Abstract
Background and purpose
Curcuma longa L. (CL), an Indian herb, has been used to treat many disorders because of its wide spectrum of pharmacological activities. It has been shown to exhibit anti-oxidant and anti-inflammatory properties, and is being used as herbal remedy since ancient times. Osteoarthritis of knee (KOA) is a chronic painful disorder in which prolong use of non-steroidal anti-inflammatory drugs (NSAIDs) or steroids may result into many serious side effects; hence, there is a need to develop herbal drugs, having good analgesia without side effects. Therefore, we planned to evaluate the efficacy of CL in KOA.
Methods
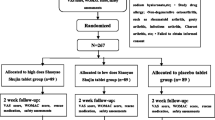
The study was designed as a randomized, double-blind, placebo-controlled trial in patients of KOA. After obtaining ethical clearance and written informed consent, a total of 160 patients of KOA were randomly enrolled into two groups to receive either CL extract or placebo along with the standard drug regimen. The patients were assessed on day 0, day 60, and day 120. On the days of their visit, the clinical prognosis was assessed by visual analog scale (VAS) and Western Ontario and McMaster Universities (WOMAC) Osteoarthritis index. On these days, the radiographs were also taken for Kellgren and Lawrence grading and blood samples were collected for assessing the changes in levels of IL-1β and biomarkers of oxidative stress, such as reactive oxygen species and malondialdehyde (MDA).
Results
Over all significant improvement was observed in the patients of CL extract group as compared to placebo group. Clinically, the VAS and WOMAC scores became better, and simultaneously, the levels of biomarkers, viz., IL-1β, ROS, and MDA, were also significantly (p < 0.05) improved.
Conclusion
It may be concluded that on chronic administration, CL suppresses inflammation and brings clinical improvement in patients of KOA, which may be observed by decreased level of IL-1β and VAS/WOMAC scores, respectively. At the same time, CL decreases the oxidative stress also.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoarthritis (OA) is a progressively debilitating, inflammatory disorder of the synovial joints. It is characterized by degradation of extracellular matrix macromolecules and decreased expression of chondrocyte protein, resulting in damage of cartilage, severe joint pain and restriction of movements (Goldring Goldring and Otero 2011). Among the joints of the body, the knee is most commonly involved and the incidence of osteoarthritis of knee (KOA) has been found to be increasing during last three decades (Ambrosia 2005; Das and Farooqi 2008; Allen et al. 2010). Moreover, the age of development of the disease has reduced in recent times (Bhatia et al. 2013). Several comorbidity factors have been associated with KOA including but not limited to previous injury, knee-bending occupations (Esser and Bailey 2011).
Therapy for KOA includes non-steroidal anti-inflammatory drugs (NSAIDs)/steroids, exercise, physiotherapy, weight relieving braces, and total knee arthroplasty (Barron and Rubin 2007; Wang et al. 2004). Modern drugs mainly relieve the symptom pain, while the damaging inflammatory process not significantly affected (Bjordal et al. 2004). NSAIDs are one of the most commonly used medications for the treatment of KOA. However, prolonged use of NSAIDs presents side effects on the kidneys and gastrointestinal system. Similarly, therapy with steroids has its known drawbacks and side effects. Furthermore, it has also been observed that in the course of progress of KOA, once the joint structures are damaged, the repair becomes increasingly difficult. Therefore, preventive strategies at an earlier stage are considered to be the best treatment for KOA (Michael et al. 2010).
Hence, an application of alternative systems of medicine with no side effects for the treatment of KOA is becoming more essential. Medicinal plants are an important source of active compounds, which have little or no side effects and a very high therapeutic index (Dharmananda 1998). All over the world, these traditionally used herbal agents are being evaluated on scientific parameters and are being advocated for OA (Altman and Marcussen 2001; Madhu et al. 2013).
