Abstract
Purpose
The aim of this pilot study was to evaluate a new semiautomatic procedure to assess in vivo changes in the crystalline lens and ciliary muscle during accommodation.
Methods
A total of 14 subjects were divided into two groups, young (aged between 20 and 25 years) and adult (aged between 35 and 40 years), and measured with an anterior segment optical coherence tomography. A semiautomatic procedure was implemented to measure the central lens thickness (CLT), anterior lens radius (ALR) and the ciliary muscle area (CMA) for the unaccommodated eye and for a vergence of − 3.00 D.
Results
The CLT increase for each population group was smaller than 5%, and the dispersion of each group was similar between them. Contrariwise, the reduction in the ALR was about 30% for both groups, although the young one showed the largest variability. The CMA increase was smaller than 30% for both groups, and the dispersion was similar between them. For each metric, differences between both groups were not statistically significant.
Conclusions
The semiautomatic procedure seems to be useful for the in vivo analysis of the accommodative system. Additionally, the results obtained showed that changes in the CLT were much smaller compared to those obtained for the ALR or CMA.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The human eye is formed by complex structures that make use of different mechanisms in order to achieve a proper visual function. Many of these functions are altered as the eye ages. One of them is the adjustment of the refractive power of the crystalline lens, which let us focus on objects at different distances, known as accommodation. According to the most widely accepted theory of accommodation of Helmholtz, this mechanism is a muscle-induced activity [1]. The crystalline lens is a transparent and flexible structure with a biconvex shape that lies in the capsular bag, which is connected through the zonular fibres to the ciliary muscle. Specifically, when the ciliary muscle contracts, it causes the release of the zonular fibres, which allows the crystalline lens to increase its size and curvature of its surfaces, increasing its refractive power.
With ageing, the lens becomes less flexible, which leads to a visual condition known as presbyopia [2]. In presbyopia, the ability of the crystalline lens to modify its size and shape is reduced, decreasing the level of accommodation. Additionally, changes in the ciliary muscle related to age have been observed [3, 4]. Furthermore, the increasingly social demand for tasks that require different working distances (far, intermediate and near vision) motivate the research on the correction of presbyopia. A better understanding of the alterations in the accommodation mechanism with ageing may lead to develop new optical solutions and/or improve the existing ones. This is particularly important, since the lack of accommodation leads to a decreasing in the quality of life [5].
To achieve this, it is crucial to image the structures of interest in the anterior segment. Recent research on anterior segment visualization and quantification of anatomical parameters has been performed using different methods such as ultrasound biomicroscopy, magnetic resonance imaging or anterior segment optical coherence tomography (AS-OCT) [6,7,8,9]. Nevertheless, during the past years, the AS-OCT has become more popular in imaging the anterior segment structures.
More specifically, Sheppard and Davies [10, 11], by mainly using AS-OCT, assessed the changes that occur during accommodation on the ciliary muscle area and contractility with age and refractive condition of the eye, finding that the overall ciliary muscle area is unchanged with age and changeable with the eye’s axial length, which is not directly related to its refractive error. Additionally, a novel approach was proposed in which two spectral-domain OCT devices were synchronized in order to assess the changes in the anterior segment including the ciliary muscle and the interaction between the crystalline lens and the ciliary muscle with accommodation [12, 13].
The numerous of ocular imaging methods that exist can be combined in order to provide us with tangible results on the accommodative changes of the crystalline lens and ciliary muscle as it is evident in the study conducted by Richdale et al. [14]. Along with these methods and future research potential accommodating IOL designs can be proposed.
The present pilot study aims to evaluate a new semiautomatic procedure to assess in vivo changes in the crystalline lens and ciliary muscle during accommodation. These results will report more evidences about changes taking place in these structures during accommodation, which could be taken into account to better understand the mechanism of accommodation at different age groups.
