Abstract
According to the principles of community ecology, sympatric species may suffer a selective pressure to decrease their niche overlap through mechanisms of niche partitioning. However, there is still a gap in knowledge of the main drivers influencing niche differentiation, particularly in communities composed by small-sized and inconspicuous species. The coastal epipelagic community structure of mid-trophic level species in the Northeast Atlantic was examined using stable isotope analysis to (1) decipher intrinsic and extrinsic factors promoting niche partitioning and (2) examine species divergence in the use of resources. A total of 474 individuals from a guild of 11 species were sampled during three years, and the influence of potential drivers of isotopic divergence was assessed. The niche partitioning was mainly determined by the taxonomic group in accordance with the fundamental principles of niche theory, which was consistent for both δ13C (i.e. habitat) and δ15N (i.e. prey). Body size was highly important in driving niche partitioning within species. Our results show that the predominant species in the guild were the most generalist, when compared with more specialised species with which they coexist. This study reveals that the intrinsic factors are determinant to decrease inter- and intra-specific niche overlap at the community level.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding the main drivers of niche partitioning among species, particularly when species belong to the same guild, i.e. share similar traits and habitat characteristics, and how their trophic interactions may evolve through time has been a central theme in community ecology (Chesson, 2000). The fundamental principles of niche theory suggest that absolute concurrent niche overlap is not evolutionarily possible (Hardin, 1960), and sympatric species must partition their limiting resources to alleviate interspecific competition (Petalas et al., 2021).
Since Darwin’s theory on the ‘The Origin of Species’, niche partitioning (also termed niche differentiation or niche segregation) was conjectured in an array of studies across a wide range of taxa, and there is a vast number of studies using models and assumptions on the maintenance of species diversity (Chesson, 2000). Niche segregation between sympatric species has been observed in several communities in the marine environment, from apex predators (e.g. Connan et al., 2019) to plankton (e.g. Williams, 1988), through small epipelagic and mesopelagic fish (e.g. Cherel et al., 2010), demersal fish assemblages (e.g. Colloca et al., 2010) or sympatric cephalopods (e.g. Golikov et al., 2020). In this sense, the long-ago existing theory of niche partitioning to decrease overlap among sympatric species is corroborated by an array of studies in the field and empirical models. However, the thorough investigation of the main drivers influencing niche differentiation in natural populations has been overlooked (Svanbäck & Bolnick, 2007). Some studies focussed on the factors influencing dietary differentiation in fish (mostly based on prey selection) using conventional and modern methodologies (e.g. Amundsen et al., 2003; Helland et al., 2008; Quevedo et al., 2009; Colloca et al., 2010; Schulze et al., 2012; Knickle and Rose 2014), but these are limited to the identification of specific drivers in one species or in the interaction of a few species and often in controlled experiments. The use of modern techniques such as biochemical analyses and robust statistical approaches can significantly increase our knowledge on the ecological niche and niche overlap (Swanson et al., 2014). Recent studies have demonstrated that both spatial (e.g. Jessopp et al., 2020) and temporal (e.g. Watabe et al., 2022) variations drive niche partitioning in natural populations. This is of main interest for the scientific community in the field of ecology, especially considering elusive and ecologically important communities composed by small-sized and very abundant organisms, such as those including mid-trophic level species in the marine realm (Brandl et al., 2020). In fact, small pelagic mid-trophic level species are important because they play a major role in neritic ecosystems as consumers and prey. Due to their high abundance, they are key species occupying intermediate trophic levels with both bottom-up and top-down control roles (Bode et al., 2007), and excellent candidates to better understand the drivers of niche differentiation. In highly productive marine ecosystems, such as shelf waters of the temperate North Atlantic influenced by upwelling regimes, trophic interactions are typically controlled by this guild of forage fish (i.e. “wasp-waist” control; Cury et al., 2000).
Sympatric fish species may partition habitat and resources in many ways (Ross, 1986), and this niche partitioning flexibility depends on the type of specialization of different species (Schulze et al., 2012). The trade-off between specialist vs. generalist species (i.e. their relative abundances in the communities and respective partition of resources) is thought to represent a central mechanism of co-existence (Wilson & Yoshimura, 1994). Thus, the width of ecological niche and how it segregates plays a central role in species adaptation to the marine environment and in trophic relationships between ecologically related species (e.g. interspecific competition). However, this also occurs among individuals of the same species (e.g. intra-specific competition) through niche segregation induced by sex, morphology (e.g. size), age (e.g. ontogenetic stages), or individual specialization (Van Valen, 1965; Bolnick et al., 2002; Amundsen et al., 2003). In fact, the combined effect of extrinsic and intrinsic factors may explain the partitioning of resources between and within species (Schoener, 1974).
Small mid-trophic level species such as sardines, anchovies, mackerels, and squids are among the most common pelagic species in upwelling systems worldwide, such as in the west Iberian coast (Bode et al., 2007). There is a relatively good knowledge of these species from a commercial use perspective (Leitão et al., 2014), and in terms of diet (Garrido et al., 2008, 2015; Garrido & Murta, 2011; Costalago et al., 2012). However, their trophic relationships within the communities and niche occupancy from an ecological point of view are less understood (Bode et al., 2007; da Silva et al., 2020, 2022). In this context, recent biochemical methods, such as analyses of stable isotopes, are relevant tools to investigate the trophic relationships of co-occurring species with a high degree of ecological similarity (da Silva et al., 2020, 2022; Golikov et al., 2020), thus presenting great potential for deciphering food web structure and function in marine systems (Ceia et al., 2022).
