdonald WA (2007) Latest advances in substrates for flexible electronics. J Soc Inf Disp 15:1075–1083. https://doi.org/10.1889/1.2825093
Chuang TH, Yu CL, Chang SY, Wang SS (2002) Phase identification and growth kinetics of the intermetallic compounds formed during In–49Sn/Cu soldering reactions. J Electron Mater 31:640–645. https://doi.org/10.1007/s11664-002-0136-1
Abtew M, Selvaduray G (2000) Lead-free solders in microelectronics. Mater Sci Eng R Rep 27:95–141. https://doi.org/10.1016/S0927-796X(00)00010-3
Kim SH, Yang S (2017) Low melting temperature solder materials for use in flexible microelectronic packaging applications. In: Recent progress in soldering materials. Intech, pp 8–37
Goldstein JLF, Morris JW (1994) The effect of substrate on microstructure and creep of eutectic In–Sn. Metall Mater Trans Trans A 323:2715–2722. https://doi.org/10.1557/PROC-323-159
Glazer J (1995) Metallurgy of low temperature Pb-free solders for electronic assembly. Int Mater Rev 40:65–93. https://doi.org/10.1179/imr.1995.40.2.65
Wang J, Mao D, Shi L et al (2019) Effect of zinc addition on the microstructure, thermal and mechanical properties of indium-Tin-xZinc alloys. J Electron Mater 48:817–826. https://doi.org/10.1007/s11664-018-6768-6
Kim SH, Yeon S-M, Kim JH et al (2019) Fine microstructured In–Sn–Bi solder for adhesion on a flexible PET substrate: its effect on superplasticity and toughness. ACS Appl Mater Interfaces. https://doi.org/10.1021/acsami.9b04159
El-Bediwi AB, El-Bahay MM (2004) Influence of silver on structural, electrical, mechanical and soldering properties of tin-indium based alloys. Radiat Eff Defects Solids 159:133–140. https://doi.org/10.1080/10420150410001670288
Jones WK, Liu Y, Shah M, Clarke R (1997) Mechanical properties of Pb/Sn Pb/In and Sn–In solders. Solder Surf Mt Technol, 37–41
Ohnuma I, Cui Y, Liu XJ et al (2000) Phase equilibria of Sn–In based micro-soldering alloys. J Electron Mater 29:1113–1121. https://doi.org/10.1007/s11664-000-0002-y
Korhonen TM, Kivilahti JK (1998) Thermodynamics of the Sn–In–Ag solder system. J Electron Mater 27:149–158. https://doi.org/10.1007/s11664-998-0205-1
Chuang TH, Huang YT, Tsao LC (2001) AgIn2/Ag2In transformations in an In–49Sn/Ag soldered joint under thermal aging. J Electron Mater 30:945–950
Callister WD Jr, Rethwisch DG (2015) Fundamentals materials science and engineering: an integrated approach, 5th edn. Wiley, London
Lu K, Lu L, Suresh S (2009) Strengthening materials by boundaries at the nanoscale. Science 80:349–353. https://doi.org/10.1126/science.1159610
Askeland DR, Fulay PP (2009) Essentials of materials science and engineering, second. Cengage Learning, Toronto ON M1K 5G4 Canada
Acknowledgements
The authors wish to thank Dr. Shiqi Zhou for help and discussions during this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest regarding the publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Le Han, D., Shen, YA., Jin, S. et al. Microstructure and mechanical properties of the In–48Sn–xAg low-temperature alloy. J Mater Sci 55, 10824–10832 (2020). https://doi.org/10.1007/s10853-020-04691-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-020-04691-7