Abstract
Microcystis, a genus of cyanobacteria that is dominant in eutrophic lakes, occurs mainly as colonial morphs under natural conditions but as single cells in laboratory cultures. Recent studies have suggested that Microcystis–bacteria interactions significantly influence Microcystis morphology, but the underlying mechanism remains unclear. In this study, a total of 48 strains of heterotrophic bacteria were purified from Microcystis mucilage. Five bacteria, Aeromonas veronii, Enterobacter aerogenes, Exiguobacterium acetylicum, Bacillus cereus and Shewanella putrefaciens, can induce unicellular Microcystis to form colonies. Heterotrophic bacteria stimulated Microcystis growth and induced the production of extracellular polymeric substances in coculture treatments. Extracellular polymeric substances, such as extracellular polysaccharides (EPS), were responsible for the mucilage formation in colonial Microcystis. We analysed extracellular metabolic compounds produced by Microcystis aeruginosa and Microcystis wesenbergii using gas chromatography mass spectrometry. Filtrate extracts from coculture treatments indicated that some compounds, such as 2-dodecen-1-yl(-) succinic anhydride and benzoic acid, 2,3-bis[(trimethylsilyl)oxy]-, trimethylsilyl ester, might play a significant role in colonial M. aeruginosa or M. wesenbergii formation. Our data suggested that the interaction of Microcystis and heterotrophic bacteria was crucial for the formation of Microcystis colony and outbreak of Microcystis blooms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Large mucilaginous cyanobacterial blooms occur worldwide in freshwater lakes and oceans (Chen et al. 2009; Kurmayer et al. 2003). Species of Microcystis, such as Microcystis aeruginosa, Microcystis wesenbergii, Microcystis flos-aquae and Microcystis viridis, are dominant bloom-forming strains in China. Human activity and economic development are considered as main factors to promote the eutrophication and Microcystis blooms (Jancula and Marsalek 2011), but the mechanism of Microcystis as dominant species in eutrophic lakes is still unknown.
Microcystis includes colonial morph types frequently inhabiting eutrophic lakes. An important strategy for sustaining the competitive advantage of Microcystis is to maintain colonial morphology in eutrophic aquatic ecosystems (Reynolds et al. 1981). The colonial Microcystis has thick mucilage around Microcystis cells compared with their unicellular forms (Plude et al. 1991). The mucilage provides protective barriers for Microcystis and their attached bacteria from stress and adverse conditions (Pereira et al. 2009). Rippka (1988) and Wu and Song (2008) also show that colonial Microcystis have a competitive advantage compared with other phytoplankton in their life cycle, related to nutrients, grazer resistance, vertical migration and oxygen damage. However, colonial Microcystis cultures lose their mucilage and convert into individual cells gradually after some generations in the laboratory (Plude et al. 1991). In addition, the morphology of colonial Microcystis can be influenced by both abiotic and biotic factors, such as zooplankton predation (Burkert et al. 2001), microcystins stress (Sedmak and Eleršek 2006), nitrogen (N), phosphorus (P) and pH (Ma et al. 2014), and water residence time (Romo et al. 2013). Importantly, there are many bacteria living in the mucilage of colonial Microcystis (Doucette 1995; Brunberg 1999), but some bacteria disappear after colonies disaggregate into unicellular forms under axenic conditions in the laboratory (Shirai et al. 1989). Shen et al. (2011) also showed that heterotrophic bacteria were an important factor related to Microcystis colony formation, but the mechanism of how bacteria impact on Microcystis colony formation remains unclear.
The mucilage is the fundamental substance responsible for colonial Microcystis formation and development (Kessel and Eloff 1975; Plude et al. 1991). It consists primarily of extracellular polymeric substances (extracellular polysaccharides [EPS], proteins, nucleic acids and so on) secreted by Microcystis or associated bacteria (Xu et al. 2013). The mucilage is a mutual microenvironment for both algae and bacteria, and some interactions such as nutrient exchange inside the mucilage could stimulate the growth of both Microcystis and bacteria (Brunberg 1999; Imai et al. 1999). On the other hand, there are also some negative interactions between Microcystis and its associated bacteria. Some Microcystis species can produce hepatotoxins, which have a negative impact on the growth of Escherichia coli and Streptoverticillium sp. (Valdor and Aboal 2007). Conversely, bacteria, such as Brevibacillus sp. and Alcaligenes denitrificans, can produce metabolites to lyse of Microcystis cells directly or indirectly (Manage et al. 2001). These studies indicate that extracellular polymeric substances secreted by Microcystis and bacteria influence the interactions of Microcystis and their associated bacteria and may be responsible for Microcystis morphology change.
Despite the large number of field and laboratory studies that have been carried out, the interactions between colonial Microcystis formation and their attached bacteria are still not fully understood. To clarify the mechanism of colonial Microcystis formation and better understand the relationship between the alga and its associated bacteria, it is necessary to rebuild the Microcystis colonies in the laboratory. We hypothesise that heterotrophic bacteria present in the mucilage of Microcystis and their associated extracellular polymeric substances are crucial for the formation and maintenance of colonial Microcystis. In the present study, heterotrophic bacteria were isolated and purified from Microcystis mucilage, and the effects of monoclonal bacteria strains on morphology change of unicellular Microcystis were investigated. During the coculture of heterotrophic bacteria and Microcystis, the growth of Microcystis and bacteria, and the contents of extracellular compounds, including extracellular polysaccharides, proteins and nucleic acids, were also determined. Our data will help understand the role of heterotrophic bacteria in the dynamics of Microcystis blooms and the relationship between alga and its associated bacteria.
