Abstract
Phycocyanin, a blue pigment, is a type of phycobiliproteins. Because of its various potential properties, phycocyanin is applied to various fields, such as nutraceutical, pharmaceutical, medicine, cosmetics, and biotechnological research. The cost and application of phycocyanin are highly dependent on its purity index. In this study, ammonium chloride is presented as a novel, effective, and inexpensive salt for phycocyanin extraction. Compared with sodium phosphate, which is commonly used during phycocyanin extraction process, ammonium chloride solution efficiently extracted phycocyanin with high purity from Arthrospira platensis FACHB-314. In addition, ammonium phosphate solution is also presented as an alternative precipitation agent in phycocyanin purification that may replace the widely used ammonium sulfate. Statistical analysis shows that there is no significant difference in phycocyanin concentration between crude extracts (overall mean of 0.208 and 0.215 for extraction using sodium phosphate and ammonium chloride, respectively). However, the difference in phycocyanin purity ratio (A620/A280) between these two extractions is significant (overall mean of 0.742 and 1.428 for extraction using sodium phosphate and ammonium chloride, respectively). With ammonium chloride, the purity indexes of phycocyanin are 1.5 and 2.81 after the optimum extraction step, and precipitation used as the primary purification step, respectively. The present study describes a novel purification method to achieve phycocyanin with analytical grade without multiple purification steps.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The microalgae have attracted attention from more researchers due to their macronutrient and micronutrient contents. In general, the microalgae produce more of these materials within a short time compared to other plants (Ullah et al. 2014). This makes them a potential economical and sustainable raw materials for many industrial fields (Parmar et al. 2011). In addition, microalgae are promising organisms to furnish novel and safer biologically active compounds (Wojtasiewicz and Stoń-Egiert 2016) and are widely used in the health food, cosmetics, and pharmaceutical industries as colorants and therapeutic agents, respectively (Liu et al. 2000; Borowitzka 2013; Kannaujiya and Sinha 2016). Arthrospira is a photosynthetic, filamentous, spiral-shaped, multicellular cyanobacterium with various therapeutic activities, including immunomodulation, anticancer, antioxidation, antiviral, antibacterial activities, and has positive effects on treatment for malnutrition, obesity, diabetes, anemia, etc. (Hoseini et al. 2013; Sagara et al. 2015; Xia et al. 2016; Wang and Zhang 2016).
The major protein constituents with significant beneficial health effects are the phycobiliproteins (PBPs), including phycocyanin, phycoerythrin, and allophycocyanin. Phycocyanin, the main type of phycobiliprotein, is a blue pigment with many potential industrial applications in different fields, including health food, biology, biotechnology, medicine, pharmaceutics, diagnostics, and cosmetics (Fernández-Rojas et al. 2014; Eriksen 2008). Phycocyanin’s applications are linked to its main functions, including natural colorants, fluorescent markers, and therapeutic agents. The light blue color is a characteristic color of phycocyanin, absorbing orange and red light, with absorption maxima between 615 and 622 nm, and emits fluorescence at about 650 nm. The phycocyanin quality is measured by purity index that describes the ratio between absorbance from phycocyanobilin at 620 nm and all proteins in the sample at 280 nm using a UV spectrometer (Wang et al. 2012). Phycocyanins with purity index of 0.7, 3.9, and greater than 4.0 are considered as food grade, reactive grade, and analytical grade, respectively (Patil and Raghavarao 2007; Walter et al. 2011). In addition, the commercial price of phycocyanin varies with increasing purity index; phycocyanin with food grade costs about US$0.13 mg−1, the one with reactive grade costs from US$1 to 5 mg−1, while the one with analytical grade can cost even more than US$15 mg−1 (Rito-Palomares et al. 2001). The absorption spectrum at 652 nm intends to ascertain the contamination of C-phycocyanin by the amount of allophycocyanin, whereas the absorbance at 280 nm intends to detect the presence of the overall concentration of noncolored proteins in the solution (Sobiechowska-Sasim et al. 2014; Eithar 2014).
