Abstract
Nutrients such as nitrogen and phosphorus are typically considered detrimental to wetland values, but waterfowl can be numerous on nutrient-rich wetlands. Waterfowl were counted three to six times per year on nine treatment lagoons and associated wetlands (2,025 ha) at the Western Treatment Plant (south-east Australia) from 2000, to help maintain ecological values of this Ramsar-listed wetland as well as treating sewage for a large city (Melbourne). Up to 185,000 waterfowl were counted, varying with season, continental rainfall and lagoon operation. Nutrient levels were reduced on Lake Borrie lagoon in 2005 (as part of an Environmental Improvement Program) and restored in 2015. Waterfowl declined on Lake Borrie lagoon from 2005 when it received treated effluent not raw sewage, and increased in 2015 when it received partially treated sewage. This pattern was highly significant for total waterfowl and most species and guilds at Lake Borrie but was not replicated on other lagoons. Modelling revealed positive relationships between waterfowl numbers and nutrient concentrations, including ammonia and nitrite, to moderate levels. This shows that with careful management nutrient enrichment can have positive benefits, allowing artificial wetlands such as wastewater treatment plants to support high densities of waterbirds and the food webs that sustain them.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
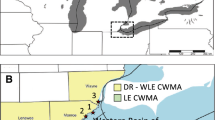
Water supply and wastewater treatment are two of the big issues facing humanity, and loss and modification of wetlands are two of the big issues in biodiversity conservation (Foote et al., 1996; Finlayson et al., 2019). When people divert water for domestic, agricultural or industrial use there is an inevitable loss of water for natural wetlands (Kingsford, 2000; Gordon et al., 2010). When wastewater is adequately treated before it is returned to the environment there may be opportunities to use treated wastewater for the benefit of biodiversity (Steele et al., 2006; Murray & Hamilton, 2010), partly alleviating the harm incurred when water was originally diverted for human use. Wastewater contains high levels of nutrients such as nitrogen and phosphorus, which can damage natural ecosystems in lakes, streams and coastal environments by encouraging excessive growth of algae (e.g. Schindler, 2006; Smith & Schindler, 2009; Chislock et al., 2013). Phosphorus is generally considered to be the limiting factor in freshwater systems, and nitrogen in coastal systems. Hence nutrient removal is a key focus of treatment plants, and the effects of nutrient enrichment and eutrophication have been a dominant theme of hydrological literature (Smith et al., 2006; Conley et al., 2009). However, those same nutrients and algae can also drive increased productivity, supporting high populations of invertebrates and waterbirds that feed on them. This paper describes a 20-year study that documents consequences of nutrient removal and re-addition at an operational scale at a plant that is used both for wastewater treatment and for waterbird conservation as part of a Ramsar-listed set of wetlands near Melbourne, south-east Australia (Fig. 1).
Sewage treatment plants that rely on pondage have long been recognised as valuable habitats for waterbirds (e.g. Fuller & Glue, 1980; Maxson, 1981; Carlisle & Mumootil, 1991), including in Australia (Loyn, 1989; Murray et al., 2013, 2014; Breed et al., 2020), and specifically the Western Treatment Plant (WTP) (Lane & Peake, 1990; Hamilton et al., 2005; Steele et al., 2006; Loyn et al., 2014a, b). Benefits can arise both from reliability of water supply and the level of nutrient enrichment, with nutrients driving the productivity of invertebrates that form the main food of most waterbird species. Usually the benefits are opportunistic, but sometimes there are ways of managing treatment plants to enhance those benefits (Murray & Hamilton, 2010). However, more needs to be known about the mechanisms involved and the effects of management interventions that have been tried. This paper examines changes in diversity and abundance of waterfowl associated with two major engineering interventions to improve both efficiency of sewage treatment and foraging habitat for waterfowl at the WTP. One part of the system (30 ponds in Lake Borrie lagoon, 454 ha) where nutrient levels were greatly reduced from 2005 to 2015 and then raised to previous levels 2015–2020, is compared with the remaining 288 ponds (1,474 ha) where such major changes did not occur (Fig. 1).
This paper has two main aims. One is to describe the changes that have occurred in water chemistry and waterbird numbers at Lake Borrie lagoon over three periods of differing sewage treatment processes: 2000–2004 (Period A) when it received raw sewage before an Environment Improvement Project (EIP); 2005–2015 (Period B) after the EIP, when it received Class C recycled water; and 2015 to 2020 (Period C) after the introduction of partly treated sewage. The second aim is to present models relating waterfowl numbers at Lake Borrie lagoon to water chemistry at the Borrie outlet, taking advantage of the unique opportunity provided by these interventions, as a step in understanding the complex mechanisms involved. Special attention is paid to nitrogen compounds as one of the principal aims of wastewater treatment is to limit discharge of nitrogen into other systems (the sea in this case), by releasing it as elemental N2 (the main constituent of air), whereas most other nutrients lack a volatile phase.
The changes in Lake Borrie lagoon are considered relative to changes in other lagoons at the WTP to control for effects of stochastic variables such as continental weather patterns. Concentrations of nitrogen and other nutrients remained reasonably constant at the two main outlets other than the Borrie outlet (Fig. 2), with increased inputs of nutrients (due mostly to Melbourne’s rising human population) largely balanced by increased treatment capacity and supply of recycled water to local farms, maintaining permitted levels of discharge to the sea. These comparisons and models may assist managers of wastewater treatment plants in balancing dual objectives of efficient sewage treatment and providing waterfowl habitat.
Mean levels of nitrogen compounds (mgN/l) and other nutrients (mg/l) at the Borrie outlet (upper graph) and the average of two main Other outlets (lower graph) at the WTP in three periods (A-C), 2000–2020. In period A (2000–2004) Lake Borrie lagoon received a mix of raw and treated sewage; in period B (2005–2015) it received fully treated recycled water, and in period C (2015–2020) it received partly treated sewage. Note that the quantities for nitrite have been multiplied by 5, and for TSS have been divided by 3, to make scales visually comparable. CBOD5 = Carbon Biological Oxygen Demand (a potential measure of bacterial activity) and TSS = Total Suspended Solids. Error bars show Standard Errors
Western Treatment Plant (WTP)
The WTP is renowned for its value as a habitat for waterbirds and it forms a key part of a wetland system, listed under the Ramsar Convention in 1982 as a wetland of international importance, known as the Port Phillip Bay (western shoreline) and the Bellarine Peninsula Ramsar site (Lane & Peake, 1990; Hale, 2010; DELWP, 2018). Ramsar values need to be maintained under Australian Commonwealth legislation including the Environment Protection and Biodiversity Conservation Act 1999 (EPBC Act). The WTP is also the source of the single largest dry-weather nitrogen discharge to Port Phillip Bay, which has necessitated a series of treatment upgrades.