Methods
Isolation and cultivation of bacteria and algae
Microcystis samples were collected from hypertrophic areas of Lake Taihu (Jiangsu, China), Lake Dianchi (Yunnan, China) and Lake Donghu (Hubei, China) in July 2011. The Microcystis biomass was greater than 90 % of total algal biomass in the study lakes. The free-living bacteria were removed from Microcystis colonies according to Shi et al. (2010). Then, Microcystis colonies were washed three times with sterile deionised water to dissolve Microcystis mucilage and release attached bacteria (Shen et al. 2011). Aliquots of water containing the released attached bacteria were spread onto sterile nutrient agar plates containing 1 g L−1 peptone, 0.5 g L−1 NaCl, 0.5 g L−1 beef extract and 20 g L−1 agar. A total of 48 strains of the attached bacteria were isolated from the mucilage of Microcystis and frozen at −80 °C in 50 % glycerol for subsequent experiments. Seven unicellular Microcystis strains were used in the present study. M. aeruginosa PCC7806 and M. aeruginosa FACHB905 were obtained from Freshwater Algae Culture Collection at the Institute of Hydrobiology (Wuhan, China). M. wesenbergii CH, M. wesenbergii TH, M. viridis, M. flos-aquae and Microcystis sp. were isolated from Lake Chaohu in 2010. To reduce contaminants and obtain axenic strains of algae, unicellular algal cells were alternately cultured in solid and liquid culture media (Parker 1982). Prior to the experiment, 4′,6′-diamidino-2-phenolindole (DAPI) staining and fluorescence microscopy were used to detect whether or not Microcystis strains were contaminated with bacteria (Porter 1980). When contaminant bacteria presented in the algal culture, the alga was discarded.
Bacteria strains screening experiment
To select bacteria which can induce the aggregation of unicellular algae into colonies, a total of 48 strains of bacteria and seven strains of algae were cocultured respectively. Seven strains of unicellular algae at the exponential stage were collected and transferred into 1-L Erlenmeyer flasks containing 500-mL autoclaved BG11 standard medium (adjust pH to 7.1 with 1 M NaOH or HCl). The formula of BG11 medium was obtained from Freshwater Algae Culture Collection at the Institute of Hydrobiology (Wuhan, China). All treatments were incubated in batch cultures of BG11 medium at 25 ± 1 °C and a 12:12-h light/dark cycle at an irradiance of 50 μmol photons m−2 s−1. Erlenmeyer flasks were shaken three times every day. Following incubation, morphological changes in Microcystis cells were observed daily by microscopic examination. The phenomenon that obvious Microcystis mucilage appeared to link Microcystis cells into macroscopic groups in culture was used for the standard of colonial Microcystis formation (Plude et al. 1991). The colony size of colonial Microcystis formed in coculture was calculated according to Ishikawa et al. (2004) and Shen et al. (2011).
Extraction of bacterial genomic DNA
The whole genomic DNA of 48 strains of bacteria was extracted using a bacterial DNA Kit (Omega, Bio-Tek, USA) according to the manufacturer’s recommendations. The 16S rDNA was amplified by PCR using the universal primer 27F, AGAGTTTGATCCTGGCTCAG, and 1492R, TACGGY TACCTTGTTACGACTT. The PCR products were submitted for sequencing using an automated ABI DNA sequencer at the Genomics Company (Beijing, China). Bacterial sequences were compared to known sequences in RDP database (http://rdp.cme.msu.edu/seqmatch/seqmatch_intro.jsp) to match the most similar sequences.
The colonial Microcystis cells which appeared in cocultures were termed Microcystis colony-inducing bacteria. These five bacteria were Aeromonas veronii (Bac-AV), Enterobacter aerogenes (Bac-EN), Exiguobacterium acetylicum (Bac-EA), Bacillus cereus (Bac-BC) and Shewanella putrefaciens (Bac-SP). The 16S rDNA sequences of these five bacteria have been submitted to NCBI with the following accession number KC166864-KC166868. Thus, in the present study, M. aeruginosa FACHB905, M. wesenbergii CH, Bac-EA and Bac-SP were selected for further investigation.
Microcystis and bacterial abundance
The screening experiment showed that both Microcystis and bacteria grew rapidly when the initial concentration of Microcystis and bacteria was 1 × 107 and 1 × 106 cells mL−1, respectively. Therefore, the above concentrations of algae and bacteria were selected as initial concentrations on day 0. Unicellular Microcystis cells were harvested at the exponential phase and added to 500-mL Erlenmeyer flasks containing 400 mL of autoclaved BG11 medium. All treatments were incubated at 25 ± 1 °C and illuminated in a 12:12-h light/dark cycle at an irradiance of 50 μmol photons m−2 s−1. Each treatment was replicated three times. Samples were collected daily, and the experiment lasted for 7 days.
Before counting the unicellular and colonial Microcystis in a haemocytometer using an Olympus BX50 light microscope (Tokyo, Japan), Microcystis cells were stained with Lugol’s iodine solution (Joung et al. 2006). For accurate counting, Microcystis cells were released from the colonial Microcystis using ultrasound. The size of colony area Microcystis was measured following the method of Ishikawa et al. (2004). Samples were preserved in 4 % (v/v) formaldehyde for determination of bacterial abundance. The fixed samples were then stained with DAPI, and cell amounts of bacteria were counted under an epifluorescence microscope (Zeiss Axioskop 20, Germany) (Porter 1980). A minimum of 10 replicates under microscope were counted for each sample.
Protein and nucleic acid determination
The samples were heated at 65 °C for 5 min to release proteins and nucleic acids from Microcystis or bacteria cells. Protein contents were measured according to Bradford (1976) using bovine serum albumin as a standard. Nucleic acid concentrations were measured by the diphenylamine colorimetric method using calf thymus DNA as a standard (Burton 1956).
