Abstract
This research studied the effect of solids retention time (SRT) on algal-activated sludge (AAS) association in carbon-nitrogen-phosphorus removal and lipid synthesis from municipal wastewater. Five 500-mL test reactors were operated at different SRTs (2, 4, 6, 8, and 10 days) parallelly, in a draw-and-fill mode with external aeration. The organic carbon (C), nitrogen (N), and phosphorus (P) removal were positively correlated with SRT. The highest C, N, and P removal rates of 89, 73, and 91% were observed at 10-day SRT. The algal percentage was more than 60% in the mixed biomass at all the SRTs at a steady state. The lipid content ranged from 15 to 30% and declined with an increase in SRT, with 60% saturated and monounsaturated fatty acid at all the SRTs. The cetane number varied from 56 to 64 indicating a good-quality biofuel. The Fourier transform infrared spectroscopy transmittance spectrum for 10-day SRT biomass showed the presence of chloroalkane, alcohols, phenols, alkane, esters, allene, alkynes, and phosphine. The AAS system can be used as the main treatment unit for treating municipal wastewater in a single reactor with SRT greater than 4 days.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The introduction of microalgae into activated sludge processes for wastewater treatment has gained a lot of attention in the last few years. The algal-activated sludge (AAS) process is easy to operate and is established on a symbiotic interaction between heterotrophic bacteria and photosynthetic algae (Lee et al. 2015). However, existing AAS systems are applied mostly as tertiary treatment plants because of their low performance (Marbelia et al. 2014; Hu et al. 2015; Xu et al. 2016). The main reasons for the low performance of AAS reactors are the insufficient retention time for algae in reactors and the undesirable C/N/P ratio of municipal wastewater (Molazadeh et al. 2019).
The C/N ratio (7:1) of municipal wastewaters is relatively low than the composition ratio of the algal biomass (15:1), resulting in low nutrient removal efficiencies in microalga-based wastewater treatment processes due to carbon deficiency (Park and Craggs 2010; Posadas et al. 2014). Van den Hende et al. (2011) reported that the algal growth rate was enhanced by raising the ratio of inorganic carbon to organic carbon. In addition, the supply of CO2 would also prevent the rise in pH due to photosynthetic activity in the mixed liquor and, therefore, reduces losses of ammonia nitrogen by stripping and phosphorus precipitation (Acien Fernandez et al. 2012; Arbib et al. 2013). The immobilization of algae is one of the most promising methods for addressing the problems associated with biomass retention (Medina and Neis 2007; Shen et al. 2017), but the materials used as media become less effective for a longer period of use.
The proliferation rates for algal cells and bacteria cells vary significantly. The maximum growth rates for algal species like cyanobacteria, green microalgae, and diatoms vary between 0.79 and 6.12 days−1 depending on growth factors such as light intensity, water temperature, carbon dioxide, and nutrients (Park et al. 2005; Sasi et al. 2011; Wu et al. 2013; Zhang et al. 2015; Huesemann et al. 2016), whereas the growth rate of Nitrosomonas, typical ammonium-oxidizing bacteria, is between 0.55 and 1.2 day−1 (Robertson et al. 1988; Gupta and Gupta 2001; Blackburne et al. 2007). Given the differences in maximum growth rates between bacteria and algae, the two foremost factors that can improve the performance of AAS systems are as follows: (1) the provision of external aeration which increases CO2 concentration and (2) the selection of suitable solids retention time (SRT). Applying a suitable SRT may reduce the concentration of bacteria in a suspended AAS system which may allow algae to have access to more nutrients, which should result in more oxygen production and nutrient removal by algae. It is important to improve the algal growth rate and study how the symbiotic association between microalgae and bacteria is established in an AAS system. If concentrations of algae can be increased significantly, the AAS system can be used as the main treatment unit for treating municipal wastewater in a single reactor. Although the main focus of using microalgae in wastewater treatment is to remove organic carbon and nutrients, utilization of the produced biomass after wastewater treatment is also important. Current knowledge on the effect of SRT on the performance of algal-bacterial reactors and the effect on lipid accumulation of microalgae is limited. The aim of this study was to (1) understand the role of SRT on the performance of AAS reactors in removing carbon and nutrients and (2) study the production and composition of lipid and FAME (fatty acid methyl esters) obtained from the AAS biomass during the treatment.
Materials and methods
Culture and cultivation
The bacteria (activated sludge) were obtained from a decentralized municipal wastewater treatment plant in Hyderabad, India. Before using in the experimental study, the activated sludge was characterized for volatile suspended solids (VSS).
