Abstract
Purpose
Cystinosis is an autosomal recessive inherited lysosomal storage disease with an incidence of 1:100.000 up to 1:200.000 caused by a gene mutation of a lysosomal transport protein resulting in deposition of cystine in lysosomes in all cells and tissues. In the cornea, crystalline, gold-dust deposition of cystine leads to visual impairment, recurrent erosions, photophobia, epiphora and blepharospasmus. Standard therapy is topical and systemic application of cysteamine which may resolve the accumulated cystine crystals.
Patient and methods
This is a case report of a thirty-one-year-old patient who already underwent renal transplantation because of nephropathic cystinosis. Visual impairment by cystine crystal deposition was aggravated by a central avascular pannus formation in his right eye. Penetrating keratoplasty was performed in intention to improve the patient’s visual acuity and life quality.
Results
After penetrating keratoplasty in the right eye, there was only a slight visual improvement. OCT scans of the macula revealed intraretinal cystine crystals and a cystoid macular edema, which was treated with a bevacizumab injection. Transmission electron microscopy of the excised cornea revealed spiky intracorneal inclusions and confocal in vivo microscopy of the left eye allowed detailed visualization of the cystine crystal deposition.
Conclusions
There is a variability of ocular manifestations of nephropathic cystinosis. Ophthalmologists have a central role in the early diagnosis of cystinosis as mostly the first manifestation are cystine crystals in the cornea. Penetrating keratoplasty may be one of the therapeutical options. Nevertheless, the patient has to be informed about the limited prognosis because of the persisting underlying disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cystinosis, also referred to as “amindiabetes,” is an autosomal recessive inherited lysosomal storage disease, which is caused by a mutation of the CTNS gene on chromosome 17p13 [1], which codes for cystinosin, a l-cystine transmembrane transport protein. This results in an impaired transport of cystine, the disulfide of the amino acid cysteine, out of the lysosomes into the cytoplasm and thereby in deposition of cystine in lysosomes in all cells and tissues. The first descriptions of its symptoms date from 1903 by Abderhalden [2]. In Germany, the total number of patients is estimated to be about 100 [3]. The disease may be subdivided in different manifestation forms [4], which are nephropathic (congenital and intermediate) or non-nephropathic (adult type). First, there is the congenital nephropathic cystinosis, which is characterized by corneal cystine deposition and renal dysfunction in the first years of life leading to a general renal tubular disorder called De-Toni-Debré-Fanconi syndrome [1]. These patients usually show growth retardation, multiple complications of cystinosis (myopathy, hypogonadism, diabetes mellitus, swallowing difficulties, pulmonary dysfunction, central nervous system deterioration) and improvement rely on renal transplantation at young age [1]. Second, in intermediate adolescent cystinosis, these characteristics do not appear until the second decade of life, and third, there is the benign adult form of cystinosis, which seems to be limited to corneal manifestations in adult life. All forms respond to therapy with cysteamine: While transport of cystine out of the lysosomes is impossible because of the defect of the transport protein, mixed disulfide of half-cystine and cysteamine complexes use another transporter (the lysine transporter) to exit the lysosomes [1]. Therefore, a therapy with cysteamine results in an efficient degradation of accumulated cystine. Early diagnosis is essential for the initiation of this treatment to prevent renal and other organ failure. When left untreated, nephropathic cystinosis patients only have a limited life expectancy up to 12 years [3].
Case report
A thirty-one-year-old male patient who already underwent renal transplantation because of nephropathic cystinosis presented in our department for the first time. In his medical history, there was renal transplantation for the first time at the age of eleven and a second time at the age of 29 years. Tacrolimus (Prograf®, Astellas Pharma Europe Ltd., Munich, Germany) was used as post-transplant immunosuppression. Visual acuity was “counting fingers” on the right eye and 20/1000 on the left eye. Neither topical therapy with cysteamine nor EDTA chelation nor phototherapeutic keratectomy (performed in another department) led to improvement of visual acuity on the right eye in the past. Slit lamp examination of both eyes showed a distinct gold-dust-like cystine crystal deposition in the cornea (Fig. 1), which made funduscopic examination and optical coherence tomography of the macula impossible (Fig. 5A). In his medical history, there was a cystoid macular edema on the right eye without treatment in the past. A retinal detachment could be excluded by sonographic examination on both eyes. Visual impairment because of cystine crystal deposition, aggravated by a central avascular pannus formation, recurrent corneal erosions, a severe limbus stem cell deficiency in the right eye (Fig. 1) and intolerance to topical cysteamine therapy in the patient’s history because of photophobia and blepharospasm required a penetrating keratoplasty first on the right eye as the therapeutic approach.
Overview and slit lamp photograph. A, Right eye, cystine crystal deposition, central avascular pannus formation, peripheral corneal neovascularization, limbus stem cell deficiency (initial presentation). B, Left eye, cystine crystal deposition, superficial limbal calcifications (initial presentation). C, Right eye after penetrating keratoplasty
In periodic-acid-Schiff-staining (PAS) of the excised cornea, small intrastromal recesses possibly resulting from cystine crystal deposition were manifest (Fig. 2A). Birefringence histology confirmed this finding by revealing cystine crystal deposition within almost all layers of the excised cornea (Fig. 2B).