Stable isotope analysis bypassed some limitations of the conventional approaches to investigate niche segregation. This method is used routinely at low costs, allowing the analysis of a large number of samples and it has a great potential to study the trophic ecology of populations and interactions within communities (Newsome et al., 2007; Ceia et al., 2022). This method also has the advantage of investigating the feeding ecology of organisms over the short- to the long-term, depending on the tissue sampled (Ceia et al., 2012). Different tissues have different isotopic turnover rates: in fish the isotopic signature of muscle is representative of the isotopic niche during the previous weeks (half-lives in the order of 2–8 weeks) preceding sampling (Boecklen et al., 2011). Thus, this is a powerful method to estimate trophic niches and overcomes the limitations of the traditional approaches (e.g. stomach analyses, direct observations). Such conventional techniques typically indicate a ‘snapshot’ of what the individual consumed recently, and may not be suitable to study a large number of elusive organisms such as small fish. However, stable isotopes rarely provide diet information at a high taxonomic resolution, i.e. to the species level (Ceia et al., 2022).
In marine systems, stable isotopes are frequently used to assess species isotopic niches (i.e. a proxy of the ecological niche), reflecting the scenopoetic (i.e. habitat; δ13C) and bionomic (i.e. prey; δ15N) axes in the multivariate Hutchinson’s space ecological niche (Hutchinson, 1957; see reviews Newsome et al., 2007; Swanson et al., 2014). There is a natural variation in stable isotope values of consumers exploiting different resources and inhabiting different ecosystems, and such variation is related to the isotopic composition of their prey and respective habitats. This variation is reflected from the base of the food web to the consumer levels, and may occur at spatial, temporal, and/or trophic dimensions (Navarro et al., 2013). Differences of isotopic composition are often linked to environmental drivers such as temperature, chlorophyll a concentration, or bathymetry (Ceia et al., 2018). In addition to these extrinsic drivers of isotopic variance, intrinsic factors, such as body mass and size, can affect the ecological role of organisms within and between species, and thus influence their isotopic niches (Colloca et al., 2010). Since mid-trophic level species commonly cluster in dense shoals, sharing similar habitats and food sources, and reflecting directly baseline variations in isotope values, a good model of the community can be achieved by analysing its’ structure and inter-specific connections (Cherel et al., 2010). This provides an excellent opportunity to study the most important drivers of niche divergence for mid-trophic level communities within complex marine food webs.
In this study, we determined stable isotopes content to investigate niche partitioning within a community of 11 co-occurring small pelagic fish and squid species in the North Atlantic. We have selected a set of potential drivers of isotopic composition, including taxonomic group (e.g. species, genus), spatial (e.g. latitudinal, distance to coast), temporal (e.g. year, season), environmental (e.g. chlorophyll a concentration, sea surface temperature) and intrinsic (e.g. size, body mass) predictors of such composition, to report the main drivers of resource partitioning. The main objectives of this study are to (1) assess the most important drivers promoting trophic dissimilarities, from the community to species level and (2) examine species divergence in the use of resources. While reporting niche partitioning related with any of the selected potential drivers, we expect to observe segregation of isotopic niches and variance in niche width of different species in accordance with the fundamental principles of niche theory (Hardin, 1960).
Materials and methods
Study area and sampling
The Portuguese continental coast, in the northeast Atlantic, is a relatively large continental shelf system characterized by shallow waters (< 200 m), through a coastal extension of around 800 km (Fig. 1). It has high productivity, especially in the north-western coast of Portugal, which is influenced by winter-intensified runoff of several rivers (Santos et al., 2004) and coastal upwelling regimes that last from April to September, although more intense in summer (i.e. from July to September) (Fiuza et al., 1982; Garrido et al., 2008; Sousa et al., 2008).
Fishes (nine species) and cephalopods (two species) were collected along the west and south Iberia (Portuguese coast), during 2014, 2016 and 2017 (Fig. 1), by the research vessel (RV) “Noruega” of the Portuguese Institute for the Sea and Atmosphere (Massé et al., 2018). Two seasonal oceanographic surveys were conducted each year to sample pelagic mid-trophic level species during spring and autumn (see Supplementary Material Table S1). Specimens were captured with trawl hauls, and information on date, time, position of capture (latitude and longitude, and respective distance to coast) and depth (m) were recorded. A total of 59 transects were performed parallel to the coast, during 47 pelagic and 12 deep trawls. All trawls were performed between 11 and 169 m depth within the continental shelf.
A total of 474 individuals from 11 species were sampled (Table 1): European pilchard Sardina pilchardus (Walbaum, 1792) (Family: Clupeidae), European anchovy Engraulis encrasicolus (Linnaeus, 1758) (Engraulidae), Atlantic horse mackerel Trachurus trachurus (Linnaeus, 1758) (Carangidae), Blue jack mackerel Trachurus picturatus (Bowdich, 1825) (Carangidae), Mediterranean horse mackerel Trachurus mediterraneus (Steindachner, 1868) (Carangidae), Atlantic mackerel Scomber scombrus Linnaeus, 1758 (Scombridae), Atlantic chub mackerel Scomber colias Gmelin, 1789 (Scombridae), Bogue Boops boops (Linnaeus, 1758) (Sparidae), European hake Merluccius merluccius (Linnaeus, 1758) (Merlucciidae), European squid Loligo vulgaris Lamarck, 1798 (Loliginidae) and Alloteuthis spp. Wülker, 1920 (Loliginidae). For each fishing event, five individuals of each species were collected, whenever possible, and immediately frozen at − 20 °C. We focussed on small and medium-sized specimens (very small and large individuals were not collected) in order to standardize sampling for a more consistent and robust comparisons of niche widths among species.