Curcuma longa L. (Family Zingiberaceae), commonly known as Turmeric or ‘Haridra’ in India, is an Asian herb. Haridra is mentioned in ‘Charaka Samhita’ (Indian traditional system of medicine) and is officially mentioned in the Ayurvedic Pharmacopoeia of India (Kaviratna and Sharma 1913; Ayurvedic Pharmacopoeia of India 1989). It contains a series of curcuminoids which are alkaloidal in nature, having 90 % curcumin as main active ingredient (Roth et al. 1998). Curcuma longa L. (CL) has a long history of use for its anti-inflammatory and anti-oxidant effects (Chandra and Gupta 1972; Sharma 1976; Selvam et al. 1995; Scartezzini and Speroni 2000). It has been suggested that curcumin acts by both direct and genomic activity on proinflammatory cytokines (Jurenka 2009). Proinflammatory (PI) cytokines are important in cell signaling and promote systemic inflammation; they are produced predominantly by activated macrophages and are involved in the upregulation of inflammatory reactions (Amico et al. 2015). IL-1β, a PI cytokine, plays a central role in the pathophysiology of cartilage damage and degradation (Ji et al. 2016). Previous studies have shown that the levels of IL-1β are raised in KOA patients compared to controls (Verma and Dalal 2013; Shahine and Elhadidi 2014).
Reactive oxygen species (ROS) are highly reactive transient chemical species or free radicals, such as nitric oxide, superoxide, and hydroxyl anions which are produced by normal cellular biochemical reactions. Production of ROS is central to the progression of many inflammatory diseases. These are produced by cells that are involved in the host-defense response and promote endothelial dysfunction by oxidation of crucial cellular signaling proteins. ROS acts both as a signaling molecule and a mediator of inflammation. When the production of ROS exceeds the capacity of the body’s anti-oxidant defense, oxidative stress (OS) develops (Mittal et al. 2014); Harma and Erel 2003). These are produced in excess by abnormal metabolism of chondrocytes and by senescence of cartilage, ultimately leading to the development of KOA (Martin et al. 2004; Yudoh et al. 2005). The prime targets of the free radicals are the polyunsaturated fatty acids in cell membranes and their interaction results in lipid peroxidation (LPO) (Lopaczynski and Zeisel 2001). Lipid peroxides are unstable and decompose to form reactive carbonyl compounds such as MDA. A recent study has also shown that the formation of ROS along with MDA is increased in different grades of KOA patients in parallel to the severity of the disease (Srivastava et al. 2015). Thus, it is evident that the levels of above-mentioned biomarkers are altered in patients of KOA. Hence, the study was planned with two aims:
-
Primarily, to observe the effect of CL extract on clinical improvement in patients of KOA as assessed by VAS and WOMAC.
-
Secondarily, to correlate between clinical improvement and biomarkers of oxidative stress and inflammation.
Methods
Trial design
The study was designed as a single centre, two-arm double-blind, randomized, placebo-controlled parallel group clinical trial conducted at King George’s Medical University, Lucknow, India. The effect of CL extract was compared with placebo in patients with primary KOA according to the CONSORT statement for reporting RCTs (Moher et al. 2010).
The study was approved by the Institutional Ethics Committee, King George’s Medical University (Ref. code:57 E.C.M. IIB/P12) and registered in Clinical Trial Registry of India (CTRI/2015/12/006438). All procedures performed in the study were in accordance with the ethical standards of the institutional ethics committee and with the 1964 Helsinki declaration and its later amendments.
Patient selection and eligibility criteria
Study was carried out on the patients who reported in the out patients’ Department of Orthopaedics in King George’s Medical University for OA of the knee joint. The criteria for establishing a patient suffering from KOA were according to the guidelines proposed/issued by ‘The American College of Rheumatology’ (Altman et al. 1991).
Inclusion criteria
The patients within the age group of 40–80 years of both sexes, who were suffering from primary KOA according to the above-mentioned eligibility criteria who accepted to participate, were included in the study.
Exclusion criteria
The patients less than 40 years and more than 80 years of age and those suffering from rheumatoid arthritis, diabetes mellitus, renal insufficiency, hepatic disease, cardiovascular disease, gout, pregnant women or with any other systematic disease were excluded from the study.
Radiographic OA of the knee was classified according to the Kellgren–Lawrence (KL) grading scale (Kellgren and Lawrence 1957). This scale involves the following grades:
-
Grade 1—Doubtful narrowing of the joint space and possible osteophytic lipping.
-
Grade 2—Definite osteophytes and possible narrowing of the joint space.
-
Grade 3—Moderate multiple osteophytes, definite narrowing of the joint space, some sclerosis and possible deformity of the bone contour.
-
Grade 4—Large osteophytes with marked narrowing of the joint space, severe sclerosis and definite deformity of the bone contour.
Study products
The extract from rhizomes of CL, developed and registered as ‘Haridra’ by Himalaya Drug Company Bangalore, India was used in this trial.
