Abstract
Purpose
To compare aneuploidy rates in early aborted tissues or blastocysts between in vitro fertilization (IVF) cycles after the gonadotropin-releasing hormone (GnRH) antagonist (GnRH-ant) protocol or the GnRH agonist (GnRH-a) long protocol.
Methods
This was a retrospective cohort study from a university-affiliated fertility center. In total, 550 early miscarriage patients who conceived through IVF/intracytoplasmic sperm injection (ICSI) after receiving the GnRH-ant or GnRH-a long protocol were analyzed to compare aneuploidy rates in early aborted tissues. To compare aneuploidy rates in blastocysts, 404 preimplantation genetic testing for aneuploidy (PGT-A) cycles with the GnRH-ant protocol or GnRH-a long protocol were also analyzed.
Results
For early miscarriage patients who conceived through IVF/ICSI, compared to the GnRH-a long protocol group, the GnRH-ant protocol group had a significantly higher rate of aneuploidy in early aborted tissues (48.51% vs. 64.19%). Regarding PGT-A cycles, the rate of blastocyst aneuploidy was significantly higher in the GnRH-ant protocol group than the GnRH-a long protocol group (39.69% vs. 52.27%). After stratification and multiple linear regression, the GnRH-ant regimen remained significantly associated with an increased risk of aneuploidy in early aborted tissues and blastocysts [OR (95% CI) 1.81 (1.21, 2.71), OR (95% CI) 1.65 (1.13, 2.42)]. Furthermore, the blastocyst aneuploidy rate in the GnRH-ant protocol group was significantly higher but only in young and normal ovarian responders [OR (95% CI) 5.07 (1.99, 12.92)].
Conclusion
Compared to the GnRH-a long protocol, the GnRH-ant protocol is associated with a higher aneuploidy rate in early aborted tissues and blastocysts. These results should be confirmed in a multicenter, randomized controlled trial.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Aneuploidy is one of the most detrimental factors causing failed implantation, miscarriage, and disordered embryo development [1]. The vast majority of human aneuploidies arise from errors in the first meiotic division of the oocyte, which is initiated prenatally and is not complete until ovulation [2]. Recent studies have confirmed that both young and older women have high rates of aneuploidy [1, 3]. However, the proportions of human preimplantation embryos with aneuploidy are increased in patients of advanced maternal age [4, 5]. In addition to maternal age, chromosome abnormalities in embryos may be induced by factors involved in assisted reproductive technology (ART), including embryo culture conditions [6]. Ovarian stimulation plays a crucial role in ART. The goal of ovarian stimulation is to induce ongoing development of multiple dominant follicles and obtain many mature oocytes to improve the chances of conception [7]. However, the potential deleterious effect of ovarian stimulation on oocyte and embryo euploidy is still the subject of lively debate. In 2020, Gang Li et al. confirmed that the long-acting gonadotropin-releasing hormone (GnRH)-a protocol after follicular phase (FP) stimulation resulted in a lower aneuploidy blastocyst rate than the short-acting GnRH agonist (GnRH-a) protocol after luteal phase (LP) stimulation [8]. It was also found that the duration of ovarian stimulation treatment was correlated with the aneuploidy rate: patients requiring more days of stimulation presented a lower rate of aneuploid embryos[9]. However, Filippo Maria Ubaldi et al. [10] demonstrated no differences in the aneuploidy blastocyst rate after FP and LP stimulations using follicle-stimulating hormone (FSH) and luteinizing hormone (LH) in combination with GnRH-ant in patients with reduced ovarian reserve.
The GnRH agonist and GnRH-ant protocols are well-established methods for controlled ovarian hyperstimulation among patients who are receiving assisted reproductive technology (ART) [11]. The GnRH-a protocol was developed in the 1980s and is used to prevent premature LH outpouring, thereby increasing the number of retrieved oocytes and pregnancy rates. The GnRH-a protocol has become the gold standard for IVF. The GnRH-ant protocol was introduced in the 1990s; it can competitively block GnRH receptors and cause a rapid suppression of Gn release. The GnRH-ant protocol results in a lower risk of severe ovarian hyperstimulation syndrome (OHSS) and is more convenient for patients because of the shorter duration of Gn administration and lower Gn dose [11, 12].
Current reports are not consistent in comparing the clinical outcomes of the GnRH-a and GnRH-ant protocols based on prior meta-analysis and system review studies. Wang et al. [12] and Xiao et al. [13] showed that compared with the standard long GnRH-a protocol, the GnRH-ant protocol resulted in similar ongoing pregnancy and live birth rates. Yang et al. [11] revealed that the long-acting GnRH-a protocol was beneficial in improving the clinical pregnancy rate and live birth rate compared with the GnRH antagonist protocol. Accounting for various patient populations, C.B. Lambalk et al. [14] systematically reviewed the GnRH-ant and GnRH-a long protocols in IVF. This meta-analysis confirmed that compared to the GnRH-a long protocol, the GnRH-ant protocol was associated with lower ongoing pregnancy rates in a general IVF population but did not seem to compromise ongoing pregnancy rates in individuals with polycystic ovarian syndrome (PCOS) and poor responders. Studies also suggest that embryos conceived after GnRH antagonists may have higher early pregnancy loss rates [15,16,17]. Furthermore, the Chinese birth cohort study on ART and birth defects published in The Lancet Regional Health, 2021, showed that the GnRH-ant regimen for ovulation induction in women was associated with an increased risk of birth defects in their offspring [18]. It is evident that aneuploidy is a major factor that results in pregnancy loss and birth defects [19,20,21]. However, to date, no study has compared the effects of the GnRH-ant protocol and GnRH-a long protocol on embryo aneuploidy.