Pond treatment involves several lagoons (series of ponds), operating in parallel with a residence time of about 60 days. Most ponds are less than 1 m deep, with average operational depths of ~ 0.8 m. Three modern lagoons (115E, 25W and 55E) built in the 1990s are up to 2 m deep.
Melbourne Water upgraded the sewage treatment system between 1998 and 2005 (the EIP) to reduce nitrogen discharge to Port Phillip Bay, convert biogas to electricity and reduce odours. The EIP upgraded two of the modern lagoons (25W and 55E, Fig. 1) and improved the primary treatment by covering initial ponds (capturing biogas and generating electricity) and installing activated sludge plants for nitrogen reduction. Land and grass filtration (a major source of odour) were discontinued, and farmland areas previously used for filtration were irrigated with Class C recycled water from the modern lagoons to produce pasture and crops. However, as the water distribution network could not supply recycled water and distribute sewage at the same time, three lagoons (Borrie, Western and T-Section) had to be decommissioned from the sewage treatment system. These three lagoons instead received recycled water intended to maintain their value as biodiversity habitat (Melbourne Water, 2002a, b). These changes became operational in 2004–2005.
Waterbird modelling (Loyn et al., 2002; Hamilton et al., 2005) predicted that lower nutrient levels would have adverse effects on waterfowl, especially on diving ducks and filter-feeding ducks that favoured ponds with moderately high nutrient levels, presumably because those nutrients helped support high densities of potential food such as oligochaete worms and larvae of chironomid midges, which were abundant in the ponds.
Lake Borrie lagoon
Lake Borrie lagoon was the main set of ponds decommissioned by the EIP in 2005 (and fed recycled water), and it was reconnected to the treatment system in 2015. It consists of two parallel systems (North and South), feeding into a common outlet.
To increase treatment capacity and potentially restore habitat value to Lake Borrie lagoon, a new pipeline was built in 2015 to deliver partially treated sewage to it. This presented an opportunity to examine effects of sewage removal from Lake Borrie lagoon and its subsequent restoration, with the Other lagoons providing control data.
Effects of the EIP (previous studies and management responses)
Monitoring showed that waterfowl numbers fluctuated in response to seasonal and continental rainfall patterns, dropping dramatically when there was plentiful water inland (Loyn et al., 2014a, b). The EIP was found to have a relatively small effect on waterfowl numbers across the whole WTP in the seven years after its implementation (2005 to 2012, Loyn et al., 2014a). However, some marked changes in distribution within the WTP were observed: more waterfowl used the old lagoons than previously, and fewer used the decommissioned lagoons (Loyn et al., 2014a). Feeding rates of waterfowl in Lake Borrie lagoon were also found to have declined (Mustoe & Waugh, 2006; Mustoe, 2009; Guay, 2013) compared with earlier measurements (Hamilton et al., 2002).
Methods
Waterfowl counts
Waterfowl were counted by species on a pond-by-pond basis across the WTP six times per year from 2000 to 2018 and then three times per year to 2020 (n = 112) (Loyn et al., 2002, 2014a). These counts focussed on ducks, swans, geese and other birds that habitually swim on open water and feed extensively in the treatment ponds at the WTP (e.g. coot and grebes). Marsh terns (Whiskered Tern Chlidonias hybrida [Pallas, 1811] and White-winged Black Tern Chlidonias leucopterus [Temminck, 1815]), which take food from the ponds’ surface, were also included. Counts were conducted mainly from a stationary vehicle (serving as a hide), using binoculars and a window-mounted telescope.
Each count aimed to cover all ponds that held water and hence provided habitat for waterfowl, along with other important habitats for waterfowl at the WTP (e.g. coasts, flooded borrow pits, areas of temporary floodwater and the Little River estuary). A natural ephemeral swamp (Ryan’s Swamp) was included on the few occasions when it held water. Altogether the counts covered 318 ponds of which 30 were in Lake Borrie lagoon (totalling 454 ha) and 288 ponds were elsewhere at the WTP (1,474 ha). Because it was not possible to count all birds at all sites in a single day, each count was conducted over 2–6 days, depending on the numbers of birds present. Observers were conscious of the risk of double-counting or overlooking some birds due to movements between sites during the surveys, and took measures to minimise those effects. Particular species or guilds of birds (e.g. dabbling ducks, diving ducks or filter-feeding ducks) were counted on different days to enable observers to cover as much area as possible for those species on a single day, and hence minimise effects of birds moving between sites. Counts disrupted by disturbance (e.g. by birds of prey) or other movements were sometimes repeated if observers were unable to assess how many birds had moved between ponds.
The counts were made in six ‘seasons’: I (January), II (late February or early March), III (April or May), IV (June or July), V (late August to October) and VI (November or December). When numbers of counts dropped from six to three a year (from 2018), those counts were conducted in February (late summer, season II), June–July (winter, season IV) and December (early summer, season VI).
Data were tabulated for analysis at the level of individual species, feeding guilds and total waterfowl. Waterfowl were considered as all Anatidae (ducks, geese and swans) along with coot and grebes, which also habitually feed while swimming on ponds. Species were assigned to guilds as shown in Table 1. Data on guilds were calculated by summing counts for all constituent species, including uncommon species that did not warrant individual analysis (e.g. Australian Wood Duck Chenonetta jubata [Latham, 1802], and rare vagrants such as Northern Shoveler Anas clypeata Linnaeus, 1758 and Tufted Duck Aythya fuligula [Linnaeus, 1758]. Australian Shelduck Tadorna tadornoides (Jardine & Selby, 1828) were classed as grazers because they often eat plant material in farmland, but they also take a range of animal food (Frith, 1982; Marchant & Higgins, 1990) and they may have been taking a wide range of food when they were feeding on treatment ponds in this study, especially when they were flightless (during moult) and unable to access food in farmland. Hence, values were calculated for the grazing guild with and without Australian Shelduck (which was often the most numerous species in the guild).
Physical and chemical data
Physical and chemical data were collected from the Borrie outlet on a weekly basis by Melbourne Water, by grab samples analysed by ALS (Australian Laboratory Services) Global Laboratories. The data were reduced by selecting dates closest to the dates of our 112 waterfowl counts. Complete information over the 20-year period was available for the following nine variables: Ammonia (NH3 or NH4+), Nitrate (NO3-), Nitrite (NO2-), TKN (Total Kjeldahl Nitrogen), DIN (Dissolved Inorganic Nitrogen), Organic Nitrogen, Total Phosphorus (P), CBOD5 (5-day Carbonaceous Biological Oxygen Demand, a measure of organic carbon and bacterial activity), and TSS (Total Suspended Solids).