The formulation/manufacturing of the Turmeric A (CL extract) capsules (Batch no. 1210001FD) and Turmeric B/Placebo capsules (Talc, batch no. 1210002FD) was carried out as per the principles of current Good Manufacturing Practices (cGMP) and quality parameters as per the pharmacopeial standards.
Each capsule composed of CL extract, which was standardized and extracted using organic solvent and then evaporated at low pressure to obtain a semisolid containing curcuminoids. The extract contained equal to or more than 95 % of total curcuminoids. The standardized CL extract was blended with rhizome powder uniformly and was processed by wet granulation method. The wet granules formed were dried in fluidized bed dryer and sizing was done to obtain uniform size of granules and the same was filled in vegetable hydroxypropyl methyl cellulose (HPMC) capsule shells. The final capsules were filled into the high density polyethylene (HDPE) containers, which served as primary packing.
The high-performance thin layer chromatography (HPTLC) finger print analysis was done; chromatogram of standard curcumin and CL extract are shown in Figs. 1 and 2a, b, respectively. Peak ratio of curcumin: demethoxy curcumin: bisdemethoxycurcumin was 0.39:0.20:0.06.
Study protocol
After obtaining ethical clearance and written informed consent from all individual participants, a total of 160 patients who were suffering from KOA were enrolled in the study.
Randomization
Eligible patients were enrolled on ‘first come first serve’ basis and were assigned into a treatment group, i.e., CL extract or placebo based on a computerized randomization schedule. After recruitment into the study, every patient was randomly allocated to receive either CL extract 500 mg or placebo 500 mg capsules which were to be taken along with the standard treatment of Diclofenac 50 mg/day as and when required for 4 months.
78 patients took CL extract along with Diclofenac twice a day and 82 patients took placebo capsules along with Diclofenac twice a day for 4 months. Both CL extract and placebo were procured in similar looking capsules from ‘The Himalaya Drug Company’, Bangalore, India.
Patients in both the treatment groups were evaluated clinically, radiologically and for biochemical changes as well as clinical outcomes on day 0, after 2 months and after fourth months. Blood samples along with the radiographs of the knee joints were also obtained from the patients on each three visits, for biochemical estimations and clinical prognosis status.
The dose of CL extract was decided based on the previous clinical studies, wherein CL showed significant symptomatic relief in KOA patients (Madhu et al. 2013; Pinsornsak and Niempoog 2012).
Sample size estimation
Based on the results of an RCT conducted in the past (Belcaro et al. 2010), we excepted to achieve 37 % reduction (effect size: D) of CL extract in WOMAC score in north Indian KOA patients in a 4 month trial in a double-blind manner. To detect this, 1 SD (standard deviation), 5 % margin of error (α = 0.05), and 90 % power (1 − β = 0.90) are required to get the sample size of 80 subjects per group. Hence, a total of 160 patients were proposed to be enrolled in study.
Outcome measures
All efficacy assessment parameters were evaluated for each visit. The primary assessment parameters were visual analog scale (VAS) and Western Ontario and McMaster Universities Osteoarthritis index (WOMAC) scores improvement level at day 0, 60, and 120, respectively. The secondary assessment parameters were the levels of the biomarkers’, such as IL-1β, ROS, and MDA at day 0, 60, and 120, respectively.
Measurement of knee pain by VAS
The severity of pain was measured on VAS. It is a 10-cm horizontal line which contains word descriptions at each end, ranging from “0 to 10” (“0” indicating “no pain” and “10” indicating “unbearable pain”). Pain was assessed by the individual patient themselves by marking “no pain, mild pain, moderate pain, and severe pain” on the pain chart on each visit (Burckhardt and Jones 2003).
WOMAC score
The functional status of KOA patients was evaluated using the WOMAC scores (Likert Version-3.0) (Bellamy et al. 1988). The index consists of three subscales: pain, stiffness and physical function (PF). A higher score on the WOMAC scale represents poorer function or greater pain; the score is directly proportional to the severity of disease. WOMAC was used for functional assessment with 24 questions (Q) to grade: pain (Q1–5), stiffness (Q6–7), and physical functional difficulty (Q8–24). The patient’s response was graded qualitatively (0 = none, 1 = mild, 2 = moderate, 3 = severe, 4 = extreme) with a maximum score of 96.
Laboratory investigations
For biochemical estimations, blood samples were allowed to clot and then centrifuged at 3000 rpm for 30 min to get serum which was stored at −80 °C and analyzed within 1 month. Furthermore, serum analysis was done to measure the levels of MDA and IL-1β.