Middle third of the excised cornea, A, period-acid-Schiff-staining (PAS), and B, birefringence histology, magnification 20×. Exfoliated epithelial cells (1) with subepithelial calcifications (2), missing Bowman’s layer (3), stroma (4), Descemet’s membrane (5) and preparation-related missing endothelium (6). Small intrastromal recesses (arrows) indicating cystine crystal deposition (A) and bright cystine crystal deposition (B) within the cornea (A. Zimpfer, R. Bohle)
Transmission electron microscopy (Fig. 3) revealed spiky inclusions and vacuoles between the collagen fibers in the whole stroma and in the endothelium of the excised cornea.
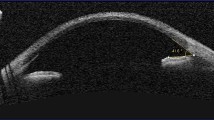
To examine the cystine deposition in the cornea of the other eye, a confocal in vivo microscopy of the left cornea was performed (Fig. 4). This examination showed an impressive epithelial and stromal cystine crystal deposition.
After penetrating keratoplasty in the right eye, there was an undisrupted postoperative healing and due to clear optic media, we found cystine crystal deposition in the iris, and optical coherence tomography (OCT) examination of the macula could be performed. The examination revealed intraretinal cystine crystals and a cystoid macular edema in the right eye (Fig. 5B), which was treated with a bevacizumab injection. After one injection, visual acuity improved up to 20/400 in the right eye (Fig. 5C). After this slight improvement with reduction in the macular edema in the OCT exam, further bevacizumab injections were performed at monthly intervals, which led to regression of the exudates and the macular edema (Fig. 5D).
Discussion
Nephropathic cystinosis is a rare disease. The diagnosis is made by measuring the leukocyte cystine content [1]. The basis of its treatment is renal transplantation and systemic cysteamine therapy [1], which may increase life expectancy dramatically. The North American Pediatric Renal Transplant Cooperative Study (NAPRTCS) reported cystinosis patients to have superior outcomes and the lowest graft failure rates among diseases leading to renal transplantation in children (familial nephritis, hemolytic uremic syndrome, congenital nephrotic syndrome, focal segmental glomerulosclerosis, [5]). Under immunosuppression with Tacrolimus (Prograf®, Astellas Pharma Europe Ltd., Munich, Germany), our patient’s post-transplant renal function was not impaired (glomerular filtration rate 138.5 ml/min). The second cornerstone of the therapy of cystinosis, the systemic cysteamine therapy, however, does not influence the corneal manifestations of the disease [6]. Therefore, topical application of cysteamine eye drops is essential to treat the anterior segment changes of the disease, which include superficial punctate keratopathy, band keratopathy, recurrent erosions, increased corneal thickness and peripheral corneal neovascularization. Intraocular manifestations, i.e., posterior synechiae, iris crystals, pigmentary retinopathy because of crystal deposition in iris, ciliary body, choroid, fundus and the optic nerve [1, 7], may also respond to systemic cysteamine application. However, about fourteen percent of patients do not tolerate systemic cysteamine therapy because of nausea, vomiting and drowsiness [1]. Topical cysteamine application in the form of eye drops may dissolve corneal cystine crystals at any age in a period of less than 1 year unless a band keratopathy [6], pannus or scars have formed. Then, penetrating keratoplasty remains the therapeutical option of first choice. In our patient, corneal cystine deposition was aggravated by recurrent erosions, photophobia, blepharospasm, a central avascular pannus formation and a severe limbus stem cell deficiency with peripheral corneal neovascularization, so that a penetrating keratoplasty was performed on his right eye. Because of clear optic media after an undisrupted postoperative healing, OCT of the macula could be performed and revealed retinal exudates with a cystoid macular edema and intraretinal cystine crystal deposition, which corresponds to other authors’ descriptions of submacular choroidal neovascularization of unknown etiology in cystinosis patients [7]. Tsilou et al. [7] speculated that the macular edema develops because of vascular endothelial growth factor release following inflammation caused by chronic cystine crystal deposition [8]. The macular edema decreased after intravitreal injection of bevacizumab and remained stable in the follow-up examinations with a slight recurrence after 10 months. The recurrence of cystine deposition in corneal allografts is controversially discussed [9].The recurrence of cystine deposition in the cornea implicates the migration of host-lysosomes within the cornea. Compared with this, renal allografts in nephropathic cystinosis show cysteine deposition but do not undergo the functional changes of cystinosis [10]. Consequently, the patient has to be informed about the possibility of a limited prognosis caused by retinal manifestations or recurrence of corneal cystine deposition in the corneal allograft because of the persisting underlying disease. If there is a recurrence of corneal cystine deposition in the corneal allograft, topical therapy with cysteamine is required again. In our patient, who did not tolerate the topical cysteamine therapy, the corneal allograft remained clear without topical cysteamine therapy in the follow-up examinations (up to now 10 months, confocal in vivo microscopy). In general, however, topical cysteamine therapy should not be stopped after corneal transplantation in nephropathic cystinosis patients to prevent a recurrence of the cystine crystal deposition in the corneal allograft.