The extraction and purification of phycocyanin can be categorized into three main processes, including the cell disruption, extraction (also known as salting-in), and purification (also known as salting-out) (Hemlata and Fareha 2011). Sodium phosphate is commonly used for phycocyanin extraction from microalgae and cyanobacteria; using it, as shown in Table 1, the purity index of the phycocyanin is lower than food grade, i.e., purity index of 0.7. Precipitation of phycocyanin with a saturated solution of ammonium sulfate is a primary purification step (Walter et al. 2011) and is commonly used as the first step in purifying proteins. The extraction of valuable active compounds (e.g., phycocyanin) with high purity is still expensive (Colla et al. 2007). Therefore, it is necessary to develop effective and economical (inexpensive) extraction techniques that contribute to the high purity and recovery yield of phycocyanin with various biological characteristics and activities.
The aim of this study was to identify an inexpensive extractive salt solution and optimize the extraction conditions that contribute to the achievement of an efficient, economical extraction method for phycocyanin with high purity from A. platensis FACHB-314.
Materials and methods
Microorganisms and culture
The strain Arthrospira platensis FACHB-314 was purchased from the Institute of Hydrobiology (IHB), Chinese Academy of Sciences, Wuhan, China. Culture was initially grown and maintained in modified Spirulina medium (SP) (Aiba and Ogawa 1977) in Erlenmeyer flasks in a photo incubator at 25 °C with light intensity of 40–50 μmol photons m−2 s−1, under 12:12 light/dark cycles. All production cultures were grown in Zarrouk medium prepared as described by Xie et al. (2014) . The culture media components were sterilized separately by autoclaving at 121 °C for 15 min and mixed afterward to achieve the final medium. For production culture, the 4-day-old culture with initial inoculum of 0.25 g L−1 was used to inoculate 1 L of liquid Zarrouk culture medium into 1-L glass vessel (Schott Duran) and incubated at 28 ± 2 °C for 12 days under continuous light of 300 μmol photons m−2 s−1 using 21 W white LED lamps. The pH of the culture was controlled at 9 ± 0∙2 with a continuous injection of 2.5 % CO2 at a rate of 0.2 v/v (Xie et al. 2014).
After cultivation period, the cells were harvested by self-sedimentation followed by centrifugation at 8960×g for 10 min at 4 °C, and the supernatant was removed. The cells in the pellet were washed once with distilled water to remove any residue of the culture medium salts and recentrifuged under the same conditions as previously described. The collected cells were called wet biomass and were stored in the refrigerator at −20 °C in dark for further processing.
Extraction phycocyanin from A. platensis FACHB-314
The selection of extracting salt solutions is mainly based on their salting-in and salting-out strength related to their constituent ions and cations positions on the Hofmeister series (also known as lyotropic series) (Baldwin 1996; Zhang and Cremer 2006). The additional criterion for selection was the cost as for industrial application, the extractive salt should be as cheap as possible. The sodium phosphate ACS reagent ≥99 %, for phycocyanin extraction, costs more than ammonium chloride ACS reagent ≥99.5 % for phycocyanin extraction (about US$234.12 kg−1 compared to US$110.32 kg−1, respectively—price in US dollars was calculated at exchange rate of 1 US$ = 1.37 Singapore $ (Sigma-Aldrich 2016)). All salt solutions were prepared utilizing deionized water (as described in supplementary material S1). In order to prevent the phycobiliproteins from degrading during experiment, 4 mM NaN3, 2 mM mercaptoethanol, and 2 mM EDTA were added in extraction salt solutions (Sun et al. 2009).
The cell disruption step and extraction step were combined in one single process unit operation, and this contributed in the reduction in extraction time. The repeated freezing and thawing cycles was reported as an effective cell disruption method in phycocyanin extraction process compared with other methods for microalgae cell disruption (Hemlata and Fareha 2011).
Wet biomass was suspended in the extraction salt solutions at different ratios, and suspended cell mass were subjected at four successive repeated freezing at −20 °C for 2 h and thawing at 4 °C for 4 h in dark (i.e., each cycle took 6 h) followed by extraction of phycocyanin. The cell lysate was subsequently centrifuged at 8960×g for 15 min at 4 °C, and blue supernatant containing phycocyanin (named crude extracts) was collected. The cell disruption and extraction processes took about 25 h while the primary purification took about 8 h (Fig. 1).