The measures of Organic Nitrogen were calculated by subtracting ammonia from TKN: these were found to agree closely with direct measurements made for part of the period, noting that TKN measures reduced forms of nitrogen (e.g. ammonia, protein or amino acids) rather than oxidised forms (nitrate or nitrite). Measures were expressed as mg/l, or mg N/l for nitrogen compounds. Salinity was measured routinely at the inlet but not the outlet, and was not expected to vary differentially between lagoons.
Statistical analysis
To analyse the effects of management in Lake Borrie lagoon, three treatment periods were compared: Period A (when it received raw sewage, October 2000 to November 2004, n = 24); B (when it received recycled water, January 2005 to June 2015, n = 65), and C (when it received partly treated sewage after reconnection, July 2015 to June 2020, n = 23). Because numbers of waterfowl vary greatly with year, season and continental rainfall patterns (Loyn et al., 2014a), it was important to allow for the effects of those variables. The effects of continental rainfall are well known in a general sense (Frith, 1982; Kingsford & Norman, 2002; Roshier et al., 2002), with numbers of waterfowl declining at coastal wetlands when there is plentiful water in inland Australia and increasing in subsequent seasons (Gosper et al., 1983; Chambers & Loyn, 2006; Breed et al., 2023), but patterns and lag times can vary between flood events (Loyn et al., 2014a, b; Clarke et al., 2015), which adds to the difficulty of controlling for those variables. For the present purpose, variables such as continental weather patterns were controlled by comparing changes in the numbers of waterfowl at Lake Borrie lagoon with those at the Other lagoons, which would be affected in the same way by those external variables. Data on water chemistry from the main two outlets from the Other lagoons showed that it was reasonably stable over time compared with the massive changes observed at the Borrie outlet (Fig. 2). Hence the approach used was to focus on any differences in patterns between Lake Borrie lagoon and the rest of the WTP (Borrie vs Other), as in a BACI design (before-after-control-impact) (Underwood, 1994; Fisher et al., 2019).
To determine whether numbers of waterfowl varied between the three treatment periods in Lake Borrie lagoon, compared with other parts of the WTP, linear mixed models were fitted to the data. This is a preferred approach for unbalanced data sets, with different numbers of observations in the categories of interest (periods in this case) and potential clustering. For each bird species and guild, models were developed with fixed terms for pond group (Borrie or Other) + period (A, B or C) + the interaction between those terms. Significant interactions would show differences in pattern between the two pond groups, which might be related to management of each part. Year, season and month (nested within season) were included as random terms. Data on waterfowl numbers were transformed by taking natural logarithms, using ln (N + 1) for species and ln (N + 10) for guilds (if there were any zero values, otherwise ln N), applied to all relevant cells within each dataset. Zero values were rare for the common species and guilds, and did not occur at all for total waterfowl, diving ducks and grazers (with or without shelduck). Plots of residuals vs fitted values were examined to test the assumption of homogeneity of variance and to pick up any patterns that may suggest a different model structure.
A similar modelling approach was used to determine whether water chemistry varied between the three periods. This was mainly expected to happen at Lake Borrie lagoon, where operational changes coincided with the periods selected. Log transformations were used for CBOD5 and all the nitrogen variables except organic nitrogen. Log transformations were found to be not necessary for organic nitrogen, total phosphorus and TSS. Conclusions from raw or log-transformed data were similar in all cases.
To investigate relationships between water chemistry and relative numbers of waterfowl in Lake Borrie lagoon (compared with elsewhere at the WTP), linear models were developed relating abundance of waterfowl species and guilds in Lake Borrie lagoon (as a percentage of total numbers at the WTP) to suites of the nine physical or chemical variables at the Borrie outlet across all three periods (112 waterfowl counts). Correlation coefficients revealed that DIN and TKN were closely correlated to ammonia (r > 0.96) and could not be included together in the same models. Ammonia was chosen as the preferred variable (of those three), as DIN and TKN include some of the chemical entities that were planned to be considered separately. Hence, the suite of explanatory variables was narrowed to seven, including four relating to discrete nitrogen entities that collectively embrace the full spectrum of dissolved nitrogen in the system, with no overlap (ammonia, nitrite, nitrate and organic nitrogen). Based on residual plots, no transformation was found necessary for dependent or explanatory variables.
Genstat software (18th Edition, VSN International, UK) was used for most of the modelling, taking a ‘best subsets regression’ approach to explore suitable combinations of explanatory variables. The best model involving only linear terms was chosen on the basis of explaining a higher proportion of variance and few if any non-significant terms. Significance was based on P < 0.05, except in relation to quadratic effects where lower levels of significance (P < 0.1) were considered appropriate as continuous linear trends were considered unlikely and potentially misleading.
When particular explanatory variables proved influential, quadratic terms were added for those variables, seeking non-linear relationships to test the hypothesis that it is possible to have either too much or too little of particular nutrients. Where such relationships were found (of the form ax2 + bx + c), those relationships were graphed and turning points (optimum levels) were calculated as TP = − b/2a, based on models including only that variable and its square. The models were also used to estimate the range of values over which waterfowl species or guilds continued to occur (densities > 0), using the standard formula for solving quadratic equations, \(=\frac{-b\pm \sqrt{{b}^{2}-4ac}}{2a}\). While it would have been possible to include quadratic terms in the initial modelling process, that would have risked obscuring the comparisons between the three periods for Borrie and Other, which were the main focus of the study. It is generally better to begin modelling relationships with linear terms, then if appropriate, explore curvilinear terms as well (Sokal & Rohlf, 1995).
Results
Changes in water chemistry
Nutrient concentrations in the Borrie outlet were comparatively high in period A (when Lake Borrie lagoon received raw sewage) and period C (when it received partly treated sewage) and low in period B (when it received recycled water) (Fig. 2). These differences between periods were significant for ammonia, nitrite, total dissolved nitrogen, organic nitrogen, TKN and total phosphorus. TSS showed a similar pattern but the differences were not significant (Fig. 2). Nitrate showed a different pattern, appearing to increase with time from periods A to B to C, but those differences were not significant. CBOD5 declined from period A to period B and only showed a modest (non-significant) increase in period C (Fig. 2).
TKN and DIN were strongly correlated with ammonia (r > 0.96) (Table S1) and so were not considered further in the modelling process. Lesser positive correlations (r values between 0.22 and 0.62) were found between ammonia, nitrite and nitrate. Phosphorus was positively correlated with ammonia and nitrite but not nitrate. Organic nitrogen was correlated positively with nitrite but not ammonia or nitrate. CBOD5 was correlated positively with organic nitrogen and negatively with nitrate. TSS was correlated positively with organic nitrogen but not with other nutrients (Table S1).
In contrast, no significant variation in these variables with period was found at the two main outlets discharging from Other lagoons (15E and 145W, Fig. 2 lower graph). A fourth outlet (Murtcaim) was not considered because it ceased operation in 2004, and it served a part of the treatment plant where grass filtration was the main treatment process, using paddocks of long grass that attracted large numbers of ibis but relatively small numbers of the waterfowl considered in this paper.