Prior to analyze the embryo aneuploidy, we first compared the abortion rates of the GnRH-a long protocol or GnRH-ant protocol in our center. In the multivariable model, when maternal age, paternal age, and potential influencing factors, including the basal estradiol (E2) level, infertility diagnosis, infertility factors, total dose of Gn, and duration of Gn, were considered, the GnRH-ant regimen remained significantly associated with an increased risk of nonpregnancy and miscarriage [(OR 1.16, 95% CI 1.04–1.29, P = 0.0094) and (OR 1.40, 95% CI 1.11–1.77, P = 0.0044)] (Supplemental Table 1).
The key of this study is aim to analyze the aneuploidy rate in early aborted tissues after receiving the GnRH-ant or GnRH-a long protocol. We elucidated whether women who received the GnRH-ant protocol for ovarian stimulation generate different aneuploid embryos than those who received the GnRH-a long protocol. These findings help to reveal the different effects of the GnRH-ant and GnRH-a long protocols on blastocyst aneuploidy and provide guidance for clinical treatment.
Materials and methods
Inclusion and exclusion criteria
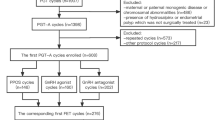
This was a retrospective study to investigate the effect of ovarian stimulation on embryo aneuploidy. The data were collected at a single tertiary reproductive medical center between January 2017 and December 2020. This study included two separate cohorts: miscarriage patients resulting from IVF/ICSI and patients underwent preimplantation genetic testing (PGT). Miscarriage patients with multiple pregnancies, second trimester abortions, unexplained infertility, basic data deficiency, and chromosomal abnormity were excluded. In total, 550 patients with a singleton pregnancy and early miscarriage (gestational age 6–12 weeks) undergoing IVF/ICSI were included. PGT patients with chromosomal abnormities who underwent PGT for chromosomal structural rearrangement (PGT-SR) and patients who underwent PGT for aneuploidy (PGT-A) but embryo amplification failure, mosaic embryos, or basic data deficiency were also excluded. In total, 1476 blastocysts from 404 cycles undergoing PGT for aneuploidy (PGT-A) were included. The GnRH-a long protocol or the GnRH-ant protocol was used for ovarian stimulation. A flow chart of the data processing procedure is provided in Supplemental Fig. 1.
This study was approved by the ethics committee of the Tang Du Hospital of the Air Force Military Medical University, China (K202108-09).
Clinical characteristics
Baseline demographic parameters, including age (in years, y) of the maternal and paternal subjects, body mass index (BMI) (kg/m2), infertility factors, and infertility diagnosis, were collected. Baseline IVF-specific data, including the basal plasma FSH level (mIU/mL), basal LH level (mIU/mL), and basal E2 level (pg/mL), all measured on menstrual cycle day 2 or day 3, were also collected. Anti-Mullerian hormone (AMH) levels (ng/mL) measured on any day of the menstrual cycle were also documented. Controlled ovarian stimulation (COS) parameters documented for all patients included the ovulation-inducing scheme, duration of ovarian stimulation (days), and total dose of gonadotropins (IU). Chromosomal aneuploidy of embryo or early aborted tissues after next-generation sequencing (NGS) diagnosis.
COS protocols and oocyte retrieval
For the GnRH-a long protocol, subcutaneous injection of 0.05–0.1 mg triptorelin acetate for injection (IPSEN, France) was started between the 18th and 21st day of the menstrual cycle before the IVF/ICSI cycle and was continued for 12–14 days. Downregulation was confirmed by a linear endometrium, as observed by ultrasonography, and suppressed ovaries, as indicated by a concentration of serum estradiol < 50 pg/mL. Recombinant human follitropin alpha for injection (rFSH, Merck Serono, USA) stimulation commenced with a dose of 50–300 IU from the 2nd to 5th days in the menstrual period in the IVF/ICSI treatment cycle and was continued until oocyte retrieval.
For the GnRH-ant protocol, the initial dose of rFSH ranged from 112.5 to 200 IU per day on day 2 or day 3 of the menstrual cycle. A gonadotropin-releasing hormone antagonist (cetrorelix acetate, Baxter Oncology GmbH, Germany) at a daily dose of 250 µg was started after 4–5 days of rFSH initiation until the day of hCG administration.
