Abstract
Periodontal disease is characterized by a microbial infection and it is one of the major causes of teeth loss. The growth of pathogenic bacteria in oral cavity, such as Aggregatibacter actinomycetemcomitans (A. actinomycetemcomitans), provides a favorable enviromment for the biofilm formation, which result in periodontal diseases. The development of new technologies to potentiate existing drugs, avoiding bacterial biofilms resistance and improving chemical stability is highly desirable. Here, we report the use of host–guest chemistry to enhance the activity of antibacterial doxycycline (DOX) in A. actinomycetemcomitans bacterial strain suspension and biofilm in vitro through complexation with hydroxypropyl-β-cyclodextrin (HPβCD) using different molar ratios of DOX/HPβCD. 2D 1H NMR confirmed the host–guest complexation of DOX and HPβCD. We assessed the colloidal characteristics of the complex DOX/HPβCD via Dynamic Light Scattering (DLS) and Zeta Potential (PZ). The mixing ratio 1:2 DOX/HPβCD significantly decreased the minimum inhibitory concentration (MIC) and improved efficacy against A. actinomycetemcomitans suspensions and biofilms, respectively, when compared to free DOX and other DOX/HPβCD complexes. Further, the interaction of different molar ratio proportions of DOX/HPβCD complex with bacterial membrane was demonstrated via Isothermal Titration Calorimetry (ITC). Thus, we suggested the enhanced efficacy of the DOX/HPβCD complexes, at molar ratio 1:2, is due the higher cyclodextrin ratio, which potentiate the interaction between drug and bacterial membrane through nonionic interactions, such as hydrogen bonding or other van der Waals interactions. Collectively, the development of these complexes enables increased efficacy against bacterial biofilms, which hold promise for the treatment of aggressive and non-responsive forms of periodontitis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Periodontal diseases are a serious healthy issue worldwide and it is one of the major causes of teeth loss [1,2,3]. More than 743 million people worldwide suffer from periodontitis diseases, and 64.7 million people only in US (~ 47% of the population), wherein the prevalence rate after 65 years old is higher than 70% [4, 5]. Periodontitis is usually associated with biofilm accumulation in periodontal tissues; characterized by loss of soft tissue attachment and alveolar bone resorption, leading in pocket formation and or gingival recession [1]. In these cases, immunologic and genetic factors might be also involved [6, 7]. Treatment is based on mechanical and/or surgical removal of accumulated biofilm, which may be also associated with adjuvant antibiotic therapy (AAT) [8,9,10].
Although different bacterial strains have been associated with periodontal diseases in adults, Aggregatibacter actinomycetemcomitans (A. actinomycetemcomitans) is considered to be a major cause of localized aggressive periodontitis [11,12,13,14]. A. actinomycetemcomitans is a Gram-negative, facultative anaerobic coccobacillus bacterium that colonizes the oral cavity with ability to form biofilm, which is a structured consortium of bacteria embedded in a self-produced polymer matrix consisting of polysaccharide, protein and DNA[15,16,17].
Bacterial biofilms can cause chronic infections, mainly because their increased resistance to antibacterials [18, 19]. A fact of considerable importance is that biofilms effectively protect bacteria from antimicrobial agents. Biofilm cells are generally said to be resistant by comparison with their planktonic counterparts [20]. Bacteria can be ~ 1000 times more resistant to an antibacterial in a biofilm when compared to the same planktonic cells [21], although the resistance mechanisms involved are still largely unknown. Among the possible mechanisms, it is believed that inactivation of the drug may occur by polymers or extracellular enzymes, or inefficiency of the drug due to very slow growth rates within the biofilm [22]. Infections associated with biofilms are generally recurring in nature, whereas conventional antimicrobial therapies eliminate predominantly planktonic forms, leaving free sessile cells to reproduce and spread in the biofilm after treatment. Also, bacteria within biofilms may also evade from the immune system response [23].
AAT may also fail due to the emergence of resistant human pathogens [12, 24]. Several antibacterial have been used as an adjunct to the basic mechanical treatment and has become the standard of care in aggressive and non-responsive forms of periodontitis, including those that directly kill bacteria, such as amoxicillin, and those that exert bacteriostatic actions and prevent their growth, such as doxycycline (DOX) [25,26,27,28].
DOX is a semi-synthetic antibacterial derived from oxytetracycline, commonly used for numerous intracellular and extracellular pathogens [29] (Fig.1). The mechanism of action is through the inhibition of protein synthesis by its interaction with 30 s ribosomal subunit and the subsequent blocking of the RNA binding to the docking sites. Studies have found that the DOX has also an anti-inflammatory activity, ability to inhibit matrix metalloproteinases actions and cell proliferation [30], and reduces the blastogenic response of lymphocytes [31].
Despite the broad spectrum of DOX against pathogenic bacteria, several bacteria groups were reported to exhibit resistance to this antimicrobial agent, such as A. actinomycetemcomitans [12, 32,33,34,35]. Although DOX have been used in a systemic administration for infection control, it is also questionable, though, if sufficient doses can reach the site of infection in commonly used therapeutic [36]. Sustained and controlled drug release formulations have recently gained interest, since they provide a long-term and effective treatment at the site of infection at much smaller doses [12, 37]. The local delivery of antimicrobials has been investigated for the possibility of overcoming the limitations of systemic therapy [38,39,40]. Furthermore, it has been demonstrated that molecular inclusion with cyclodextrins (CDs) could prevent degradation of the associated drugs, improving stability [41]. The DOX complexed in hydroxypropyl-β-cyclodextrin (HPβCD) has been already studied in a hydrogel for ophthalmic delivery [42]; however, there isn’t approved formulation, for local and controlled release of antibacterial using cyclodextrins, for periodontitis treatment in humans.