The seed microalgal culture used in this study was collected from the nearby lake located in Sangareddy (Hyderabad, India). The culture was adapted to synthetic BG11 medium for about a year at room temperature under the direct sunlight with approx. 12-h:12-h light/dark cycle. Before starting the experiment, a correlation curve was plotted between the chlorophyll and mixed liquor volatile suspended solids (MLVSS, which represents the weight of microorganisms) as shown in Fig. 1, for mixed algal culture. The MLVSS of algal culture was observed to be linearly correlated with the chlorophyll concentration (Fig. 1). The correlation equation was used in this study to estimate the algal biomass in AAS reactors.
Municipal wastewater
A gated community with 2016 families, located in Hyderabad, Telangana, India, was selected as a municipal wastewater source. Fresh wastewater was collected on weekends every week. The collected wastewater was mixed thoroughly and passed through a filter with a 2.36-mm pore size to remove large solid particles. The total solids concentration in the wastewater was less than 50 mg L−1. The wastewater was stored at − 20 °C and used directly without any sterilization (Cho et al. 2017).
Experimental scheme
Transparent glass bottles of 500 mL volume with wide mouth were used as the test reactors. Five reactors were operated parallelly each at different SRTs, i.e., 2, 4, 6, 8, and 10 days, in a semi-continuous mode (draw and fill once per day). The semi-continuous mode is recommended because it provides better control of solids wasting and feedstock input. The draw-and-fill feed schedule also conditions the culture to accept the initial high substrate doses that are used in batch tests (Young and Cowan 2004). The recycling of biomass was not done. Therefore, the hydraulic retention time (HRT) was equal to the solids retention time (SRT). The feed volume was calculated by the volume of reactor/SRT (= HRT for the present study). For SRT = 2 days, feed volume was 250 mL (= 500/2) per day. Every day, the calculated volume of mixed liquor (which contains both biomass and water) was withdrawn and the same amount of fresh wastewater or substrate was fed to the reactors. A similar procedure was followed for all the other SRTs. The test reactors were kept at an ambient temperature (25–30 °C) under direct sunlight with approx. 12:12 (light:dark) cycle. Continuous supply of air was provided to the reactors by means of aerators at a constant airflow of 0.2 L min−1. Each reactor was operated for three times its SRT, to provide sufficient time for the biomass to reach the steady states. Equal proportion (by mass) of algae and bacteria were introduced in the reactors at the beginning of the experiment. The initial cell density (VSS = 1500 mg L−1) was kept the same for all the test reactors.
Analytical methods
Determination of wastewater parameters (COD, P, TN, and TOC)
The influent and effluent samples were characterized for chemical oxygen demand (COD), phosphorus (P), total nitrogen (TN), and total organic carbon (TOC) thrice a week. The withdrawn mixed liquor samples were filtered by using 0.45-μm Whatman filter paper. The filtered samples were analyzed for COD using method 5220 - C and P using method 4500 P - C by following the Standard Methods for the Examination of Water and Wastewater (APHA/AWWA/WEF 2012). The total dissolved organic carbon (TOC) and nitrogen (TN) were measured using a TOC-L analyzer (Shimadzu). The removal efficiency was calculated by subtracting the effluent concentration from the influent concentration, and the value was divided by influent concentration. The obtained decimal value was multiplied with 100 to convert it to percentage.
Determination of biomass concentration
The chlorophyll (method 10200 H) and VSS (method 2540 E) were estimated by following the Standard Methods for Water and Wastewater (APHA/AWWA/WEF 2012). For chlorophyll estimation, 2 mL of mixed liquor sample was collected from the test reactors prior to feeding. The co-relation equation of chlorophyll and algal MLVSS (described in “Culture and cultivation”) was used to estimate the fraction of algal concentration in the total biomass (algae + activated sludge). The chlorophyll concentration was substituted in the chlorophyll-VSS co-relation curve to estimate algal biomass (as VSS). The algal biomass was subtracted from the total biomass to estimate the VSS of the heterotrophic biomass (activated sludge) in the reactors.
Determination of lipid content
The mixed liquor, drawn out of the test reactors during feeding, was centrifuged at 3000 rpm (2960×g) for 15 min. The biomass pellet was dried at 60 °C in a hot air oven. The dried biomass was finely powdered using a mortar and pestle. The lipid content in the biomass was measured weekly. The modified Bligh and Dyer method was used for the lipid extraction process (Bligh and Dyer 1959; Piasecka et al. 2014). Lipid content (%) was calculated from the expression shown in Eq. (3)
FAME analysis using gas chromatography
The direct in situ transesterification method was used to convert algal lipids into FAME (Van Wychen and Laurens 2013). The FAME composition was estimated by gas chromatography (Bruker; model: GC-MS 400 series) using a flame ionization detector. A BR-5MS capillary column (Bruker) was used for separation and helium was used as carrier gas (flow rate, 1 mL min−1). The temperature of the oven was initially maintained at 80 °C for 2 min and then ramped up to 250 °C at the rate of 4 °C min−1. The injector and detector temperatures were maintained at 230 and 300 °C, respectively. A 100:1 split ratio was used. The internal calibration was prepared by using a mixed standard solution of 37 (C4-C24) FAME compounds (Supelco, USA).