For both protocols, the initial dose of rFSH was based on age, antral follicle count, and basal FSH levels, and the ovarian response was monitored through serum sex steroids and serial transvaginal ultrasound examinations during stimulation. Gonadotropin doses were adjusted when needed. Human menopausal gonadotropin (Lizhu, China) or recombinant Human Lutropin Alfa for Injection (Merck Serono, USA) could be added. Urinary human chorionic gonadotropin was administered at a dose of 5000 to 10,000 IU to induce oocyte maturation when two or more follicles measured 17 mm or more. Oocyte retrieval was performed 34 to 36 h later. A subsequent frozen-thawed embryo transfer (FET) under hormone replacement treatment (HRT) was subsequently undertaken.
IVF and embryo culture
Semen samples were collected by masturbation after 3–7 days of sexual abstinence, on the day of ovum pick-up. After sperm density gradient centrifugation (Vitrolif, Göteborg, Sweden), fertilization was performed by IVF, intracytoplasmic sperm injection (ICSI), or a combination of IVF and ICSI, either by an active choice of half IVF and half ICSI or passive use of rescue ICSI; the latter was applied when conventional IVF failure was confirmed by the fact that two polar bodies could not be observed 6 h after insemination. All normally fertilized embryos were cultured in sequential medium, and the culture fluid was replaced on the third day of embryo culture (CM/BM, Cook, Sydney). Embryo vitrification and warming were performed in accordance with the method reported by Ozgur and colleagues [22].
Biopsy and PGT
In PGT patients, blastocyst biopsy was performed by removing five to eight trophectoderm cells from day 5 or day 6 fully expanded blastocysts through a small opening in the zona pellucida, which was created by laser, with a pipette. The biopsied trophoblasts were lysed, and DNA was extracted and amplified by the SurePlex DNA amplification system (Illumina, USA). Chromosomal aneuploidy screening was performed using NGS-based technology with preimplantation chromosomal aneuploidy detection kit-semiconductor sequencing (Suzhou Basecare Medical Co., Ltd., China). Each sample met the 0.1-fold average sequencing depth.
Acquisition of and testing early aborted tissues
The early aborted tissues of early miscarriage patients as long as fetal or villous tissue is present were rinsed with 0.9% saline solution, and then DNA was extracted from 0.5 cm3 sections with a QIAamp DNA Mini Kit (Qiagen, Germany). Chromosomal aneuploidy screening was performed using a MiSeq® Dx Reagent Kit V3 (Illumina, USA). Each sample met the 0.25-fold average sequencing depth.
Statistical analysis
Data processing and statistical analysis were performed using EmpowerStats software (www.empowerstats.com) and the statistical software package R.
Categorical variables are expressed as the number (n) and percentage (%), and continuous variables are expressed as the mean ± standard deviation (SD). Normality was assessed through the Shapiro–Wilk normality test in addition to visual inspection of the distribution. Subject characteristics in the GnRH-a long protocol group and the GnRH-ant protocol group are described separately. Student’s t test was applied for the primary comparison between the two groups. To assess the odds ratio (OR) of aneuploidy in the two groups, a multivariable regression model was established with potential confounding factors as the variables and adjusted for maternal age, maternal BMI, paternal age, infertility diagnosis, infertility factors, basal plasma levels of FSH, LH and E2, menstrual cycle day 2 or day 3, AMH, duration of ovarian stimulation, and total dose of gonadotropins. Stratified analysis by AMH (< 1.3 ng/mL, ≥ 1.3 ng/mL < 3.36 ng/mL, ≥ 3.36 ng/mL) and maternal age (< 35 years, ≥ 35 years) was also conducted to assess the primary outcome of embryo aneuploidy in PGT patients between the two COS groups. Differential types of chromosome aneuploidy in early aborted tissues from the two COS groups were examined using chi-square analysis.
Results
Effect of the ovarian stimulation protocol on the rate of aneuploidy in early aborted tissues in patients receiving IVF/ICSI
Baseline characteristics of early miscarriage patients
In total, 550 early miscarriage patients conceived through IVF/ICSI, and the demographics and clinical characteristics of those who received the GnRH-ant and GnRH-a long protocols were compared. A total of 402 patients in the GnRH-a long protocol group and 148 patients in the GnRH-ant protocol were analyzed. The baseline characteristics of the patients are presented in Table 1. As shown in Table 1, the rate of aneuploidy in early aborted tissues in patients in the GnRH-ant protocol group was significantly higher than that in patients in the GnRH-a long protocol group [95 (64.19%) vs. 195 (48.51%)]. In addition, women in the GnRH-ant protocol group had a higher basal plasma FSH level but received a shorter duration of ovarian stimulation. In addition, there was a statistically significant distribution of infertility factors between the two groups.
GnRH antagonists increase the aneuploidy rate in early aborted tissues in patients receiving IVF/ICSI
We assessed whether any specific infertility diagnosis or clinical characteristic was associated with the risk of aneuploidy in aborted tissues. We observed significant associations of aneuploidy with maternal and paternal ages (Supplemental Tables 2). When assessing specific ovarian stimulation procedures, the GnRH-ant protocol was associated with an increased risk of aneuploidy compared with the GnRH-a long protocol (OR 1.90, 95% CI 1.29–2.81, P = 0.0012) (Supplemental Table 2). We observed weak associations of aneuploidy with other exposures.