Chemicals and reagents
2′,7′-dichlorofluorescein diacetate, Histopaque-1077 solution and phosphate-buffered saline (Sigma-Aldrich, St. Louis, MO, USA), thiobarbituric acid (Loba Chemei, India), glacial acetic acid, and trichloroacetic acid (Biobasic, India) and standard curcumin (CDH Pvt, Ltd. New Delhi, India).
Biochemical estimations
Estimation of intracellular ROS
2′,7′-dichlorofluorescein diacetate is a stable non-fluorescent, cell permeable compound, which on penetrating the cell is converted to DCFH2 by intracellular esterases which is trapped within the cell and is stable for a few hours. The de-esterified product on oxidation by ROS is converted to the highly fluorescent 2′,7′dichlorofluorescein (DCF) and upon excitation at 488 nm emits green fluorescence which is proportional to the intracellular level of ROS. Thus, change in DCF fluorescence reflects mainly the intracellular accumulation of ROS (Boldyrev 2000).
Briefly, 1 ml of heparinized blood was carefully layered over 1-ml density gradient histopaque-1077 solution (Sigma-Aldrich), and then, it was centrifuged for 30 min at 300 ×g. The interface band of peripheral blood mononuclear cells (PBMCs) containing lymphocytes was isolated and washed with phosphate-buffered saline (PBS) and centrifuged at 3000 rpm. The supernatant was discarded, and the PBMCs so obtained were incubated with (10 µM) 2′,7′dichlorofluorescein diacetate (DCF-DA) dye for 30 min at 37 °C in dark.
A minimum of 10,000 events were acquired and lymphocyte population was focused on an ASSIST calibrated Image Stream X Mark II flow cytometer (AMNIS Corporation, Seattle, USA). RAW image files (rif) were acquired and adjusted for spectral overlap using IDEAS analysis software (AMNIS v.6.1.602).
Evaluation of serum MDA
The quantitative measurement of LPO in the terms of MDA equivalent was measured using TBARS assay according to the modified method (Ohkawa et al. 1979; Wade and van Rij 1988).
200 µl of trichloroacetic acid (TCA) (25-g TCA in 10 ml distilled water) was added to 1 ml of serum. The mixture was centrifuged at 1000 ×g for 10 min and the precipitate was reacted with 1 ml of 0.67 % TBA (w/v). The samples were heated at 90 °C for 30 min. After centrifugation, the absorption of MDA-TBA chromogen was measured at 532 nm on UV-spectrophotometer; using 1,1,3,3-tetramethoxy propane as standard. The results are expressed as nmol/ml using molar extinction coefficient of 1.56 × 105 M−1 cm−1.
Estimation of serum IL-1β
The quantitative estimation of serum IL-1β was done using a commercially available Enzyme Linked Immunosorbent Assay (ELISA) kit, according to the manufacturer’s protocol (Diaclone, Human serum ELISA Kit) with sensitivity of <5 pg/ml.
Statistical analysis
The results are presented as mean ± SE. Chi-square test was used to compare the categorical variables, and an unpaired t test was used to compare the discrete variables between the groups. Paired t test was used to compare the mean change in discrete variables from day 0 to day 60, day 60 to day 120, and day 0 to day 120. Study analysis was adjusted for the covariables, such as age, gender, and BMI. A p value <0.05 was considered to be significant. All the analysis was carried on SPSS 16.0 version (Chicago, Inc., USA).
Results
The demographic characteristics of the patients are given in Table 1, which shows that the mean age of the patients was 50.27 ± 8.63 for CL extract and 50.23 ± 8.08 for placebo group. In this study, number of female patients was 53 and 50 for CL extract and placebo group, respectively, while male patients were 25 and 32 for CL extract and placebo group, respectively. Radiographs of the knee of enrolled patients were taken, and the patients were graded according to KL grading scale revealing severity of the disease. On the basis of KL grade; KL-I = 7 and 4 patients, KL-II comprised of 11 and 19 patients, KL-III = 32 and 34 patients and KL-IV = 28 and 25 patients were there in group A and group B, respectively. The mean BMI of the patients was 28.32 ± 5.06 for test group and 27.40 ± 5.76 for placebo group patients.