There is some uncertainty about the efficacy of DOX when used as the only therapy to treat periodontal diseases. However, some studies have shown that DOX adjunct therapeutic scheme have more favorable antimicrobial effects than mechanical therapy alone [14, 36]. The complexation of DOX with HPβCD has been proposed as a strategy to improve the drug properties such as stability, solubility, and efficacy.
Herein, different molar ratios of DOX/HPβCD complexes were prepared and assessed for enhanced efficacy against bacterial A. actinomycetemcomitans suspensions and biofilms. An optimized complex was identified, through complexation of HPβCD with DOX at molar ratio of 1:2, with enhanced stability and greater efficacy against A. actinomycetemcomitans suspension and biofilm.
Materials and methods
Chemicals and bacterial strain
Doxycycline hydrochloride (DOX) was obtained from Sigma-Aldrich (St Louis, MO) and hydroxypropyl-beta-cyclodextrin (HPβCD) was obtained from Cerestar®, Co. (Milwaukee, WI, USA). A. actinomycetemcomitans was obtained from the American Type Culture Collection (ATCC 29,522). Brain Heart Infusion (BHI) broth and yeast extract were purchased from Biobras S.A® (MG, Brazil). All other materials and solvents were of analytical grade. Dimethyl sulfoxide 99.9% ACS reagent was obtained from Sigma-Aldrich (Milwaukee, WI, USA). Phosphate buffered saline, pH 7.4 was obtained from ThermoFisher Scientific (Cambridge, MA, USA). Deuterium Oxide (D2O) was obtained from Cambridge Isotope Laboratories, Inc. (Cambridge, MA, USA).
DOX/HPβCD complexation
The 1:2, 1:1 and 2:1 DOX/HPβCD mixing ratio inclusion complexes were prepared by freeze-drying method using the same methods described elsewhere [41]. Briefly, different proportions of DOX and HPβCD were weighted at given molar ratios and dissolved in ultrapure water. DOX solution was protected of direct light. The aqueous solution of DOX and HPβCD were mixed and stirred for 2 h at room temperature and then submitted to freeze-drying process to achieve a solid inclusion complex, before spectroscopic characterizations.
Solid state analysis: Fourier transformed infrared spectroscopy (FTIR)
Solid state characterization of DOX, HPβCD, DOX/HPβCD and a physical mixture (PM) of DOX and HPβCD at a molar ratio of 1:1 was performed using FTIR. The samples were prepared as potassium bromide (KBr) pellets and scans were performed from 4000 to 400 cm−1 at a resolution of 4 cm−1 using 32 scans per sample. FTIR spectra were recorded using a Perkin Elmer spectrometer (Spectrum GX; Perkin Elmer, Boston, MA, USA).
Solution analysis: nuclear magnetic resonance (NMR) spectroscopy
The DOX/HPβCD inclusion complex in solution was characterized using NMR spectroscopy at a molar ratio of 1:1. 1H NMR chemical shifts (δ) and 2D 1H-1H ROESY experiments were performed using a Brucker DPX-400 Avance (400 MHz) spectrometer, at 300 K. The solutions analyzed included 2.0 mM of DOX and DOX/HPβCD (1:1). Both solutions were prepared in D2O (Cambridge Isotope Laboratories, Inc.; 99.9% isotopic purity). The HOD signal at δ = 4.80 was used as a reference. DOX/HPβCD complexes were determined via 2D 1H-1H ROESY correlation. Briefly, DOX/HPβCD complexes were dissolved in D2O (concentration: ~15 mg/mL) and then 2D 1H-1H ROESY (ROESY, mix time of either 400 or 650 ms, 256 FID) was performed. Complexation and structure were determined by observing the proton cross-peaks between HPβCD and DOX.
Microcalorimetric measurements
Calorimetric titrations were carried out with a VP-ITC Microcalorimeter (Microcal Company, Northampton, MA, USA) at 25 degrees Celsius. To investigate the of drug-membrane direct interaction, calorimetric titrations for each DOX/HPβCD solution, at 1:0 (free DOX solution), 1:2, 1:1 and 2:1 mixing ratio in A. actinomycetemcomitans suspensions were performed. For each titration, the nominal concentration of DOX was constant (15 mM), only varying the HPβCD proportion. Each titration was performed by 51 injections of 5.0 µL of titrants in 1.5 mL of the titer bacterial solution in sterile saline prepared with turbidity (0.08 OD at 600 nm), with around 0.8 × 108 cells/mL. The raw data was analyzed using the “one site model” set forth by Microcal Origin 7.0 for ITC after the subtraction of the blank experiment.
Dynamic light scattering analysis
The hydrodynamic diameter titrations for DOX/HPβCD complex were performed using a Malvern Zetasizer Nano ZS instrument (Malvern Instruments, Malvern, UK) and polyethylene square cells. Briefly, HPβCD (4 mM) was titrated in with the addition of ~ 30 aliquots (10.0 µL each) of aqueous solutions of DOX (60 mM). Sample suspensions were exposed to monochromatic light (10 mW He-Ne laser, wavelength 632.4 nm) and scattered light intensity was measured at 90°. Hydrodynamic diameters (Dh) for each titration were calculated from the average of five independent measurements that represented the mean of thirty runs. The reported Dh is the average of two independent titrations and error bars represent the standard deviation (SD) of these two experiments.
Zeta potential (ZP) titrations
ZP values were determined using the Laser Doppler Velocimetry technique and disposable cell folded capillaries (Malvern DPS1060) at a scattering angle of 90° at 25 °C. Titrations were conducted using ~ 25 successive injections of 10 µL aliquots of of DOX (60 mM) solution, into 1.0 mL of HPβCD (4 mM). ZP values were obtained from the average of five measurements, with each one representing the mean of five runs. Reported ZP values are the average of two independent titrations and error bars represent the SD of these two experiments.