Extractions and determination of EPS
EPS were classified into soluble EPS (SL-EPS), loosely bound EPS (LB-EPS) and tightly bound EPS (TB-EPS). The samples were centrifuged at 2500×g for 15 min, and the supernatant was used to measure SL-EPS. The harvested precipitates were suspended in 0.05 % NaCl solution and centrifuged at 5000×g for 15 min. The supernatant was carefully collected for measurement of LB-EPS. The remaining precipitates were resuspended with 0.05 % NaCl solution and treated for TB-EPS extraction using the method of Xu et al. (2013). All solutions were filtered using 0.45-μm membrane filters (Millipore, USA). As a blank, BG11 medium was used for SL-EPS, and 0.05 % NaCl solutions were used for LB-EPS and TB-EPS. Polysaccharide contents were determined by the phenol–sulphuric acid method using glucose as a standard (Dubois et al. 1956).
Dichloromethane extraction
Cultures from control (Microcystis or bacteria) and coculture (Microcystis and bacteria) treatments were collected 4 days after inoculation. The filtrates were separated from Microcystis and bacteria cells using the 0.22-μm membrane filters (Millipore, USA). The extracellular compound extraction used the method described by Zhang et al. (2013). Dichloromethane (dichloromethane/culture filtrates = 2:1) was used to extract metabolites from the above filtrates. The dichloromethane extracts were analysed on a Gas Chromatograph Mass Spectrometer (GC/MS 7890A+5975C, Agilent). Chemical types of compounds were identified with NIST08.1 and Wiley 275 libraries. Authentic samples were also compared with the mass spectra and retention times of chromatographic peaks.
Statistical analysis
Results are expressed as mean ± standard deviation. Statistical analysis was carried out using one-way ANOVA, followed by Tukey’s post hoc test to determine any significant differences among different treatments. Standard statistical analysis and graphics were carried out using Microcal™ Origin 8.0 (Microcal Software Inc., USA).
Results
Bacterial identification by phylogenetic analysis
A total of 48 bacterial strains were isolated from the mucilage of Microcystis colonies. The affiliations of isolated bacteria originating from Microcystis mucilage were mainly dominated by Firmicutes (17/48) and Gammaproteobacteria (13/48), followed by Epsilonproteobacteria (7/48) (Fig. S1). When unicellular strains Microcystis spp. were exposed to the above 48 strains, five bacterial strains Bac-AV, Bac-EN, Bac-EA, Bac-BC and Bac-SP were observed to induce a change from unicellular Microcystis into colonies (Table 1). According to the molecular identification, Bac-AV was classified under Gammaproterobacteria, and Bac-BC was classified under Firmicutes, which were obtained from Lake Taihu. Bac-EN and Bac-SP were classified under Gammaproterobacteria, which were obtained from Lake Dianchi. Bac-EA was classified under Firmicutes and obtained from Lake Donghu.
Table 1 shows a possible species-specific relationship between Microcystis and bacteria. However, only four heterotrophic bacteria strains Bac-AV, Bac-EN, Bac-EA and Bac-BC could induce colony formation of unicellular M. aeruginosa FACHB905 and M. aeruginosa PCC7806. These bacteria could not induce colony formation of other algae species. In addition, only Bac-SP induced colony formation of unicellular M. wesenbergii CH and M. wesenbergii TH. At the end of the experiment, most of unicellular Microcystis aggregated to colonies in the coculture treatments of Microcystis and bacteria (Fig. S2). No morphology changes of other algae were observed.
Interaction between bacteria and Microcystis
As shown in Fig. 1a, d, Microcystis cells in controls still maintained the unicellular form during the whole experiment. Upon exposure to Bac-EA and Bac-SP, unicellular Microcystis showed a tendency to aggregate. Colonial M. aeruginosa and M. wesenbergii, formed in the present experiment, did not show significant differences in morphology, and the morphology of colonial Microcystis was different from that of colonies under field conditions (Fig. 1b, e). Both of them were lobate and reticulated, with distinct colony margins and prominent slime composed of colloid substances around the colonies. The cell surfaces of Microcystis colonies turned blue after staining with Alcian Blue (Fig. 1c, f), indicating the presence of acidic polysaccharides in the colonies.
Micrographs of M. aeruginosa and M. wesenbergii on day 4. a Control M. aeruginosa, b unicellular M. aeruginosa inoculated with Bac-EA, c colonial M. aeruginosa formed stained with Alcian Blue, d control M. wesenbergii, e unicellular M. wesenbergii inoculated with Bac-SP, and f colonial M. wesenbergii formed stained with Alcian Blue. Scale bar = 10 μm
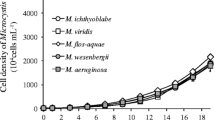
M. aeruginosa FACHB 905 started to aggregate 2 or 3 days after inoculation with the bacterium Bac-EA. The colony sizes reached a maximum aggregation size of 800 μm2 after 5 days (Fig. 2). Other three bacteria (Bac-AV, Bac-EN and Bac-BC) also stimulated aggregation in M. aeruginosa, and the formed colonies exhibited similar morphological changes. Unicellular M. wesenbergii CH started to form colonies in the presence of Bac-SP and reached the maximum colony area 4 or 5 days after inoculation.
Size of colonial Microcystis after incubating with associated bacteria. Ma and Mw represent M. aeruginosa FACHB905 and M. wesenbergii CH, respectively. Bac-SP, Bac-EA, Bac-EN, Bac-AV and Bac-BC represent Shewanella putrefaciens, Exiguobacterium acetylicum, Enterobacter aerogenes, Aeromonas veronii and Bacillus cereus, respectively. Mean ± std dev (n = 3)
Growth of Microcystis and bacteria
The growth characteristics of Microcystis showed pronounced differences between control and coculture treatments during the course of experiment. As shown in Fig. 3, the solid line represents the growth of Microcystis and bacteria in the coculture treatments, and the hollow line represents the growth of Microcystis and bacteria in controls. In Fig. 3a, the growth of M. aeruginosa in coculture increased remarkably under Bac-EA at the beginning of the experiments (P < 0.05), but cell numbers declined after day 4. The growth of Bac-EA in cocultures increased much more quickly than control Bac-EA. The growth of M. wesenbergii in coculture was significantly increased by Bac-SP (P < 0.05) compared to the control M. wesenbergii at 3 days and remained unchanged thereafter (Fig. 3b). The cell numbers of Bac-SP in coculture treatments also grew faster than in the control Bac-SP.