The cetane number (CN) of the extracted algal oil was calculated based on the FAME composition using the formula reported by Mishra et al. (2016).
where
Fourier transform infrared spectroscopy
Fourier transform infrared spectroscopy (FTIR) analysis can be used to determine the organic compounds present in the sludge. Once the test reactors reached the steady states, the biomass drawn from the test reactors during feeding was dried, ground into a powder, and then analyzed in FTIR. The analysis was performed using an FTIR spectrometer (make: Bruker, model: Alpha II). The dried AAS biomass was scanned at a resolution of 4 cm−1 in the range of 4000–300 cm−1.
Statistical analyses
The correlation matrix for six parameters—SRT, Chl/MLVSS, TOC removal, TN removal, P removal, and FAME (mg g−1)—was plotted to analyze the interaction between the parameters.
Results
Wastewater characteristics
The composition of wastewater plays a major role in the efficiency of the AAS consortium in nutrient (N and P) uptake mainly initial characteristics of the wastewater such as pH, concentrations, and bioavailability of nutrients (Aslan and Kapdan 2006; Delgadillo-Mirquez et al. 2016). The pH of municipal wastewater was found to be 7.2 ± 0.5 which is suitable for algal growth. The TN concentration was 101.3 ± 2.8 mg L−1. The level of P was 5.2 ± 1.3 mg L−1 as orthophosphate. COD and TOC were 492 ± 10 and 245.6 ± 16 mg L−1, respectively. The C:N:P ratio of wastewater used in this study was 47:19:1. The optimal C:N:P molar ratio suitable for the growth of microalgae is 56:9:1 (Phang and Kim-Chong 1988). The N:P molar ratio for significant nutrient removal ranges from 11:1 to 18:1 (Xin et al. 2010).
AAS biomass dynamics
The total chlorophyll concentration was measured to estimate algal growth in test reactors. The variation in biomass concentration at different SRTs is shown in Fig. 2. At the beginning of the experiment, all the reactors were seeded with an equal quantity of algal and bacterial biomass, measured in terms of MLVSS. From the figure, it was observed that the ratio of chlorophyll concentration (Chl) to total MLVSS concentration increased at all the SRTs. As shown in Fig. 2 a, the Chl/MLVSS in the reactor increased until day 4 and reached a steady state. At steady state, the lower SRTs showed lower concentrations of chlorophyll. Chl/MVLSS ratio increased from an initial value of 1.1 to 1.58 for 2-day SRT and to 2 for all the other SRTs. The relative percentage distribution of algal and bacterial biomass (MLVSS) in the reactors at different SRTs is shown in Fig. 2 b. The algal concentration in the reactors increased with increasing SRT. The algal biomass was observed to be 70% for 2-day and 90% for 10-day SRT. This indicates that SRT has a great influence on algal-bacterial population dynamics.
Performance of the reactors in removing carbon and nutrients
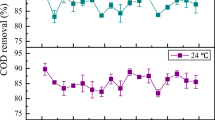
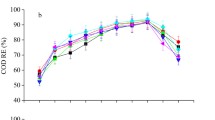
Figure 3 a shows the performance of the system in removing organic pollutants (as TOC), TN, and P from municipal wastewater. Both carbon and nutrients were successfully reduced in the test reactors. The reactor efficiency for removing C, N, and P increased with the SRT. The highest TOC, TN, and P removal rates, observed in the 10-day SRT, were 89.2%, 73.1%, and 91%, respectively. Even at a low SRT of 2 days, AAS reactors showed 85% TOC, 65.9% TN, and 78.9% P removal. The effluent carbon and nitrogen concentrations at different SRTs are shown in Fig. 3 b. The TOC and TN in wastewater were significantly decreased from 259 to 28 mg L−1 and from 102 to 27 mg L−1, respectively, at 10-day SRT. Significant removal of P, from 5 to 0.4 mg L−1, was also observed at the same SRT.
Lipid production by AAS biomass
Lipids are biosynthetically or functionally related to fatty acids and their derivatives. The lipid content based on total biomass and algal biomass at different SRTs is shown in Fig. 4 a. The highest lipid content (31%) was observed at 2-day SRT and it decreased with increasing SRT. The lipid content at 2-day SRT was twofold the lipid content at 10-day SRT. The lipid accumulation is triggered mainly in nutrient (N and P)-deficient stress conditions. The highest lipid assimilation at 2-day SRT could be due to the bacterial stress. The higher proportion of bacterial biomass at 2-day SRT could increase external stress on microalgae, such as oxidative stress, triggering lipid synthesis, and storage (Cho et al. 2015).