Minimum inhibitory concentration (MIC)
MIC for planktonic bacteria was determined by microdilution method, according to the standards of NCCLS [43, 44]. Briefly, A. actinomycetemcomitans was anaerobically seeded in BHI broth supplemented with 0.5% yeast extract and incubated anaerobically (10% CO2, 90% N2) at 37 oC for 24 h. After incubation, bacteria suspension was adjusted to an optical density determined spectrophotometrically of 0.08–0.10 at 600 nm to obtain 1.5 × 108 CFU, added to double serial dilution of DOX, HPβCD, DOX/HPβCD (128 µg/mL to 0.25 µg/mL), and then transferred to 96-well plates and incubated at 37 oC for a period of 24 h. MIC was defined as the lowest concentration that prevented visible growth of the microorganism, determined with a spectrophotometer at 610 nm [43, 44]. All MICs assays were performed in triplicates of two independent experiments.
Scanning electron microscopy (SEM)
Standard procedures of SEM have been used to verify biofilm bacterial growth and to assess cellular membrane integrity and morphologic changes caused by the drug treatments [43]. Briefly, biofilms were grown under anaerobic conditions for 24 h on coverslips treated with poly-L-lysine in 24-well plates. After incubation the antimicrobial agents replaced the medium in the concentration of 1 µg/mL for DOX and DOX/HPβCD. For positive control, medium was replaced by fresh BHI supplemented medium. After 24 h of incubation under anaerobic conditions at 37 oC, the coverslips were fixed for 24 h in a solution containing 2.5% glutaraldehyde. Coverslips were washed in 0.1 M cacodylate buffer (pH 7.3) and dehydrated in a graded series of ethanol. Subsequently, the cover slips were subjected to critical point drying with CO2, sputter-coated with gold (thickness approx. 10 nm), and examined with a Zeiss DSM 960A electron microscope.
Biofilm formation, quantification of Biomass and Minimum Biofilm inhibitory concentration (MBIC)
Overnight cultures were diluted to an optical density at 600 nm of 0.10 ± 0.02 absorbance (between 107 and 107.4 CFU/mL) in fresh BHI supplemented with 0.5% yeast extract. In brief, 0.2 mL of the diluted culture was added to a 96-well plate and incubated for 24 h. After incubation, solutions of DOX or DOX/HPβCD at different stoichiometric ratio replaced the BHI broth. The nominal DOX concentrations tested were: 2, 20 and 200 µg/mL. Fresh BHI broth with bacteria was used as positive control and BHI broth supplemented without bacteria as a negative control. Following 24 h of incubation, biofilm biomass was assessed by crystal violet assay as described elsewhere [45, 46]. Briefly, after 24 h incubation with the antimicrobial agents the medium was removed and the microtiter plate wells were washed with 200 µL of PBS (0.1 M, pH 7.4) buffer and allowed to dry for 15 min. Microtiter wells were stained with 200 µL of 0.1% crystal violet stain for 30 min at room temperature. The unbound crystal violet stain was removed and the wells were washed with 200 µL of PBS buffer. Wells were air-dried for 15 min and the remaining crystal violet was solubilized in acetic acid (200 µL; 33% w:w). Biofilm formation was represented by the analysis of the absorbance carried out with a spectrophotometer at 620 nm. Biofilm biomass was given by the equation modified from Pereira et al. [47]:
where % B.b is the percentage of biofilm biomass, ODw is the OD620nm value for tested wells and ODc is the OD620nm value for the positive control.
Reproducibility and statistics
The results were analyzed by the nonparametric test One Way Anova. Results were presented as the means ± SD (standard deviation) and P < 0.05 was considered statistically significant.
Results and discussion
To enhance efficacy against A. actinomycetemcomitans biofilms, DOX was complexed with HPβCD in a range of mixing ratios (1:2, 1:1, 2:1). Inclusion complexes can provide a long-term, effective treatment at the site of infection at much smaller doses [40], prevent drug degradation [48], enhance drug absorption as well as facilitate drug transport across physiological barriers and biological membranes [41]. Furthermore, it has been shown the hydrophilic outer surface of cyclodextrin molecules forms a weak interaction with biologic membranes [49]. We hypothesized that the increase of cyclodextrin ratio, at constant concentration of DOX, potentiate the interaction of drug with bacteria membrane, probably by increase its contact time on surface of membrane through no ionic interactions, such as hydrogen bonding or other van der Waals interactions. Here, we showed the antibacterial activity of DOX/HPβCD against A. actinomycetemcomitans biofilms compared throughout different times. The efficacy of these complexes proportions was demonstrated by the significantly decreasing in MIC and the percentage of the biofilm biomass.
DOX/HPβCD complexation
To characterize the host–guest interactions and to gain insight into the molecular interactions between DOX and HPβCD, FTIR and 2D ROESY NMR were performed (Figs. 2 and 3). For these studies, only the molar ratio of 1:1 was chosen, because these techniques are not sensible enough to furnish information about different topologies resultant from different proportions of the compounds.
FTIR provides data consisting of the molecular structure of the complex, functional groups and intermolecular interactions between DOX and HPβCD in the solid state. The FTIR spectra of free drug, PM, DOX/HPβCD inclusion complex and HPβCD are shown in Fig. 2. The interactions between DOX and HPβCD were verified with changes observed in N–H and O–H absorption bands. The DOX/HPβCD spectrum inclusion complex did not overlay the FTIR spectra of either free species or with the PM of DOX/HPβCD. These included sharper peaks for DOX/HPβCD compared with free HPβCD and PM, suggesting that there is a reduction in the total number of hydrogen bonds upon complexation. Reduction of angular and axial DOX C-O bends at 1175 cm−1 and 1130 cm−1 were observed, as well as sharpening of C=O and C=C DOX stretches at 1700 and 1600 cm−1. These data suggest that strong disturbance of the CONH2 group of DOX occurs in the solid state because of both the binding and breaking of hydrogen bonds upon inclusion of DOX in HPβCD. Moreover, these results are in accordance previous study [42].