The role of penetrating keratoplasty in cystinosis patients is controversially discussed. Fahey et al. [11] published a case report of a twenty-one-years-old male corneal cystinosis patient, who underwent cataract surgery and achieved a best corrected visual acuity of 20/20 postoperatively [11]. They concluded that visual acuity is rather affected by macular involvement than by corneal crystals in corneal cystinosis patients [11]. Indeed, retinal abnormalities including retinal crystals, submacular neovascular membranes, bone spicules and pigment clumps resembling retinitis pigmentosa and as the most common finding retinal depigmentation have been reported in cystinosis patients [8]. The retinal manifestations of cystinosis described above correlated inversely with consequent application of systemic cysteamine [8]. Our patient’s right eye was already pseudophakic, and the patient reported a progressive visual loss. His severe corneal symptoms and the impossibility of funduscopy led to the decision for penetrating keratoplasty as promising therapy, which up to now, increased our patient’s visual acuity from “counting fingers” to 20/400 on the right eye.
As in most inborn metabolic disorders, the prognosis of the nephropathic cystinosis depends on an early diagnosis. The pigmentary retinopathy, which has been seen at an age of 5 weeks [7], and the corneal cystine deposition developing in the first 16 months of life are the first symptoms of congenital nephropathic cystinosis even before the onset of renal dysfunction [6]. Consequently, ophthalmologists have a central role in the early diagnosis of congenital nephropathic cystinosis and the initiation of its appropriate treatment to prevent renal failure and resulting systemic complications.
References
Gahl WA, Thoene JG, Schneider JA (2002) Cystinosis. N Engl J Med 347:111–121
Abderhalden E (1903) Familiäre Cystindiathese. Hoppe-Seylers Z f Physiol Chem 38(5-6):557–561
Hohenfellner K, Harms E (2015) Spezielle Cystinosesprechstunde. Dtsch Ärztebl. 112:B 1160
Gräf M, Grote A, Wagner F (1992) Cysteamine eyedrops for treatment of corneal cysteine deposits in infantile cystinosis. Klin Monatsbl Augenheilkd 201:48–50
Kashtan CE, McEnery PT, Tejani A, Stablein DM (1995) Renal allograft survival according to primary diagnosis: a report of the North American Pediatric Renal Transplant Cooperative Study. Pediatr Nephrol 9:679–684
Gahl WA, Kuehl EM, Iwata F, Lindblad A, Kaiser-Kupfer MI (2000) Corneal crystals in nephropathic cystinosis: natural history and treatment with cysteamine eyedrops. Mol Genet Metab 71:100–120
Tsilou E, Zhou M, Gahl W, Sieving PC, Chan CC (2007) Ophthalmic manifestations and histopathology of infantile nephropathic cystinosis: report of a case and review of the literature. Surv Ophthalmol 52:97–105
Tsilou ET et al (2006) Nephropathic cystinosis: posterior segment manifestations and effects of cysteamine therapy. Ophthalmology 113:1002–1009
Katz B, Melles RB, Schneider JA (1960) Crystal deposition following keratoplasty in nephropathic cystinosis. Arch Ophthalmol Chic Ill 1989(107):1727–1728
Mahoney CP, Striker GE, Hickman RO, Manning GB, Marchioro TL (1970) Renal transplantation for childhood cystinosis. N Engl J Med 283:397–402
Fahey DK, Fenton S, Mohamed Q, Logan P (2001) Cystinosis, cataract surgery, and corneal erosions. J Cataract Refract Surg 27:2041–2043
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Elias Flockerzi has received a travel grant from Orphan Europe Germany GmbH to present the corneal slit lamp findings of the patient described in this manuscript at the second Ophthalmology Cystinosis Forum (OCF II) Conference in Berlin, Germany, June 30, 2017 and a travel grant from Orphan Europe Germany GmbH to attend the third Ophthalmology Cystinosis Forum (OCF III) Conference in London, United Kingdom, June 15, 2018. The other authors declare that there is no conflict of interest.
Ethical approval
The local ethics committee of Saarland (Ethikkommission bei der Ärztekammer des Saarlandes) was informed. Because this article does not contain any studies with human participants or animals performed by any of the authors as it describes the required ophthalmological treatment of one nephropathic cystinosis patient, the ethics committee of Saarland approved the publication of this article.
Informed consent
Informed consent to publish this case study was obtained from the patient.
Rights and permissions
About this article
Cite this article
Flockerzi, E., Daas, L., Schlötzer-Schrehardt, U. et al. Ocular changes in nephropathic cystinosis: The course of the gold-dust. Int Ophthalmol 39, 1413–1418 (2019). https://doi.org/10.1007/s10792-018-0954-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-018-0954-7