Due to the significant relationship between aneuploidy in early aborted tissues and the GnRH-ant protocol observed, multivariable regression analysis was performed to corroborate the impact of the GnRH-ant protocol on aneuploidy. In the multivariable model, when maternal age, paternal age, and potential influencing factors, including the basal E2 level, infertility diagnosis, and total dose of Gn, were considered, the GnRH-ant regimen remained significantly associated with an increased risk of aneuploidy in early aborted tissues (P = 0.0037) with an adjusted odds ratio of 1.81 (95% confidence interval, 1.21–2.71) (Table 3).
Effect of the ovarian stimulation protocol on blastocyst aneuploidy in PGT cycles
Baseline characteristics of blastocysts from PGT cycles
In the present study, a total of 1476 blastocysts from 404 cycles were obtained, of which 1212 blastocysts were obtained from 323 cycles receiving the GnRH-a long protocol and 264 blastocysts were obtained from 81 cycles receiving the GnRH-ant protocol. The baseline characteristics of the patients are presented in Table 2. As shown in Table 2, significant differences were observed in the rate of aneuploidy between the GnRH-a long protocol and GnRH-ant protocol groups, and the rate of aneuploidy was significantly higher in the GnRH-ant protocol group [138(52.27%) vs. 481(39.69%)].
GnRH antagonists increase the blastocyst aneuploidy rate in PGT cycles
We assessed whether any specific infertility diagnosis or clinical characteristic was associated with the risk of blastocyst aneuploidy. We observed significant associations of blastocyst aneuploidy with maternal and paternal ages (Supplemental Table 3). When assessing specific ovarian stimulation procedures, the GnRH-ant protocol was associated with an increased risk of aneuploidy compared with the GnRH-a long protocol (OR 1.66, 95% CI 1.13–2.44, P = 0.0094) (Supplemental Table 3). We observed null associations of aneuploidy with other clinical factors.
Multivariable regression analysis was performed to corroborate the impact of the GnRH-ant protocol on the aneuploidy of blastocysts. In the multivariable model, when maternal age, paternal age, and potential influencing factors, including basal E2 level, PGT indications, and total dose of Gn, were considered, the GnRH-ant regimen remained significantly associated with an increased risk of aneuploidy in blastocysts from PGT cycles (P = 0.0102) with an adjusted odds ratio of 1.65 (95% confidence interval, 1.13–2.42) (Table 3).
Comparison of blastocyst aneuploidy in PGT patients receiving the two stimulation protocols stratified by AMH and age using multivariable regression analysis
The ovarian response to gonadotropins may be related to oocyte aneuploidy, and advanced maternal age is the determining factor of embryo aneuploidy. AMH is a key predictive biomarker for the ovarian response. The aneuploidy rate between the two groups was stratified by AMH and maternal age. We stratified the patients into expected low (AMH < 1.30 ng/mL), medium (AMH ≥ 1.30 ng/mL, and AMH < 3.36 ng/mL), and high (AMH ≥ 3.36 ng/mL) response groups and two age groups (< 35 years and ≥ 35 years).
As shown in Table 4, the multivariable regression stratified analysis showed that women younger than 35 years and with an AMH concentration between 1.30 and 3.36 ng/mL had a significantly higher rate of blastocyst aneuploidy after receiving the GnRH-ant protocol (P = 0.0007) with an adjusted odds ratio of 5.07 (95% confidence interval, 1.99–12.96). This tendency was not observed in other groups.
Interactions between the AMH subgroups and the stimulation protocols were tested in two subgroups according to maternal age, and the results showed that all the AMH subgroups failed to have an interaction effect with the stimulation protocol on blastocyst aneuploidy.
Discussion
The present study suggested that the GnRH-ant protocol group had a significantly higher rate of aneuploidy in early aborted tissues than the GnRH-a long protocol group. This study also demonstrated that the GnRH-ant protocol was associated with a higher blastocyst aneuploidy rate than the GnRH-a long protocol in patients receiving PGT-A cycles. Furthermore, this study showed that the blastocyst aneuploidy rate in the GnRH-ant protocol was significantly higher than that in the GnRH-a long protocol in young and normal ovarian responders. These results suggest that an increased blastocyst aneuploidy rate may account for the higher early pregnancy loss rate associated with the GnRH-ant protocol. This is the first study that evaluated whether a difference in aneuploidy rates exists between the GnRH-ant protocol and GnRH-a long protocol.