Fatty acid methyl ester profile
Microalgal lipid is mainly used as a raw material for biodiesel synthesis, but the quality of biodiesel is evaluated based on the fatty acid (FA) composition and the total FAME content. In this study, the algal oil was converted into FAME through transesterification to examine the composition of the FA profile using GC. The saturated and unsaturated fatty acids from both algal and bacterial lipids with carbon chain lengths ranging from C4:0 to C24:0 were observed in the FAME profile. Table 1 shows the effect of SRT on the FAME profile of algal consortium in the algal-bacterial system. The profile showed the presence of 16:0, 18:0, 18:1, 20:0, and 22:2 series of FA in all SRTs (Table 1). The most desirable fatty acids for the production of high-quality biodiesel with superior properties of stability, cold flow, viscosity, and ignitability are C16:0, C18:0, and C18:1 (Knothe 2008; Prathima Devi et al. 2012). The C18:0 and C18:1 percentage was observed to be less than 10% at 2-day SRT and ranged from 23 to 43% at SRTs > 4 days. The highest percentage (32.6%) of C18:1 was observed at 10-day SRT which is potentially more favorable for the production of biodiesel.
The percentage distribution of saturated fatty acid (SFA), monounsaturated fatty acid (MUFA), and polyunsaturated fatty acid (PUFA) at different SRTs is shown in Fig. 4 b. The SRT exerted a significant effect on the FAME composition. The percentage of PUFA decreased with increasing SRT. The SFA accounted for more than 63% of fatty acids at 10-day SRTs. Though the lipid productivity was higher at 2-day SRT, the conversion efficiency was found to be less which shows that bacterial stress in the reactors enhanced the lipid productivity but shifted the fatty acid profile towards unsaturation.
The distribution of the dominant fatty acids—palmitic acid (C16:0), stearic acid (C18:0), elaidic acid (C18:1), and arachidic acid (C20:0), at different SRTs—is shown in Fig. 4 c. The figure suggests that the algal-bacterial dynamics had shown a significant effect on the composition of the fatty acid profile, especially at lower SRT. The dominant microalgal species in the reactors have a significant effect on the fatty acid profile. The dominant species observed in the reactors were mainly Chlorella and Scenedesmus. The FAME composition of the algal-bacterial culture obtained in this study was compared with the values of the literature on the mixed algal culture during wastewater treatment and is presented in Table 2. The FAME profile (C16:0, C18:0) observed in this study was similar to an algal mixed culture reported in the literature.
The properties of biodiesel fuel, including its ignition quality, combustion heat, cold filter plugging point, oxidative stability, viscosity, and lubricity, are determined by the structure of its component fatty esters. High levels of saturated fatty acids tend to increase the stability of biodiesel while unsaturated fatty acids result in poor oxidative stability. The CN plays a major role in indicating the ignition properties of diesel fuel. The CN values were calculated based on Eqs. 2–4 and are given in Table 3. The FAME mg g−1 dried biomass and CN increased with an increase in SRT. The CN in this study varied from 56 to 64; these values are in the range of CN values reported for different algal species (Islam et al. 2013).
FTIR analysis
The FTIR analysis of the AAS dried biomass at 2-day and 10-day SRT is shown in Fig. 5. The FTIR spectra were observed to be different at low and high SRTs. The transmittance band at 1009.2 cm−1 which is majorly observed in 2-day SRT shows the presence of polysaccharides or lipids (Grube et al. 2006; Deepayan and Karar 2017). The peak intensity decreased with an increase in SRT. The next highest peaks were observed at 1399.17 cm−1 (S=O, sulfur compound), 1641.6 cm−1 (C=C, alkene), and 1541.4 cm−1 (N-H, amides). The FTIR transmittance spectrum for 10-day SRT biomass showed the presence of chloroalkane, alcohols, phenols, alkane, esters, allene, alkynes, and phosphine. Similar kind of groups was also observed in Chlorella and Scenedesmus species of green algae which shows the dominance of algal biomass in total biomass at higher SRT in AAS reactors (Duygu et al. 2012; Sudhakar and Premalatha 2015).
Correlation of parameters
The visual presentation of the correlation matrix for six parameters—SRT, Chl/MLVSS, TOC removal, TN removal, P removal, and FAME (mg g−1)—is shown in Fig. 6. It was observed that almost all the parameters are positively interrelated to each other for enhancing the performance of AAS reactors. The TOC removal is related to SRT and Chl/MLVSS indicating the role of microalgae in utilizing the carbon. The TN removal was observed to be less related to Chl/MLVSS and P removal and more related to SRT. The P removal was more related to Chl/MLVSS. The FAME (mg/g) was also less related to Chl/MLSS. It was also observed that the lipid content of the biomass was negatively related to SRT.
