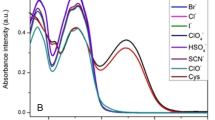
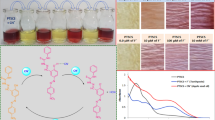
Phenylboronic acids (PBAs) form stable complexes with fluoride. The effect of type (methoxy-, formyl-, and acetyl-) and position (ortho-, meta-, and para-) of electron-donating substitutions on the hydrolytic stability and acidity of PBAs, as well as their spectroscopic and physicochemical properties and their usage in spectrofluorimetric fluoride determination, were investigated. Thermal stabilities, relative predisposition, and resistance to dehydroboronation of related PBA isomers were investigated in detail and compared with the use of thermogravimetric analysis and differential scanning calorimetry profiles. Dehydroboronation reaction leads to the synthesis of related new cyclic anhydric forms — specifically named boroxines — which are clearly distinguished by Fourier transform infrared spectroscopy. PBAs were used for the complexation of fluoride for spectrofluorimetric fluoride determination in dental formulations [toothpastes (TPs) and mouth rinses (MRs)] and beverages [mineral waters (MiWs)]. Determination was done by fluorescence quenching of PBAs in response to increasing fluoride concentration. The regression equation was y = –15.336x + 983.17 (R2 = 0.9931), and was linear in the 1.4–3.0-mM range. Determinations were performed with relative errors (%) in a range of –5.60 to +1.23, –2.01 to +5.69, and –4.16 to +2.54 for MRs, TPs, and MiWs, respectively, relative to fluoride levels of commercial samples. Chemometric analyses (cluster analysis, CA, principal component analysis, PCA) were performed on the same real samples. Raw fluorescence data was investigated by PCA to check their significance in chemometric discrimination. Dendograms and score plots successfully discriminated samples in related groups. This is the first demonstration of spectrofluorometric fluoride determination based on the quenching of related isomers of PBAs thus far, also the potential of raw fluorescence data of these PBAs for chemometric discrimination studies on related pharmaceutical samples was highlighted for the first time.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S. Pillitteri, P. Ranjan, E. V. Van der Eycken, and U. K. Sharma, Adv. Synth. Catal., 364, 1643–1665 (2022), doi: https://doi.org/10.1002/adsc.202200204.
D. G. Hall, Chem. Soc. Rev., 48, 3475–3496 (2019), doi: https://doi.org/10.1039/c9cs00191c.
W. L. A. Brooks and B. S. Sumerlin, Chem. Rev., 116, 1375–1397 (2016), doi: https://doi.org/10.1021/acs.chemrev.5b00300.
R. Wang, Z. Bian, D. Zhan, Z. Wu, Q. Yao, and G. Zhang, Dyes Pigments, 185, Article ID 108885 (2021), doi: https://doi.org/10.1016/j.dyepig.2020.108885.
M. P. Silva, L. Saraiva, M. Pinto, and M. E. Sousa, Molecules, 25, 4323 (2020), doi: https://doi.org/10.3390/molecules25184323.
B. J. Graham, I. W. Windsor, B. Gold, and R. T. Raines, PNAS, 118, e2013691118 (2021), doi: https://doi.org/10.1073/pnas.2013691118.
Y. Y. Aung, A. N. Kristanti, H. V. Lee, and M. Z. Fahmi, ACS Omega, 6, 17750–17765 (2021), doi: https://doi.org/10.1021/acsomega.1c01352.
D. Zarzeczańska, A. Adamczyk-Wozńiak, A. Kulpa, T. Ossowski, and A. Sporzyński, Eur. J. Inorg. Chem., 4493–4498 (2017), doi: https://doi.org/10.1002/ejic.201700546.
A. Adamczyk-Wozńiak, E. Kaczorowska, J. Kredatusova, I. Madura, P. H. Marek, A. Matuszewska, A. Sporzyński, and M. Uchman, Eur. J. Inorg. Chem., 2018, 1492–1498 (2018), doi: https://doi.org/10.1002/ejic.201701485.
A. L. Korich and P. M. Iovine, Dalton Trans., 39, 1423–1431 (2010), doi: https://doi.org/10.1039/b917043j.
N. Dicesare and J. R. Lakowicz, Anal. Biochem., 301, 111–116 (2002), doi: https://doi.org/10.1006/abio.2001.5476.
R. Badugu, J. R. Lakowicz, and C. D. Geddes, Sens. Actuators B Chem., 104, 103–110 (2005), doi: https://doi.org/10.1016/j.snb.2004.04.119.
K. M. K. Swamy, Y. J. Lee, H. N. Lee, J. Chun, Y. Kim, S. J. Kim, and J. Yoon, J. Org. Chem., 71, 8626–8628 (2006), doi: https://doi.org/10.1021/jo061429x.