Methods
Subjects
A total of 14 subjects were included in this study. These were divided into two groups. The first group consisted of young healthy patients (three males and four females) aged from 20 to 25 (mean 23.0 ± 2.2 years) whose mean spherical equivalent refractive error was − 0.6 ± 2.8 D. The second group was compound of adult subjects (six males and one female) aged from 35 to 40 (mean 39.0 ± 1.5 years) with a spherical equivalent error of − 0.2 ± 2.0 D. For all cases, the refractive error was obtained with dry autorefraction.
The subjects had enough amplitude of accommodation (at least 3.0 D), which was assessed monocularly by the Donders’ method [15]. Their best-corrected visual acuity evaluated with the ETDRS chart (Precision Vision, USA) was at least 20/20 in Snellen equivalent. The subjects had no ocular abnormality or systemic condition and no ocular surgery history, and they all presented clear intraocular media. The study followed the Declaration of Helsinki and was approved by the Ethics Committee of our Institution. The patients were informed about the details of the study and signed a formal consent after written and verbal explanation of the implications.
Measurement device
The Visante Omni was the equipment used to visualize the ocular structures, which combines OCT technology together with Placido disc topography to obtain advanced anterior segment measurements. Furthermore, it is a non-contact diagnostic instrument which acquires and analyses detailed cross-sectional tomographic images of the anterior eye segment. In order to obtain the captures, it makes use of low-coherence interferometry. Specifically, infrared light of 1310 nm is sent along an optical path that reaches the eye together with another reference path of the interferometer. Then, both paths are combined at the sensor to determine the axial depth of the tissue thanks to the reflectivity signal. Furthermore, the vergence of the fixation target can be adjusted through a set of internal lenses, which were used to evaluate changes in the anterior eye with accommodation.
Experimental procedure
The laboratory of the Grupo de Investigación en Optometría (University of Valencia, Spain) held all measurements, which were taken by a skilled operator on one eye of each subject. The ambient lightning conditions were kept constant during the whole procedure in order not to have significant variations in the pupil diameter. Furthermore, any physical contact in the eye was not allowed before the measurement with the OCT, and all measurements were taken in the same session.
The patients were requested to keep their fixation on the instrument visual target before capturing. They were also requested to blink before starting the exam, and the acquisition was taken 4 s after the subject’s last blink in order to achieve a proper tear film spread over the cornea [16]. In this study, each acquisition was taken with the fixation target placed at 0.0 D with respect to each patient’s far point and at − 3.0 D of vergence. Furthermore, the order of each vergence was presented randomly, and all captures were performed at the horizontal meridian.
The “Raw Image” mode was used to obtain the central lens thickness (CLT). Concretely, this mode is composed of 256 primitive A-Scans that represent a total area of 16 mm in width and 6 mm in depth and gets images deep into the eye. On the other hand, the “Raw Image HR” was used to capture the central anterior lens radius of curvature (ALR) and the anterior ciliary muscle area (CMA) at the nasal region. This mode yields images of 512 primitive A-Scans which represent a total area of 10 mm in width and 3 mm in depth.
Image analysis
Crystalline lens thickness
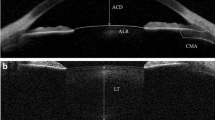
The captured image of the crystalline lens was exported to the ImageJ software (National Institute of Health, USA). For the aims of the present study, the CLT was considered as the axial distance between the central anterior surface and central posterior surface, which was recognized by the posterior reflex. Figure 1, panels A and B, displays the procedure followed to carry out this measure.
Representative images showing the procedure followed in the semiautomatic algorithm to analyse the central lens thickness. A Original image. B Crystalline lens thickness considered as the axial distance between the central anterior surface and central posterior surface (recognized by the posterior reflex in this contrast-enhanced image)
Anterior lens radius of curvature
Figure 2 (panels A to E) displays the procedure followed to measure the ALR. Concretely, this procedure consisted of: on the one hand, the original image (panel A) was converted into its complementary (panel B), whose histogram was equalized to enhance its contrast. Then, a Gaussian filter was applied to smooth the image, and a Sobel one was applied to remark the borders. After all this, a curve was applied to fit the selected points that delimit the crystalline lens border (panel C), and then, the observer had to mark the centre of the lens border (panel D), which will be used to calculate the radius of the circumference that fits the anterior lens (green line on the image). Finally, the final fit (panel E) was displayed to ensure that the final circumference (red line) fits the selected points.