Sodium phosphate buffers with different concentrations (with different pHs) are frequently used for extraction of phycocyanin from algae (Patel et al. 2005; Walter et al. 2011; Moraes et al. 2011). The suitable concentration of sodium phosphate for phycocyanin extraction from A. platensis FACHB-313 was evaluated by mixing 250 mg of wet biomass with 10 mL of sodium phosphate (0.05, 0.075, 0.1, and 0.125 M, pH = 7.0) in 15-mL test tubes. Then, the suspended cell mass was subjected to successive series of freezing and thawing cycles under the same conditions as previously described. Ammonium chloride was chosen (as an available and inexpensive salt) to be evaluated for its effectiveness in phycocyanin extraction from A. platensis FACHB-314. Different ammonium chloride concentrations (0.05, 0.075, 0.1, 0.125, and 4.1 M) were evaluated by the same procedure as used for sodium phosphate. Finally, the optimum concentration was utilized in further experiments, where sodium phosphate was considered as a control.
Once extraction salt solution concentration was evaluated, effective ratio of wet biomass and extraction salt solution was also examined in the following phases. In the first phase (screening phase), wet biomass in the range between 250 and 2500 mg was mixed with 10 mL of 0.125 M sodium phosphate (pH 7.0); in the second phase, wet biomass (effective range from the first screening phase) was used to evaluate the optimum ratio and effectiveness of extraction salt solutions, which are 0.125 M sodium phosphate (pH 7.0), and 0.05 ammonium chloride (pH 4.39). All suspended cell mass solutions were exposed to four successive series of freezing and thawing cycles under the same conditions as described in previous paragraphs. The supernatants rich in phycocyanin were collected by centrifugation at 8960×g for 30 min at 4 °C. The obtained crude extracts were named sodium phosphate crude extract (SP) and ammonium chloride crude extract (AC).
Primary purification of phycocyanin
The process of phycocyanin purification is to remove impurities. The use of ammonium sulfate precipitation as the first purification step exhibited high efficiency in removing most of the protein contamination (Eithar 2014). Therefore, precipitation was used for primary purification of phycocyanin from crude extracts to examine whether the extraction salt solutions used in extraction process hindered further purification processes. Primary purification of phycocyanin was conducted by checking the crude extract to ammonium sulfate precipitation salt-solution ratio and evaluating effectiveness of different precipitation salt-solutions for phycocyanin purification from crude extracts (as described in supplementary material S2).
Phycocyanin concentration measurement
The concentration and purity of phycocyanin, after each step, were measured by reading absorbance at 280 nm (A280), 562 nm (A562), 620 nm (A620), and 652 nm (A652) against blanks using a UV-1780 spectrophotometer (Shimadzu, Japan). Phycocyanin (PC) concentration was calculated using Eq. (1) while allophycocyanin (APC) and phycoerythrin (PE) concentrations were calculated from Eqs. (2) and (3), respectively (Bennett and Bogorad 1973).
The phycocyanin purity ratio (purity index) was calculated by absorbance ratio, Eq. (4) (Walter et al. 2011):
Characterization of phycocyanin
The characterization of phycocyanin was conducted by evaluating the A620/A280 ratio, absorption spectrum (spectrophotometric analysis), and gel electrophoresis (sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE)). Absorption spectrum at 652 nm is to detect the presence of allophycocyanin, and the absorbance at 280 nm intends to check the presence of an amount of noncolored proteins (Eithar 2014). For spectrophotometric analysis, the absorption spectra were measured by scanning from 700 to 200 nm with a UV-1780 spectrophotometer (Shimadzu, Japan).
SDS-PAGE was accomplished using a 15 % polyacrylamide slab gel (5 % stacking gel) with the thickness of 1 mm. Electrophoresis was run at room temperature at 80 V for initial 30 min and at 120 V till the end of the run (about 1 h 45 min) and 12.5 mA. The bands were visualized with Coomassie brilliant blue R250. The sizes of the phycocyanin subunit bands were determined using a Thermo Scientific PageRuler Prestained Protein Ladder with molecular weight with broad range of 170–10 kDa (Thermo Scientific, Lithuania).
Statistical analysis
Apart of preliminary screenings, each essay was conducted twice and all values are shown as mean ± standard deviation. The phycocyanin purity indexes from sodium phosphate extraction were compared to those from ammonium chloride extraction by one-way analysis of variance (ANOVA), using Fisher LSD test for comparing means (OriginPro 9, OriginLab Corporation, USA). The level of significance was set at p < 0.05.