Aneuploidy is one of the most detrimental factors causing failed implantation, miscarriage, and disordered embryo development [23]. Many researchers have explored the influencing factors of embryonic aneuploidy. Several studies in animal oocytes have suggested increased aneuploidy rates after ovulation induction [24, 25]. In humans, studies have also reported that the proportions of human preimplantation embryos with aneuploidy are increased by ovarian stimulation [26, 27]. Studies have also suggested that a higher daily dose of gonadotropin during IVF is associated with greater aneuploidy rates than a lower daily dose of gonadotropin [28, 29]. It was also found that patients requiring more days of stimulation presented a lower rate of aneuploidy embryos [9]. Conversely, Labarta et al. showed that ovarian stimulation did not significantly increase the embryo aneuploidy rate in IVF-derived human embryos compared with an unstimulated cycle (24). Three retrospective studies of Preimplantation Genetic Screening (PGS) outcome data showed that the total dose of exogenous gonadotropins was not significantly associated with blastocyst aneuploidy and that a high dosage of gonadotropin did not affect euploidy or pregnancy rates [30,31,32]. A retrospective cohort study that included 2230 embryos conceived from IVF that underwent PGT-A also demonstrated that the gonadotropin dosage, duration of ovarian stimulation, estradiol level, follicle size at ovulation trigger, and number of oocytes retrieved, within certain ranges, do not appear to significantly influence euploidy rates, regardless of the woman’s age [33]. Therefore, the potential deleterious effect of ovarian stimulation on oocyte and embryo euploidy is still the subject of lively debate. The conflicting results may be attributed to the operation of different physicians in different IVF centers. It has been found that euploidy rates for embryos created using donor oocytes can vary significantly between different IVF centers and even between donors treated by different physicians at the same IVF center [26, 34]. Studies have also demonstrated different mosaicism rates between IVF centers, implicating differences in stimulation protocols as a potential reason. Therefore, in the present study, we compared the effects of the GnRH-ant protocol and GnRH-a long protocol on embryo aneuploidy.
It has been reported that in a general IVF population, the GnRH-ant protocol is associated with lower ongoing pregnancy rates than the GnRH-a long protocol [14, 35]. Studies have also suggested that embryos conceived after receiving GnRH antagonists may be associated with higher early pregnancy loss rates [15,16,17]. It is evident that aneuploidy is a major factor that results in early pregnancy loss [19]. However, the rate of aneuploidy in early aborted tissues was not evaluated in patients who conceived after receiving the GnRH-ant or GnRH-a long protocol. In the present study, we noted a difference in aneuploidy rates between our two cohorts and found that the GnRH-ant protocol was associated with a significantly higher aneuploidy rate. Univariable analysis revealed that maternal age and paternal age were associated with the aneuploidy rate in aborted tissues. Furthermore, after regression analysis controlling for age, the basal E2 level, infertility diagnosis, and the total dose of Gn, this difference in aneuploidy rates between the GnRH-ant and GnRH-a long protocol groups was still statistically significant. The higher aneuploidy rate may be a major detrimental factor that results in the increased risk of miscarriage in the GnRH-ant group.
We proposed that the higher aneuploidy rates in aborted tissues in the GnRH-ant protocol group may be derived from the preimplantation embryo. Based on this hypothesis, we compared the rates of aneuploidy in blastocysts treated with GnRH antagonists and long agonists in PGT-A cycles. Given the potential variation in different PGT-A testing platforms, we evaluated blastocysts using NGS only [34]. The rate of blastocyst aneuploidy was significantly higher in the GnRH-ant protocol group. In the multivariable model, when maternal age, paternal age, and potential influencing factors, including PGT indications, the basal E2 level, and the total dose of Gn, were considered, the GnRH-ant regimen remained significantly associated with an increased risk of blastocyst aneuploidy in women receiving PGT cycles. It also found that women younger than 35 years and with a normal AMH concentration had a significantly higher rate of blastocyst aneuploidy after receiving the GnRH-ant protocol than after receiving the GnRH-a long protocol. Previous studies reported that embryonic aneuploidy rates were not influenced by the dose of gonadotropins used in ovarian stimulation [30, 31]. Our study results reinforce the idea of their findings, showing that blastocyst aneuploidy is independent of the dose of gonadotropins in both the GnRH-ant and GnRH-a long protocols. Previous data have indicated that the aneuploidy rates in embryos produced from eggs collected from ovarian stimulation are between 39.1 and 53.2%, which are higher than the proportion of eggs with abnormal chromosomes in young women during the natural cycle (17%) [4, 36]. Our present data indicate that the aneuploidy rate in embryos produced from the GnRH-ant protocol is 52.27%, excluding mosaic and amplification failure.
The effect of the hormones administered during stimulation at the cellular level is still unknown. It has been reported that mitochondrial dysfunction that causes a decrease in ATP and/or an increase in reactive oxygen species (ROS) is sufficient to disrupt meiotic spindles [37, 38]. The percentage of mitochondria that were vacuolated in oocytes was significantly increased after ovarian stimulation in mice [39]. Moreover, the ratio of activated mitochondria to inactivated mitochondria and ATP synthesis in mouse oocytes decreased after ovarian stimulation [39]. Evidence has also demonstrated that repeated superovulation has adverse effects on the mitochondrial function of cumulus cells in rhesus monkeys or mice [40, 41]. Evidence has also indicated that repeated superovulation induces oxidative stress by elevating ROS levels in oocytes [42]. A significant decrease in ATP generation and increase in ROS caused by ovarian stimulation may adversely affect chromosomal segregation and meiotic spindle adjunction, which result in embryo aneuploidy [43, 44]. It has been reported that the use of antagonists may present an endocrinologically unfavorable scenario in which the suppression of endogenous pituitary gonadotropin secretion may be insufficient [14]. The nonphysiological microenvironment caused by GnRH-ant may result not only in abnormal follicular fluid biochemistry but also aberrant oocyte cytoplasmic development, which compromises mitochondrial function. However, no report has compared the effects of the GnRH-ant and GnRH-a long protocols on mitochondria and metabolism in human oocytes. In the future, it will be necessary to analyze the effects of GnRH antagonists on oocyte mitochondria and ATP synthesis.