Representative images showing the procedure followed in the semiautomatic algorithm to analyse the anterior lens radius. A Original image. B Equalized enhanced complementary image with a Sobel filter. C Curve (green line) applied to fit the selected points that delimit the crystalline lens border. D Observer marking the centre of the lens border. E Final fit applied to ensure that the circumference (red line) fits the selected points
Ciliary muscle area
The ImageJ software was used to measure the CMA of each image, and the procedure is displayed in Fig. 3 panels A to D for reference purposes. To measure this metric, the original image was converted into an 8-bits image (panel A). Then, the whole image was smoothed (panel B) using a 3 × 3 average filter (each pixel is replaced with the average of its 3 × 3 neighbourhood), and the contrast was increased (panel C) using a nonuniform histogram equalization to enhance the limits of the area of interest. Then, the observer eased the delimitation of the ciliary muscle through two lines: one of them was along the border between the sclera and ciliary muscle, and the other one went through the scleral spur and was perpendicular to the previous line. After all this, an automatic selecting tool was applied to the enhanced image to select the ciliary muscle limits (panel D), in which the muscle area was calculated.
Representative images showing the procedure followed in the semiautomatic algorithm to analyse the ciliary muscle area. A Original image. B Smoothed image using a 3 × 3 average filter. C Enhanced contrast image using a nonuniform histogram equalization. D Automatic selecting tool applied to the enhanced image to obtain the ciliary muscle limits
The relative change with respect to the relaxed condition was obtained for all these parameters and for each age group. It should be pointed out that all measurements were taken directly in pixels because we were initially interested in reporting the ratio of change.
Statistical analysis
The statistical analysis was performed with SPSS software (version 22.0, SPSS Inc., Chicago, IL, USA). Each measurement was obtained three times per image, and a mean and a standard deviation values were calculated. Due to the limited number of subjects in each sample, the nonparametric Mann–Whitney U test for two independent samples was used. This procedure allows getting the significance level between two different groups or samples for a given parameter. The statistical significance level was set as p < 0.05. Prior to analysis of the parameters of interest, the spherical equivalent of the groups was checked for significant differences, which were not found in our samples. Additionally, box-and-whisker plots were generated and represented for each parameter. Finally, the correlation among all biometric parameters for both population groups was also calculated.
Results
Young group
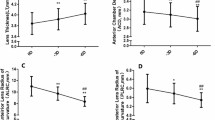
Figure 4 displays the box-and-whisker plots obtained with each population group for the relative changes in the CLT (panel A), ALR (panel B) and CMA (panel C) during accommodation. All in all, these plots show that for the young group, the CLT and CMA tend to increase at 3.0 D of accommodation; meanwhile, the ALR tends to decrease. Concretely, the CLT resulted with the smallest relative change (4%), and the ALR did the largest one (− 30%). Additionally, the CLT showed the most compacted outcomes with maximum relative difference of 4%, and the ALR did the most spread outcomes with a difference of 44% between the maximum and minimum relative difference.
Adult group
Regarding the results obtained for the adult group (Fig. 4), the CLT (panel A) showed almost no change from 0.0 to − 3.0 D of vergence; meanwhile, the ALR (panel B) decreased and the CMA (panel C) increased. Concretely, for the CLT, the median change was 3%; for the ALR, it was − 28%; and for the CMA, it was 29%, where negative sign denotes a reduction in the parameter. At the same time, the difference between the lowest and highest relative difference was 5% for the CLT, 23% for the ALR and 15% for the CMA.
Young versus adult groups
Finally, the comparison between the young and the adult groups showed similar relative changes in the CLT between both groups, in which the median change for the former group was 4% and for the latter one was 3%. Furthermore, the difference between the maximum and minimum relative change in the CLT was at the most 5% for both groups. At the same time, the relative change in the CLT was not statistically significant different between both population groups (p = 0.277).