The body mass (to 0.01 g) and total length (in fishes; to 1 mm) or mantle length (in squids; to 1 mm) were recorded for each individual (Table 1). Total length of fish specimens ranged from 6.8 to 33.1 cm and mantle length of squids from 3.2 to 9.8 cm. The body mass varied between 2.4 and 264.3 g in fish, and between 1.1 and 25.4 g in cephalopods.
Stable isotope analyses (SIA)
Stable isotopes of carbon (δ13C) and nitrogen (δ15N) were analysed in the muscle of each individual (n = 474). Muscle tissue was grounded to fine powder in a mill (Mixed Mill MM 400 (Retsch, Haan, Germany)). Lipids were removed from samples by successive rinses in a 2:1 chloroform–methanol solution (Cherel et al., 2005) and dried at 50 °C for 48 h. The average C:N mass ratios of the delipidated samples were 3.1 ± 0.2 (mean ± SD), thus below the 3.5 threshold value that corresponds to low lipid concentrations in tissues (Post et al., 2007). However, individual values ranged from 2.8 to 4.1. Hence, we included C:N mass ratio as an intrinsic explanatory variable (see Supplementary Material Table S2) to check for potential effects of remaining lipids on δ13C and δ15N values, as there are evidences that variation in lipid content has the potential to introduce bias into stable isotope values, particularly δ13C (Cherel et al., 2005; Post et al., 2007).
Stable isotopes were performed on approximately 0.3 mg of homogenized muscle tissue by loading it into tin cups and combusting it at 1,800 °C in a Flash EA1112 Series elemental analyser (Thermo Italy, Rhodano, Italy) coupled on line via a Finnigan conflo II interface to a Thermo Delta V mass spectrometer (Bremen, Germany). Isotope ratios are presented in the usual δ notation based on the Vienna-PeeDee Belemnite (V-PDB) for carbon and atmospheric N2 (AIR) for nitrogen and expressed as ‰. δ13C or δ15N = [(Rsample/Rstandard) − 1], where R = 13C/12C or 15N/14N, respectively. Replicate measurements of internal laboratory standards (acetanilide STD, Thermo scientific-PN 33836700) in every batch indicate precision < 0.2 ‰ for both δ13C and δ15N.
Data analyses
A set of 17 predictor variables (see Supplementary Material Table S2), including three taxonomic group categories (species, genus and order), three spatial (latitude, distance to coast and depth), three environmental (chlorophyll a concentration, Chl a; sea surface temperature, SST; and type of trawl, i.e. pelagic or demersal), three temporal (year, season and continuous time) and five intrinsic variables (total length, body mass, body mass index, lipid content and sample size), were selected to identify the most important drivers promoting isotopic differences at the community and species level. Three different taxonomic group categories were chosen to test the influence of taxon in resource partitioning because closely related species typically show similar morphological, physiological and behavioural characteristics. Monthly composites of remote sensing data (i.e. Chl a and SST), from the corresponding months of capture between 2014 and 2017 (see Supplementary Material Table S2), were used in the model to test the effect of these environmental variables on isotopic niche of individuals. The type of trawl was used as a potential proxy of habitat use in the water column, noticing however that most fish species can move vertically and the type of trawl does not necessarily correspond to the vertical position of the shoals. Universal (i.e. continuous) time was chosen in complement to categorical year and season variables to evaluate potential variations of niche partitioning throughout time. The standard residuals of the relationship between body mass and total length within each species were used to estimate body mass index, an index primarily reflecting body condition of individuals. Sample size was included as an intrinsic variable to control for the potential effect of different sampling among species in the community structure.
Random Forest modelling was used to evaluate which variables should be more prone to drive niche partitioning among and within species (i.e. in both δ13C and δ15N values). Only appropriate explanatory variables were included in each model. We ran models for the pooled data (i.e. the community) and also separately for the five most representative species (i.e. S. pilchardus, E. encrasicolus, T. trachurus, S. scombrus and S. colias). Other species were in insufficient number to run models independently. Random Forest is a classification technique based on regression trees, which is able of modelling and showing a consensus prediction of the response variable from a set of explanatory variables (Breiman, 2001). The number of trees needs to be sufficiently high, therefore a total of 5,000 trees were performed for each model. Random Forest models handle quite well-correlated/redundant variables, having the flexibility in capturing non-linear relationships. Overall, this method attributes a rank of importance of each variable for each model performed, enabling the evaluation of the most important drivers of niche divergence, considering the 17 above nominated explanatory variables. These analyses were performed using Statistica 10.0.