Results
Extraction of phycocyanin from A. platensis FACHB-314
The results from the screening phase for effective sodium phosphate concentration (data presented in supplementary material Fig. S1) showed that 0.125 M sodium phosphate (pH 7.0) was the best concentration and was chosen as the control to be used subsequent experiments. The same procedure was applied to determine the suitable concentration of ammonium chloride. Different ammonium chloride concentration (0.05, 0.075, 0.1, and 0.125 M) achieved the same phycocyanin concentration (Fig. 2a), while 0.05 M extracted phycocyanin with a higher purity ratio compared with the other samples (Fig. 2b). Thus, 0.05 M ammonium chloride was selected to be used in subsequent experiments.
The effective ratio of wet biomass to extraction salt solution was evaluated in two successive phases. In the first phase, wet biomass (in the range between 250 and 2500 mg) was mixed with 10 mL of 0.125 M sodium phosphate (pH 7.0). The results showed that the effective range was 250–500 mg per 10 mL of 0.125 M sodium phosphate (pH 7.0) (data presented in supplementary ma-terial Fig. S2).
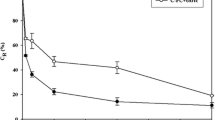
In the second phase, wet biomass (in the range of 100 and 500 mg was mixed with 10 mL of 0.125 M sodium phosphate (pH 7.0) or 0.05 M ammonium chloride (pH 4.39). The highest phycocyanin concentration was observed at wet biomass-extraction salt solution ratio of 350 mg: 10 mL in both extraction processes (i.e., sodium phosphate extraction and ammonium chloride extractions), and a high phycocyanin purity ratio was detected in the ammonium chloride crude extracts (Fig. 2).
There was no significant difference in phycocyanin concentration between crude extracts obtained from sodium phosphate extraction and ammonium chloride extraction (ANOVA: F (1,12) = 0.454, p = 0.51, α = 0.05) (Fig. 3). However, the difference in phycocyanin purity ratio between crude extracts obtained by both extraction methods is significant (ANOVA: F (1,12) = 849.08, p < 0.001, α = 0.05). The results in Fig. 2 demonstrate that extraction in 0.05 M ammonium chloride (pH 4.39) is best in achieving a high phycocyanin purity ratio of 1.5, compared to 0.79 achieved in sodium phosphate extraction. These results were also confirmed by spectroscopic analysis (Fig. 6a), where the absorbance at 280 nm shows the significant difference of an additional large amount of noncolored proteins between crude extracts in sodium phosphate and ammonium chloride.
Wet biomass and extraction salt solution ratio for efficient phycocyanin extraction from A. platensis FACHB-314. SP sodium phosphate, AC ammonium chloride; 100, 250, 300, 350, 400, 450, and 500 are amounts of wet biomass into 10 mL of extraction salt solution. Values are mean of two independent experiments
Primary purification of phycocyanin
The preliminary study revealed that the ratio of 1:2 (v/v: crude extract-precipitation salt solution) is a suitable ratio to precipitate phycocyanin with a high purity ratio and concentration. The ratio of 1:1 showed persistence of the blue color in the supernatant indicating the presence of some remaining phycocyanins in the supernatant. The ratio of 1:4 resulted in less quantity of phycocyanin, while the ratios of 1:2 and 1:3 showed a clear supernatant, indicating that the precipitation was completed. A high purity index was obtained when the ratio of 1:2 was used (Fig. 4b). Therefore, the 1:2 (v/v: crude extract volume per ammonium sulfate volume) was chosen for the next experiments.
Results from precipitation as primary purification of phycocyanin from A. platensis FACHB-314. a The comparison of efficiencies about different salts in phycocyanin precipitation. b The ratio of crude extract to precipitation salt solutions for phycocyanin precipitation. Values are mean of two independent experiments
The efficiency of salts was evaluated in terms of reduction of impurities (including mainly noncolored proteins) and recovery of high amount of phycocyanin. This evaluation was estimated by purity ratio A620/A280 (Fig. 4a) and absorption spectrum of precipitated samples, where there was a notable reduction in absorption at 280 nm i.e., A280 (Fig. 5). It proved the efficiency of one single precipitation process to remove a considerable amount of noncolored contaminated proteins. These observations were supported by the A620/A280 values that increased from 0.853 (in crude extracts) to 2.373 and 2.364 after precipitation by ammonium phosphate and ammonium sulfate, respectively (Fig. 4a). The above optimum conditions were applied in preliminary primary purification of crude extracts presented in Fig. 3 (SP-400 and AC-400); the phycocyanin purity ratio increased from 0.78 and 1.50 to 1.95 and 2.81 after one single step ammonium phosphate precipitation, and increased to 1.94 and 2.78 after one single step ammonium sulfate precipitation, respectively. The absorption spectra are presented in Fig. 6b, c.