Both groups showed similar changes in the ALR with the accommodation, in which the median change for the young group was − 30% and for the adult one was − 28%. Nonetheless, the difference between the maximum and minimum relative change in the ALR was larger in the young group than in the adult one, and the value was 44% for the former group and 23% for the latter one. Furthermore, no statistically significant differences were obtained in the relative change between both population groups (p = 0.848).
Regarding changes in the CMA with accommodation, both population groups showed similar results between them, in which the median difference between them was 9%. At the same time, the young group showed more disperse relative changes than the presbyopic group. Finally, the statistical analysis showed no significant differences between both groups (p = 0.142).
Correlations among parameters
Figure 5 displays the correlation diagrams and the Pearson correlation coefficients among all absolute biometric measurements for the young and adult groups. All in all, the tendency line for most of the correlations obtained in both groups was as expected, i.e. CLT decreased when ALR increased, CLT increased when CMA increased, and ALR decreased when CMA increased. Nevertheless, the correlation between the CLT and ALR showed the strongest value and steepest tendency for the young group (− 0.519), while the correlation between the ALR and CMA showed the strongest value and steepest tendency for the adult group (− 0.748).
Discussion
The aim of this pilot study was to evaluate a new semiautomatic procedure to assess in vivo changes in the eye’s accommodative system in terms of crystalline lens radius, thickness and ciliary muscle area in two population groups. From a clinical point of view, this procedure could help clinicians to have objective evidences of some dynamic changes taking place in the anterior segment during accommodation as a function of age.
With ageing, the human body goes through several changes that have an impact on daily functions and thus, in the quality of life. The crystalline lens suffers from several transformations as part of the ageing process. These changes include a loss of the elasticity of the capsule. Additionally, the cortex decreases its elasticity and the nucleus hardens, modifying the ability of the lens to change its shape and achieve the proper refractive state required in order to focus on objects at different distances [2]. Some in vivo studies that have been carried out decades ago already showed that the total volume of the lens increases with age, thus increasing the CLT [17].
The results obtained in the present study showed similar relative changes in the CLT between both population groups, in which the lens thickness increased about 5% for both groups. At the same time, both groups showed similar dispersion values in the relative changes in the CLT with the accommodation, which values were smaller than 5%, and differences between groups were not statistically significant. As was expected, no significant differences were obtained because both population groups could be classified as pre-presbyopic. Thus, from the results obtained in the present study, it could be concluded that the CLT varies with the accommodation independently of its thickness in non-presbyopic eyes.
The results obtained in the current study show the same tendency as those obtained previously [18,19,20,21,22], where the lens thickness increased with the vergence. At the same time, the posterior lens surface has been reported to move backwards with accommodation, because the decrease in the anterior chamber depth is smaller than the increase in the lens thickness [23]. Furthermore, it is known that with age the crystalline lens thickness increases, the anterior chamber depth shallows and the anterior lens curvature steeps [19]. Additionally, another study reported that the lens reshaping is much slower in ageing eyes [24].
Regarding the ALR, both population groups showed a relative median reduction about 30%, although the young group showed larger variability than the adult group, which might reflect that the crystalline lens in the latter group could be losing elasticity. Additionally, and as was expected, no statistically significant differences were obtained in the relative ALR changes with the accommodation between both population groups. Thus, according to the results obtained in the present study, it could be concluded that the anterior crystalline lens radius becomes steeper with the accommodation.
The results obtained in the present study about the ALR agree with those obtained previously [21, 23]. Concretely, one study measured changes in the crystalline shape during accommodation by means of a Scheimpflug camera [23]. That study obtained that the anterior and posterior lens radius decreased with the accommodation, and the anterior lens surface became more hyperbolic. On the other hand, another study imaged the anterior segment of the eye from the cornea to the posterior lens surface with a custom built and dual-focus spectral-domain OCT [21]. According to that study, the anterior and posterior radii of curvature significantly decreased with accommodation, where the greatest steepening took place on the anterior surface. From those results, the authors suggested that changes in the anterior lens surface might play greater role than those taking place on the posterior one.