Stable isotope data were analysed in the context of isotopic niche width using seven isotopic metrics accounting for variability between individuals. Three isotopic richness metrics (i.e. accounting for the amount of isotopic space filled by a group of organisms) were estimated using a Bayesian framework (Stable Isotope Bayesian Ellipses in R: SIBER; Jackson et al., 2011). The area of the standard ellipse (SEAc, an ellipse obtained by Bayesian inference containing 40% of the data regardless of sample size and corrected for small sample sizes) was adopted to compare niche width and overlap between species. The overlap in isotopic niches between species ranges from 0 to 1, where 0.00–0.29 indicate no overlap, 0.30–0.60 a medium overlap, and 0.61–1.00 a large overlap and indicating potential competition (Langton, 1982; Golikov et al., 2020). Bayesian standard ellipse areas (SEAB) were calculated from 10,000 iterations of Markov-chain Monte Carlo (MCMC) simulation using rjags to test for differences in niche widths among species (i.e. the proportion of draws of the posterior distribution of the SEAB in which the area of one species was smaller than that of other species). The Layman metric of convex hull area (TA; Layman et al., 2007, also termed isotopic richness, i.e. IRic; Cucherousset & Villéger, 2015) was calculated as a measure of total isotopic niche area. Since TA (or IRic) is influenced by the number of individuals due to the increasing probability of having extreme values and therefore higher convex hulls, a bootstrapping approach, using 10 samples per species, was applied (Cucherousset & Villéger, 2015). Additionally, four isotopic diversity metrics (i.e. accounting for the distribution of points within the convex hull) complementary to the above stated isotopic richness metrics were estimated following Cucherousset and Villéger (2015). These metrics included isotopic divergence (IDiv), dispersion (IDis), evenness (IEve) and uniqueness (IUni), and are independent of the number of individuals analysed. All these metrics range from 0 to 1 and quantify different components of isotopic diversity among individuals within species (see Cucherousset & Villéger, 2015). Succinctly, IDiv is close to 0 when most individuals are close to the centre of gravity of the convex hull. IDis equals 0 when all organisms have the same stable isotope values. IEve tends to 0 when most individuals concentrate within a small region of convex hull, while a few others are far from this cluster. IUni tends to 0 when each organism has at least one other organism with the same stable isotope values. All isotopic metrics were calculated in R 4.0.3 (R Core Team, 2020) by adapting the computational codes supplied by Jackson et al. (2011) and Cucherousset and Villéger (2015). A principal component analysis (PCA) was then run considering six isotopic metrics: SEAc, IRic (bootstrapped), IDiv, IDis, IEve and IUni, to help in the assessment of species divergence in the use of resources and identification of ecological grouping based on isotopic richness and isotopic diversity. Differences in SIA were assessed among species (11), genus (8) and orders (4), using an ANOVA and followed by Fisher's Least Significant Difference test for multiple comparisons between species. Data were tested for normality (Shapiro–Wilk test) and homogeneity of variances (Levene's test).
Results
Random forest models and predictor variables
The two models reflecting scenopoetic (i.e. habitat; δ13C, Fig. 2A) and bionomic (i.e. prey; δ15N, Fig. 2B) components of niche width showed similar outputs in the identification of the main predictors as elementary taxonomic groups (i.e. species followed by genus). This clearly suggests that taxa (at the lowest taxonomic level) are the most important driver of trophic dissimilarities within the community. Overall, environmental variables (SST and Chl a), universal time and latitude showed a moderate influence on both δ13C and δ15N values. However, the remaining spatial (distance to coast and depth), and temporal (year and season) variables had lower importance, according to the same models. The sample size and C:N mass ratio had little influence on both models, indicating, respectively, that these outputs were not influenced by different number of individuals per species nor by the lipid content that may have remained in the samples after delipidation.
Considering each species separately, Random Forest models identified, in general, the intrinsic biometric variables (i.e. total length and body mass) as the main predictors of both δ13C and δ15N values (Supplementary Material Fig. S1). This suggests that, at the species level, the size of organisms has an important influence on the resource partitioning of the populations. However, this pattern was not always observed for the five species considered in these analyses. Specifically, environmental (SST and Chl a) variables, and, sporadically, spatial (latitude and depth) and temporal variables (time, year) showed strong and/or moderate influence on δ13C (for S. scombrus and S. colias) and δ15N values (for S. pilchardus, E. encrasicolus, T. trachurus and S. colias) (Supplementary Material Fig. S1).
Trophic ecology and isotopic niche
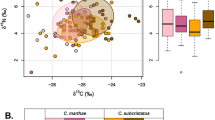
Overall, the isotopic niche space of this community of mid-trophic level organisms ranged from -20.5 to -16.1 ‰ in δ13C, and from 8.5 to 13.5 ‰ in δ15N values (Table 1; Fig. 3A). Species differed in both δ13C (ANOVA: F10,463 = 21.8, P < 0.001; Alloteuthis spp. > L. vulgaris > B. boops > M. merluccius > T. trachurus > S. pilchardus > T. mediterraneus > T. picturatus > S. scombrus > S. colias > E. encrasicolus) and δ15N values (ANOVA: F10,463 = 38.4, P < 0.001; T. trachurus > T. mediterraneus > M. merluccius > B. boops > S. scombrus > T. picturatus > E. encrasicolus > S. pilchardus > Alloteuthis spp. > S. colias > L. vulgaris) (see Supplementary Material Table S3 for multiple comparisons between species). Muscle δ13C and δ15N values also differed among genus (ANOVA: δ13C, F7,466 = 25.7, P < 0.001; δ15N, F7,466 = 42.0, P < 0.001; Supplementary Material Fig. S2) and orders (ANOVA: δ13C, F3,470 = 12.0, P < 0.001; δ15N, F3,470 = 44.6, P < 0.001; Supplementary Material Fig. S3).