Tables 2 and 5 and Fig. 4 show the primary outcome parameters of the study, i.e., VAS and WOMAC scores. The levels of VAS (4.96 ± 0.07 and 4.03 ± 0.08) and WOMAC score which included variables like pain (11.19 ± 0.26 and 9.48 ± 0.17), stiffness (4.51 ± 0.21 and 4.08 ± 0.17) and PF (41.28 ± 0.51 and 32.14 ± 0.40) were also significantly (p < 0.05) reduced in the test drug group than placebo group at day 60 and 120, when compared with baseline values. Moreover, the secondary outcome parameters, i.e., biochemical analysis of CL extract and placebo group patients are shown in Tables 3 and 6 and Figs. 3 and 4. There was significant (p < 0.05) reduction in the levels of IL-1β (65.61 ± 21.59 and 21.11 ± 1.176), ROS (2553 ± 775.67 and 1200 ± 864.08), and MDA (3.85 ± 0.12 and 3.69 ± 0.12) in the test drug group at day 60 and day 120, respectively, when compared with baseline values. The effect of CL extract and placebo treatment on clinical parameters is shown in Table 4. Adverse effects in CL extract group were 2 in 78 patients and in placebo group were 4 in 82 patients as detailed in Table 7.
Measurement of intracellular ROS production a and b are the gating strategies for focused cells and single cell population (lymphocyte), respectively. c and d the quantitative estimation of intracellular ROS formation in the two treatment groups, and its subsequent level at day 0, 60, and 120, respectively
Discussion
The study was envisaged primarily to evaluate the efficacy of CL extract in patients of KOA. Simultaneously, a correlation between severity of the disease and the levels of IL-1β, ROS, and MDA (biomarkers of oxidative stress) was investigated. Thus, this double-blind, randomized, placebo-controlled clinical trial was designed.
The incidence of OA is increasing, where the global prevalence of KOA has become 3.8 % among population between age 50–80 years; incidence being higher in females than in males (Cross et al. 2014). As the knee is the most affected (41 %) synovial joint (Cushnaghan and Dieppe 1991), the maximum research has been carried out on KOA; hence, we also targeted KOA in our study, more so due to easy availability of the patients.
In our study, we found that there was overall clinical improvement by CL in all three parameters of the WOMAC score (Pain, Stiffness, and PF) and VAS in the patients of KOA as compared to the placebo treated patients. This finding is in concurrence with the findings of many other workers (Belcaro et al. 2010; Kuptniratsaikul et al. 2014). Moreover, apart from clinical improvement, there was a decrease in disease-related biomarkers; the biomarker of inflammation (IL-1β) and OS (ROS and MDA) showed significant reduction. Various workers have worked with CL on KOA, but they have not estimated such biomarkers (Pinsornsak and Niempoog 2012; Madhu et al. 2013).
The clinical improvement in WOMAC score and VAS may be subjective, but the levels of biomarkers are certain parameters depicting status of anti-inflammatory activity and oxidative stress. It has been established that there is a rise in these markers in various diseases and lowering of these markers strongly correlates with the disease level (Mateen et al. 2016; Attur et al. 2015).
We evaluated the effect of CL extract on biochemical parameters, viz., IL-1β, ROS, and MDA and found that there was a significant reduction in the levels of these biomarkers in the CL extract group in all four grades of patients as compared to patients who received placebo. OS is related to inflammation (Martin et al. 2004); hence, it can be concluded that CL not only improves clinical parameters but also reduces OS and inflammatory processes related with KOA.
Madhu et al. (2013) have used curcuminoids free CL extract in a dose of 1000 mg/day, while Belcaro et al. (2010) have used a complex of curcumin in dose of 200 mg. It is claimed by the later workers that the complex increases bioavailability of curcumin, while the adverse events if any are decreased to a great extent. Madhu et al. (2013) have not estimated any biomarker in their study, while Belcaro et al. (2010) evaluated IL-1β and found reduction in elevated levels as has been observed in our study also. Paracetamol was given as rescue medicine, by Madhu et al. (2013); it is not clear whether given regularly or on SOS basis. In the study conducted by Belcaro et al. (2010), one group of patients was given ‘Best possible treatment’ and other group received ‘Best possible treatment’ plus the complex of curcumin (Meriva). However, both sets of workers have noted that the need for rescue medication was decreased. During our study a fixed regimen of diclofenac 50 mg BD and omeprazole 20 mg once day was given. As far as AE are concerned, very few AE were reported by the patients and these too were quite mild and of benign in nature. Other workers have also reported mild AEs in their studies (Kuptniratsaikul et al. 2014; Madhu et al. 2013).