Our study has several strengths and a few limitations. Strengths included its relatively large sample size from a single state over several years, which included 550 early miscarriage patients and a total of 404 cycles after receiving the GnRH-ant or GnRH-a long protocol. We controlled for maternal age, paternal age, basal E2 level, infertility diagnosis, PGT indications, and total dose of Gn as confounders in the study.
This study has some limitations. First, it was retrospective in nature, and some key statistical parameters may not have been calculated. Second, the analyses performed in the study showed associations between an increased rate of embryonic aneuploidy and the GnRH-ant protocol, but they did not establish causality. In addition, this study was performed at a single IVF center, which may limit its generalizability. As euploidy rates for embryos created using donor oocytes can vary significantly between different IVF centers and even between donors treated by different physicians at the same IVF center [26, 34], a multicenter, randomized controlled trial would be the optimal strategy to confirm these results.
Conclusion
This study demonstrated that during IVF, the aneuploidy rate in early aborted tissues and blastocysts was significantly higher after receiving the GnRH-ant protocol than after receiving the GnRH-a long protocol. GnRH antagonists may be associated with detrimental effects on embryo quality, and the higher early pregnancy loss rates associated with GnRH antagonists may originate from the higher aneuploidy rate. Prospective, multicenter, and randomized controlled trials are needed to corroborate these findings and obviate the problems of bias and confounding by interdependent variables.
Data availability
All datasets generated for this study are included in the article/Supplementary Material.
References
Gruhn JR, Zielinska AP, Shukla V, Blanshard R, Capalbo A, Cimadomo D, Nikiforov D, Chan AC, Newnham LJ, Vogel I, et al. Chromosome errors in human eggs shape natural fertility over reproductive life span. Science (New York, NY). 2019;365(6460):1466–9.
Rai R, Regan L. Recurrent miscarriage. Lancet (London, England). 2006;368(9535):601–11.
Franasiak JM, Forman EJ, Hong KH, Werner MD, Upham KM, Treff NR, Scott RT Jr. The nature of aneuploidy with increasing age of the female partner: a review of 15,169 consecutive trophectoderm biopsies evaluated with comprehensive chromosomal screening. Fertil Steril. 2014;101(3):656-663.e651.
Haddad G, Deng M, Wang CT, Witz C, Williams D, Griffith J, Skorupski J, Gill J, Wang WH. Assessment of aneuploidy formation in human blastocysts resulting from donated eggs and the necessity of the embryos for aneuploidy screening. J Assist Reprod Genet. 2015;32(6):999–1006.
Mikwar M, MacFarlane AJ, Marchetti F. Mechanisms of oocyte aneuploidy associated with advanced maternal age. Mutat Res. 2020;785:108320.
Derrick R, Hickman C, Oliana O, Wilkinson T, Gwinnett D, Whyte LB, Carby A, Lavery S. Perivitelline threads associated with fragments in human cleavage stage embryos observed through time-lapse microscopy. Reprod Biomed Online. 2017;35(6):640–5.
Macklon NS, Stouffer RL, Giudice LC, Fauser BC. The science behind 25 years of ovarian stimulation for in vitro fertilization. Endocr Rev. 2006;27(2):170–207.
Li G, Wu Y, Niu W, Xu J, Hu L, Shi H, Sun Y. Analysis of the number of euploid embryos in preimplantation genetic testing cycles with early-follicular phase long-acting gonadotropin-releasing hormone agonist long protocol. Front Endocrinol. 2020;11:424.
Cascales A, Lledo B, Ortiz JA, Morales R, Ten J, Llacer J, Bernabeu R. Effect of ovarian stimulation on embryo aneuploidy and mosaicism rate. Syst Biol Reprod Med. 2021;67(1):42–9.
Ubaldi FM, Capalbo A, Vaiarelli A, Cimadomo D, Colamaria S, Alviggi C, Trabucco E, Venturella R, Vajta G, Rienzi L. Follicular versus luteal phase ovarian stimulation during the same menstrual cycle (DuoStim) in a reduced ovarian reserve population results in a similar euploid blastocyst formation rate: new insight in ovarian reserve exploitation. Fertil Steril. 2016;105(6):1488-1495.e1481.