(A) Individual muscle δ13C and δ15N values and the corresponding standard ellipses (SEAc, 40%) and (B) standard ellipse areas (SEAb, 50%, 75% and 95% credible intervals) estimated with a Bayesian framework from 11 fish and squid species from the western Iberian shelf. Crosses in (B) represent the area of the standard ellipse (SEAc)
Although the bulk of species displayed their isotopic niches at around an overall mean (and median) of -17.8 ‰ in δ13C and 11.0 ‰ in δ15N, SIBER outputs showed a general segregation based on SEAc (Fig. 3A). The overlap between species was generally low or medium, and there was no overlap in numerous instances (see Supplementary Material Table S4 for overlap between species SEAc). A large overlap only occurred between: T. mediterraneus, M. merluccius and B. boops with T. trachurus; S. scombrus with T. picturatus; and B. boops with M. merluccius. The niche width also varied among species, with S. colias presenting a significantly higher niche width than all the other species (Fig. 3B). On the other hand, L. vulgaris showed a significantly smaller niche width than S. pilchardus, T. trachurus, S. colias, E. encrasicolus and Alloteuthis spp.
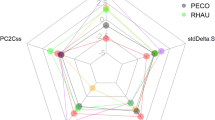
Isotopic diversity metrics indicated that all species exhibited relatively similar isotopic divergence (min.– max.: 0.70–0.79), moderately variable dispersion (0.35–0.64) and evenness (0.68–0.85), and relatively high variable isotopic uniqueness (0.18–0.70) (Table 2). The PCA did not highlight any structuring of the 11 species into taxonomic groups (Fig. 4 and Supplementary Material Fig. S4); in fact, species belonging to the same genus, such as Trachurus spp. and Scomber spp., grouped in different clusters. On the other hand, the most abundant species from the same trawls, i.e. S. pilchardus, E. encrasicolus, T. trachurus and S. colias, clustered together, sharing high isotopic richness and moderate isotopic diversity, denoting almost similar foraging strategies.
Discussion
Although the isotopic divergence and niche partitioning among species can be driven by spatial, temporal and/or environmental factors, this study illustrates that trophic dissimilarities within a community of marine mid-trophic level species are mostly determined by the taxonomic group, in accordance with the fundamental principles of niche theory (Hardin, 1960). Furthermore, when considering species-specific isotopic niches, intrinsic variables such as the size of organisms must be carefully evaluated because these are important drivers of niche partitioning within species, a trait which helps to reduce competition between conspecifics (Van Valen, 1965). Ultimately, the combined effect of both intrinsic and extrinsic factors is the key to explain partitioning of resources in ecological communities, as suggested by Schoener (1974).
Drivers of niche partitioning
Species can segregate along both the spatial and temporal dimensions of their ecological niche (e.g. Jessopp et al., 2020; Watabe et al., 2022), but those belonging to the same guild can pursue specific microhabitats and foods as the principal factors that regulate the structure of the community (Ross, 1986; Moreno & Castro, 1995; Brandl et al., 2020; Colloca et al., 2010). The feeding and foraging preferences of different taxa were potentially driven by specific feeding capabilities and behavioural traits (see below), supported by evidence that species followed by genus were the most important drivers of niche partitioning within the community. This was consistent for both scenopoetic (i.e. habitat; δ13C) and bionomic (i.e. prey; δ15N) dimensions in Hutchinson’s space ecological niche (Hutchinson, 1957). However, environmental variables (SST and Chl a) also had a moderate to high influence on both δ13C and δ15N values, and this was also evident in the models conducted for each species separately. Temperature and chlorophyll a concentration are known to influence the distribution of both isotopes in marine environments (MacKenzie et al., 2011, 2014; Magozzi et al., 2017), especially in areas where enhanced primary productivity is supported by nutrients from continental runoff and upwelling events like our study region (Sousa et al., 2008; Ceia et al., 2018).
Considering temporal drivers, and although the year and the season had little influence on the dynamics of isotopic values for most of the models performed, the continuous timeline (i.e. universal time) cannot be neglected. Ceia et al. (2018) show that at a much smaller spatial scale (i.e. < 100 km), both δ13C and δ15N values can be highly driven by temporal drivers in the study region. The influence of universal time in driving isotopic variance, but not the year or season, was somehow unexpected because both δ13C and δ15N values can be highly driven by different years in top predators (Ceia et al., 2018). Consequently, temporal drivers associated with a continuous timeline (during the course of this study), and not to year or season, might be related with stabilizing mechanisms of maintenance of species diversity in the community through time (Chesson, 2000). The population dynamics within the communities results from balance mechanisms of inter- and intra-specific interactions (e.g. competition, predation), and availability and specificity of resources (de Villemereuil & López-Sepulcre, 2011; Tadesse, 2018). Thus, stabilizing mechanisms are essential for species co-existence, and may drive isotopic variance and resource partitioning in species from the same guild in order to allow them to adapt to each other and to environmental factors in space and time (Chesson, 2000).