The CMA of the young and adult groups increased with the accommodation in a similar amount (about 30%), in which the results dispersion of both groups was similar between them. Furthermore, the statistical analysis showed no significant differences between both groups. Thus, from all these results, it could be concluded that the ciliary muscle contractility does not deteriorate during the pre-presbyopic stage.
These results are in agreement with those obtained in a previous study that assessed the effect of ageing on the morphology and contractility of the human ciliary muscle in seventy-nine subjects aged between 19 and 70 years [11]. In that study, the authors used high-resolution images of the nasal and temporal muscle by means of an anterior segment OCT. From the results obtained, the authors concluded that the morphology of the ciliary muscle suffers an antero-inwards displacement, particularly in emmetropic eyes. Nevertheless, those morphologic changes did not appear to affect the muscle ability to contract during the accommodation. More recently, another study assessed the ciliary muscle activity in a small group of pseudophakic subjects, whose ages ranged around 80 years, by means of a developed method based on high-speed recording of lens wobbling [25]. Concretely, this system combined an open-view wavefront sensor and Purkinje meter. According to the results obtained, the authors showed that the muscle had a significant activity, which was able to contract under binocular stimulation of accommodation.
The correlations obtained among the ALR, CMA and CLT showed the expected tendency for both age groups. Thus, an accommodative effort implying an increase in the CMA was accompanied by an increase in the CLT and a decrease in the ALR, as expected from the change in the geometry of the radii of curvature of the crystalline lens, while a relaxation of the accommodation implying a reduction in the CLT was accompanied by an increase in the ALR (Fig. 5). These results reflect that the thickness and anterior curvature of the lens vary with the accommodation depending on the age group, as has been previously reported [24].
Originally, the information about the crystalline lens and ciliary muscle was mostly extracted from in vitro studies due to the limitation on the available in vivo devices [26,27,28]. For example, one study evaluated variations in the lens thickness capsule as a function of age on donor human lenses aged between 12 and 103 years [26], and obtained that the lens capsule thickness increased with age. Concretely, the results showed that the anterior midperiphery central to the zonular insertion yielded the maximum thickness, whereas the midperipheral zone decreases after the seventh decade. On the other hand, another study determined how accommodation and age affect the optical properties of the lens in enucleated human lenses of subjects whose age ranged from 10 to 87 years [27]. Concretely, those authors measured the focal length and spherical aberration of the crystalline lenses, while the lenses were subjected to stretching forces applied through the ciliary body. The results obtained showed that the focal length of all unstretched lenses increased linearly with age, and the younger the lens, the more significant the change in the focal length with stretching. At the same time, the profile of 19 pairs of isolated human eye bank was digitalized to measure the lens thickness and lens surface curvatures, from which the equivalent refractive indices were calculated [28]. That study revealed no age dependent change in equivalent refractive index of the isolated lens. Finally, information about the ciliary muscle was derived from histological studies performed post-mortem in monkeys [29, 30], specifically in rhesus monkeys due to similarities in accommodative structures [30, 31], mechanism [32, 33] and presbyopia development related to timescales [34, 35]. Furthermore, in humans, analysis of post-mortem ciliary body tissue from eyes exposed to high doses of pharmacologic agents before dissection has demonstrated that the muscle maintains its ability to contract throughout life [36].
However, with the advent of high-resolution UBM [37, 38] and OCT [10, 39, 40], in vivo studies visualized and recorded the dynamic of the accommodative mechanism. For example, one study quantified the movements of the zonule and ciliary muscle during accommodation by means of UBM and identified their age-related changes that could impact the optical change that occurs during accommodation [37]. Similarly, another study assessed the dynamic movements of the vitreous membrane and peripheral choroid during accommodation with UBM and also assessed age-related changes in the anterior sclera [38]. The results obtained in that study showed that the hyaloid bent posteriorly during accommodation in proportion to accommodative amplitude and the sclera bowed inward with increase in age.