The absorption spectra of crude extract in sodium phosphate, crude extract in ammonium chloride, and precipitated samples by ammonium sulfate and ammonium phosphate. SP: crude extract extracted by using 0.125 M sodium phosphate (pH 7.0); AC: crude extract extracted by using 0.05 M ammonium chloride (pH 4.39); 400: biomass-extraction salt solution ratio where 400 mg of wet biomass was mixed with 10 mL of extraction salt solution; AS: precipitated sample by using ammonium sulfate; AC: precipitated sample by using ammonium phosphate
Characterization of phycocyanin
The characterization of phycocyanin in crude extracts and precipitated samples was conducted by spectroscopic and the SDS-PAGE electrophoresis analysis. Absorption spectra of phycocyanin showed that there was a difference in purity of phycocyanin obtained from crude extracts in sodium phosphate and ammonium chloride extractions (Fig. 6a), with a significant difference in amount of noncolored protein contamination between those crude extracts as indicated by absorption at 280 nm, while the absorption peaks in between 550 and 650 nm presented almost the same value.
Figure 6b depicts the comparison between absorption spectra of the crude extract displayed in sodium phosphate and its derived precipitated samples, and Fig. 6c presents the comparison between absorption spectrum of the crude extract in ammonium chloride and its derived precipitated samples. It can be seen clearly from the absorption at 280 nm in Fig. 6b, c that the preliminary purification by ammonium sulfate and ammonium phosphate precipitation resulted in a considerable reduction of noncolored proteins.
The SDS-PAGE electrophoresis analysis (Fig. 7) indicated that phycocyanin migrated as two bands with apparent molecular weight of about 14 and 16 kDa corresponding to the two phycocyanin subunits α and β, respectively.
Coomassie Brilliant Blue R250 stained 15 % SDS-PAGE of crude extracts of phycocyanin from A. platensis FACHB-314 after primary purification by precipitation. Lane 1, SP-400; lane 2, AC-400; lane 3, SP-400-AS; lane 4, SP-400-AP; lane 5, AC-400-AS; lane 6, AC-400-AP. Abbreviations have the same meanings as in Fig. 6
Discussion
The downstream process (i.e., extraction, purification processes) is one of the most important required processes to obtain phycocyanin with high purity index from algal biomass (Manirafasha et al. 2016). The cost of phycocyanin is frequently determined by the quality (measured in terms of purity index) and concentration of desired end product, which is strongly dependent on series steps used in the process (Moraes and Kalil 2009). The phycocyanin recovery from microalgal biomass combines various steps, which can be summarized into four main steps: cell disruption, extraction, purification, and characterization of phycocyanin. In addition, each step may be carried out using various techniques (Reis et al. 1998). Moraes and Kalil (2009) demonstrated that the rearrangement of steps in the downstream process may lead to different end product yields and purity indexes. The high purity index of phycocyanin has been achieved by combing a series of steps of extraction and purification methods that lead to the loss of the product yield and high downstream process cost. A minimization of production steps may contribute to a reduction in of process costs and an increase in product yield. Thus, optimization of extraction step is one strategy for boosting phycocyanin purity index in crude extract and contributing to the minimization of the series steps for downstream processing.
Sodium phosphate extraction is commonly used in extraction of phycocyanin from microalgae (Su et al. 2014); unfortunately, the purity ratio of this crude extraction method is still low and does not reach food grade (A620/A280 = 0.7) in some cases. Sodium phosphate extraction of phycocyanin from microalgae has been reported by Martelli et al. (2014) with a 0.45 purity ratio in crude extract; Walter et al. (2011) with 0.7, 0.8, and 1.1 purity ratio in crude extracts; Santiago-Santos et al. (2004) with 0.4 phycocyanin purity ratio in crude extract. Moreover, besides sodium phosphate, other salt solutions (including distilled water, calcium chloride, sodium chloride) for phycocyanin extraction have been reported with low phycocyanin purity ratio in the crude extract Soni et al. (2008).