Our outputs show an overall small importance of spatial variables in influencing isotopic variance within the community. Several studies have demonstrated that latitude at large spatial scales and contrast inshore versus offshore environments are often associated with isotopic baseline patterns (especially δ13C values) that are reflected along the food webs (Roscales et al., 2011; Chouvelon et al., 2012; Ceriani et al., 2014). Although there is a slight latitudinal gradient in δ13C values along the Portuguese coast and especially between inshore vs. offshore environments (Ceia et al., 2018), spatial variables were not very meaningful in the current study. The fact that all organisms inhabit the continental shelf supposedly did not drive a higher isotopic variance in the community (Chouvelon et al., 2014), which is consistent with more homogeneous values in this region (Ceia et al., 2018).
Body size is an important driver of niche partitioning within species. In fact, niche partitioning between fish species can be much lower than the intra-specific dissimilarities between conspecifics (e.g. between ontogenetic stages, Amundsen et al., 2003). There is evidence that the trophic level is positively related with length in myctophid fishes (Cherel et al., 2010), or that fish size and prey size are strongly correlated (Colloca et al., 2010), thus pointing out for a structuring effect of body size. The effect of body size driving niche partitioning in our study was expected, although extrinsic variables such as SST and Chl a showed to have a high influence on some models, a pattern also verified in predatory fish such as blue sharks Prionace glauca (Linnaeus, 1758) (Maxwell et al., 2019). However, the effect of body size should be much higher when considering a large spectrum of size classes, at least for species experiencing ontogenetic niche shifts and long-life span with continuous growth.
Species divergence in the use of resources
The productive shelf environment of the western Iberian waters supports an abundant trophic pyramid, as well as large fisheries targeting mid-trophic level species. The studied species occur here in large densities and abundances and are often commercially exploited (Leitão et al., 2014). Consequently, this is a potential competitive environment for small pelagic fishes inhabiting this ecosystem (Bode et al., 2007), which should favour the evolution of adaptations to reduce niche overlap (Hardin, 1960). Our results denote a relatively low overall variation in the isotopic values of the whole community within the study area (i.e. 4.4 ‰ in δ13C and 5.0 ‰ in δ15N), considering the large number of samples analysed in this study (i.e. 474 individuals from 11 species). However, the observed segregation of isotopic niches and variance in niche width resulted in species divergence in the use of resources (see below), a mechanism to reduce niche overlap and allow co-existence (Schulze et al., 2012; Knickle & Rose, 2014; Petalas et al., 2021).
Our results point to an overall segregation in isotopic niches and variance in niche width among most species. In a community where several mid-trophic level species coexist in large abundances, even small divergences in dietary foraging characteristics would potentially alleviate overlap in isotopic niches and hence competition, which can result in ecological differentiation among closely related species (Brandl et al., 2020). This gains further relevance in a community where species of the same genus coexist because they have similar structure and habits, which increases competition if they share the same spatiotemporal and trophic dimensions (Hardin, 1960; Knickle & Rose, 2014). For instance, the Atlantic horse mackerel (T. trachurus), the blue jack mackerel (T. picturatus) and the Mediterranean horse mackerel (T. mediterraneus) share morphological characteristics and have overlapping habitats. However, T. trachurus is much more abundant in the study area, although T. picturatus occurs in higher densities in the southwest of Portugal, and T. mediterraneus is found especially in the south (which may explain its higher degree of foraging specialization, Fig. 4) (Gonçalves et al., 2013). Similarly, the Atlantic mackerel (S. scombrus) and the Atlantic chub mackerel (S. colias) are very common pelagic fish species in the Iberian Peninsula coast, but S. scombrus is more abundant in the colder waters of the north and S. colias in the south (Martins et al., 2013). Thus, along with physiological adaptative traits such as species adaptations to warm versus cold waters, species-specific foraging strategies (either spatiotemporal or trophic) are crucial to allow the cohabitation of different species within the same guild. In this context, we highlight that the opportunistic behaviour and feeding plasticity should favour the co-existence of these species.
All the most captured species in this study, i.e. Trachurus spp., Scomber spp., S. pilchardus and E. encrasicolus are pelagic/semi-pelagic and opportunistic planktivorous species that show specific feeding traits (da Silva et al., 2020, 2022; Garrido et al., 2015). Species-specific feeding characteristics should be relevant for survival when sharing the same environment. For instance, the predominant consumption of meso and macrozooplankton by Trachurus spp. and S. scombrus (such as copepods and teleost larvae, but also shrimps, cephalopods, crustaceans and small pelagic fish), in contrast with a higher importance of microzooplankton and higher occasionally phytoplankton (such as diatoms and dinoflagellates) by S. pilchardus, E. encrasicolus and S. colias (Garrido & Murta, 2011; Costalago et al., 2012; Garrido et al., 2015), is reflected in higher δ15N values (and respective trophic level) in the former group. Moreover, S. pilchardus and S. colias also exert intraguild predation, actively preying on each other’s eggs and larvae (Garrido et al., 2015). Some species such as S. pilchardus, E. encrasicolus and Scomber spp. can use two feeding modes, i.e. filter feeding in the presence of smaller or highly concentrated prey and particulate feeding for larger particles of food or less abundant prey (Macy et al., 1998; Garrido et al., 2008; Costalago et al., 2012), which will increase their potential food spectrum. These feeding characteristics will alleviate competition by differentiating trophic ecology as our results highlight the general small potential competition (i.e. small niche overlap) between the species within this community. Thus, the flexibility to niche differentiation depends also on the type of specialization of each species, and its extent might be dependent on the species’ trophic specialisation along the specialists-to-generalists axis (Schulze et al., 2012).