EPS, proteins and nucleic acids determination
Concentrations of proteins, total polysaccharides and nucleic acids are greatly increased in the coculture treatments of Microcystis and bacteria on day 4 (P < 0.05) and rose with prolonged incubation time (Table 2). Besides increased EPS concentration in the coculture treatments, the EPS compositions also varied after M. aeruginosa cells became aggregated. SL-EPS in control M. aeruginosa was the major component (>50 %) of total EPS throughout the experiment. However, when Microcystis was cocultured with heterotrophic bacteria, the percentage of TB-EPS in all coculture treatments quickly increased and became the major component (>60 %) (Fig. 4). There were no obvious differences between two coculture treatments (M. aeruginosa and Bac-EA, M. wesenbergii CH and Bac-SP).
Extracellular polysaccharides (EPS) compositions in control and coculture treatments. Con and Co- represent control M. aeruginosa FACHB905, control M. wesenbergii CH, coculture of M. aeruginosa FACHB905 and Exiguobacterium acetylicum, and coculture of M. wesenbergii CH and Shewanella putrefaciens, respectively. TB-EPS, LB-EPS and SL-EPS represent tightly bound EPS, loosely bound EPS and soluble EPS, respectively. Mean ± std dev (n = 3)
Dichloromethane extraction
To explore the effects of potential compounds secreted by Microcystis and bacteria on colonial formation, the filtrates of the control and coculture treatments were extracted with CH2Cl2. For M. aeruginosa and Bac-EA, a total of 37 compounds were identified from the CH2Cl2 extract and grouped into 11 categories: alkane (12 compounds), ester (11), acetate (3), amine (3), amide (2), phenol (1), anhydride (1), anthracene (1), acid (1), alcohol (1) and alkali (1) (Table 3). A total of 28 compounds were identified in the coculture extracts, 15 compounds in the control M. aeruginosa extracts and 12 compounds in the control Bac-EA extracts. Only six compounds were observed in all the three filtrate extracts, seven compounds were observed in two extracts of control M. aeruginosa and their cocultures and nine compounds were observed in two extracts of control Bac-EA and their cocultures. Importantly, 18 compounds were identified in coculture extracts but not in two control extracts. In the two control extracts, we identified nine compounds which were not found in coculture treatments. Extracellular substances of a colonial M. aeruginosa FACHB939 obtained from Freshwater Algae Culture Collection at the Institute of Hydrobiology (Wuhan, China) were also extracted for extracellular substance detection. Six compounds were identified in control M. aeruginosa, Bac-SP, cocultures and colonial M. aeruginosa FACHB939. Only one compound was identified in cocultures and colonial M. aeruginosa FACHB939 but not others.
For M. wesenbergii, a total of 97 compounds from the control, cocultures and a colonial M. wesenbergii isolated from lakes filtrates were identified using GC/MS, which could be classified into eight categories, including alkane (10 compounds), ester (8), amine (2), phenol (2), nitrile (1), ketone (1), acetate (1) and other unknown compounds (72), not described in the subsequent article (Table 4). A total of 24 compounds were identified in the dichloromethane extraction of cocultures, 23 compounds were identified in the control M. wesenbergii CH and 25 compounds were identified in the control Bac-EA. Twelve compounds were identified in cocultures but not control M. wesenbergii and Bac-SP. Six compounds were identified in cocultures and control M. wesenbergii but not in control Bac-SP. Six compounds were identified in cocultures and control Bac-SP. A colonial M. wesenbergii isolated from Lake Chaohu in 2014 was also extracted extracellular substance. There was no compound identified in all extracts (control M. wesenbergii, control Bac-SP, cocultures and colonial M. wesenbergii), but two compounds were identified in cocultures and colonial M. wesenbergii but not others.
Discussion
The interactions between bacteria and Microcystis can be better explained by coculture of Microcystis and monoclonal bacteria strains, compared with our previous experiment on Microcystis and complex bacteria community (Shen et al. 2011). In this study, five bacteria strains were found to stimulate colonial M. aeruginosa and M. wesenbergii formation. Four bacteria, Aeromonas veronii, Enterobacter aerogenes, Exiguobacterium acetylicum and Bacillus cereus can induce unicellular M. aeruginosa into colonies, and Shewanella putrefaciens can induce unicellular M. wesenbergii into colonies. All of the five strains have been found in eutrophy lakes and were associated with cyanobacterial blooms. Liu et al. (2013) demonstrated that Aeromonas sp. can inhibit the growth of Microcystis. Enterobacter sp. related with Microcystis cells is a potential pathogenic member for humans and usually appears in the cyanobacterial blooms (Dziallas and Grossart 2011). Exiguobacterium sp. inhibits the growth of Microcystis in the cocultures (Tian et al. 2012). Bacillus cereus is a common cyanobacteriolytic bacterium (Nakamura et al. 2003). Shewanella putrefaciens strains can secret enzymes related with intermediary carbon metabolism and influence cyanobacteria growth (Scott and Nealson 1994). Thus, taken together with these studies, we speculated that species-specific relationships might exist in colonial Microcystis formation and their bacteria. Shi et al. (2012) also found a specific relationship between colonial Microcystis and their assoicated bacteria living in the mucilage. In addition, Parveen et al. (2013) showed a clear distinction between Microcystis-attached and free-living bacterial communities. These results provided evidence for specfic bacteria–Microcystis relationships. Specific coorpetation between bacteria and phytoplankton may enhance their survival in eutrophic ecosystems (Bratbak and Thingstad 1985). Wu and Song (2008) also found that the colonial Microcystis phenotype may possess a higher ability to tolerate enhanced stress conditions compared to unicellular (noncolonial) phenotypes. Interestingly, one given bacterium could not induce all the unicellular Microcystis species to form colonies. For example, in the present study, Enterobacter aerogenes (Bac-EA) induced colonial formation of M. aeruginosa FACHB 905 and M. aeruginosa PCC7806 but could not induce M. wesenbergii CH, M. wesenbergii TH or other Microcystis species, to form colonies. The findings in this study raise the question of interactions between bacteria and colonial Microcystis formation, and an understanding of these will facilitate the current understanding on the ecological success of Microcystis in eutrophic lakes.