Observation of 2D NMR 1H-1H ROESY, inter- and intra-molecular interactions of functional groups within 5 Å of each other can be detected [50, 51]. In this study, ROESY experiments identified the host–guest specific interactions that occur between DOX and HPβCD upon formation of the supramolecular complex and provided the possible mechanism of both inclusions in the HPβCD cavity. Dipolar proton interactions were found between DOX and HPβCD, indicating the DOX complexation. The expanded contour map of ROESY showed cross peak correlations between aromatic H7D, H8D, and H9D DOX hydrogens (in the region of δ ≈ 6.5 to 7.6) and H3, H5, and H6 HPβCD hydrogens (Fig. 3 and supplementary material). Furthermore, cross peak correlations, which could suggest inclusion by the aliphatic side, were not observed. These results indicate that DOX and HPβCD effectively form supramolecular complexes, with preferential inclusion of the aromatic side of DOX into HPβCD cavity.
DLS and zeta potential titrations
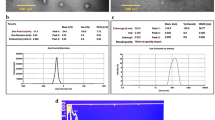
To assess the colloidal characterization of the DOX/HPβCD complex, a titration of DOX 60 mM in HPβCD at 4 mM was performed and monitored via DLS (Fig. 4) or ZP (Fig. 5).
In DLS experiment, it was found an increase of size, since from 56 nm (in the beginning of the titration) up to 245 nm, at molar ratio close to 1:2 (Fig. 4). This data suggest the hypothesis that the system suffer an aggregation, forming high order supramolecular structures. It is important highlight that the aggregation phenomenon of cyclodextrins and their inclusion complexes, forming supramolecular structures with 100–400 nm, may be a natural phenomenon, which have been observed in several others systems [52, 53]. After this molar ratio, the supramolecular structures need to suffer a disaggregation, forming structures with smaller sizes.
In ZP experiment, it was found a reduction of |ZP| values upon titration, reaching the isoelectric point close to molar ratio of 0.5, corresponding to the molar ration of 1:2 (Fig. 5). These lower ZP values matches with the higher size values found at this DOX/HPβCD proportion, so that lower surface charges favor the attractive interactions and consequently the aggregation phenomenon. With the increase of DOX concentration, the positive ZP values are observed after the isoelectric point (and close to 1:2 stoichiometry), suggesting the increasing of surface charges of aggregates. This can favor the breakdown of aggregates in lower species, explaining thereby the lower stoichiometry at excess of DOX.
Minimum inhibitory concentration (MIC)
As discussed above, different molar proportions of DOX/HPβCD are subject to different level of aggregation. Thus, considering that each molar proportions might cause differences in biological environment, we have assessed the effect of free DOX and the same nominal concentration of DOX in presence of different proportions of HPβCD (at 2:1, 1:1 and 1:2), on the growth of the microorganism A. actinomycetemcomitans.
DOX/HPβCD at 1:2 proportion showed the lowest MIC (0.125–0.25 µg/mL), when compared with free DOX (2 µg/mL) and DOX/HPβCD at 1:1 (0.5 µg/mL) and DOX/HPβCD at 2:1 (1.0 µg/mL) proportions. Moreover, the free HPβCD did not show antimicrobial activity (Table 1). Optimization of antimicrobial activity of drugs associated with cyclodextrins, with no increased cytotoxic effects, is consistent with previous studies using other antimicrobials [38, 39, 54, 55]. Collectively, these results indicate that DOX/HPβCD at 1:2, which presented the most stable stoichiometry according to DLS and ZP titrations, induce higher antimicrobial activity against A. actinomycetemcomitans. Furthermore, these data corroborate the hypothesis that the excess of cyclodextrin, at constant concentration of DOX, potentiate the interaction of drug with membrane, probably by enhanced contact time on surface of microorganism through nonionic interactions, such as hydrogen bonding or other van der Waals interactions. Therefore, as described below, we have studied the direct interactions of DOX and DOX/HPβCD at 2:1, 1:1 and 1:2 proportions, against A. actinomycetemcomitans cells, by isothermal calorimetric experiments.
Drug–membrane interaction studies by ITC
To assess the interactions between DOX or different proportions of DOX/HPβCD (at 1:2, 1:1 and 2:1) with A. actinomycetemcomitans, ITC experiments were performed by direct titration of these compositions in A. actinomycetemcomitans suspensions (Fig. 6). The results are expressed after the subtraction of the respective blank experiments (dilution curves of the compounds in saline).
In these experiments, it was monitored the heat of injection upon titration of free DOX or each of the DOX/HPβCD proportions, concerning the stepwise change in the heat of the system, normalized to ligand total concentration added per injection, at constant pressure (enthalpy of injection at constant pressure, (dQ/d[DOX]tot)P = ΔinjH°), to the absolute concentration of DOX at any point during the course of the titration [56].
As observed, it was found clear differences between free DOX and the DOX/HPβCD compositions (Fig. 6). For the titration of free DOX in A.a microorganisms, the profile of curve is similar to “one set of sites” interaction [57], with a trend of saturation close to 0.2 mM of nominal concentration of DOX. Moreover, exothermic values were observed until ≈ 0.07 mM. As described in literature, exothermic character is usually found in systems subject to ionic interactions [57, 58]. Thus, this partially exothermic profile was attributed to the presence of ionic interactions between cationic DOX and anionic components of membrane.