The isotopic diversity and richness metrics here calculated provide a comprehensive assessment of the type of specialisation based on organisms functional traits and variability between individuals (Jackson et al., 2011; Cucherousset and Villéger 2015). Although we did not find any structuring of the 11 species into taxonomic groups along the specialists-to-generalists axis, this is thought to represent a central mechanism in the co-existence of species (Wilson & Yoshimura, 1994; Schulze et al., 2012). As discussed before, species from the same genus are unlikely to share the same spatiotemporal and trophic dimensions (Hardin, 1960), and thus to display similar strategies in their trophic ecology that would increase competition. In this study, we found that the most abundantly caught species (S. pilchardus, E. encrasicolus, T. trachurus and S. colias) showed higher generalisation of foraging strategies, suggesting that these four species represent an established piscivorous guild in the epipelagic neritic habitats of the North Atlantic. The other less abundant species showed their own trophic strategies in direction to a lower isotopic richness and/or diversity, and respective higher specialization. These results suggest that in a community of mid-trophic level species, the more generalist species, with higher flexibility to niche partitioning, should be predominant in the guild. However, more specialized species living in the same environment might have their own differentiated niches able to coexist and form a multi-species assemblage.
Conclusions
In this study, we examined the drivers of isotopic divergence and niche partitioning in a community of mid-trophic level species, considering both intrinsic (taxa, body size) and extrinsic (environmental, spatial, temporal) factors. Using a guild of common species from the North Atlantic composed by nine fish and two squid species, this study illustrates that the main driver of niche partitioning is determined by the taxonomic group, in accordance with the fundamental principles of niche theory (Hardin, 1960). Potential competition was limited to a very few interactions between species within the community. Moreover, the fact that body size can have a strong influence in driving isotopic variability within species, highlights that intrinsic factors are essential in the partitioning of resources within communities since these surpassed any environmental, temporal and/or spatial drivers considered here. However, the environment is central in determining isotopic variance of the food webs, and extrinsic factors should have a crucial role in the partitioning of resources of species/populations from different ecological communities, habitats and during long timeframes (as a stabilizing mechanism of maintenance of species diversity through time). These results suggest that intrinsic variables should be more relevant than extrinsic variables in driving niche partitioning at a smaller spatiotemporal scale. However, at a larger scale, the combined effect of both intrinsic and extrinsic factors is essential to support partitioning of resources and respective isotopic variance in ecological communities, and drive the adaptation and evolution of species.
Data and code availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Amundsen, P. A., T. Bøhn, O. A. Popova, F. J. Staldvik, Y. S. Reshetnikov, N. A. Kashulin & A. A. Lukin, 2003. Ontogenetic niche shifts and resource partitioning in a subarctic piscivore fish guild. Hydrobiologia 497: 109–119. https://doi.org/10.1023/A:1025465705717.
Bode, A., M. T. Alvarez-Ossorio, M. E. Cunha, S. Garrido, J. B. Peleteiro, C. Porteiro, L. Valdés & M. Varela, 2007. Stable nitrogen isotope studies of the pelagic food web on the Atlantic shelf of the Iberian Peninsula. Progress in Oceanography 74: 115–131. https://doi.org/10.1016/j.pocean.2007.04.005.
Boecklen, W. J., C. T. Yarnes, B. A. Cook & A. C. James, 2011. On the use of stable isotopes in trophic ecology. Annual Review of Ecology, Evolution, and Systematics 42: 411–440. https://doi.org/10.1146/annurev-ecolsys-102209-144726.
Bolnick, D. I., L. H. Yang, J. A. Fordyce, J. M. Davis & R. Svanbäck, 2002. Measuring individual-level resource specialization. Ecology 83: 2936–2941. https://doi.org/10.1890/0012-9658.
Brandl, S. J., J. M. Casey & C. P. Meyer, 2020. Dietary and habitat niche partitioning in congeneric cryptobenthic reef fish species. Coral Reefs 39: 305–317. https://doi.org/10.1007/s00338-020-01892-z.
Breiman, L., 2001. Random forests. Machine Learning 45: 5–32. https://doi.org/10.1023/A:1010933404324.
Ceia, F. R., R. A. Phillips, J. A. Ramos, Y. Cherel, R. P. Vieira, P. Richard & J. C. Xavier, 2012. Short- and long-term consistency in the foraging niche of wandering albatrosses. Marine Biology 159: 1581–1591. https://doi.org/10.1007/s00227-012-1946-1.
Ceia, F. R., Y. Cherel, V. H. Paiva & J. A. Ramos, 2018. Stable isotope dynamics (δ13C and δ15N) in neritic and oceanic waters of the North Atlantic inferred from GPS-tracked Cory’s shearwaters. Frontiers in Marine Science 5: 377. https://doi.org/10.3389/fmars.2018.00377.
Ceia, F. R., J. C. Xavier, A. R. Carreiro, I. dos Santos & Y. Cherel, 2022. Conventional and modern approaches to study seabird trophic ecology and diet. In Ramos, J. A. & L. Pereira (eds), Seabird Biodiversity and Human Activities 1st ed. CRC Press, Boca Raton: 19–35.
Ceriani, S. A., J. D. Roth, C. R. Sasso, C. M. McClellan, M. C. James, H. L. Haas, R. J. Smolowitz, D. R. Evans, D. S. Addison, D. A. Bagley, L. M. Ehrhart & J. F. Weishampel, 2014. Modeling and mapping isotopic patterns in the Northwest Atlantic derived from loggerhead sea turtles. Ecosphere 5: 122. https://doi.org/10.1890/ES14-00230.1.