Differences between Lake Borrie lagoon and Other lagoons
Most species and guilds were well represented both on Lake Borrie lagoon and on Other lagoons. Over the whole period three species had higher densities on Lake Borrie lagoon than elsewhere (Freckled Duck Stictonetta naevosa [Gould, 1841], Pink-eared Duck Malacorhynchus membranaceus [Latham, 1802] and White-winged Black Tern), along with the guild of filter-feeding ducks which includes the first two species (Table 1). Mean densities of other species and guilds did not differ greatly between Lake Borrie lagoon and Other lagoons.
Differences between the three periods
Six species and three guilds showed significant differences between the three periods, averaged across both lagoon groups (Table S2). One of the species (Australasian Shoveler Anas rhynchotis Latham, 1802) was most numerous in period A; two species (Australasian Grebe Tachybaptus novaehollandiae [Stephens, 1826] and Cape Barren Goose Cereopsis novaehollandiae Latham, 1802) were most numerous in period B and two species (Australian Shelduck and Hardhead Aythya australis [Eyton, 1838]) were most numerous in period C. Of the three guilds, diving ducks and grazers were most numerous in period C and least numerous in period B and filter-feeding ducks were least numerous in period B, with little difference between periods A and C (Table S2).
Changes in waterfowl numbers
Most species and all but two guilds showed significant interactions between lagoon group and period (Table S2), all showing positive responses on Lake Borrie lagoon after reconnection (Fig. 4). The exceptions (showing no significant interactions) were the guild dabbling ducks and two of its constituent species (Chestnut Teal Anas castanea [Eyton, 1838] and Pacific Black Duck Anas superciliosa J.F. Gmelin, 1789); marsh terns and both constituent species (Whiskered Tern and White-winged Black Tern); and three other species whose numbers varied greatly (Australasian Grebe, Great Crested Grebe Podiceps cristatus [Linnaeus, 1758] and Pink-eared Duck). Apart from the two grebes mentioned above, even the exceptions generally increased on Lake Borrie lagoon after reconnection, but they also increased in the Other lagoons. Patterns of variation between the three time periods were much more marked for Lake Borrie lagoon than for the Other lagoon group.
The most common pattern on Lake Borrie lagoon was for species and guilds to be relatively scarce in period B (when it received recycled water) compared with period A (when it received raw sewage) or period C (when it received partly treated sewage), in line with the changes in nutrient levels already described (Figs. 2, 3). At the guild level this applied most clearly to total waterfowl, filter-feeding ducks, diving ducks and grebes (Fig. 4) and most of their constituent species (Table S2), and in all those cases the pattern differed significantly from patterns observed on the Other lagoon group. Marsh terns showed a similar pattern but their numbers were highly variable and the differences were not significant. Dabbling ducks showed a similar increase in period C on Lake Borrie lagoon but did not decline significantly between periods A and B; they also increased on Other lagoons, and collectively did not show an interaction with the Other lagoon group. However, one of their three constituent species (Grey Teal Anas gracilis Buller, 1869) did show such an interaction, with the increase in period C being significantly more marked on Lake Borrie lagoon than on Other lagoons. Grazers (swan, coot, shelduck, etc.) increased more on Other lagoons than they did on Lake Borrie lagoon, and they were the only guild to show that pattern (Fig. 4).
Mean numbers of waterfowl and waterfowl guilds on Lake Borrie lagoon (left) and Other lagoons (right) at WTP in three periods (A-C) 2000–2020. The interaction between pond group and period was significant for filter-feeding ducks, diving ducks, grebes, grazers and 12 individual species (Table S2), with all except grazers and Australian Shelduck increasing more in the Borrie lagoon after reconnection than on Other lagoons. Error bars show Standard Errors
Most species and guilds on Lake Borrie lagoon increased in period C to levels that approached or exceeded levels observed in period A (Fig. 4), indicating close to complete recovery from the low levels in period B. For total waterfowl, there was no significant difference between numbers on Lake Borrie lagoon in period C compared with period A, with both being higher than in period B (Fig. 4). At the guild level, dabbling ducks, diving ducks, grazers and grebes were all more numerous on Lake Borrie lagoon in period C than they had been in period A. The same applied to ten of the 19 species analysed (Table S2). Numbers of filter-feeding ducks did not differ significantly between periods A and C (both being higher than in period B), and the same applied to two of its constituent species (Pink-eared Duck and Freckled Duck). The third species in the guild (Australasian Shoveler) had also increased from levels observed in period B, but they were still not as numerous as they had been in period A.
Among the diving ducks, the two most numerous species (Hardhead and Blue-billed Duck Oxyura australis Gould, 1837) followed the same pattern as the guild, declining on Lake Borrie lagoon in period B and then increasing to mean levels in period C that exceeded those observed in period A (Table S2). However, the third species in the guild (Musk Duck Biziura lobata [Shaw, 1796]) declined in period B and showed no significant recovery in period C (Table S2).
Relationships between effluent chemistry and waterfowl on Lake Borrie lagoon
Over the 20 years, numbers of total waterfowl and various guilds and species on Lake Borrie lagoon (as a percentage of those on the entire WTP) were positively correlated with ammonia, nitrite, organic nitrogen, CBOD5 and phosphorus as measured at the Borrie outlet (Table S1), whereas nitrate and TSS appeared to be less influential. Correlations with nitrate tended to be small or sometimes negative, and TSS was correlated with numbers of just two species (Hoary-headed Grebe Poliocephalus poliocephalus [Jardine & Selby, 1827] and Pacific Black Duck, both positively) and no guilds (Table S1). The best regression models are shown in Table 2 (for guilds and total waterfowl) and Table 3 (for individual species). Quadratic relationships were found for nitrite (with total waterfowl, diving ducks, grebes, Freckled Duck and Hardhead), ammonia (for Musk Duck, Australian Shelduck, Australasian Shoveler and Chestnut Teal), organic nitrogen (for Australasian Shoveler) and phosphorus (for Blue-billed Duck and Hoary-headed Grebe) (Tables 2, 3), implying that there were optimum levels (turning points) for those nutrients (Table 4).
The best combined model for total waterfowl included positive terms for nitrite, CBOD5 and phosphorus, and a negative term for nitrite squared (Table 2), with a turning point for nitrite at 0.7 mg N/l (Table 4; Fig. 5). The quadratic relationship for nitrite implies that waterfowl increase with increasing levels of nitrite up to the turning point and then decline with further increases. Models for grebes were similar to those for total waterfowl (Table 2), with a turning point for nitrite also at 0.7 mg N/l (Table 4; Fig. 5). CBOD5 did not emerge as a significant variable in models for any waterfowl guild or species other than total waterfowl, grebes and the most numerous grebe species (Hoary-headed Grebe).