However, in the presence of different proportions of HPβCD, the injection enthalpy values were always endothermic and showed an inflexion which is characteristic of a stepwise process [58, 59], necessarily with new mechanisms of interaction. However, as the proportion of HPβCD was increased, the number of points after inflexion also increased, indicating that cyclodextrin is able to intensify the new mode of interaction.
Thus, the ITC data obtained in the present study corroborate the hypothesis that the excess of cyclodextrin could provide an efficient adhesion with A. actinomycetemcomitans membrane, especially because the MIC was reduced as the proportion of HPβCD was increased. It is important highlight that CDs are rich in hydroxyl groups, and these can spontaneously form hydrogen bonds with several components of the cellular membrane, creating new a mechanism based on non-covalent interactions.
Scanning electron microscopy (SEM)
To assess cellular membrane integrity and changes in morphology caused by the drug treatments, SEM of A. actinomycetemcomitans biofilms treated with 1 µg/mL DOX or 1 µg/mL of DOX in DOX/HPβCD or untreated for 24 h were performed. A. actinomycetemcomitans biofilms consist of a matrix composed of exopolysaccharides and are found in small clusters of cells (islet-like), different stages of bacterial growth (Fig. 7A1, A2). It was found morphological changes and the appearance of indentations on the surface of some cells or outer membrane residues around the cells after treatment with DOX or DOX/HPβCD compared to untreated group (Fig. 7), which indicates that the bacterial outer membrane was disrupted with leakage of intracellular material (white arrows). DOX/HPβCD enhanced the antimicrobial effect, and leakage in cellular membrane, probably due to a direct interaction between the cellular membrane and the complex, which corroborated that cyclodextrins increased drug uptake through biological membranes [60].
SEM micrographs of A. actinomycetemcomitans biofilms (A1, A2), A actinomycetemcomitans biofilms treated with 1 µg/mL of DOX (B1, B2) and the same nominal concentration of DOX as DOX /HPβCD (C1, C2), after 24 h of exposure. Micrographs of A1–C1 were presented in a magnification of 1000×, and A2–C2 were presented in a magnification of 10,000×. White arrows indicate the integrity or disruption of the membrane with leakage of intracellular material
Quantification of biofilm biomass and minimum biofilm inhibitory concentration (MBIC)
The ability to form biofilm may be a virulence factor of bacteria and contribute to the serious course of the disease as well as the lack of response to antimicrobial therapy [19, 22]. In this study, A. actinomycetemcomitans was chosen as a test organism in this study because it is associated with localized and generalized aggressive periodontitis, including chronic periodontitis [12, 15]. Also, this bacteria exhibits high resistance to antimicrobial agents when associated with other bacteria in biofilms [12, 32,33,34,35]. To assess the effect of DOX/HPβCD against A. actinomycetemcomitans biofilm after 24 h of exposure, we treated the biofilms with free DOX or DOX/HPβCD at different proportions (1:2, 1:1 and 2:1). DOX/HPβCD at 1:2 showed the higher efficacy against A. actinomycetemcomitans biofilms at dosage of 20 µg/mL when compared with other DOX/HPβCD proportions and free DOX (Fig. 8). It was not found significant differences between 1:1 and 2:1 DOX/HPβCD compositions and free DOX. At higher concentration (200 µg/mL) the antibacterial activity was not significant. This is possibly due to that a surfactant reaches the equilibrium between the adsorption of the drug-surfactant complex. This phenomenal interaction is related with the surface tension as a dynamic process in bacterial biofilms, which has specific characteristics including their structure and physiological mechanism of protection by the formation of a gel-like network against noxious environmental conditions [61]. Otherwise, the manner as drugs act after application can comprise a diffused phase, electrical organization, adsorption, equilibrium and desorption [62]. Therefore, these results indicate that DOX/HPβCD complexes at molar ratio 1:2, enhanced the antimicrobial activity against A. actinomycetemcomitans biofilms at 20 µg/mL. The results showed that lower doses of DOX would be required against A. actinomycetemcomitans when DOX/HPβCD complexes is applied. Although the exact mechanism behind enhanced efficacy against biofilm will require further study, we suggest the DOX/HPβCD complexes at molar ratio 1:2, which has greater amount of cyclodextrin, potentiate the interaction between drug and bacterial membrane through nonionic interactions, such as hydrogen bonding or other van der Waals interactions as discussed above.
Biofilm Biomass (%) of A. actinomycetemcomitans after treatment with DOX and DOX/HPβCD at different molar ratios (1:2, 1:1 and 2:1) after 24 h of exposure assessed with crystal violet assay. DOX was normalized in all groups at 2, 20 and 200 µg/mL. Mean values ± SD for at least two replicates are illustrated, *P < 0.05, by One-Way ANOVA with Tukey’s multiple comparisons test (among all compounds at 20 µg/mL)
Conclusions
The development of the DOX/HPβCD complexes enable increased efficacy against bacterial biofilms, which hold promise for the treatment of aggressive and non-responsive forms of periodontitis. Also, this study would enable further fundamental studies on the role of DOX/HPβCD compositions in the treatment of aggressive and non-responsive forms of periodontitis in vivo. Furthermore, additional studies should be conducted with more complex bacterial biofilms and releasing devices with proper technology for long-term use in vivo.
Data availability
All data and material available upon request.