Discussion
In typical wastewater treatment plants, the bacterial consortium mainly mineralizes organic carbon by using DO through heterotrophic metabolism. In contrast, microalgae use photoautotrophic (light and CO2 (or inorganic carbon)) and mixotrophic (organic carbon and light) mechanisms (Chen et al. 2011). When both algae and activated sludge are together, organic carbon can be utilized through mixotrophic or heterotrophic metabolism (Abeliovich and Weisman 1978). The carbon and nutrient metabolisms in microalgae are interconnected (Huppe and Turpin 1994; Liu et al. 2015). When algae and activated sludge coexist, organic carbon can be utilized through mixotrophic or heterotrophic metabolism (Abeliovich and Weisman 1978). AAS culture might have used different mechanisms in removing nutrients from wastewater such as bacterial and algal cell uptake, nitrification-denitrification, volatilization, and precipitation (Perez-Garcia et al. 2011; Su et al. 2011). Nitrogen removal by ammonia volatilization could be negligible because the pH in the reactors was below 8.5. Nitrogen uptake by the cells is the most common mechanism observed in algal species. Su et al. (2011) also found that 40.3 to 53% of nitrogen was accumulated in filamentous blue-green algae when light to the dark period of 12 h was applied. Denitrification was observed to be a major removal mechanism by a mixed population of microalgae and bacteria while treating municipal wastewater (Su et al. 2011). The N/P ratio is another main parameter of wastewater that affects biomass growth and removal of total inorganic nutrients from municipal wastewater (Gupta et al. 2016). Aslan and Kapdan (2006) reported that at the N/P ratio of 2:1, 78% of P was removed in 10-day retention time by Chlorella vulgaris culture from synthetic wastewater. Su et al. (2011) observed 56 to 73% of P removal at N/P = 3:1 in batch cultures (8 days of incubation). High levels of P removal (90%) by Chlorella sp. were reported by Wang et al. (2010) with an increase in N/P (6:1) from wastewater after 9 days of the incubation period. Similar results (> 95%) were reported by Delgadillo-Mirquez et al. (2016) when N/P = 17:1. In this study, the N/P of wastewater was observed to be 19:1 which might have favored P removal.
The lipid accumulation is triggered mainly in nutrient (N and P)-deficient stress conditions. The highest lipid assimilation at 2-day SRT could be due to bacterial stress. Chlorella sp. cultivated in anaerobically digested dairy manure had shown lipid content ranging from 9.0 to 13.7% of the dry weight of the biomass (0.141–0.233 g L−1) (Wang et al. 2010). Chlorellaellipsoidea YJ1 achieved a higher lipid content of about 40% in the domestic secondary effluent (Yang et al. 2011). Abreu et al. (2012) reported lipid productivity of about 250 mg L−1 day−1 by C. vulgaris cultivated in hydrolyzed cheese whey (5 g L−1 glucose and galactose). Farooq et al. (2013) reported that by using the two-stage autotrophic-heterotrophic cultivation for C. vulgaris the lipid productivity was increased from 31 to 108 mg L−1 day. Chlorella vulgaris cultivated in an artificial wastewater medium showed about 20–42% of lipid content (Feng et al. 2011). Although the literature suggests that the lipid storing capacity of Chlorella pyrenoidosa is only 2% (Becker 1994), Hongyang et al. (2011) reported that C. pyrenoidosa can accumulate lipid up to 37% of its cell weight and can exhibit lipid productivity up to 0.4 g L−1 day−1 using soybean processing wastewater. Moreover, very few studies have been performed to analyze the effect of retention time on lipid accumulating capacity of microalgae. Lipid content for pure cultures of algae was reported in several publications. However, there has been less documentation on lipid percentages in mixed cultures of algae in algal-bacterial systems. Many studies presented that biomass and lipid production significantly improved in heterotrophic or mixotrophic conditions (Perez-Garcia et al. 2011). However, there are many other factors affecting lipid production like microalgal species, wastewater characteristics (like pH, dissolved solids, C/N ratio), and light-dark cycle (Qin et al. 2016). It was evident that the characteristics of wastewater, SRT, and algal/bacterial ratio had shown a significant effect on lipid accumulation of algal-bacterial biomass.
A fatty acid with a high saturation fraction, i.e., with a less unsaturated fatty acid (MUFA + PUFA) and high SFA, indicates excellent fuel properties with less risk of combustion and ignition interruption (Chandra et al. 2014). As per European Standard EN 14214, the C18:3 and polyunsaturated (≥ 4 double bonds) contents should be less than 12 and 1%, respectively, for good-quality biodiesel (Gouveia and Oliveira 2009). In this study, C18:3 was observed to be less than 0.3% at all the SRTs. The fuel with CN greater than 50 is recommended as a quality parameter. The biodiesel derived from microalgae with more saturation (SFA %) will result in high CN; this fuel exhibits shorter ignition delay time and fewer NOx emissions (Cheirsilp and Torpee 2012). It was evident that the characteristics of wastewater, SRT, and algal/bacterial ratio had shown a significant effect on lipid accumulation of co-culture biomass. The study indicates that the culturing algae and activated sludge together are capable of effectively treating domestic wastewater, rich in carbon and nutrient, and can simultaneously produce good-quality lipids which are desirable for biodiesel production.