Nowadays, the AS-OCT has become more popular in imaging the anterior segment structures. With this regard, an anterior segment OCT has been used to analyse the morphology and changes in the ciliary muscle during accommodation, in which the results obtained showed an anterior and centripetal contractile shift of ciliary muscle mass [10]. Similarly, another study evaluated the morphology of the ciliary muscle during the accommodation in children and obtained that the thickness of 1 mm from the scleral spur and the maximum thickness increased with the accommodation; meanwhile, it was reduced to 3 mm. Furthermore, the OCT allowed to objectively analyse the effect of phenylephrine in the ciliary muscle, in which it was demonstrated that small doses do not affect the muscle dimensions [41].
There are several algorithms developed to extract information about the ciliary muscle or crystalline lens from OCT images [12, 13, 40, 42]. Concretely, a semiautomatic algorithm was developed for segmentation and morphological assessment of the dimensions of the ciliary muscle in images taken with the Visante OCT [42]. The authors compared the results of the algorithm with the measurement done with the instrument calliper and concluded that the new procedure successfully segmented the ciliary muscle. Furthermore, this algorithm was considered as a measurement protocol to assess changes in the ciliary muscle with accommodation [40].
On the other hand, a novel approach for imaging the anterior segment and ciliary muscle with the accommodation was developed combining and synchronizing two spectral-domain OCTs, one for imaging the anterior segment and the other one for the ciliary muscle [12]. Furthermore, a semiautomatic segmentation of the anterior segment and ciliary muscle was included in the image analysis and was demonstrated to be capable to image both the anterior segment and ciliary muscle in vivo during accommodation.
Similarly, two spectral-domain OCTs were combined and precisely synchronized with a dual channel accommodation target to simultaneously image the anterior segment and ciliary muscle during accommodation [13]. Actually, that system included an image analysis to measure automatically intraocular distances such as central corneal thickness, anterior chamber depth and lens thickness. In fact, that system allowed the precise correlation of anatomical changes occurring in the crystalline lens and ciliary muscle at identical time points during accommodation.
The present pilot study assessed changes in the crystalline lens and ciliary muscle area with accommodation by means of a semiautomatic procedure. However, a presbyopic group was not included in the study population, and changes in these metrics were assessed between 0.0 and − 3.0 D of vergence. With this regard, further studies could apply this procedure in a presbyopic population. As a pilot study, only seven subjects per group were included, and measurements were made only on the horizontal meridian. Further studies could use this procedure in a larger sample and take measurements in additional meridians. The novelty of this pilot study lies mainly in the image processing algorithms that are proposed to extract the relevant information from the OCT images. In this sense, it is important to consider that even a small change in the image processing procedures with respect to the information that exists in the literature could be of relevant interest for those cases in which classical methods do not work properly.
To sum up, the results obtained in the present pilot study showed that the semiautomatic procedure was useful for the in vivo analysis of the accommodative eye system. At the same time, those results revealed that changes in the CLT were much smaller compared to those obtained for the ALR or CMA.