References
Tonetti, M.S., Greenwell, H., Kornman, K.S.: Staging and grading of periodontitis: framework and proposal of a new classification and case definition. J. Periodontol. 89(Suppl 1), S159–S172 (2018)
Ramseier, C.A., Anerud, A., Dulac, M., Lulic, M., Cullinan, M.P., Seymour, G.J., Faddy, M.J., Burgin, W., Schatzle, M., Lang, N.P.: Natural history of periodontitis: disease progression and tooth loss over 40 years. J. Clin. Periodontol. 44, 1182–1191 (2017)
Houshmand, M., Holtfreter, B., Berg, M.H., Schwahn, C., Meisel, P., Biffar, R., Kindler, S., Kocher, T.: Refining definitions of periodontal disease and caries for prediction models of incident tooth loss. J. Clin. Periodontol. 39, 635–644 (2012)
Marcenes, W., Kassebaum, N.J., Bernabé, E., Flaxman, A., Naghavi, M., Lopez, A., Murray, C.J.L.: Global burden of oral conditions in 1990–2010: a systematic analysis. J. Dent. Res. 92, 592–597 (2013)
Papapanou, P.N.: The prevalence of periodontitis in the US: forget what you were told. J. Dent. Res. 91, 907–908 (2012)
Hajishengallis, G.: Periodontitis: from microbial immune subversion to systemic inflammation. Nat. Rev. Immunol. 15, 30–44 (2015)
Laine, M.L., Loos, B.G., Crielaard, W.: Gene polymorphisms in chronic periodontitis. Int J Dent. 2010, 1–22 (2010)
Deas, D.E., Moritz, A.J., Sagun, R.S.J., Gruwell, S.F., Powell, C.A.: Scaling and root planing vs. conservative surgery in the treatment of chronic periodontitis. Periodontology 71, 128–139 (2016)
Larsson, L., Decker, A.M., Nibali, L., Pilipchuk, S.P., Berglundh, T., Giannobile, W.V.: Regenerative medicine for periodontal and peri-implant diseases. J. Dent. Res. 95, 255–266 (2016)
Habashneh, Al, R., Alsalman, W., Khader, Y.: Ozone as an adjunct to conventional nonsurgical therapy in chronic periodontitis: a randomized controlled clinical trial. J Periodontal Res. 50, 37–43 (2015)
Amano, A., Chen, C., Honma, K., Li, C., Settem, R.P., Sharma, A.: Genetic characteristics and pathogenic mechanisms of periodontal pathogens. Adv. Dent. Res. 26, 15–22 (2014)
Dhamecha, H.R.R., Jagwani, D., Rao, S., Jadhav, M., Shaikh, K., Puzhankara, S., Jalalpure, L.: S.: Local drug delivery systems in the management of periodontitis: a scientific review. J Control Release 307, 393–409 (2019)
Cortelli, J.R., Aquino, D.R., Cortelli, S.C., Roman-Torres, C.V.G., Franco, G.C.N., Gomez, R.S., Batista, L.H.B., Costa, F.O.: Aggregatibacter actinomycetemcomitans serotypes infections and periodontal conditions: a two-way assessment. Eur. J. Clin. Microbiol. Infect. Dis. 31, 1311–1318 (2012)
Slots, J., Ting, M.: Actinobacillus actinomycetemcomitans and Porphyromonas gingivalis in human periodontal disease: occurrence and treatment. Periodontology 20, 82–121 (1999)
Mira, A., Simon-Soro, A., Curtis, M.A.: Role of microbial communities in the pathogenesis of periodontal diseases and caries. J. Clin. Periodontol. 44(Suppl 18), S23–S38 (2017)
Yoshida, A., Ennibi, O.-K., Miyazaki, H., Hoshino, T., Hayashida, H., Nishihara, T., Awano, S., Ansai, T.: Quantitative discrimination of Aggregatibacter actinomycetemcomitans highly leukotoxic JP2 clone from non-JP2 clones in diagnosis of aggressive periodontitis. BMC Infect. Dis. 12, 253–210 (2012)
Haubek, D., Ennibi, O.-K., Poulsen, K., Vaeth, M., Poulsen, S., Kilian, M.: Risk of aggressive periodontitis in adolescent carriers of the JP2 clone of Aggregatibacter (Actinobacillus) actinomycetemcomitans in Morocco: a prospective longitudinal cohort study. Lancet 371, 237–242 (2008)
Penesyan, A., Nagy, S.S., Kjelleberg, S., Gillings, M.R., Paulsen, I.T.: Rapid microevolution of biofilm cells in response to antibiotics. NPJ Biofilms Microbiomes. 5, 34–14 (2019)
Høiby, N., Bjarnsholt, T., Givskov, M., Molin, S., Ciofu, O.: Antibiotic resistance of bacterial biofilms. Int. J. Antimicrob. Agents 35, 322–332 (2010)
Bridier, A., Briandet, R., Thomas, V., Dubois-Brissonnet, F.: Resistance of bacterial biofilms to disinfectants: a review. Biofouling. 27, 1017–1032 (2011)
Walker, C., Sedlacek, M.J.: An in vitro biofilm model of subgingival plaque. Oral Microbiol. Immunol. 22, 152–161 (2007)
Mah, T.F., O’Toole, G.A.: Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol. 9, 34–39 (2001)
Arciola, C.R., Campoccia, D., Speziale, P., Montanaro, L., Costerton, J.W.: Biofilm formation in Staphylococcus implant infections. A review of molecular mechanisms and implications for biofilm-resistant materials. Biomaterials. 33, 5967–5982 (2012)
Heitz-Mayfield, L.J.A.: Systemic antibiotics in periodontal therapy. Aust. Dent. J. 54(Suppl 1), S96–S101 (2009)
Ghangurde, A.A., Ganji, K.K., Bhongade, M.L., Sehdev, B.: Role of chemically modified tetracyclines in the management of periodontal diseases: a review. Drug Res (Stuttg) 67, 258–265 (2017)