Growth of Microcystis and bacteria
The present study clearly shows that Microcystis and their associated bacteria affected the growth significantly of Microcystis or bacteria growth in coculture treatments. The growth of Microcystis and bacteria displayed different interactions in different coculture treatments (M. aeruginosa and Bac-EA, M. wesenbergii and Bac-SP). For the coculture treatment of M. aeruginosa and Bac-EA, the cell amount of Bac-EA increased significantly in coculture treatments at the beginning of experiment compared with the controls (single culture of M. aeruginosa or Bac-EA). Extracellular organic matter released by photosynthesis from Microcystis cells can supply nutrients for bacteria growth (Bell 1984), and phytoplanktons are considered as bacterial ‘incubator’ to supply habitat and nutrients for bacteria (Worm and Sondergaard 1998). M. aeruginosa cells also increased at the beginning of experiment and may be stimulated by some substances secreted by bacteria (Brunberg 1999). However, in the later period, both the growth of Microcystis and bacteria were suppressed. This phenomenon could be explained by the combined effects of complex extracellular metabolic substances by Microcystis or bacteria (Table 3). These extracellular substances are secreted by bacteria or algae, and some compounds play a protective role in microbe growth (Armstrong et al. 2001), while others may have an algicidal role (Middelboe et al. 1995; Walker and Higginbotham 2000). For the coculture treatments of M. wesenbergii and Bac-SP, the cell count of M. wesenbergii and Bac-SP increased throughout the experiment, and metabolic substances related with growth stimulation may account for the majority of the substances produced by Microcystis or Bac-SP in coculture. In any case, colony formation may have some connection with the concentration of Microcystis cells (Shen et al. 2011), and the growth of Microcystis provided enough cells for the formation colonial Microcystis. Furthermore, the bacteria present in colony Microcystis mucilage may be responsible for the growth of Microcystis cells in eutrophic lakes.
EPS, proteins and nucleic acids determination
Microcystis and bacteria in coculture treatments were enhanced to secrete extracellular substances compared with the controls in this study. Extracellular substances, especially EPS, play important roles in mucilage formation of colonial M. aeruginosa (Parveen et al. 2013), and mucilage that served to link Microcystis and their associated bacterial cells into colonies was rebuilt by Microcystis and/or bacteria in cocultures. In the present study, extracellular substances may guarantee the mucilage formation. These results accord with previous findings that total extracellular substances in the cells and sheaths of colonial Microcystis are significantly higher than in disaggregated cells (Zhang et al. 2007). Sundh (1992) and Giroldo et al. (2007) also show that EPS released by phytoplankton is a major component of dissolved organic carbon. The EPS secreted by Microcystis and bacteria contributes organic carbon to lakes and may play an important role in the freshwater ecosystems.
EPS was classified into three types, i.e. SL-EPS, LB-EPS and TB-EPS. TB-EPS may be the crucial extracellular substance which supported colonial M. aeruginosa mucilage compared with SL-EPS and LB-EPS (Klock et al. 2007; Xu et al. 2013). As shown in Table 2 and Fig. 4, TB-EPS in coculture treatments were higher than those in controls. Therefore, TB-EPS increased by interaction between Microcystis and Bac-EA and provided the material basis for colonial Microcystis mucilage. In addition, the polysaccharides produced by cyanobacteria reportedly form the hydrogen bonds with proteins, lipids and DNA to protect cyanobacterial cells from the harsh external environment. The levels of TB-EPS, proteins and nucleic acids significantly increased in cocultures of Microcystis and bacteria, implying that Microcystis cells reached the possibly condition for colonial Microcystis formation.