The biomarkers were found to be significantly raised in the subjects of all four grades in our study on day 0, i.e., at the time of commencement of the study. Similarly, other scientists have found raised levels of ROS in KOA (Srivastava et al. 2015) and in other disorders also (Amico et al. 2015; Sarkar et al. 2005) and the level of MDA has also been reported to be elevated by some workers in OA and other inflammatory disorders (Paliwal et al. 2012).
Our study showed that the treatment with CL brought a decrease in baseline values of IL-1β, ROS, and MDA after 2 months’ treatment. This decrease was further reduced as the treatment was continued up to 4 months. Therefore, this study shows that CL is able to decrease biomarkers of inflammation.
KL grading scale is an established radiological method of grading the KOA patients according to radiological findings of the joint. In this study, it was used to grade the patients at the beginning and twice during study period. It was observed that the radiographic appearance of the joints was not improved even after 4 months treatment; however, significant symptomatic relief was observed after 2 months of treatment as assessed by VAS and WOMAC scores. This relief was further significantly increased after 4 months of treatment. This explains that although radiographically there was no change in the appearance of damaged joint, however, as there was arrest of inflammatory process and OS, the patients were relieved from pain and inflammation.
Conclusion
This study showed that adjuvant therapy of CL extract along with Diclofenac produces overall significant improvement in patients of KOA. It also improves the quality of life in patients and can be taken as herbal supplement. Thus, it is proposed that in the patients of KOA, CL should be given as soon as the diagnosis is made. Any NASID, if required may be given for a short period but CL may be prescribed for long durations, without fear of damaging GIT or kidneys of patient.
References
Allen KD, Chen JC, Callahan LF, Golightly YM, Helmick CG, Renner JB, Jordan JM (2010) Associations of occupational tasks with knee and hip osteoarthritis: the Johnston County Osteoarthritis Project. J Rheumatol 37(4):842–850
Altman RD, Marcussen KC (2001) Effects of a ginger extract on knee pain in patients with osteoarthritis. Arthr Rheum 44:2531–2538. doi:10.1002/1529-0131(200111)44:11<2531:AID-ART433>3.0.CO;2-J
Altman R, Alarcón G, Appelrouth D, Bloch D, Borenstein D, Brandt K (1991) The American College of Rheumatology criteria for the classification and reporting of osteoarthritis of the hip. Arthr Rheum 34:505–514
Ambrosia RD (2005) Epidemiology of osteoarthritis. Orthopedics 28:S201–S205
Amico D, Spadoni T, Rovinelli M, Serafini M, D’Amico G, Campelli N, Baroni SS, Gabrielli A (2015) Intracellular free radical production by peripheral blood T lymphocytes from patients with systemic sclerosis: role of NADPH oxidase and ERK1/2. Arthr Res Ther 17:68. doi:10.1186/s13075-015-0591-8
Attur M, Statnikov A, Samuels J, Li Z et al (2015) Plasma levels of interleukin-1 receptor antagonist (IL1Ra) predict radiographic progression of symptomatic knee osteoarthritis. Osteoarthr Cartil 23(11):1915–1924
Barron MC, Rubin BR (2007) Managing osteoarthritic knee pain. J Am Osteopath Assoc 107:21–27
Belcaro G, Cesarone MR, Dugall M, Pellegrini L, Ledda A, Grossi MG, Togni S, Appendino G (2010) Efficacy and safety of Meriva, a curcumin-phosphatidylcholine complex, during extended administration in osteoarthritis patients. Altern Med Rev 15:337–343
Bellamy N, Buchanan WW, Goldsmith CH, Campbell J, Stitt LW (1988) Validation study of WOMAC: a health status instrument for measuring clinically important patient relevant outcomes to anti-rheumatic drug therapy inpatients with osteoarthritis of the hip or knee. J Rheumatol 15:1833–1840