Preshaw, P.M., Hefti, A.F., Novak, M.J., Michalowicz, B.S., Pihlstrom, B.L., Schoor, R., Trummel, C.L., Dean, J., Van Dyke, T.E., Walker, C.B., Bradshaw, M.H.: Subantimicrobial dose doxycycline enhances the efficacy of scaling and root planing in chronic periodontitis: a multicenter trial. J. Periodontol. 75, 1068–1076 (2004)
Mohammad, A.R., Preshaw, P.M., Bradshaw, M.H., Hefti, A.F., Powala, C.V., Romanowicz, M.: Adjunctive subantimicrobial dose doxycycline in the management of institutionalised geriatric patients with chronic periodontitis. Gerodontology. 22, 37–43 (2005)
Novak, M.J., Johns, L.P., Miller, R.C., Bradshaw, M.H.: Adjunctive benefits of subantimicrobial dose doxycycline in the management of severe, generalized, chronic periodontitis. J. Periodontol. 73, 762–769 (2002)
Golub, L.M., McNamara, T.F., Ryan, M.E., Kohut, B., Blieden, T., Payonk, G., Sipos, T., Baron, H.J.: Adjunctive treatment with subantimicrobial doses of doxycycline: effects on gingival fluid collagenase activity and attachment loss in adult periodontitis. J. Clin. Periodontol. 28, 146–156 (2001)
Gueders, M.M., Bertholet, P., Perin, F., Rocks, N., Maree, R., Botta, V., Louis, R., Foidart, J.-M., Noel, A., Evrard, B., Cataldo, D.D.: A novel formulation of inhaled doxycycline reduces allergen-induced inflammation, hyperresponsiveness and remodeling by matrix metalloproteinases and cytokines modulation in a mouse model of asthma. Biochem. Pharmacol. 75, 514–526 (2008)
Griffin, M.O., Fricovsky, E., Ceballos, G., Villarreal, F.: Tetracyclines: a pleitropic family of compounds with promising therapeutic properties. Review of the literature. Am. J. Physiol. Cell Physiol. 299, C539–C548 (2010)
Madinier, I.M., Fosse, T.B., Hitzig, C., Charbit, Y., Hannoun, L.R.: Resistance profile survey of 50 periodontal strains of Actinobacillus actinomyectomcomitans. J. Periodontol. 70, 888–892 (1999)
Kulik, E.M., Lenkeit, K., Chenaux, S., Meyer, J.: Antimicrobial susceptibility of periodontopathogenic bacteria. J. Antimicrob. Chemother. 61, 1087–1091 (2008)
Ardila, C.M., Lopez, M.A., Guzman, I.C.: High resistance against clindamycin, metronidazole and amoxicillin in Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans isolates of periodontal disease. Med Oral Patol Oral Cir Bucal. 15, e947–e951 (2010)
Feres, M., Haffajee, A.D., Goncalves, C., Allard, K.A., Som, S., Smith, C., Goodson, J.M., Socransky, S.S.: Systemic doxycycline administration in the treatment of periodontal infections (II). Effect on antibiotic resistance of subgingival species. J. Clin. Periodontol. 26, 784–792 (1999)
Chadha, V.S., Bhat, K.M.: The evaluation of doxycycline controlled release gel versus doxycycline controlled release implant in the management of periodontitis. J Indian Soc Periodontol. 16, 200–206 (2012)
Joshi, D., Garg, T., Goyal, A.K., Rath, G.: Advanced drug delivery approaches against periodontitis. Drug Delivery. 23, 363–377 (2014)
Pataro, A.L., Franco, C.F., Santos, V.R., Cortés, M.E., Sinisterra, R.D.: Surface effects and desorption of tetracycline supramolecular complex on bovine dentine. Biomaterials. 24, 1075–1080 (2003)
Denadai, ÂM.L., Teixeira, K.I., Santoro, M.M., Pimenta, A.M.C., Cortés, M.E., Sinisterra, R.D.: Supramolecular self-assembly of beta-cyclodextrin: an effective carrier of the antimicrobial agent chlorhexidine. Carbohydr. Res. 342, 2286–2296 (2007)
Franco, C.F., Pataro, A.L., Souza, E., Santos, L.C.R., Cortes, V.R., Sinisterra, M.E.: R.D.: In vitro effects of a chlorhexidine controlled delivery system. Artif. Organs 27, 486–491 (2003)
Guimarães, P.P.G., Tan, M., Tammela, T., Wu, K., Chung, A., Oberli, M., Wang, K., Spektor, R., Riley, R.S., Viana, C.T.R., Jacks, T., Langer, R., Mitchell, M.J.: Potent in vivo lung cancer Wnt signaling inhibition via cyclodextrin-LGK974 inclusion complexes. J Control Release. 290, 75–87 (2018)
He, Z.-X., Wang, Z.-H., Zhang, H.-H., Pan, X., Su, W.-R., Liang, D., Wu, C.-B.: Doxycycline and hydroxypropyl-β-cyclodextrin complex in poloxamer thermal sensitive hydrogel for ophthalmic delivery. Acta Pharm. Sin. B. 1, 254–260 (2011)
National Committee for Clinical Laboratory Standards: Performance Standards for Antimicrobial Susceptibility Testing; Twenty First International Supplement. Wayne, PA: (2011)
Wiegand, I., Hilpert, K., Hancock, R.E.W.: Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nature Protocols. 3, 163–175 (2008)
Burton, E., Yakandawala, N., LoVetri, K., Madhyastha, M.S.: A microplate spectrofluorometric assay for bacterial biofilms. J. Ind. Microbiol. Biotechnol. 34, 1–4 (2007)
Erriu, M., Genta, G., Tuveri, E., Orrù, G., Barbato, G., Levi, R.: Microtiter spectrophotometric biofilm production assay analyzed with metrological methods and uncertainty evaluation. Measurement. 45, 1083–1088 (2012)
Margarida Pereira, A., Cristina Abreu, A., Simões, M.: Action of kanamycin against single and dual species biofilms of Escherichia coli and Staphylococcus aureus. J. Microbiol. Res. 2, 84–88 (2012).