Models for diving ducks included positive terms for ammonia as well as nitrite, and negative terms for ammonia squared as well as nitrite squared (Table 2), with turning points for ammonia at 27.5 mg N/l and nitrite at 0.7 mg N/l (Table 4; Fig. 5). They also included negative terms for nitrate (Table 2). All these models explained > 30% of variance in numbers of the respective guilds as a percentage of numbers over the entire WTP (Table 2). Models for other guilds were much weaker, explaining < 10% of variance (Table 2). No useful models were found for dabbling ducks or coot in relation to effluent discharge.
At the species level, models explaining > 30% of variance were found for Hoary-headed Grebe (similar to grebes as a guild) and two species of diving ducks (Blue-billed Duck and Hardhead) (Table 3). The latter models were similar to the models for diving ducks as a guild, but the best model for Blue-billed Duck also included a positive term for phosphorus and a negative term for phosphorus squared, with a turning point at 18 mg/l (Table 4). An alternative model for Hoary-headed Grebe also included a negative term for phosphorus squared, with an even higher turning point of 40.9 mg/l (Table 4). However, the curves against phosphorus were almost linear, suggesting that the effect of phosphorus was positive over a wide range of concentrations.
Models explaining 10–20% of variance were also found for the third species of diving duck (Musk Duck), the most common species of dabbling duck (Chestnut Teal), the second commonest filter-feeding duck (Australasian Shoveler) and the second commonest grazing waterbird (Black Swan Cygnus atratus [Latham, 1790]) (Table 3). The best models for Musk Duck, Chestnut Teal and Australasian Shoveler included positive terms for ammonia and ammonia squared with turning points of 20–22 mg N/l in all cases (Table 4; Fig. 5). The model for Musk Duck also included a non-significant positive term for organic nitrogen, and the model for Australasian Shoveler included a positive term for organic nitrogen and a negative term for organic nitrogen squared (Table 3), with a turning point of 7.4 mg N/l for organic nitrogen (Table 4; Fig. 5). The best model for Black Swan was quite different, with positive terms for organic nitrogen and phosphorus but not ammonia (Table 3).
Weaker models explaining less than 10% of variance were found for most of the remaining species analysed, and they included positive terms for ammonia or nitrite (at least up to a certain turning point), and various relationships with other nutrients (generally positive with organic nitrogen and negative with nitrate) (Table 3). No significant models were found for Eurasian Coot Fulica atra Linnaeus, 1758. Two species (Australasian Grebe and Pacific Black Duck) showed negative relationships with ammonia, and they were the only species to do so. In addition, Australasian Grebes showed a weak negative relationship with organic nitrogen.
Quadratic relationships between effluent chemistry and waterfowl
When waterfowl species or guilds were examined in relation to single physical or chemical variables, quadratic relationships were revealed in 11 cases (Fig. 5). These implied optimum levels of nutrients (measured at the Borrie outlet) of 20–30 mg N/l for ammonia, 0.7 mg N/l for nitrite, 7.4 mg N/l for organic nitrogen, and 18–41 mg/l for phosphorus (Table 4). The values for ammonia and nitrite were remarkably consistent between species and guilds. The models implied that these species and guilds could tolerate a wide range of nutrient levels (up to 45–62 mg N/l for ammonia, 1.5–1.8 mg N/l for nitrite, 15 mg N/l for organic nitrogen and 37–83 mg/l for phosphorus). The minimum levels of these nutrients were close to zero in all cases, implying that pure water would support negligible densities of these birds.
Discussion
Effects of sewage management on waterfowl numbers
The EIP caused a redistribution of waterfowl at the WTP, with increases in ponds that received partially treated sewage (vs raw sewage) and decreases in ponds that were decommissioned from the treatment process (Loyn et al., 2014a). Many ponds would have received high levels of various chemicals including ammonia and sulphides before the EIP when they received raw sewage, reducing biological productivity. This accords with observations on smaller treatment plants in north-east Victoria, where the highest bird densities were found on winter-storage and maturation ponds at later stages in the treatment process (Murray et al., 2014). The observed decrease in the decommissioned Lake Borrie lagoon accords with the common observation that waterbird densities are higher on treatment plants than on many other wetlands (e.g. Murray et al., 2013; Breed et al., 2020), presumably because high nutrient levels increase productivity and provide a rich food resource for waterfowl.
This view was reinforced when partly treated sewage was restored to Lake Borrie lagoon and waterfowl numbers increased to levels comparable with the period before the EIP (Fig. 4). Feeding rates of waterfowl on Lake Borrie lagoon were also found to increase (Loyn et al., 2017). Two guilds (dabbling ducks and grazers) also increased in number on Other lagoons, and both these guilds included species that feed extensively away from ponds (Chestnut Teal from tidal mudflats; Pacific Black Duck and Australian Shelduck in nearby farmland). For most species (including all those that feed primarily from the treatment ponds), the increases on Lake Borrie lagoon were greater than changes observed on Other lagoons, in accord with our expectations. This hypothesis was strongly supported for total waterfowl, filter-feeding ducks, diving ducks, grebes and ten of 17 individual species, and similar patterns were observed for marsh terns. Hence it seems that the investment in the new pipeline to deliver carefully planned, moderate, nutrient concentrations achieved its objective for waterfowl habitat restoration. It also delivered the desired improvements to sewage treatment capacity, and it is an excellent example of integrated water management delivering multiple benefits.
Roles of nutrients
Studies of natural wetlands have shown variable results for the effects of nutrients and other variables on waterfowl numbers. Salinity was a key variable affecting waterfowl in 95 wetlands in southern Western Australia (Halse et al., 1993), and phosphorus had little effect; nitrogen was not assessed in that study. Murphy et al. (1984) found positive relationships between duck numbers and both phosphorus and nitrite in taiga ponds in central Alaska. Hansson et al. (2005) found that wetland birds were positively related to wetland surface area (up to ~ 5 ha) on 32 small recently constructed wetlands in southern Sweden, showing no clear relationship with nutrient status. Hamilton et al. (2005) and Murray et al. (2014) showed that waterfowl favoured intermediate levels of nutrients at wastewater treatment plants. Our results support that finding and provide a unique demonstration that major changes in nutrient status (reduction and subsequent restoration) can have direct effects on waterfowl abundance. Hence interventions of that sort can be highly effective in improving the habitat value of wastewater treatment plants (or potentially other artificial wetlands) for waterfowl by providing appropriate levels of nitrogen and phosphorus.
Sewage contains a cocktail of chemicals, including some that are beneficial for aquatic biota and others that are toxic. Some may be beneficial at low concentrations but toxic at high concentrations. Concentrations of chemical constituents are often correlated with each other (as observed in this study), adding to the difficulty of identifying which ones may have most influence on the biota of interest. As with all observational studies, influence in a statistical model does not necessarily imply causal effects.