Conclusion
Solids retention time has a significant influence on algal-activated sludge population dynamics, lipid, and fatty acid composition. High SRT causes an increase in algal biomass, which results in nutrient removal and favors the formation of good-quality lipid, desirable for biodiesel production. Palmitic acid (C16:0) and stearic acid (C18:0) were observed at all SRTs which are the desired substrates for synthesizing biodiesel. The study indicates that the addition of algae into activated sludge has the potential to directly treat municipal wastewater in a single reactor if operated at higher SRT (> 4 days) with simultaneous biodiesel production.
References
Abeliovich A, Weisman D (1978) Role of heterotrophic nutrition in growth of the alga Scenedesmus obliquus in high-rate oxidation ponds. Appl Environ Microbiol 35:32–37
Abreu AP, Fernandes B, Vicente AA, Teixeira J, Dragone G (2012) Mixotrophic cultivation of Chlorella vulgaris using industrial dairy waste as organic carbon source. Bioresour Technol 118:61–66
APHA/AWWA/WEF (2012) Standard methods for the examination of water and wastewater. Stand Methods 541
Arbib Z, Ruiz J, Álvarez-Díaz P, Garrido-Pérez C, Barragan J, Perales J (2013) Effect of pH control by means of flue gas addition on three different photo-bioreactors treating urban wastewater in long-term operation. Ecol Eng 57:226–235
Aslan S, Kapdan IK (2006) Batch kinetics of nitrogen and phosphorus removal from synthetic wastewater by algae. Ecol Eng 28:64–70
Becker EW (1994) Microalgae: biotechnology and microbiology. Cambridge University Press, Cambridge
Blackburne R, Vadivelu VM, Yuan Z, Keller J (2007) Determination of growth rate and yield of nitrifying bacteria by measuring carbon dioxide uptake rate. Water Environ Res 79:2437–2445
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Chandra R, Rohit MV, Swamy YV, Venkata Mohan S (2014) Regulatory function of organic carbon supplementation on biodiesel production during growth and nutrient stress phases of mixotrophic microalgae cultivation. Bioresour Technol 165:279–287
Cheirsilp B, Torpee S (2012) Enhanced growth and lipid production of microalgae under mixotrophic culture condition: effect of light intensity, glucose concentration and fed-batch cultivation. Bioresour Technol 110:510–516
Chen CY, Yeh KL, Aisyah R, Lee DJ, Chang JS (2011) Cultivation, photobioreactor design and harvesting of microalgae for biodiesel production: a critical review. Bioresour Technol 102:71–81
Cho DH, Ramanan R, Heo J, Lee J, Kim BH, Oh HM, Kim HS (2015) Enhancing microalgal biomass productivity by engineering a microalgal-bacterial community. Bioresour Technol 175:578–585
Cho HU, Cho HU, Park JM (2017) Enhanced microalgal biomass and lipid production from a consortium of indigenous microalgae and bacteria present in municipal wastewater under gradually mixotrophic culture conditions. Bioresour Technol 228:290–297
Deepayan P, Karar PK (2017) Utilising FTIR and gas chromatograph for optimizing lipid extraction for biodiesel production from domestic sewage sludge and food waste. Res J Chem Environ 21:26–36
Delgadillo-Mirquez L, Lopes F, Taidi B, Pareau D (2016) Nitrogen and phosphate removal from wastewater with a mixed microalgae and bacteria culture. Biotechnol Reports 11:18–26
Duygu DY, Udoh AU, Akbulut A, Erkaya IA, Yildiz K, Guler D (2012) Fourier transform infrared (FTIR) spectroscopy for identification of Chlorella vulgaris Beijerinck 1890 and Scenedesmus obliquus (Turpin) Kützing 1833. Afr J Biotechnol 11:3817–3824
Farooq W, Lee YC, Ryu BG, Kim BH, Kim HS, Choi YE, Yang JW (2013) Two-stage cultivation of two Chlorella sp. strains by simultaneous treatment of brewery wastewater and maximizing lipid productivity. Bioresour Technol 132:230–238
Feng Y, Li C, Zhang D (2011) Lipid production of Chlorella vulgaris cultured in artificial wastewater medium. Bioresour Technol 102:101–105
Acien-Fernandez GF, González-López CV, Fernández Sevilla JM, Molina Grima E (2012) Conversion of CO2 into biomass by microalgae: how realistic a contribution may it be to significant CO2 removal? Appl Microbiol Biotechnol 96:577–586