Bisson-Boutelliez, C., Fontanay, S., Finance, C., Kedzierewicz, F.: Preparation and physicochemical characterization of amoxicillin beta-cyclodextrin complexes. AAPS PharmSciTech 11, 574–581 (2010)
Abdelwahed, W., Degobert, G., Dubes, A., Parrot-Lopez, H., Fessi, H.: Sulfated and non-sulfated amphiphilic-beta-cyclodextrins: impact of their structural properties on the physicochemical properties of nanoparticles. Int. J. Pharm. 351, 289–295 (2008)
Denadai, ÂM.L., Santoro, M.M., Texeira, A.V., Sinisterra, R.D.: New insights regarding the cyclodextrin/AAS self-assembly: a molar ratio dependent system. Mater. Sci. Eng. C. 30, 417–422 (2010)
Lula, I., De Sousa, F.B., Denadai, ÂM.L., de Lima, G.F., Duarte, H.A., Mares Guia, dos, Faljoni-Alario, T.R., Santoro, A., de Camargo, M.M., Santos, A.C.M., dos, Sinisterra, R.A.S., R.D.: Interaction between bradykinin potentiating nonapeptide (BPP9a) and β-cyclodextrin: a structural and thermodynamic study. Mater. Sci. Eng. C. 32, 244–253 (2012)
Turnbull, W.B., Daranas, A.H.: On the value of c: can low affinity systems be studied by isothermal titration calorimetry? J. Am. Chem. Soc. 125, 14859–14866 (2003)
Messner, M., Kurkov, S.V., Flavià-Piera, R., Brewster, M.E., Loftsson, T.: Self-assembly of cyclodextrins: the effect of the guest molecule. Mater. Sci. Eng. C. 408, 235–247 (2011)
Teixeira, K.I.R., Araujo, P.V., Neves, B.R.A., Mahecha, G.A.B., Sinisterra, R.D., Cortes, M.E.: Ultrastructural changes in bacterial membranes induced by nano-assemblies beta-cyclodextrin chlorhexidine: SEM, AFM, and TEM evaluation. Pharm Dev Technol. 18, 600–608 (2013)
Consuegra, J., de Lima, M.E., Santos, D., Sinisterra, R.D., Cortes, M.E.: Peptides: beta-cyclodextrin inclusion compounds as highly effective antimicrobial and anti-epithelial proliferation agents. J. Periodontol. 84, 1858–1868 (2013)
Suárez, D.F., Consuegra, J., Trajano, V.C., Gontijo, S.M.L., Guimarães, P.P.G., Cortés, M.E., Denadai, ÂL., Sinisterra, R.D.: Structural and thermodynamic characterization of doxycycline/β-cyclodextrin supramolecular complex and its bacterial membrane interactions. Colloids Surf. B 118, 194–201 (2014)
Denadai, ÂM.L., de Oliveira, A.M., Daniel, I.M.P., Carneiro, L.A., Ribeiro, K.C., Beraldo, H., de O., da Costa, da Cunha, K.J.R., Cortés, V.C., Sinisterra, M.E.: R.D.: Chlorhexidine/losartan ionic pair binding and its nanoprecipitation: physico-chemical characterisation and antimicrobial activity. Supramol. Chem. 24, 204–212 (2012)
de Carvalho, F.G., Magalhães, T.C., Teixeira, N.M., Gondim, B.L.C., Carlo, H.L., Santos, dos, R.L., de Oliveira, A.R., Denadai, A.M.L.: Synthesis and characterization of TPP/chitosan nanoparticles: Colloidal mechanism of reaction and antifungal effect on C. albicans biofilm formation. Mater. Sci. Eng. C. 104, 109885 (2019)
Denadai, ÂM.L., Da Silva, J.G., Guimarães, P.P.G., Gomes, L.B.S., Mangrich, A.S., de Rezende, E.I.P., Daniel, I.M.P., Beraldo, H., Sinisterra, R.D.: Control of size in losartan/copper(II) coordination complex hydrophobic precipitate. Mater. Sci. Eng. C. 33, 3916–3922 (2013)
Loftsson, T., Duchene, D.: Cyclodextrins and their pharmaceutical applications. Int. J. Pharm. 329, 1–11 (2007)
Wingender, J., Neu, T.R., Flemming, H.-C.: What are bacterial extracellular polymeric substances? In: Microbial Extracellular Polymeric Substances, pp. 1–19. Springer, Berlin (1999)
Eastoe, J., Dalton, J.S.: Dynamic surface tension and adsorption mechanisms of surfactants at the air–water interface. Adv. Colloid Interface. Sci. 85, 103–144 (2000)
Acknowledgements
We are grateful for CNPq, CAPES, FAPEMIG, INCT/Nanobiofar that support this investigation. The authors also thank Professor Gustavo Menezes, Immunobiophotonics Lab, Universidade Federal de Minas Gerais (UFMG) for technical support and constructive advice.
Funding
CNPq, CAPES, FAPEMIG, INCT/Nanobiofar that support this investigation.
Author information
Authors and Affiliations
Contributions
PPGG, ACM, MEC and RDS conceived the ideas, designed the experiments, interpreted the data and wrote the manuscript. PPGG, ACM, KIRT, AMLD, and RAF conducted the experiments and analyzed the data. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interests to declare.
Consent for publication
All authors have read and approved this version of the article. The author signs for and accepts responsibility for releasing this material on behalf of any and all co-authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the Supplementary Information.
Rights and permissions
About this article
Cite this article
Guimarães, P.P.G., de Menezes, A.C., Teixeira, K.I.R. et al. Enhanced efficacy against bacterial biofilms via host:guest cyclodextrin‐doxycycline inclusion complexes. J Incl Phenom Macrocycl Chem 99, 197–207 (2021). https://doi.org/10.1007/s10847-020-01041-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-020-01041-7