Bhatia D, Bejarano T, Novo M (2013) Current interventions in the management of knee osteoarthritis. J Pharm Bioallied Sci 5:30–38. doi:10.4103/0975-7406.106561
Bjordal JM, Ljunggren AE, Klovning A, Slordal L (2004) Non-steroidal anti-inflammatory drugs, including cyclo-oxygenase-2 inhibitors, in osteoarthritic knee pain: meta-analysis of randomised placebo controlled trials. BMJ 329(7478):1317. doi:10.1136/bmj.38273.626655.63
Boldyrev AA (2000) Discrimination between apoptosis and necrosis of neurons under oxidative stress. Biochemistry 65:834–842
Burckhardt CS, Jones KD (2003) Adult measures of pain: The McGill Pain Questionnaire (MPQ), Rheumatoid Arthritis Pain Scale (RAPS), Short-Form McGill Pain Questionnaire (SF-MPQ), Verbal Descriptive Scale (VDS), Visual Analog Scale (VAS), and West Haven-Yale Multidisciplinary Pain Inventory (WHYMPI). Arthr Care Rheum 49:96–104
Chandra D, Gupta SS (1972) Anti-inflammatory and anti-arthritic activity of volatile oil of Curcuma longa (Haldi). Indian J Med Res 60:138–142
Cross M, Smith E, Hoy D, Nolte S, Ackerman I, Fransen M, Bridgett L, Williams S, Guillemin F, Hill CL, Laslett LL, Jones G, Cicuttini F, Osborne R, Vos T, Buchbinder R, Woolf A, March L (2014) The global burden of hip and knee osteoarthritis: estimates from the global burden of disease 2010 study. Ann Rheum Dis 73(7):1323–1330
Cushnaghan J, Dieppe P (1991) Study of 500 patients with limb joint osteoarthritis. Analysis by age, sex, and distribution of symptomatic joint sites. Ann Rheum Dis 50:8–13
Das SK, Farooqi A (2008) Osteoarthritis. Best Pract Res Clin Rheumatol 22:657–675. doi:10.1016/j.berh.2008.07.002
Dharmananda S (1998) Countering side effects of modern medical therapies with Chinese herbs. Institute for Traditional Medicine, Portland
Esser S, Bailey A (2011) Effects of exercise and physical activity on knee osteoarthritis. Curr Pain Headache Rep 15:423–430
Goldring MB, Otero M (2011) Inflammation in osteoarthritis. Curr Opin Rheumatol 23:471–478
Harma M, Erel O (2003) Increased oxidative stress in patients with hydatidiform mole. Swiss Med Wkly 133:563–566
Ji Q, Xu X, Zhang Q, Kang L, Xu Y, Zhang K, Li L, Liang Y, Hong T, Ye Q, Wang Y (2016) The IL-1beta/AP-1/miR-30a/ADAMTS-5 axis regulates cartilage matrix degradation in human osteoarthritis. J Mol Med (Berl) 94(7):771–785. doi:10.1007/s00109-016-1418-z
Jurenka JS (2009) Anti-inflammatory properties of curcumin, a major constituent of Curcuma longa: a review of preclinical and clinical research. Altern Med Rev 14:141–153
Kaviratna AC, Sharma P (1913) The Charaka Samhita 5 Vols., Sri Satguru, ISBN81-7030. pp 471–477
Kellgren JH, Lawrence JS (1957) Radiological assessment of osteo-arthrosis. Ann Rheum Dis 16:494–502
Kuptniratsaikul V, Dajpratham P, Taechaarpornkul W, Buntragulpoontawee M, Lukkanapichonchut P, Chootip C, Saengsuwan J, Tantayakom K, Laongpech S (2014) Efficacy and safety of Curcuma domestica extracts compared with ibuprofen in patients with knee osteoarthritis: a multicenter study. Clin Interv Aging 9:451–458. doi:10.2147/CIA.S58535
Lopaczynski W, Zeisel SH (2001) Antioxidants, programmed cell death and cancer. Nutr Res 21:295–307
Madhu K, Chandan K, Saji MJ (2013) Safety and efficacy of Curcuma longa extract in the treatment of painful knee osteoarthritis: a randomized placebo-controlled trial. Inflammopharmacology 21:129–136. doi:10.1007/s10787-012-0163-3
Martin JA, Brown TD, Heiner AD et al (2004) Chondrocyte senescence, joint loading and osteoarthritis. Clin Orthop Relat Res 427:S96–S103
Mateen S, Moin S, Khan AQ, Zafar A, Fatima N (2016) Increased Reactive Oxygen Species Formation and Oxidative Stress in Rheumatoid Arthritis. PLoS One 11(4):e0152925. doi:10.1371/journal.pone.015292
Michael JW, Schlüter-Brust KU, Eysel P (2010) The epidemiology, etiology, diagnosis, and treatment of osteoarthritis of the knee. Dtsch Arztebl Int 107:152–162. doi:10.3238/arztebl.2010.0152