Yang R, Guan Y, Perrot V, Ma J, Li R. Comparison of the long-acting GnRH agonist follicular protocol with the GnRH antagonist protocol in women undergoing in vitro fertilization: a systematic review and meta-analysis. Adv Ther 2021.
Wang R, Lin S, Wang Y, Qian W, Zhou L. Comparisons of GnRH antagonist protocol versus GnRH agonist long protocol in patients with normal ovarian reserve: a systematic review and meta-analysis. PLoS ONE. 2017;12(4):e0175985.
Xiao JS, Su CM, Zeng XT. Comparisons of GnRH antagonist versus GnRH agonist protocol in supposed normal ovarian responders undergoing IVF: a systematic review and meta-analysis. PLoS ONE. 2014;9(9):e106854.
Lambalk CB, Banga FR, Huirne JA, Toftager M, Pinborg A, Homburg R, van der Veen F, van Wely M. GnRH antagonist versus long agonist protocols in IVF: a systematic review and meta-analysis accounting for patient type. Hum Reprod Update. 2017;23(5):560–79.
Bahceci M, Ulug U, Sismanoglu A, Tosun S, Cengiz B. Early pregnancy loss rates were different among singleton gestations conceived by ICSI using GnRH agonist and antagonist. J Assist Reprod Genet. 2009;26(4):227–9.
Maldonado LG, Franco JG Jr, Setti AS, Iaconelli A Jr, Borges E Jr. Cost-effectiveness comparison between pituitary down-regulation with a gonadotropin-releasing hormone agonist short regimen on alternate days and an antagonist protocol for assisted fertilization treatments. Fertil Steril. 2013;99(6):1615–22.
Hu L, Du J, Lv H, Zhao J, Chen M, Wang Y, Wu F, Liu F, Chen X, Zhang J, et al. Influencing factors of pregnancy loss and survival probability of clinical pregnancies conceived through assisted reproductive technology. Reprod Biol Endocrinol. 2018;16(1):74.
Lv H, Diao F, Du J, Chen T, Meng Q, Ling X, Li H, Song C, Xi Q, Jiang Y et al. Assisted reproductive technology and birth defects in a Chinese birth cohort study. Lancet Reg Health - West Pac. 2021;7.
Colley E, Hamilton S, Smith P, Morgan NV, Coomarasamy A, Allen S. Potential genetic causes of miscarriage in euploid pregnancies: a systematic review. Hum Reprod Update. 2019;25(4):452–72.
Hartman RJ, Rasmussen SA, Botto LD, Riehle-Colarusso T, Martin CL, Cragan JD, Shin M, Correa A. The contribution of chromosomal abnormalities to congenital heart defects: a population-based study. Pediatr Cardiol. 2011;32(8):1147–57.
Davis AR, Horvath SK, Castano PM. Trends in gestational age at time of surgical abortion for fetal aneuploidy and structural abnormalities. Am J Obstet Gynecol. 2017;216(3):278 e271-278 e275.
Ozgur K, Berkkanoglu M, Bulut H, Humaidan P, Coetzee K. Perinatal outcomes after fresh versus vitrified-warmed blastocyst transfer: retrospective analysis. Fertil Steril. 2015;104(4):899-907.e893.
Thorne J, Loza A, Kaye L, Nulsen J, Benadiva C, Grow D, Engmann L. Euploidy rates between cycles triggered with gonadotropin-releasing hormone agonist and human chorionic gonadotropin. Fertil Steril. 2019;112(2):258–65.
Spielmann H, Vogel R. Genotoxic and embryotoxic effects of gonadotropin hyperstimulated ovulation on murine oocytes, preimplantation embryos and term fetuses. Ann Ist Super Sanita. 1993;29(1):35–9.
Roberts R, Iatropoulou A, Ciantar D, Stark J, Becker DL, Franks S, Hardy K. Follicle-stimulating hormone affects metaphase I chromosome alignment and increases aneuploidy in mouse oocytes matured in vitro. Biol Reprod. 2005;72(1):107–18.
McCulloh DH, Alikani M, Norian J, Kolb B, Arbones JM, Munne S. Controlled ovarian hyperstimulation (COH) parameters associated with euploidy rates in donor oocytes. Eur J Med Genet. 2019;62(8):103707.
Sachdeva K, Upadhyay D, Discutido R, Varghese MM, Albuz F, Almekosh R, Bouhafs L, Solkar S, Stevikova M, Peramo B. Low gonadotropin dosage reduces aneuploidy in human preimplantation embryos: first clinical study in a UAE population. Genet Test Mol Biomarkers. 2018;22(10):630–4.
Baart EB, Martini E, Eijkemans MJ, Van Opstal D, Beckers NG, Verhoeff A, Macklon NS, Fauser BC. Milder ovarian stimulation for in-vitro fertilization reduces aneuploidy in the human preimplantation embryo: a randomized controlled trial. Hum Reprod (Oxford, England). 2007;22(4):980–8.
Katz-Jaffe MG, Trounson AO, Cram DS. Chromosome 21 mosaic human preimplantation embryos predominantly arise from diploid conceptions. Fertil Steril. 2005;84(3):634–43.