The chemical measurements used in this study were made at the Borrie outlet, and can be expected to differ in some respects from those that applied in the treatment ponds at the time (probably with lower concentrations of total nitrogen, and higher concentrations of nitrate as the most oxidised form of nitrogen, as a result of the longer retention time). So, while the models show positive responses of waterfowl on Lake Borrie lagoon to ammonia or nitrite at the outlet, the chemistry driving the waterfowl response will be somewhat different in the ponds from that observed at the outlet.
Ammonia and nitrite both featured strongly in our models, showing positive responses up to turning points of ~ 20–29 mgN/l for ammonia or 0.7 mgN/l for nitrite (as measured at the outlet). These levels are higher than those generally tolerated by freshwater fish. Phosphorus and CBOD5 featured less often, also positively with a weak turning point identified for phosphorus at ~ 18 mg/l. Those turning points are all at the high end of the observed scale of concentrations at the outlet, so the general effects of those nutrients can be viewed as positive for waterfowl within the common prevailing range. The prominence of nitrite is somewhat surprising, as it is generally found at low concentrations, occurring as an intermediate product in the oxidation of ammonia or organic nitrogen to nitrate. However, previous modelling at the WTP also found that nitrite featured prominently, especially in models for diving ducks (Loyn et al., 2002). A possible explanation is that the level of nitrite reflects the productivity or turnover of biota such as algae, and the rate at which ammonia is oxidised to nitrate in the system. In view of the low concentrations, it is unlikely that nitrite per se has especially beneficial properties for waterfowl or their food supplies.
In terms of practical advice to managers, our models suggest that waterfowl will benefit from running the system to deliver high rather than low levels of ammonia, nitrite, phosphorus and CBOD5. Although optimum levels (turning points) were identified for these nutrients, they were found to be at the upper range of recorded levels and do not appear to be sharp, so for most species there is a wide range of nutrient levels that can provide good conditions, varying along the sequence of treatment ponds. Previous modelling also recognised the value of intermediate nutrient levels (Loyn et al., 2002, 2014c; Hamilton et al., 2005; Murray et al., 2014).
Possible mechanisms
Further work is needed to identify the chemical and biological pathways that may be involved. Invertebrates are a dominant food source for most ducks (including all the duck species that declined on Lake Borrie lagoon in Period B and increased again in Period C) as well as other waterbirds that showed that pattern (Hoary-headed Grebe and Whiskered Tern). Hence it is likely that the effects of nutrients were mediated through their effects on invertebrate abundance. Chironomid midges were often visibly abundant at Lake Borrie lagoon and other treatment ponds in summer and autumn (when adults emerged from aquatic larvae), though no quantitative surveys were undertaken. Oligochaete worms have been found to be abundant in the sludge in these ponds (Hamilton et al., 2005; Rogers et al., 2010).
Parallel studies to this have provided evidence that waterfowl may reduce populations of phytoplankton in Lake Borrie lagoon (unpublished data), perhaps contributing to rapid turnover and production of nitrite. It is likely that waterfowl play an active role along with other biota in the chemical processes of sewage treatment, with different guilds such as filter-feeders or diving ducks and grebes consuming phytoplankton or benthic invertebrates and stirring the surface waters or benthic sludge respectively.
Fish are generally considered scarce in treatment ponds, but some are known to have entered Lake Borrie lagoon and Other lagoons in 2005, when the adjacent Little River overflowed its banks (McGuckin, 2008, 2009). The comparatively low nutrient status of Lake Borrie lagoon at that time may have helped these fish populations thrive, perhaps reducing food supplies for birds through competition (Bouffard & Hanson, 1997; Potthoff et al., 2008; Stamation & Loyn, 2009). Reintroduction of treated effluent from 2015 may have reversed that process, to the detriment of fish but with positive benefits for waterbirds. Fish may be more sensitive to nutrients than waterbirds because they depend on the aquatic system for respiration as well as nutrition. Specialist fish-eating birds generally made little use of the WTP ponds for feeding (assuming that Hoary-headed Grebes were mainly taking invertebrate prey), though many cormorants (five species), Australian Darters Anhinga novaehollandiae (Gould, 1847), Australian Pelicans Pelecanus conspicillatus Temminck, 1824 and coastal species of tern feed commonly in tidal waters nearby, and cormorants, darters and pelicans use some of the ponds for roosting or nesting in dead trees (Loyn et al., 2014a).
Managers of wastewater treatment plants (and other wetlands) devote much effort to reducing levels of nitrogen in the outlets, as excessive amounts of nitrogen, phosphorus or other nutrients can cause serious pollution in freshwater or coastal ecosystems, with eutrophication producing algal blooms and reduced biodiversity (e.g. Smith & Schindler, 2009; Chislock et al., 2013; Dodds & Smith, 2016). However, this study confirms results of previous work showing that, within limits, nutrients including nitrogen enhance habitat for waterfowl on treatment plants, and this should be considered in managing those plants and fine-tuning requirements for discharge licences. If licence requirements are too onerous (demanding unnecessarily low levels of nutrients in the discharge), the negative effects on waterfowl may outweigh any benefits to the health of recipient waters. In the case of the WTP, this is not a trivial matter as the numbers of waterfowl involved are consistently large (up to 185,000) and include listed threatened species and Australian endemic waterfowl.
Conclusions
The study shows that replacing sewage effluent with treated water reduced the value of a major habitat and treatment lagoon for waterfowl (presumably by reducing invertebrate food), and returning partially treated sewage saw a corresponding resurgence of waterfowl to previous numbers. These effects were most marked for filter-feeding ducks, diving ducks, Hoary-headed Grebe and marsh terns (Chlidonias spp.), which feed largely on invertebrates. Models showed that suitable levels of nutrients such as nitrogen from sewage effluent can have a positive influence on waterfowl numbers and diversity at wastewater treatment plants. This is a rare demonstration of these effects (in both directions) at an operational scale.
Data availability
Data are held by Melbourne Water and the Arthur Rylah Institute for Environmental Research. Specific data can be provided on request.
Code availability (software application or custom code)
Not applicable.
References
Bouffard, S. H. & M. A. Hanson, 1997. Fish in waterfowl marshes: waterfowl managers’ perspective. Wildlife Society Bulletin 25: 146–157.
Breed, W. G., J. H. Hatch, C. Rogers, W. Brooker, A. C. Breed, M. H. K. Marklund, H. Roberts & M. F. Breed, 2020. Bolivar Wastewater Treatment Plant provides an important habitat for South Australian ducks and waders. Australian Field Ornithology 37: 190–199. https://doi.org/10.20938/afo37190199.