Mittal M, Siddiqui MR, Tran K, Reddy SP, Malik AB (2014) Reactive oxygen species in inflammation and tissue injury. Antioxid Redox Signal 20(7):1126–1167
Moher D, Hopewell S, Schulz KF, Montori V, Gotzsche PC, Devereaux PJ (2010) CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. BMJ 340:c869. doi:10.1136/bmj.c869
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Paliwal MN, Sontakke AN, Paliwal P (2012) Study of enzymatic antioxidant marker and lipid peroxidation status in patients with osteoarthritis. J Evol Med Dent Sci 1:634–638
Pinsornsak P, Niempoog S (2012) The efficacy of Curcuma Longa L. extract as an adjuvant therapy in primary knee osteoarthritis: a randomized control trial. J Med Assoc Thail 95(Supp 1):S51–S58
Roth G, Chandra A, Nair M (1998) Novel bioactivities of Curcuma longa constituents. J Nat Prod 61:542–545
Sarkar M, Varshney R, Chopra M, Sekhri T, Adhikari JS, Dwarakanath BS (2005) Flow-cytometric analysis of reactive oxygen species in peripheral blood mononuclear cells of patients with thyroid dysfunction. Cytom B Clin Cytom 70:20–23
Scartezzini P, Speroni E (2000) Review on some plants of Indian traditional medicine with antioxidant activity. J Ethnopharmacol 71:23–43
Selvam R, Subramanian L, Gayathri R, Angayarkanni N (1995) The anti-oxidant activity of turmeric (Curcuma longa). J Ethnopharmacol 47:59–67
Shahine EM, Elhadidi AS (2014) Efficacy of glucosamine sulfate in lowering serum level of interleukin-1β in symptomatic primary knee osteoarthritis: Clinical and laboratory study. Alex J Med 50(2):159–163
Sharma OP (1976) Antioxidant activity of curcumin and related compounds. Biochem Pharmacol 25(15):1811–1812
Srivastava S, Saksena AK, Khattri S, Kumar S, Dagur RS (2015) Status of oxidative stress biomarkers in osteoarthritis patients in North Indian population. Osteoarthr Cartil 23:A84–A85. doi:10.1016/j.joca.2015.02.785
The Ayurvedic Pharmacopoeia of India (1989) Part I, Department of AYUSH, Ministry of Health and Family Welfare, Govt. of India
Verma P, Dalal K (2013) Serum cartilage oligomeric matrix protein (COMP) in knee osteoarthritis: a novel diagnostic and prognostic biomarker. J Orthop Res 31(7):999–1006. doi:10.1002/jor.22324
Wade CR, van Rij AM (1988) Plasma thiobarbituric acid reactivity: reaction conditions and the role of iron, antioxidants and lipid peroxy radicals on the quantitation of plasma lipid peroxides. Life Sci 43:1085–1093
Wang CT, Lin J, Chang CJ, Lin YT, Hou SM (2004) Therapeutic effects of hyaluronic acid on osteoarthritis of the knee. A meta-analysis of randomized controlled trials. J Bone Jt Surg 86:538–545
Yudoh K, Nguyen T, Nakamura H et al (2005) Potential involvement of oxidative stress in cartilage senescence and development of osteoarthritis: oxidative stress induces chondrocyte telomere instability and down regulation of chondrocyte function. Arthr Res Ther 7(2):R380–R391
Acknowledgments
We are thankful to COUNCIL OF SCIENTIFIC AND INDUSTRIAL RESEARCH (CSIR), India for providing Senior Research Fellowship to MR. SHOBHIT SRIVASTAVA vide file no. 09/910/0006/2012-EMR-I for the study. Our sincere thanks to The Himalaya Drug Company, Bangalore, India for providing capsules of CL extract and placebo for the study. We are also thankful to Dr. A.K.S. Rawat, Scientist and Head, Department of Pharmacognosy and Ethnopharmacology, CSIR-National Botanical Research Institute, Lucknow for carrying out the finger print analysis of the CL extract capsules.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Srivastava, S., Saksena, A.K., Khattri, S. et al. Curcuma longa extract reduces inflammatory and oxidative stress biomarkers in osteoarthritis of knee: a four-month, double-blind, randomized, placebo-controlled trial. Inflammopharmacol 24, 377–388 (2016). https://doi.org/10.1007/s10787-016-0289-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-016-0289-9