Gouveia L, Oliveira AC (2009) Microalgae as a raw material for biofuels production. J Ind Microbiol Biotechnol 36:269–274
Grube M, Lin JG, Lee PH, Kokorevicha S (2006) Evaluation of sewage sludge-based compost by FT-IR spectroscopy. Geoderma 130:324–333
Gupta AB, Gupta SK (2001) Simultaneous carbon and nitrogen removal from high strength domestic wastewater in an aerobic RBC biofilm. Water Res 35:1714–1722
Gupta PL, Choi HJ, Lee SM (2016) Enhanced nutrient removal from municipal wastewater assisted by mixotrophic microalgal cultivation using glycerol. Environ Sci Pollut Res 23:10114–10123
Hongyang S, Yalei Z, Chunmin Z, Xufei Z, Jinpeng L (2011) Cultivation of Chlorella pyrenoidosa in soybean processing wastewater. Bioresour Technol 102:9884–9890
Hu W, Yin J, Deng B, Hu Z (2015) Application of nano TiO2 modified hollow fiber membranes in algal membrane bioreactors for high-density algae cultivation and wastewater polishing. Bioresour Technol 193:135–141
Huesemann M, Crowe B, Waller P, Chavis A, Hobbs S, Edmundson S, Wigmosta M (2016) A validated model to predict microalgae growth in outdoor pond cultures subjected to fluctuating light intensities and water temperatures. Algal Res 13:195–206
Huppe HC, Turpin DH (1994) Integration of carbon and nitrogen metabolism in plant and algal cells. Annu Rev Plant Physiol 45:577–607
Islam MA, Magnusson M, Brown RJ, Ayoko GA, Nabi MN, Heimann K (2013) Microalgal species selection for biodiesel production based on fuel properties derived from fatty acid profiles. Energies 6:5676–5702
Katam K, Bhattacharyya D (2019) Simultaneous treatment of domestic wastewater and bio-lipid synthesis using immobilized and suspended cultures of microalgae and activated sludge. J Ind Eng Chem 69:295–303
Knothe G (2008) “Designer” biodiesel: optimizing fatty ester composition to improve fuel properties. Energy Fuel 22:1358–1364
Lee CS, Lee SA, Ko SR, Oh HM, Ahn CY (2015) Effects of photoperiod on nutrient removal, biomass production, and algal-bacterial population dynamics in lab-scale photobioreactors treating municipal wastewater. Water Res 68:680–691
Liu N, Li F, Ge F, Tao N, Zhou Q, Wong M (2015) Mechanisms of ammonium assimilation by Chlorella vulgaris F1068: isotope fractionation and proteomic approaches. Bioresour Technol 190:307–314
Mahapatra DM, Chanakya HN, Ramachandra TV (2014) Bioremediation and lipid synthesis through mixotrophic algal consortia in municipal wastewater. Bioresour Technol 168:142–150
Marbelia L, Bilad MR, Passaris I, Discart V, Vandamme D, Beuckels A, Muylaert K, Vankelecom IF (2014) Membrane photobioreactors for integrated microalgae cultivation and nutrient remediation of membrane bioreactors effluent. Bioresour Technol 163:228–235
Medina M, Neis U (2007) Symbiotic algal bacterial wastewater treatment: effect of food to microorganism ratio and hydraulic retention time on the process performance. Water Sci Technol 55:165–171
Mishra S, Anand K, Mehta PS (2016) Predicting the cetane number of biodiesel fuels from their fatty acid methyl ester composition. Energy Fuel 30:10425–10434
Mohan SV, Devi MP, Mohanakrishna G, Amarnath N, Babu ML, Sarma PN (2011) Potential of mixed microalgae to harness biodiesel from ecological water-bodies with simultaneous treatment. Bioresour Technol 102:1109–1117
Molazadeh M, Ahmadzadeh H, Pourianfar HR, Lyon S, Rampelotto PH (2019) The use of microalgae for coupling wastewater treatment with CO2 biofixation. Front Bioeng Biotechnol 7:42
Park JBK, Craggs RJ (2010) Wastewater treatment and algal production in high rate algal ponds with carbon dioxide addition. Water Sci Technol 61:633–639
Park K, Jung HS, Kim HS, Ahn SM (2005) Three-dimensional hydrodynamic-eutrophication model (HEM-3D): application to Kwang-Yang Bay, Korea. Mar Environ Res 60:171–193
Perez-Garcia O, Escalante FME, de-Bashan LE, Bashan Y (2011) Heterotrophic cultures of microalgae: metabolism and potential products. Water Res 45:11–36
Phang SM, Kim-Chong O (1988) Algal biomass production in digested palm oil mill effluent. Biol Wastes 25:177–191
Piasecka A, Krzemiñska I, Tys J (2014) Physical methods of microalgal biomass pretreatment. Int Agrophys 28:341–348