Based on the Hofmeister series salts (HSS), ammonium chloride could be more effective than sodium phosphate in phycocyanin extraction from A. platensis. Hofmeister series salt (HSS) solutions have been applied in extraction and purification of biomolecules due to their salting-in and salting-out properties. The salting-in and salting-out properties of HSS are more pronounced for anionic salt component compared to cations (Lezin et al. 2011), the reason why the present work has focused on Cl− and HPO4 2− than their respective cations to explain their effect on phycocyanin extraction. Cl− is a stronger chaotrope than HPO4 2− (i.e., HPO4 2− is kosmotrope), thus attributing ammonium chloride more salting-in ability than sodium phosphate. Thus, it would be better to use a salt solution containing HPO4 2− anion for purification (example: precipitation) instead of extraction.
The ammonium chloride utilized in this work was found to be an effective and suitable salt solution for raising the phycocyanin purity index in crude extracts. Furthermore, it reduces the extraction cost because it is cheaper. The optimum wet biomass to extraction ratio was in the range between 25 and 50 mg to 10 mL of extraction salt solution. These results were in the same range with the ratio used by Setyoningrum and Nur (2015), who used 40 mg of wet biomass.
The extraction in 0.05 M ammonium chloride (pH 4.39) was the best because of the high purity ratio A620/A280 in the crude extract. The crude extract in ammonium chloride (Fig. 3) achieved an optimum phycocyanin purity ratio of 1.5 with overall mean of 1.428 in seven assays and 2.81 after single-step precipitation. These results clearly demonstrate that ammonium chloride reduce the series of phycocyanin purification steps, thus reducing the production cost while achieving the same or better results than ones obtained after several purification steps. Martelli et al. (2014) obtained the purity ratio of 1.4 after triple filtration steps by standard 5-μm filter paper membrane, microfiltrated using a 0.2-μm syringe filter and ultrafiltration (using a membrane cutoff of 10 kDa). Santiago-Santos et al. (2004) reported 0.4 phycocyanin purity ratio in crude extract and 2.2 after ion exchange chromatography. Soni et al. (2008) reported 0.42 in crude extract in Tris-HCl buffer and 0.86 phycocyanin purity ratio after ammonium chloride fractionation. Kumar et al. (2014) reported a 0.75 phycocyanin purity ratio in crude extract and 1.5 phycocyanin purity ratio after ammonium sulfate precipitation. Kamble et al. (2013) reported a 0.161 phycocyanin purity ratio in crude extract and 0.248, 0.628, 1.08, and 2.317 phycocyanin purity ratio after 25 % ammonium sulfate precipitation, 50 % ammonium sulfate precipitation, dialysis, and Sephadex G-25, respectively. Patel et al. (2005) reported a 0.80 phycocyanin purity ratio in crude extract, 0.82 after fractional precipitation with 25 % ammonium sulfate, and 2.66 after fractional precipitation with 50 % ammonium sulfate with Arthrospira, and the value was less by using other strains.
The results from A620/A280 values, spectroscopic, and SDS-PAGE analysis have shown that ammonium chloride does not affect the structural characteristics of phycocyanin from A. platensis FACHB-314, and the achieved phycocyanin purity ratio is relatively high compared to that achieved with sodium phosphate extraction. Additional advantage of the present method is that two steps have been combined in one unity operation, thus contributing to the reduction of extraction steps and time (for example, comparing the present work to the previous work done by Kannaujiya and Sinha (2016) sonication step has been eliminated from the process).
In conclusion, ammonium chloride was demonstrated as an effective and suitable extraction salt solution for phycocyanin extraction from A. platensis FACHB-314. Ammonium sulfate and ammonium phosphate precipitation as the preliminary primary purification step has proven that ammonium chloride as extraction salt solution does not hinder subsequent downstream processing of phycocyanin. Furthermore, if more extraction factors are well evaluated, ammonium chloride would be an excellent salt solution to provide phycocyanin with high purity index. The ammonium phosphate results have shown that it is an effective precipitation agent that can be used as an alternative method to ammonium sulfate in biomolecules precipitation. The ammonium chloride extraction can be associated with the novel purification method to obtain analytical grade phycocyanin without many purification steps.