Breed, W. G., J. H. Hatch, C. Rogers, H. Roberts & I. Ahmer, 2023. Effects of rainfall in the arid zone on waterbird abundance at a wastewater treatment plant in coastal South Australia. Australian Field Ornithology. https://doi.org/10.20938/afo40000000.
Carlisle, T. J. & G. Mulamootil, 1991. Artificial wetlands for the treatment of stormwater. Canadian Water Resources Journal 16: 331–343. https://doi.org/10.4296/cwrj1604331.
Chambers, L. & R. H. Loyn, 2006. The influence of climate on numbers of three waterbird species in Western Port, Victoria, 1973–2002. Journal of International Biometeorology 50: 292–304.
Chislock, M. F., E. Doster, R. A. Zitomer & A. E. Wilson, 2013. Eutrophication: causes, consequences and controls in aquatic ecosystems. Nature Education Knowledge 4: 10.
Clarke, R. H., A. Herrod, R. H. Loyn, M. J. Carter, A. Silcocks, P. Menkhorst & C. Johnstone, 2015. Waterbird Fluctuations in Response to Murray-Darling Basin Streamflow and Rainfall. Report prepared for Melbourne Water by Monash University, Clayton, Victoria.
Conley, D. J., H. W. Paeri, R. W. Howarth, D. F. Boesch, S. P. Seitzinger, K. E. Havens, C. Lancelot & G. E. Likens, 2009. Controlling eutrophication: nitrogen and phosphorus. Science 323: 1014–1015.
DELWP, 2018. Port Phillip Bay (Western Shoreline) and Bellarine Peninsula Ramsar Site Management Plan. Department of Environment, Land, Water and Planning, Melbourne.
Dodds, W. K. & V. H. Smith, 2016. Nitrogen, phosphorus and eutrophication in streams. Inland Waters 6: 155–164.
Finlayson, C. M., G. T. Davies, W. R. Moomaw, G. L. Chmura, S. M. Natali, J. E. Perry, N. Roulet & A. E. Sutton-Grier, 2019. The Second Warning to Humanity – providing a context for wetland management and policy. Wetlands 39: 1–5. https://doi.org/10.1007/s13157-018-1064-z.
Fisher, R., G. R. Shiell, R. J. Sadler, K. Inostroza, G. Shedrawi, T. H. Holmes & J. M., McGree, 2019. ePower: an R package for power analysis in Before-After-Control-Impact (BACI) designs. Methods in Ecology and Evolution 10: 1843–1853. https://doi.org/10.1111/2041-210X.13287.
Fjeldsa, J. F., 1988. Comparative ecology of the Australasian grebes (Aves: Podicepidae). Royal Australasian Ornithologists Union Report, 30.
Foote, A. L., S. Pandey & N. T. Krogman, 1996. Processes of wetland loss in India. Environmental Conservation 23: 45–54.
Frith, H., 1982. Waterfowl in Australia, 2nd ed. Angus & Robertson, Sydney:
Fuller, R. J. & D. E. Glue, 1980. Sewage works as bird habitat in Britain. Biological Conservation 17: 165–181. https://doi.org/10.1016/0006-3207(80)90054-3.
Gordon, L. J., C. M. Finlayson & M. Falkenmark, 2010. Managing water in agriculture for food production and other ecosystem services. Agricultural Water Management 97: 512–519.
Gosper, D. G., S. V. Briggs & S. M. Carpenter, 1983. Waterbird dynamics in the Richmond Valley, New South Wales, 1974–77. Australian Wildlife Research 10: 319–327.
Guay, P.-J., 2013. Activity Budget Analysis of Waterfowl at the WTP. Unpublished report, School of Engineering and Science, Victoria University, Melbourne.
Hale, J., 2010. Ecological Character Description of the Port Phillip Bay (Western Shoreline) and Bellarine Peninsula Ramsar Site. Report prepared for the Department of Environment, Water, Heritage and the Arts, Canberra.
Halse, S. A., M. R. Williams, R. P. Jaensch & J. A. K. Lane, 1993. Wetland characteristics and waterbird use of wetlands in south-western Australia. Wildlife Research 20: 103–126.
Hamilton, A., I. Taylor & G. Hepworth, 2002. Activity budgets of waterfowl (Anatidae) on a waste-stabilisation pond. Emu 102: 171–179.
Hamilton, A. J., W. Robinson, I. R. Taylor & B. P. Wilson, 2005. The ecology of sewage treatment gradients in relation to their use by waterbirds. Hydrobiologia 534: 91–108. https://doi.org/10.1007/s10750-004-1415-z.
Hansson, L.-A., C. Bronmark, P. A. Nilsson & K. Abjornsson, 2005. Conflicting demands on wetland ecosystem services: nutrient retention, biodiversity or both? Freshwater Biology 50: 705–714. https://doi.org/10.1111/j.1365-2427.2005.01352.x.
Higgins, P. J. & S. J. J. F. Davies (eds), 1996. Handbook of Australian, New Zealand and Antarctic Birds. Volume 3 - Snipe to Pigeons. Oxford University Press, Melbourne.
Kingsford, R. T., 2000. Ecological impacts of dams, water diversions and river management on floodplain wetlands in Australia. Austral Ecology 25: 109–127.
Kingsford, R. T. & F. I. Norman, 2002. Australian waterbirds–products of the continent’s ecology. Emu 102: 47–69. https://doi.org/10.1071/MU01030.
Lane, B. A. & P. Peake, 1990. Nature Conservation at the Werribee Treatment Complex. Environment Series. No. 91/008, Melbourne and Metropolitan Board of Works, Melbourne.
Loyn, R. H., 1989. The management of duck hunting in Victoria – a review. Arthur Rylah Institute for Environmental Research. Technical Report Series 256. Department of Sustainability and Environment, Heidelberg, Victoria.
Loyn, R. H., E. S. G. Schreiber, R. J. Swindley, K. Saunders & B. A. Lane, 2002. Use of Sewage Treatment Lagoons by Waterfowl at the Western Treatment Plant – An Overview. Report prepared for Melbourne Water by the Arthur Rylah Institute in association with Brett Lane & Associates Pty Ltd and Water ECOscience.
Loyn, R. H., D. I. Rogers, R. J. Swindley, K. Stamation, P. Macak & P. Menkhorst, 2014a. Waterbird monitoring at the Western Treatment Plant, 2000–12: the effects of climate and sewage treatment processes on waterbird populations. Arthur Rylah Institute for Environmental Research. Technical Report Series 256. Department of Sustainability and Environment, Heidelberg, Victoria.
Loyn, R. H., R. J. Swindley & K. Stamation, 2014b. Waste water not wasted: the Western Treatment Plant as a habitat for waterfowl. Victorian Naturalist 131: 146–149.