Wu Q, Li H, Zhu Y, Jiang W, Lu J, Wei D, Yan J, Chen ZJ. Dosage of exogenous gonadotropins is not associated with blastocyst aneuploidy or live-birth rates in PGS cycles in Chinese women. Hum Reprod. 2018;33(10):1875–82.
Barash OO, Hinckley MD, Rosenbluth EM, Ivani KA, Weckstein LN. High gonadotropin dosage does not affect euploidy and pregnancy rates in IVF PGS cycles with single embryo transfer. Hum Reprod. 2017;32(11):2209–17.
Sekhon L, Shaia K, Santistevan A, Cohn KH, Lee JA, Beim PY, Copperman AB. The cumulative dose of gonadotropins used for controlled ovarian stimulation does not influence the odds of embryonic aneuploidy in patients with normal ovarian response. J Assist Reprod Genet. 2017;34(6):749–58.
Irani M, Canon C, Robles A, Maddy B, Gunnala V, Qin X, Zhang C, Xu K, Rosenwaks Z. No effect of ovarian stimulation and oocyte yield on euploidy and live birth rates: an analysis of 12 298 trophectoderm biopsies. Hum Reprod (Oxford, England). 2020;35(5):1082–9.
Munne S, Alikani M, Ribustello L, Colls P, Martinez-Ortiz PA, McCulloh DH, Referring Physician G. Euploidy rates in donor egg cycles significantly differ between fertility centers. Hum Reprod. 2017;32(4):743–9.
Yang R, Guan Y, Perrot V, Ma J, Li R. Comparison of the long-acting GnRH agonist follicular protocol with the GnRH antagonist protocol in women undergoing in vitro fertilization: a systematic review and meta-analysis. Adv Ther. 2021;38(5):2027–37.
Battaglia DE, Goodwin P, Klein NA, Soules MR. Influence of maternal age on meiotic spindle assembly in oocytes from naturally cycling women. Hum Reprod. 1996;11(10):2217–22.
Zeng HT, Ren Z, Yeung WS, Shu YM, Xu YW, Zhuang GL, Liang XY. Low mitochondrial DNA and ATP contents contribute to the absence of birefringent spindle imaged with PolScope in in vitro matured human oocytes. Hum Reprod. 2007;22(6):1681–6.
Zhang X, Wu XQ, Lu S, Guo YL, Ma X. Deficit of mitochondria-derived ATP during oxidative stress impairs mouse MII oocyte spindles. Cell Res. 2006;16(10):841–50.
Lee M, Ahn JI, Lee AR, Ko DW, Yang WS, Lee G, Ahn JY, Lim JM. Adverse effect of superovulation treatment on maturation, function and ultrastructural integrity of murine oocytes. Mol Cells. 2017;40(8):558–66.
Xie JK, Wang Q, Zhang TT, Yin S, Zhang CL, Ge ZJ. Repeated superovulation may affect mitochondrial functions of cumulus cells in mice. Sci Rep. 2016;6:31368.
Dong G, Guo Y, Cao H, Zhou T, Zhou Z, Sha J, Guo X, Zhu H. Long-term effects of repeated superovulation on ovarian structure and function in rhesus monkeys. Fertil Steril. 2014;102(5):1452-1457 e1451.
Xiao P, Nie J, Wang X, Lu K, Lu S, Liang X. Melatonin alleviates the deterioration of oocytes from mice subjected to repeated superovulation. J Cell Physiol. 2019;234(8):13413–22.
Al-Zubaidi U, Adhikari D, Cinar O, Zhang QH, Yuen WS, Murphy MP, Rombauts L, Robker RL, Carroll J. Mitochondria-targeted therapeutics, MitoQ and BGP-15, reverse aging-associated meiotic spindle defects in mouse and human oocytes. Hum Reprod. 2021;36(3):771–84.
Perkins AT, Das TM, Panzera LC, Bickel SE. Oxidative stress in oocytes during midprophase induces premature loss of cohesion and chromosome segregation errors. Proc Natl Acad Sci U S A. 2016;113(44):E6823–30.
Acknowledgements
The authors gratefully acknowledge the patients who participated in this study.
Funding
This work was supported by the National Natural Science Foundation of China (81671463) and the Shaanxi Province Key R & D Program General Project (2021SF-012).
Author information
Authors and Affiliations
Contributions
Jun Wang collected the data and prepared all the tables and wrote the main manuscript, Jing Zhang and Nan Zhao collected the data, Shuqiang Chen searched the literature and designed the study, and Xiaohong Wang revised the manuscript. The author(s) read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This study was approved by the ethics committee of the Tang Du Hospital of the Air Force Military Medical University, China (K202108-09). All patients provided written informed consent.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, J., Zhang, J., Zhao, N. et al. The effect of ovarian stimulation on aneuploidy of early aborted tissues and preimplantation blastocysts: comparison of the GnRH agonist long protocol with the GnRH antagonist protocol. J Assist Reprod Genet 39, 1927–1936 (2022). https://doi.org/10.1007/s10815-022-02557-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-022-02557-2