References
Von Helmholtz H (1855) Über die Akkommodation des Auges. Arch Ophthalmol 1:1–74
Strenk S, Strenk L, Koretz J (2005) The mechanism of presbyopia. Prog Retin Eye Res 24:379–393
Strenk S, Semmlow J, Strenk L, Munoz P, Gronlund-Jacob J, DeMarco J (1999) Age-related changes in human ciliary muscle and lens: a magnetic resonance imaging study. Invest Ophthalmol Vis Sci 40:1162–1169
Tamm S, Tamm E, Rohen J (1992) Age-related changes of the human ciliary muscle. A quantitative morphometric study. Mech Ageing Dev 62:209–221
Luo B, Brown G, Luo S, Brown M (2008) The quality of life associated with presbyopia. Am J Ophthalmol 145:618–622
Leung C, Chan W, Ko C, Chui S, Woo J, Tsang M, Tse R (2005) Visualization of anterior chamber angle dynamics using optical coherence tomography. Ophthalmology 112:980–984
Dada T, Sihota R, Gadia R, Aggarwal A, Mandal S, Gupta V (2007) Comparison of anterior segment optical coherence tomography and ultrasound biomicroscopy for assessment of the anterior segment. J Cataract Refract Surg 33:837–840
Strenk S, Strenk L, Guo S (2010) Magnetic resonance imaging of the anteroposterior position and thickness of the aging, accommodating, phakic, and pseudophakic ciliary muscle. J Cataract Refract Surg 36:235–241
Ursea R, Silverman R (2010) Anterior-segment imaging for assessment of glaucoma. Expert Rev Ophthalmol 5:59–74
Sheppard A, Davies L (2010) In vivo analysis of ciliary muscle morphologic changes with accommodation and axial ametropia. Invest Ophthalmol Vis Sci 51:6882–6889
Sheppard A, Davies L (2011) The effect of ageing on in vivo human ciliary muscle morphology and contractility. Invest Ophthalmol Vis Sci 52:1809–1816
Shao Y, Tao A, Jiang H, Shen M, Zhong J, Lu F, Wang J (2013) Simultaneous real-time imaging of the ocular anterior segment including the ciliary muscle during accommodation. Biomed Opt Express 4:466–480
Ruggeri M, de Freitas C, Williams S, Hernandez V, Cabot F, Yesilirmark N, Alawa K, Chang Y, Yoo S, Gregori G, Parel J, Manns F (2016) Quantification of the ciliary muscle and crystalline lens interaction during accommodation with synchronous OCT imaging. Biomed Opt Express 17:1351–1364
Richdale K, Sinnott L, Bullimore M, Wassenaar P, Schmalbrock P, Kao C, Patz S, Mutti D, Glasser A, Zadnik K (2013) Quantification of age-related and per diopter accommodative changes of the lens and ciliary muscle in the emmetropic human eye. Invest Ophthalmol Vis Sci 54:1095–1105
Benjamin WJ (2006) Borish’s clinical refraction, 2nd edn. Butterworth-Heinemann, London, p 1003
Montés-Micó R, Alió J, Muñoz G, Charman W (2004) Temporal changes in optical quality of air-tear film interface at anterior cornea after blink. Invest Ophthalmol Vis Sci 45:1752–1757
Brown N (1974) The change in lens curvature with age. Exp Eye Res 19:175–183
Du C, Shen M, Li M, Zhu D, Wang M, Wang J (2012) Anterior segment biometry during accommodation imaged with ultralong scan depth optical coherence tomography. Ophthalmology 119:2479–2485
Richdale K, Bullimore M, Sinnott L, Zadnik K (2016) The effect of age, accommodation, and refractive error on the adult human eye. Optom Vis Sci 93:3–11
Neri A, Ruggeri M, Protti A, Leaci R, Gandolfi S, Macaluso C (2015) Dynamic imaging of accommodation by swept-source anterior segment optical coherence tomography. J Cataract Refract Surg 41:501–510
Sun Y, Fan S, Zheng H, Dai C, Ren Q, Zhou C (2014) Noninvasive imaging and measurement of accommodation using dual-channel SD-OCT. Curr Eye Res 39:611–619
Ramasubramanian V, Glasser A (2015) Objective measurement of accommodative biometric changes using ultrasound biomicroscopy. J Cataract Refract Surg 41:511–526
Dubbelman M, van der Heijde G, Weeber H (2005) Change in shape of the aging human crystalline lens with accommodation. Vis Res 45:117–132
Shao Y, Tao A, Jiang H, Mao X, Zhong J, Shen M, Lu F, Xu Z, Karp C, Wang J (2015) Age-related changes in the anterior segment biometry during accommodation. Invest Ophthalmol Vis Sci 56:3522–3530