Loyn, R. H., J. Potts, D. Duncan, K. Stamation & P. Menkhorst, 2014c. Relationships between waterfowl numbers and water chemistry of ponds at the Western Treatment Plant, Victoria. Arthur Rylah Institute for Environmental Research. Technical Report Series 260. Department of Sustainability and Environment, Heidelberg, Victoria.
Loyn, R. H., G. Dutson, J. Davies, G. Hepworth & D. I. Rogers, 2017. Waterfowl Activity at Lake Borrie (Western Treatment Plant) in 2017: Assessing whether Management Interventions have helped Restore the Value of Pond 9 as Foraging Habitat for Waterfowl. Report prepared for Melbourne Water by Eco Insights, Melbourne.
Marchant, S. & P. J. Higgins (eds), 1990. Handbook of Australian, New Zealand and Antarctic birds. Ratites to Ducks. Vol. 1. Oxford University Press, Melbourne.
Maxson, G.-A., 1981. Waterfowl use of a municipal sewage lagoon. The Prairie Naturalist 13: 1–12.
McGuckin, J., 2008. A fish survey of the western treatment plant. Report prepared for Melbourne Water by Streamline Research Pty Ltd, Melbourne.
McGuckin, J., 2009. A fish survey of the Lake Borrie ponds and Little River estuary. Report prepared for Melbourne Water by Streamline Research Pty Ltd, Melbourne.
Melbourne Water, 2002a. Western Treatment Plant Environment Improvement Project (Post-effluent Reuse Stage 2). EPBC Act Referral to Environment Australia, Canberra, prepared by Melbourne Water, Melbourne.
Melbourne Water, 2002b. Preliminary Documentation—EPBC Referral 2002/688: Environment Improvement Project (Post Effluent Reuse Stage 2) Western Treatment Plant, Werribee, Victoria, Melbourne Water submission to Environment Australia, Canberra.
Murphy, S. M., B. Kessel & L. J. Vining, 1984. Waterfowl populations and limnological characteristics of taiga ponds. Journal of Wildlife Management 48: 1156–1163.
Murray, C. & A. Hamilton, 2010. Perspectives on wastewater treatment wetlands and waterbird conservation. Journal of Applied Ecology 47: 976–985.
Murray, C. G., S. Kasel, R. H. Loyn, G. Hepworth & A. J. Hamilton, 2013. Waterbird use of artificial wetlands in an Australian urban landscape. Hydrobiologia 736: 131–146. https://doi.org/10.1007/s10750-013-1558-x.
Murray, C. G., S. Kasel, E. Szantyr, R. Barratt & A. J. Hamilton, 2014. Waterbird use of different treatment stages in waste-stabilisation pond systems. Emu - Austral Ornithology 114: 30–40.
Mustoe, S., 2009. Waterfowl activity budgets at Lake Borrie Pond 9, Western Treatment Plant - Comparison between years. Report prepared for Melbourne Water by Applied Ecology Solutions Pty Ltd, Melbourne.
Mustoe, S. & N. Waugh, 2006. Waterfowl activity budgets at Lake Borrie Pond 9, Western Treatment Plant. Report prepared for Melbourne Water by Applied Ecology Solutions Pty Ltd, Melbourne.
Potthoff, A. J., B. R. Herwig, M. A. Hanson, K. D. Zimmer, M. G. Butler, J. R. Reed, B. G. Parsons & M. C. Ward, 2008. Cascading food-web effects of piscivore introduction in shallow lakes. Journal of Applied Ecology 45: 1170–1179.
Rogers, D. I., R. H. Loyn, B. Cumbo, P. Papas & W. K. Steele, 2010. Abundance of benthos in non-tidal ponds of the Western Treatment Plant, Werribee, and implications for shorebird management. Arthur Rylah Institute for Environmental Research, Heidelberg, Victoria.
Roshier, D., A. Robertson & R. Kingsford, 2002. Responses of waterbirds to flooding in an arid region of Australia and implications for conservation. Biological Conservation 106: 399–411.
Schindler, D. W., 2006. Recent advances in the understanding and management of eutrophication. Limnology and Oceanography 51: 356–363.
Smith, V. H. & D. W. Schindler, 2009. Eutrophication science: where do we go from here? Trends in Ecology and Evolution 24: 201–207.
Smith, V. H., S. B. Joye & R. W. Howarth, 2006. Eutrophication of freshwater and marine ecosystems. Limnology and Oceanography 51: 351–355.
Sokal, R. R. & F. J. Rohlf, 1995. Biometry – the Principles and Practice of Statistics in Biological Research, 3rd ed. Freeman, New York: 665.
Stamation, K. & R. Loyn, 2009. Impact of recreational access and stocking with fish on waterbirds and shorebirds – Devilbend Reservoir. Arthur Rylah Institute client report for Parks Victoria.
Steele, W. K., A. J. Hamilton, I. R. Taylor & R. H. Loyn, 2006. Balancing wastewater treatment and waterbird conservation at a major sewage treatment plant. Proceedings of the 5th World Water Congress, 10–14 September 2006, Beijing, China. International Water Association.
Underwood, A. J., 1994. On beyond BACI– Sampling designs that might reliably detect environmental disturbances. Ecological Applications 4: 3–15. https://doi.org/10.2307/1942110.
Wetlands International, 2020. Waterbird population estimates. [available on internet at www.wpe.wetlands]. Accessed 22 September 2020.
Acknowledgements
The programs described here were initiated and funded by Melbourne Water (MW). The authors are grateful to many MW staff for their support and advice including Brad McLean, Trevor Gulovsen, Peter Nilon, Ben Pratt and Kevin Gillett. Dr Judy Blackbeard (MW) and Dr Dave Ramsey (ARI) made valuable comments on a draft. Members of the WTP Biodiversity Conservation Advisory Committee (BCAC) provided strong support at all times: special thanks to John Barkla (chair 1985-2021) and the late Dr Clive Minton for their support and valuable advice over >20 years. The task of counting thousands of waterfowl was undertaken mainly by Bob Swindley (2000–2015) and Dr Danny Rogers and Jeff Davies (2014–2020), with occasional help from others including Maarten Hulzebosch and Dr Ian Norman. Dr Stuart Halse and anonymous referees made valuable suggestions to improve the manuscript.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. The project was funded by Melbourne Water.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Three authors, as indicated above, are Melbourne Water staff, and the others were employed by the Arthur Rylah Institute for Environmental Research, which received funding from Melbourne Water to undertake this study.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors have consented to submitting this paper for publication.
Additional information
Handling editor: Stuart Halse
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Loyn, R.H., Rogers, D.I., Swindley, R.J. et al. Waterfowl populations decline with nutrient reduction and increase with nutrient restoration: 20 years of adaptive management at a Ramsar-listed wastewater treatment plant. Hydrobiologia 850, 4127–4147 (2023). https://doi.org/10.1007/s10750-023-05265-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-023-05265-9