Extracellular metabolic substances
To clarify the mechanism of colonial Microcystis formation, filtrate extracts of different treatments were analysed using the GC/MS. Our results showed that Microcystis and bacteria in cocultures might secret some extracellular metabolites to induce colonial Microcystis formation. We identified 18 compounds in coculture treatments but not in their controls. Some of these compounds, such as 11,13-dimethyl-12-tetradecen-1-ol acetate, methyl 2-octylcyclopropene-1-octanoate, docosane, di(2-ethylhexyl) ester, 2,7-dimethyl-2,7-octadien-1-amine, isomenthylamine and 2-ethylhexyl-hexyl ester, are antioxidants (Adams 1978; Ivanovic et al. 2012; Liu et al. 2007; Ma et al. 2011; Malloy et al. 2011; Ngang et al. 2014; Sharman et al. 1994) and may protect Microcystis or Bac-EA cells from adverse environmental conditions. In particular, N-benzyl-N-ethy-P-isopropylbenzamide, 2-dodecen-1-yl(-) succinic anhydride and 9-(methylaminomethyl) anthracene were identified in coculture extracts but not from the control group (single culture of M. aeruginosa FACHB905 or Bac-EA). These compounds might play a significant role in colonial Microcystis formation due to their polymer function (Lee et al. 2005; Lin and Chen 2006). Wang et al. (2015) also reported that N-benzyl-N-ethy-P-isopropylbenzamide, detected in the extracts of a colonial M. aeruginosa FACHB939, was responsible for organic aerosols and mucilage formation. For M. wesenbergii, a total of 24 compounds were identified in coculture extractions. A total of 12 compounds, such as 1-(2-methylallyl) azetidine, [2-pyrrolidinyl] methylamine, 2-bromopropionic acid, pentyl ester, l-valine, n-propargyloxycarbonyl, pentadecyl ester and undecanenitrile, were only identified in cocultures but not in control M. wesenbergii and Bac-SP, and these compounds may provide help for Microcystis morphology change. We found that eight compounds, including octamethylcyclotetrasiloxane, tetradecamethylhexasiloxane, benzoic acid, 2,3-bis[(trimethylsilyl)oxy], trimethylsilyl ester, tetradecanedioic acid and bis(tert-butyldimethylsilyl) ester, appear in two extracts related with colonial M. wesenbergii formed in coculture and a colonial M. wesenbergii isolated from the lake. Among the above compounds, tetradecamethylhexasiloxane and octamethylcyclotetrasiloxane, which play a crucial role in polymer formation (Tajima and Yamamoto 1987), were also identified in extracts of control Bac-SP. Thus, Bac-SP was further confirmed to be responsible for M. wesenbergii colony formation. Two compounds, benzoic acid, 2,3-bis[(trimethylsilyl)oxy], trimethylsilyl ester and (-)-epinephrine, tris(trimethylsilyl) ether, identified in cocultures and a colonial M. wesenbergii but not in other extracts, were helpful to high plant growth (Mohamed et al. 2009). For other compounds, hentriacontane, identified in cocultures and control M. wesenbergii, is a biological promoter (Kolattukudy 1968) and may be crucial for Microcystis growth. Hence, these compounds, identified in the coculture, may be responsible for Microcystis morphology change.
Comparing the extracellular metabolic substances that only appeared in two cocultures (Table S1), we found 18 compounds, including ester (4 compounds), amine (3), acetate (3), alkane (2), amide (2), acid (1), alcohol (1), anhydride (1) and anthracene (1), which were identified in the coculture of M. aeruginosa and Bac-EA (Ma and EA) but not their controls. Furthermore, 12 compounds, including esters (7), acetates (1), alkane (1), amine (1), nitrile (1) and ketone (1), were also identified in coculture of M. wesenbergii and Bac-SP (Mw and SP) but not their controls. Though the same chemical types, such as ester and acetate, were found in extracts of two cocultures (Ma and EA, and Mw and SP), there were no identical compounds in cocultures. This phenomenon can be explained by the study of Forni et al. (1997); the different polysaccharide compositions were confirmed in different Microcystis species. The different substance compositions in cocultures might be related to the different features of colonial Microcystis. Finding the extracellular metabolic of colonial Microcystis formation may contribute to a better understanding of the taxonomy and ecology of Microcystis. Moreover, variety compounds secreted by Microcystis or bacteria may release into freshwater to influence the biochemical cycle lakes.
Apart from associated bacteria and extracellular compounds, many studies reported that other factors (zooplankton, nutrients and so on) could also influence Microcystis colony formation and maintenance (Burkert et al. 2001; Ma et al. 2014). However, these factors influencing the morphological changes of Microcystis in laboratory cultures are often quite different from that in natural water bodies. Though pH and N/P ratio significantly affected the growth and colony size of Microcystis (Ma et al. 2014), the formation of colonies was independent of changes in N or P concentration and pH value in the present study. The N and P concentration in BG11 medium was sufficient for the growth of Microcystis and bacteria. The concentration of P in the medium was 7.13 and 6.77 mg L−1 before and after the experiment, respectively. The concentration of N in the medium was 247.06 and 241.42 mg L−1 before and after the experiment, respectively. The pH of each treatment did not change significantly, and pH value was approximately 6.95–7.10 during the course of experiment. In the present study, colonies rebuilt by the single bacteria strains isolated from the Microcystis mucilage. It is evident that heterotrophic bacteria play an important role in Microcystis colony formation. Anyhow, the morphological and physiological change of Microcystis may be as a result of regulating to the change of environmental conditions.
In conclusion, we obtained five key heterotrophic bacteria from the mucilage of Microcystis colonies and found five bacteria that can induce the aggregation of unicellular M. aeruginosa or M. wesenbergii into colonies for the first time. The results suggested that key heterotrophic bacteria played an important role in Microcystis colony formation. Heterotrophic bacteria and/or Microcystis produced and secreted EPS and organic/inorganic compounds change into the culture, affecting the morphology and physiology of Microcystis. The aggregation of unicellular Microcystis into colonies may be an adaptive response for Microcystis to environmental stress, that is, a regulation mechanism that enables the Microcystis cells to overcome bacteria interference and other stimulation. Therefore, the interaction of Microcystis and heterotrophic bacteria is responsible for the formation of Microcystis colony and outbreak of Microcystis blooms. More work is required to elucidate the relationship between the alga and its associated bacteria and the mechanism of Microcystis bloom in eutrophic lakes.