Tabernero J, Chirre E, Hervella L, Prieto P, Artal P (2016) The accommodative ciliary muscle function is preserved in older humans. Sci Rep 6:25551
Barraquer R, Michael R, Abreu R, Lamarca J, Tresserra F (2006) Human lens capsule thickness as a function of age and location along the sagittal lens perimeter. Invest Ophthalmol Vis Sci 47:2053–2060
Glasser A, Campbell M (1998) Presbyopia and the optical changes in the human crystalline lens with age. Vis Res 38:209–229
Glasser A, Campbell M (1999) Biometric, optical and physical changes in the isolated human crystalline lens with age in relation to presbyopia. Vis Res 39:1991–2015
Koretz J, Neider M, Kaufman P, Bertasso A, DeRousseau C, Bito L (1987) Slitlamp studies of the rhesus monkey eye I: survey of the anterior segment. Exp Eye Res 44:307–318
Koretz J, Bertasso A, Neider M, True-Gabelt B, Kaufman P (1987) Slit-lamp studies of the rhesus monkey eye: II. Changes in crystalline lens shape, thickness and position during accommodation and aging. Exp Eye Res 45:317–326
Ostrin L, Glasser A (2007) Effects of pharmacologically manipulated amplitude and starting point on Edinger-Westphal-stimulated accommodative dynamics in rhesus monkeys. Invest Ophthalmol Vis Sci 48:313–320
Glasser A, Wendt M, Ostrin L (2006) Accommodative changes in lens diameter in rhesus monkeys. Invest Ophthalmol Vis Sci 47:278–286
Glasser A, Kaufman P (1999) The mechanism of accommodation in primates. Ophthalmology 106:863–872
Wasilewski R, McDonald J, Heatley G, Lütjen-Drecoll E, Kaufman P, Croft M (2008) Surgical intervention and accommodative responses II: forward ciliary body accommodative movement is facilitated by zonular attachments to the lens capsule. Invest Ophthalmol Vis Sci 49:5495–5502
Kaufman P, Bito L, DeRousseau C (1982) The development of presbyopia in primates. Trans Ophthalmol Soc UK 102:323–326
Pardue M, Sivak J (2000) Age-related changes in human ciliary muscle. Optom Vis Sci 77:204–210
Croft M, McDonald J, Katz A, Lin T, Lütjen-Drecoll E, Kaufman P (2013) Extralenticular and lenticular aspects of accommodation and presbyopia in human versus monkey eyes. Invest Ophthalmol Vis Sci 54:5035–5048
Croft M, Nork T, McDonald J, Katz A, Lütjen-Drecoll E, Kaufman P (2013) Accommodative movements of the vitreous membrane, choroid, and sclera in young and presbyopic human and nonhuman primate eyes. Invest Ophthalmol Vis Sci 54:5049–5058
Lewis H, Kao C, Sinnott L, Bailey M (2012) Changes in ciliary muscle thickness during accommodation in children. Optom Vis Sci 89:727–737
Lossing L, Sinnott L, Kao C, Richdale K, Bailey M (2012) Measuring changes in ciliary muscle thickness with accommodation in young adults. Optom Vis Sci 89:719–726
Richdale K, Bailey M, Sinnott L, Kao C, Zadnik K, Bullimore M (2012) The effect of phenylephrine on the ciliary muscle and accommodation. Optom Vis Sci 89:1507–1511
Kao C, Richdale K, Sinnott L, Grillot L, Bailey M (2011) Semiautomatic extraction algorithm for images of the ciliary muscle. Optom Vis Sci 88:275–289
Funding
This study was funded by Ministerio de Economía y Competitivad Research Project SAF2013-44510-R with ERDF funds (European Regional Development Fund) from European Union. Aikaterini I. Moulakaki is a “Marie Curie Early Stage Researcher” from the AGEYE Initial Training Network Program FP7-People-2013-608049 Project, Daniel Monsálvez-Romín is a “Formación de Profesorado Universitario” grantee (FPU13/05332, Ministerio de Educación, Cultura y Deporte), and Alberto Domínguez-Vicent is an “Atracció de Talent” grantee (UV-INV-PREDOC13-110412, University of Valencia, Spain).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Moulakaki, A.I., Monsálvez-Romín, D., Domínguez-Vicent, A. et al. Semiautomatic procedure to assess changes in the eye accommodative system. Int Ophthalmol 38, 2451–2462 (2018). https://doi.org/10.1007/s10792-017-0752-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-017-0752-7