References
Aiba S, Ogawa T (1977) Assessment of growth yield of a blue—green alga, Spirulina platensis, in axenic and continuous culture. J Gen Microbiol 102:179–182
Baldwin RL (1996) How Hofmeister ion interactions affect protein stability. Biophys J 71:2056–2063
Bennett A, Bogorad L (1973) Complementary chromatic adaptation in a filamentous blue-green alga. J Cell Biol 58:419–435
Borowitzka MA (2013) High-value products from microalgae—their development and commercialisation. J Appl Phycol 25:743–756
Colla LM, Oliveira Reinehr C, Reichert C, Costa JAV (2007) Production of biomass and nutraceutical compounds by Spirulina platensis under different temperature and nitrogen regimes. Bioresource Technol 98:1489–1493
Eithar E-M (2014) Efficient methods for fast, producible, C-phycocyanin from Thermosynechococcus elongatus. J Res Biol 3:1132–1146
Eriksen NT (2008) Production of phycocyanin—a pigment with applications in biology, biotechnology, foods and medicine. Appl Microbiol Biot 80:1–14
Fernández-Rojas B, Hernández-Juárez J, Pedraza-Chaverri J (2014) Nutraceutical properties of phycocyanin. J Funct Foods 11:375–392
Hemlata GP, Fareha B (2011) Studies on Anabaena sp. nccu-9 with special reference to phycocyanin. J Algal Biomass Util 2:30–51
Hoseini S, Khosravi-Darani K, Mozafari M (2013) Nutritional and medical applications of Spirulina microalgae. Mini Rev Med Chem 13:1231–1237
Kamble SP, Gaikar RB, Padalia RB, Shinde KD (2013) Extraction and purification of C-phycocyanin from dry Spirulina powder and evaluating its antioxidant, anticoagulation and prevention of DNA damage activity. J Appl Pharmaceut Sci 3:149–153
Kannaujiya VK, Sinha RP (2016) Thermokinetic stability of phycocyanin and phycoerythrin in food-grade preservatives. J Appl Phycol 28:1063–1070
Kumar D, Dhar DW, Pabbi S, Kumar N, Walia S (2014) Extraction and purification of C-phycocyanin from Spirulina platensis (CCC540). Indian J Plant Physiol 19:184–188
Lezin G, Kuehn MR, Brunelli L (2011) Hofmeister series salts enhance purification of plasmid DNA by non-ionic detergents. Biotechnol Bioeng 108:1872–1882
Liu Y, Xu L, Cheng N, Lin L, Zhang C (2000) Inhibitory effect of phycocyanin from Spirulina platensis on the growth of human leukemia K562 cells. J Appl Phycol 12:125–130
Manirafasha E, Ndikubwimana T, Zeng X, Lu Y, Jing K (2016) Phycobiliprotein: potential microalgae derived pharmaceutical and biological reagent. Biochem Eng J 109:282–296
Martelli G, Folli C, Visai L, Daglia M, Ferrari D (2014) Thermal stability improvement of blue colorant C-phycocyanin from Spirulina platensis for food industry applications. Process Biochem 49:154–159
Moraes C, Sala L, Cerveira G, Kalil S (2011) C-phycocyanin extraction from Spirulina platensis wet biomass. Braz J Chem Eng 28:45–49
Moraes CC, Kalil SJ (2009) Strategy for a protein purification design using C-phycocyanin extract. Bioresource Technol 100:5312–5317
Parmar A, Singh NK, Pandey A, Gnansounou E, Madamwar D (2011) Cyanobacteria and microalgae: a positive prospect for biofuels. Bioresource Technol 102:10163–10172
Patel A, Mishra S, Pawar R, Ghosh P (2005) Purification and characterization of C-phycocyanin from cyanobacterial species of marine and freshwater habitat. Protein Expres Purif 40:248–255
Patil G, Raghavarao KSMS (2007) Aqueous two phase extraction for purification of C-phycocyanin. Biochem Eng J 34:156–164
Reis A, Mendes A, Lobo-Fernandes H, Empis JA, Novais JM (1998) Production, extraction and purification of phycobiliproteins from Nostoc sp. Bioresource Technol 66:181–187
Rito-Palomares M, Nuñez L, Amador D (2001) Practical application of aqueous two-phase systems for the development of a prototype process for c-phycocyanin recovery from Spirulina maxima. J Chem Technol Biot 76:1273–1280
Sagara T, Nishibori N, Kishibuchi R, Itoh M, Morita K (2015) Non-protein components of Arthrospira (Spirulina) platensis protect PC12 cells against iron-evoked neurotoxic injury. J Appl Phycol 27:849–855
