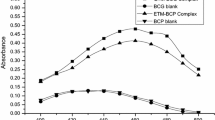
Two simple and sensitive extraction-free spectrophotometric methods are described for the determination of flunarizine dihydrochloride. The methods are based on the ion-pair complex formation between the nitrogenous compound flunarizine (FNZ), converted from flunarizine dihydrochloride (FNH), and the acidic dye phenol red (PR), in which experimental variables were circumvented. The first method (method A) is based on the formation of a yellow-colored ion-pair complex (1:1 drug:dye) between FNZ and PR in chloroform, which is measured at 415 nm. In the second method (method B), the formed drug-dye ion-pair complex is treated with ethanolic potassium hydroxide in an ethanolic medium, and the resulting base form of the dye is measured at 580 nm. The stoichiometry of the formed ion-pair complex between the drug and dye (1:1) is determined by Job's continuous variations method, and the stability constant of the complex is also calculated. These methods quantify FNZ over the concentration ranges 5.0–70.0 in method A and 0.5–7.0 μg/mL in method B. The calculated molar absorptivities are 6.17 × 103 and 5.5 × 104 L/mol·cm–1 for method A and method B, respectively, with corresponding Sandell sensitivity values of 0.0655 and 0.0074 μg/cm2. The methods are applied to the determination of FNZ in pure drug and human urine.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
P. A. Todd and P. Benfield, Drugs, 38, 481 (1989).
M. R. Alan, D. S. Fred, and J. T. Stewart, Conquering Headache, 4th edn., Decker DTC, Hamilton, London (2003).
European Pharmacopoeia, Vol. II, European Department for the Quality of Medicines, Council of Europe, Stranbourg, France (2005).
British Pharmacopoeia, Vol. II, Her Majesty's Stationary Office, London (2009).
A. A. M. Wahbi, M. E. Abdel-Fattah, E. M. Hassan, F. G. Saliman, and El-Gendi, J. Pharm. Biomed. Anal., 13, 777 (1995).
W. F. Kartinasari, H. Chufianty, and G. Indrayanto, J. Liq. Chromatogr. Related Technol., 26, 1059 (2003).
S. P. Amod, A. A. Shirkhedkar, J. S. Surana, and S. P. Nawale, J. Chil. Chem. Soc., 57, 1033 (2012).
M. Abdul-Azim Mohammad, Bull Fac. Pharm. Cairo Univ., 42, 27 (2004).
S. A. Patil, A. A. Shirkhedkar, J. S. Surana, and S. P. Nawale, Pharma Chem., 3, 404 (2011).
A. K. Doshi, B. N. Patel, and C. N. Patel, Int. J. Pharm. Sci. Res., 3, 1741 (2012).
K. Busaranon, W. Suntornsuk, and L. Suntornsuk, J. Pharm. Biomed. Anal., 41, 158 (2006).
V. S. S. P. Adapa, C. Ravi, C. S. P. Sastry, and U. V. Prasad, Acta Cienc. Indica, Chem., 27, 105 (2001).
V. S. S. P. Adapa, C. S. R. Lakshmi, C. S. P. Sastry, and V. P. Uppuleti, J. Anal. Chem., 58, 937 (2003).
M. Y. El-Maamli, Egypt. J. Biomed. Sci., 11, 122 (2003).
K. Kelani, I. L. Bebawy, and L. A. Fattah, J. Pharm. Biomed. Anal., 18, 985 (1999).
M. J. Badalani, K. R. Mahadik, H. N. More, and S. S. Kadam, East Pharm., 38, 133 (1995).
M. S. Elazazy, M. El-Mammli, A. Shalaby, and M. A. Magda, Biosci., Biotechnol. Res. Asia, 5, 107 (2008).
V. S. S. P. Adapa, G. P. V. Mallikarjuna Rao, C. S. P. Sastry, and U. V. Prasad, J. Ind. Council Chem., 18, 36 (2001).
S. S. Zarapkar and R. K. Bapat, Indian Drugs, 31, 170 (1994).
V. S. S. P. Adapa and C. S. P. Sastry, Sci. Asia, 33, 119 (2007).
A. F. M. E. Walily, A. E. Gindy, and A. A. M. Wahbi, J. Pharm. Biomed. Anal., 13, 53 (1995).
K. N. Prashanth, K. Basavaiah, and M. S. Raghu, Chem. Sci. J., 80, 1–14 (2012).
K. N. Prashanth, K. Basavaiah, and K. B. Vinay, Arab. J. Chem. (2011); doi:https://doi.org/10.1016/j.arabjc.2011.10.006.
M. S. Raghu and K. Basavaiah, Proc. Natl. Acad. Sci., India A: Phys. Sci., 82, 187 (2012).
K. B. Vinay, H. D. Revannasiddappa, N. Rajendraprasad, P. J. Ramesh, M. X. Cijo, and K. Basavaiah, Drug Test Anal., 4, 116 (2012).
H. Zavis, D. Ludvik, K. Milan, S. Ladislaw, and V. Frantisck, Handbook of Organic Reagents in Inorganic Analysis, Chichester, A Division of John Wiley & Sons IC, New York, London, Sydney, Toronto (1976).
International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use, ICH Harmonised Tripartite Guideline. Validation of Analytical Procedures: Text and Methodology, Q2 (R 1), Complementary Guideline on Methodology dated 06 November 1996, London, incorporated in November 2005.
J. Inczedy, T. Lengyel, and A. M. Ure, IUPAC Compendium of Analytical Nomenclature: Definitive Rules, Blackwell Science Inc., Boston (1998).
Author information
Authors and Affiliations
Corresponding author
Additional information
Abstract of article is published in Zhurnal Prikladnoi Spektroskopii, Vol. 84, No. 6, p. 1017, November–December, 2017.
Rights and permissions
About this article
Cite this article
Prashanth, K.N., Basavaiah, K. Modified Extraction-Free Ion-Pair Methods for the Determination of Flunarizine Dihydrochloride in Bulk Drug, Tablets, and Human Urine. J Appl Spectrosc 84, 1114–1123 (2018). https://doi.org/10.1007/s10812-018-0596-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10812-018-0596-6