References
Adams JH (1978) Synergistic combinations of hydrated potassium borate, antiwear agents, and organic sulfide antioxidants. US Patent 4089790A
Armstrong E, Yan L, Boyd KG, Wright PC, Burgess JG (2001) The symbiotic role of marine microbes on living surfaces. Hydrobiologia 461:37–40
Bell WH (1984) Bacterial adaptation to low-nutrient conditions as studied with algal extracellular products. Microb Ecol 10:217–230
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bratbak G, Thingstad TF (1985) Phytoplankton-bacteria interactions: an apparent paradox? Analysis of a model system with both competition and commensalism. Mar Ecol Prog Ser 25:23–30
Brunberg AK (1999) Contribution of bacteria in the mucilage of Microcystis spp. (Cyanobacteria) to benthic and pelagic bacterial production in a hypereutrophic lake. FEMS Microbiol Ecol 29:13–22
Burkert PH, Drakare S, Blomqvist P (2001) Effects of the mixotrophic flagellate Ochromonas sp. on colony formation in Microcystis aeruginosa. Aquat Ecol 35:11–17
Burton K (1956) A study of the conditions and mechanism of the diphenylamine reaction for the colorimetric estimation of deoxyribonucleic acid. Biochem J 62:315
Chen W, Peng L, Wan N, Song L (2009) Mechanism study on the frequent variations of cell-bound microcystins in cyanobacterial blooms in Lake Taihu: implications for water quality monitoring and assessments. Chemosphere 77:1585–1593
Doucette GJ (1995) Interactions between bacteria and harmful algae: a review. Nat Toxins 3:65–74
Dubois M, Gilles KA, Hamilton JK, Rebers P, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Dziallas C, Grossart HP (2011) Temperature and biotic factors influence bacterial communities associated with the cyanobacterium Microcystis sp. Environ Microbiol 13:1632–1641
Forni C, Telo' FR, Caiola MG (1997) Comparative analysis of the polysaccharides produced by different species of Microcystis (Chroococcales, Cyanophyta). Phycologia 36:181–185
Giroldo D, Ortolano PI, Vieira AA (2007) Bacteria–algae association in batch cultures of phytoplankton from a tropical reservoir: the significance of algal carbohydrates. Freshw Biol 52:1281–1289
Imai A, Fukushima T, Matsushige K (1999) Effects of iron limitation and aquatic humic substances on the growth of Microcystis aeruginosa. Can J Fish Aquat Sci 56:1929–1937
Ishikawa K, Walker R, Tsujimura S, Nakahara H, Kumagai M (2004) Estimation of Microcystis colony size in developing water blooms via image analysis. J Jpn Soc Water Environ 4:69–72
Ivanovic J, Misic D, Zizovic I, Ristic M (2012) In vitro control of multiplication of some food-associated bacteria by thyme, rosemary and sage isolates. Food Control 25:110–116
Jancula D, Marsalek B (2011) Critical review of actually available chemical compounds for prevention and management of cyanobacterial blooms. Chemosphere 85:1415–1422
Joung SH, Kim CJ, Ahn CY, Jang KY, Boo SM, Oh HM (2006) Simple method for a cell count of the colonial cyanobacterium, Microcystis sp. J Microbiol 44:562–565
Kessel M, Eloff JN (1975) The ultrastructure and development of the colonial sheath of Microcystis marginata. Arch Microbiol 106:209–214
Klock JH, Wieland A, Seifert R, Michaelis W (2007) Extracellular polymeric substances (EPS) from cyanobacterial mats: characterisation and isolation method optimisation. Mar Biol 152:1077–1085
Kolattukudy P (1968) Tests whether a head to head condensation mechanism occurs in the biosynthesis of n-hentriacontane, the paraffin of spinach and pea leaves. Plant Physiol 43:1466
Kurmayer R, Christiansen G, Chorus I (2003) The abundance of microcystin-producing genotypes correlates positively with colony size in Microcystis sp. and determines its microcystin net production in Lake Wannsee. Appl Environ Microbiol 69:787–795
Lee JA, Kontopoulou M, Parent JS (2005) Synthesis and characterization of polyethylene-based ionomer nanocomposites. Polymer 46:5040–5049
Lin LH, Chen KM (2006) Preparation and surface activity of gelatin derivative surfactants. Colloids Surf A 272:8–14
Liu X, Dong M, Chen X, Jiang M, Lv X, Yan G (2007) Antioxidant activity and phenolics of an endophytic Xylaria sp. from Ginkgo biloba. Food Chem 105:548–554
Liu YM, Chen MJ, Wang MH, Jia RB, Li L (2013) Inhibition of Microcystis aeruginosa by the extracellular substances from an Aeromonas sp. J Microbiol Biotechnol 23:1304–1307
Ma J, Brookes JD, Qin B, Paerl HW, Gao G, Wu P, Zhang W, Deng J, Zhu G, Zhang Y, Xu H, Niu H (2014) Environmental factors controlling colony formation in blooms of the cyanobacteria Microcystis spp. in Lake Taihu, China. Harmful Algae 31:136–142
Ma QZ, Wu FJ, Zhang DQ, Peng WX (2011) Py-GC-MS analysis on benzene-alcohol extractives of Phyllostachys pubescens for biomedical engineering. Key Eng Mater 480:211–214
Malloy KL, Suyama TL, Engene N, Debonsi H, Cao Z, Matainaho T, Spadafora C, Murray TF, Gerwick WH (2011) Credneramides A and B: neuromodulatory phenethylamine and isopentylamine derivatives of a vinyl chloride-containing fatty acid from cf. Trichodesmium sp. nov. J Nat Prod 75:60–66
Manage PM, Kawabata Z, Nakano S-i (2001) Dynamics of cyanophage-like particles and algicidal bacteria causing Microcystis aeruginosa mortality. Limnology 2:73–78
Middelboe M, Søndergaard M, Letarte Y, Borch N (1995) Attached and free-living bacteria: production and polymer hydrolysis during a diatom bloom. Microb Ecol 29:231–248
Mohamed H, Ons M, Yosra ET, Rayda S, Neji G, Moncef N (2009) Chemical composition and antioxidant and radical‐scavenging activities of Periploca laevigata root bark extracts. J Sci Food Agric 89:897–905
Nakamura N, Nakano K, Sugiura N, Matsumura M (2003) A novel cyanobacteriolytic bacterium, Bacillus cereus, isolated from a eutrophic lake. J Biosci Bioeng 95:179–184