Santiago-Santos MC, Ponce-Noyola T, Olvera-Ramı́rez R, Ortega-López J, Cañizares-Villanueva RO (2004) Extraction and purification of phycocyanin from Calothrix sp. Process Biochem 39:2047–2052
Setyoningrum Tutik M, Nur MMA (2015) Optimization of C-phycocyanin production from S. platensis cultivated on mixotrophic condition by using response surface methodology. Biocatal Agricult Biotechnol 4:603–607
Sigma-Aldrich (2016) http://www.sigmaaldrich.com/catalog/search?&interface=All&N=0+14577477&mode=partialmax&lang=en®ion=SG&focus=product. Accessed 12 March 2016
Sobiechowska-Sasim M, Stoń-Egiert J, Kosakowska A (2014) Quantitative analysis of extracted phycobilin pigments in cyanobacteria—an assessment of spectrophotometric and spectrofluorometric methods. J Appl Phycol 26:2065–2074
Soni B, Trivedi U, Madamwar D (2008) A novel method of single step hydrophobic interaction chromatography for the purification of phycocyanin from Phormidium fragile and its characterization for antioxidant property. Bioresource Technol 99:188–194
Su C-H, Liu C-S, Yang P-C, Syu K-S, Chiuh C-C (2014) Solid–liquid extraction of phycocyanin from Spirulina platensis: kinetic modeling of influential factors. Sep Purif Technol 123:64–68
Sun L, Wang S, Gong X, Zhao M, Fu X, Wang L (2009) Isolation, purification and characteristics of R-phycoerythrin from a marine macroalga Heterosiphonia japonica. Protein Expres Purif 64:146–154
Ullah K, Ahmad M, Sofia SVK, Lu P, Harvey A, Zafar M, Sultana S, Anyanwu CN (2014) Algal biomass as a global source of transport fuels: overview and development perspectives. Prog Nat Sci 24:329–339
Walter A, Carvalho JC, Soccol VT, Faria ABB, Ghiggi V, Soccol CR (2011) Study of phycocyanin production from Spirulina platensis under different light spectra. Braz Arch Biol Technol 54:675–682
Wang Z, Zhang X (2016) Characterization and antitumor activity of protein hydrolysates from Arthrospira platensis (Spirulina platensis) using two-step hydrolysis. J Appl Phycol. 28:3379–3385
Wang WJ, Zhang XL, Xu CB, Cheng HY (2012) Purification and concentration of C-phycocyanin from Spirulina platensis using aqueous two-phase system. Appl Mech Mater 138-139:995–1001
Wojtasiewicz B, Stoń-Egiert J (2016) Bio-optical characterization of selected cyanobacteria strains present in marine and freshwater ecosystems. J Appl Phycol 28:2299–2314
Xia D, Liu B, Xin W, Liu T, Sun J, Liu N, Qin S, Du Z (2016) Protective effects of C-phycocyanin on alcohol-induced subacute liver injury in mice. J Appl Phycol 28:765–772
Xie Y, Jin Y, Zeng X, Chen J, Lu Y, Jing K (2014) Fed-batch strategy for enhancing cell growth and C-phycocyanin production of Arthrospira (Spirulina) platensis under phototrophic cultivation. Bioresource Technol 180:281–287
Zhang Y, Cremer PS (2006) Interactions between macromolecules and ions: the Hofmeister series. Curr Opin Chem Biol 10:658–663
Acknowledgments
The authors are grateful to the Government of Rwanda through University of Rwanda-College of Education for giving a Ph.D. study leave to Mr. Manirafasha Emmanuel and the Government of People’s Republic of China for financially supporting his Ph.D. studies. This work was supported by the National Natural Science Foundation of China (No. 31071488), the National High Technology Research and Development Program 863, China (No. 2014AA021701), and the State Key Program of National Natural Science Foundation of China (No. 21336009).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Manirafasha, E., Murwanashyaka, T., Ndikubwimana, T. et al. Ammonium chloride: a novel effective and inexpensive salt solution for phycocyanin extraction from Arthrospira (Spirulina) platensis . J Appl Phycol 29, 1261–1270 (2017). https://doi.org/10.1007/s10811-016-0989-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-016-0989-y