S. Arimori, M. G. Davidson, T. M. Fyles, T. G. Hibbert, T. D. James, and G. I. Kociok-Köhn, Chem. Commun., 40, 1640–1641 (2004), doi: https://doi.org/10.1039/b404937c.
C. R. Cooper, N. Spencer, and T. D. James, Chem. Commun., 24, 1365–1366 (1998), doi: https://doi.org/10.1039/A801693C.
J. M. Koskela, T. M. Fyles, and T. D. James, Chem. Commun., 41, 945–947 (2005), doi: https://doi.org/10.1039/B415522J.
W. Tan, D. Zhang, and D. Zhu, Bioorg. Med. Chem. Lett., 17, 2629–2633 (2007), doi: https://doi.org/10.1016/j.bmcl.2007.01.099.
J. K. Day, C. Bresner, N. D. Coombs, I. A. Fallis, L. L. Ooi, and S. Aldridge, Inorg. Chem., 47, 793–804 (2008), doi: https://doi.org/10.1021/ic701494p.
M. Nicolas, B. Fabre, and J. Simonet, Electrochim. Acta, 46, 1179–1190 (2001), doi: https://doi.org/10.1016/S0013-4686(00)00694-0.
M. Nicolas, B. Fabre, and J. Simonet, Chem. Commun., 25, 1881–1882 (1999), doi: https://doi.org/10.1039/A905136H.
S. Sole and F. P. Gabbai, Chem. Commun., 40, 1284–1285 (2004), doi: https://doi.org/10.1039/B403596H.
M. Melami, S. Solé, C. W. Chiu, H. Wang, and F. P. Gabbai, Inorg. Chem., 45, 8136–8143 (2006), doi: https://doi.org/10.1021/ic060709s.
W. Tan, D. Zhang, Z. Wang, C. Liu, and D. Zhu, J. Mater. Chem., 17, 1964–1968 (2007), doi: https://doi.org/10.1039/b618183j.
S. Yamaguchi, S. Akiyama, and K. Tamao, J. Am. Chem. Soc., 123, 11372–11375 (2001). doi:https://doi.org/10.1021/ja015957w.
Y. Sert, F. Ucun, and M. Boyukata, Indian J. Phys., 87, 113–119 (2013), doi: https://doi.org/10.1007/s12648-012-0202-3.
O. Alver and G. Dikmen, J. Mol. Struct., 1108, 103–111 (2016), doi: https://doi.org/10.1016/j.molstruc.2015.11.041.
C. Liao and M. C. Nicklaus, J. Chem. Inf. Model., 49, 2801–2812 (2009), doi: https://doi.org/10.1021/ci900289x.
C. Dardonville, Drug Discov., 27, 49–58 (2018), doi: https://doi.org/10.1016/j.ddtec.2018.04.001.
K. Mioduszewska, J. Dolzonek, D. Wyrzykowski, L. Kubik, P. Wiczling, C. Sikorska, M. Tonski, Z. Kaczyński, P. Stepnowski, and A. Bialk-Bielińska, Trend. Anal. Chem., 97, 283–296 (2017), doi: https://doi.org/10.1016/j.trac.2017.09.009.
W. A. Marinaro, L. J. Schieber, E. J. Munson, V. W. Day, and V. J. Stella, J. Pharm. Sci., 101, 310–318 (2012), doi: https://doi.org/10.1002/jps.23207.
C. Liu, Q. Yin, X. Li, L. Hao, W. Zhang, Y. Bao, and J. Ma, Adv. Comp. Hyb. Mater., 4, 138–149 (2021), doi: https://doi.org/10.1007/s42114-021-00206-3.
A. Adamczyk-Wozńiak, M.K. Cyrański, K. Durka, J.T. Gozdalik, P. Klimentowska, R. Rusiecki, A. Sporzyński, and D. Zarzeczańska, Crystals., 9, 109–125 (2019), doi: https://doi.org/10.3390/cryst9020109.
F. G. Der, G. Yalcin, E. Ozcan Bulbul, H. İleri, B. Cihan, M. Gokcan, Y. C. Esitmez, and E. Kilinc, Electroanalysis, 34, 1711–1734 (2022), doi: https://doi.org/10.1002/elan.202200012.
Author information
Authors and Affiliations
Corresponding author
Additional information
Abstract of article is published in Zhurnal Prikladnoi Spektroskopii, Vol. 90, No. 6, p. 969, November–December, 2023.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kilinc, E. Resistance of Acetyl-, Formyl-, and Methoxy-Phenylboronic Acids to Boroxine Formation and Their Employment in Fluoride Determination of Dental Formulations and Beverages by Fluorescence Quenching. J Appl Spectrosc 90, 1358–1371 (2024). https://doi.org/10.1007/s10812-024-01674-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10812-024-01674-3