Ngang JEJ et al (2014) Characterization of Mexican coriander (Eryngium foetidum) essential oil and its inactivation of Listeria monocytogenes In vitro and during mild thermal pasteurization of pineapple juice. J Food Protect 77:435–443
Parker DL (1982) Improved procedures for the cloning and purification of Microcystis cultures (Cyanophyta) 1. J Phycol 18:471–477
Parveen B, Ravet V, Djediat C, Mary I, Quiblier C, Debroas D, Humbert JF (2013) Bacterial communities associated with Microcystis colonies differ from free living communities living in the same ecosystem. Environ Microbiol Rep 5:716–724
Pereira S, Zille A, Micheletti E, Moradas‐Ferreira P, De Philippis R, Tamagnini P (2009) Complexity of cyanobacterial exopolysaccharides: composition, structures, inducing factors and putative genes involved in their biosynthesis and assembly. FEMS Microbiol Rev 33:917–941
Plude JL, Parker DL, Schommer OJ, Timmerman RJ, Hagstrom SA, Joers JM, Hnasko R (1991) Chemical characterization of polysaccharide from the slime layer of the cyanobacterium Microcystis flos-aquae C3-40. Appl Environ Microbiol 57:1696–1700
Porter KG (1980) The use of DAPI for identifying and counting aquatic microflora. Limnol Oceanogr 25:943–948
Reynolds CS, Jaworski G, Cmiech H, Leedale G (1981) The formation of colonies or aggregates might be phenotypic response of individuals to current environmental conditions. Phil Trans R Soc B 4:419–477
Rippka R (1988) Isolation and purification of cyanobacteria. Method Enzymol 167:3
Romo S, Soria J, Fernandez F, Ouahid Y, Barón-Solá Á (2013) Water residence time and the dynamics of toxic cyanobacteria. Freshw Biol 58:513–522
Scott JH, Nealson KH (1994) A biochemical study of the intermediary carbon metabolism of Shewanella putrefaciens. J Bacteriol 176:3408–3411
Sedmak B, Eleršek T (2006) Microcystins induce morphological and physiological changes in selected representative phytoplanktons. Microb Ecol 51:508–515
Sharman M, Read WA, Castle L, Gilbert J (1994) Levels of di-(2-ethylhexyl) phthalate and total phthalate esters in milk, cream, butter and cheese. Food Addit Contam 11:375–385
Shen H, Niu Y, Xie P, Tao M, Yang X (2011) Morphological and physiological changes in Microcystis aeruginosa as a result of interactions with heterotrophic bacteria. Freshw Biol 56:1065–1080
Shi L, Cai Y, Kong F, Yu Y (2012) Specific association between bacteria and buoyant Microcystis colonies compared with other bulk bacterial communities in the eutrophic Lake Taihu, China. Environ Microbiol Rep 4:669–678
Shi L, Cai Y, Wang X, Li P, Yu Y, Kong F (2010) Community structure of bacteria associated with Microcystis colonies from cyanobacterial blooms. J Freshw Ecol 25:193–203
Shirai M, Matumaru K, Ohotake A, Takamura Y, Aida T, Nakano M (1989) Development of a solid medium for growth and isolation of axenic Microcystis strains (cyanobacteria). Appl Environ Microbiol 55:2569–2571
Sundh I (1992) Biochemical composition of dissolved organic carbon derived from phytoplankton and used by heterotrophic bacteria. Appl Environ Microbiol 58:2938–2947
Tajima I, Yamamoto M (1987) Characterization of plasma polymers from tetramethylsilane, octamethylcyclotetrasiloxane, and methyltrimethoxysilane. J Polym Sci Polym Chem 25:1737–1744
Tian C, Liu X, Tan J, Lin S, Li D, Yang H (2012) Isolation, identification and characterization of an algicidal bacterium from Lake Taihu and preliminary studies on its algicidal compounds. J Environ Sci 24:1823–1831
Valdor R, Aboal M (2007) Effects of living cyanobacteria, cyanobacterial extracts and pure microcystins on growth and ultrastructure of microalgae and bacteria. Toxicon 49:769–779
Walker HL, Higginbotham LR (2000) An Aquatic bacterium that lyses cyanobacteria associated with off-flavor of channel catfish Ictalurus punctatus. Biol Control 18:71–78
Wang X, Cotter E, Iyer KN, Fang J, Williams BJ, Biswas P (2015) Relationship between pyrolysis products and organic aerosols formed during coal combustion. Proc Combust Inst 35:2347–2354
Worm J, Sondergaard M (1998) Dynamics of heterotrophic bacteria attached to Microcystis spp. (Cyanobacteria). Aquat Microb Ecol 14:19–28
Wu ZX, Song LR (2008) Physiological comparison between colonial and unicellular forms of Microcystis aeruginosa Kütz. (Cyanobacteria). Phycologia 47:98–104
Xu H, Yu G, Jiang H (2013) Investigation on extracellular polymeric substances from mucilaginous cyanobacterial blooms in eutrophic freshwater lakes. Chemosphere 93:75–81
Zhang M, Kong FX, Tan X, Yang Z, Cao HS, Xing P (2007) Biochemical, morphological, and genetic variations in Microcystis aeruginosa due to colony disaggregation. World J Microb Biotechnol 23:663–670
Zhang P, Zhai C, Wang X, Liu C, Jiang J, Xue Y (2013) Growth competition between Microcystis aeruginosa and Quadrigula chodatii under controlled conditions. J Appl Phycol 25:555–565
Acknowledgments
We thank Dr. J. S. Owen for his constructive suggestions and professional editing, Mr Liang Chen for his help in revising the manuscript, and Dr. Yuan Niu and Juan Lin for their help in sample preparation. This study was supported by the National Natural Science Foundation of China (31400407), the National High Technology Research and Development Program of China (Grant no. 2012ZX07105-004) and State Key Laboratory of freshwater ecology and biotechnology (2014FB15 and 2014FBZ02).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, W., Shen, H., Shi, P. et al. Experimental evidence for the role of heterotrophic bacteria in the formation of Microcystis colonies. J Appl Phycol 28, 1111–1123 (2016). https://doi.org/10.1007/s10811-015-0659-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-015-0659-5