Posadas E, Bochon S, Coca M, Coca M, García-González MC, García-Encina PA, Muñoz R (2014) Microalgae-based agro-industrial wastewater treatment: a preliminary screening of biodegradability. J Appl Phycol 26:2335–2345
Prathima Devi M, Venkata Subhash G, Venkata Mohan S (2012) Heterotrophic cultivation of mixed microalgae for lipid accumulation and wastewater treatment during sequential growth and starvation phases: effect of nutrient supplementation. Renew Energy 43:276–283
Qin L, Wang Z, Sun Y, Shu Q, Feng P, Zhu L, Xu J, Yuan Z (2016) Microalgae consortia cultivation in dairy wastewater to improve the potential of nutrient removal and biodiesel feedstock production. Environ Sci Pollut Res 23:8379–8387
Robertson LA, van Niel EW, Torremans RA, Kuenen JG (1988) Simultaneous nitrification and denitrification in aerobic chemostat cultures of Thiosphaera pantotropha. Appl Environ Microbiol 54:2812–2818
Sasi D, Mitra P, Vigueras A, Hill GA (2011) Growth kinetics and lipid production using Chlorella vulgaris in a circulating loop photobioreactor. J Chem Technol Biotechnol 86:875–880
Shen Y, Yang T, Zhu W, Zhao Y (2017) Wastewater treatment and biofuel production through attached culture of Chlorella vulgaris in a porous substratum biofilm reactor. J Appl Phycol 29:833–841
Soydemir G, Keris-Sen UD, Sen U, Gurol MD (2016) Biodiesel production potential of mixed microalgal culture grown in domestic wastewater. Bioprocess Biosyst Eng 39:45–51
Su Y, Mennerich A, Urban B (2011) Municipal wastewater treatment and biomass accumulation with a wastewater-born and settleable algal-bacterial culture. Water Res 45:3351–3358
Sudhakar K, Premalatha M (2015) Characterization of micro algal biomass through FTIR/TGA /CHN analysis: application to Scenedesmus sp. Energy Sourc A 37:2330–2337
Van Den Hende S, Vervaeren H, Saveyn H, Saveyn H, Maes G, Boon N (2011) Microalgal bacterial floc properties are improved by a balanced inorganic/organic carbon ratio. Biotechnol Bioeng 108:549–558
Van Wychen S, Laurens LML (2013) Determination of total lipids as fatty acid methyl esters (FAME) by in situ transesterification: laboratory analytical procedure (LAP) NREL Technical report NREL/TP-5100-60958
Wang L, Li Y, Chen P, Min M, Chen Y, Zhu J, Ruan RR (2010) Anaerobic digested dairy manure as a nutrient supplement for cultivation of oil-rich green microalgae Chlorella sp. Bioresour Technol 101:2623–2628
Wu YH, Li X, Yu Y, Hu HY, Zhang TY, Li FM (2013) An integrated microalgal growth model and its application to optimize the biomass production of Scenedesmus sp. LX1 in open pond under the nutrient level of domestic secondary effluent. Bioresour Technol 144:445–451
Xin L, Hong-ying H, Ke G, Ying-xue S (2010) Effects of different nitrogen and phosphorus concentrations on the growth, nutrient uptake, and lipid accumulation of a freshwater microalga Scenedesmus sp. Bioresour Technol 101:5494–5500
Xu M, Xu S, Bernards M, Hu Z (2016) Evaluation of high density algal cultivation for secondary wastewater polishing. Water Environ Res 88:47–53
Yang J, Li X, Hu H, Zhang X, YU Y, Chen Y (2011) Growth and lipid accumulation properties of a freshwater microalga, Chlorella ellipsoidea YJ1, in domestic secondary effluents. Appl Energy 88:3295–3299
Young JC, Cowan RM (2004) Respirometry for environmental science and engineering. SJ Enterprises, Springdale
Zhang D, Dechatiwongse P, del Rio-Chanona EA, Maitland GC, Hellgardt K, Vassiliadis VS (2015) Dynamic modelling of high biomass density cultivation and biohydrogen production in different scales of flat plate photobioreactors. Biotechnol Bioeng 112:2429–2438
Funding
This research was funded by the Ministry of Human Resource and Development, Government of India, under the FAST program.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Katam, K., Bhattacharyya, D. Effect of solids retention time on the performance of alga-activated sludge association in municipal wastewater treatment and biofuel production. J Appl Phycol 32, 1803–1812 (2020). https://doi.org/10.1007/s10811-020-02076-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-020-02076-6