Cherel, Y., K. A. Hobson & S. Hassani, 2005. Isotopic discrimination between food and blood and feathers of captive penguins: implications for dietary studies in the wild. Physiological and Biochemical Zoology 78: 106–115. https://doi.org/10.1086/425202.
Cherel, Y., C. Fontaine, P. Richard & J. P. Labat, 2010. Isotopic niches and trophic levels of myctophid fishes and their predators in the Southern Ocean. Limnology and Oceanography 55: 324–332. https://doi.org/10.4319/lo.2010.55.1.0324.
Chesson, P., 2000. Mechanisms of maintenance of species diversity. Annual Review of Ecology and Systematics 31: 343–366. https://doi.org/10.1146/annurev.ecolsys.31.1.343.
Chouvelon, T., J. Spitz, F. Caurant, P. Mèndez-Fernandez, A. Chappuis, F. Laugier, E. Le Goff & P. Bustamante, 2012. Revisiting the use of δ15N in meso-scale studies of marine food webs by considering spatio-temporal variations in stable isotopic signatures – The case of an open ecosystem: The Bay of Biscay (North-East Atlantic). Progress in Oceanography 101: 92–105. https://doi.org/10.1016/j.pocean.2012.01.004.
Chouvelon, T., A. Chappuis, P. Bustamante, S. Lefebvre, F. Mornet, G. Guillou, L. Violamer & C. Dupuy, 2014. Trophic ecology of European sardine Sardina pilchardus and European anchovy Engraulis encrasicolus in the Bay of Biscay (north-east Atlantic) inferred from δ13C and δ15N values of fish and identified mesozooplanktonic organisms. Journal of Sea Research 85: 277–291. https://doi.org/10.1016/j.seares.2013.05.011.
Colloca, F., P. Carpentieri, E. Balestri & G. Ardizzone, 2010. Food resource partitioning in a Mediterranean demersal fish assemblage: the effect of body size and niche width. Marine Biology 157: 565–574. https://doi.org/10.1007/s00227-009-1342-7.
Connan, M., B. J. Dilley, T. O. Whitehead, D. Davies, C. D. Mcquaid & P. G. Ryan, 2019. Multidimensional stable isotope analysis illuminates resource partitioning in a sub-Antarctic island bird community. Ecography (cop) 42: 1–12. https://doi.org/10.1111/ecog.04560.
Costalago, D., J. Navarro, I. Álvarez-Calleja & I. Palomera, 2012. Ontogenetic and seasonal changes in the feeding habits and trophic levels of two small pelagic fish species. Marine Ecology Progress Series 460: 169–181. https://doi.org/10.3354/meps09751.
Cucherousset, J. & S. Villéger, 2015. Quantifying the multiple facets of isotopic diversity: new metrics for stable isotope ecology. Ecological Indicators 56: 152–160. https://doi.org/10.1016/j.ecolind.2015.03.032.
Cury, P., A. Bakun, R. J. M. Crawford, A. Jarre, R. A. Quiñones, L. J. Shannon & H. M. Verheye, 2000. Small pelagics in upwelling systems: patterns of interaction and structural changes in ‘“wasp-waist”’ ecosystems. ICES Journal of Marine Science 57: 603–618. https://doi.org/10.1006/jmsc.2000.0712.
da Silva, J. M., L. M. F. Alves, M. I. Laranjeiro, A. Silva, M. M. Angélico, A. C. Norte, M. F. L. Lemos, J. A. Ramos, S. C. Novais & F. R. Ceia, 2020. Mercury levels in commercial mid-trophic level fishes along the Portuguese coast – Relationships with trophic niche and oxidative damage. Ecology Indicators 116: 106500. https://doi.org/10.1016/j.ecolind.2020.106500.
da Silva, J. M., L. M. F. Alves, M. I. Laranjeiro, F. Bessa, A. V. Silva, A. C. Norte, M. F. L. Lemos, J. A. Ramos, S. C. Novais & F. R. Ceia, 2022. Accumulation of chemical elements and occurrence of microplastics in small pelagic fish from a neritic environment. Environmental Pollution 292: 118451. https://doi.org/10.1016/j.envpol.2021.118451.
de Villemereuil, P. B. & A. López-Sepulcre, 2011. Consumer functional responses under intra- and inter-specific interference competition. Ecological Modelling 222: 419–426. https://doi.org/10.1016/j.ecolmodel.2010.10.011.
Fiuza, A. F. G., M. E. Macedo & M. R. Guerreiro, 1982. Climatological space and time variation of the Portuguese coastal upwelling Upwelling Portugal Coastal winds Sea surface temperature Sardine Upwelling Portugal Vents côtiers Température de surface de la mer Sardine ABSTRACT RÉSUMÉ. Oceanologica Acta 5: 31–40.
Garrido, S. & A. G. Murta, 2011. Interdecadal and spatial variations of diet composition in horse mackerel Trachurus trachurus. Journal of Fish Biology 79: 2034–2042. https://doi.org/10.1111/j.1095-8649.2011.03148.x.
Garrido, S., R. Ben-Hamadou, P. Oliveira, M. Cunha, M. Chícharo & C. van der Lingen, 2008. Diet and feeding intensity of sardine Sardina pilchardus: correlation with satellite-derived chlorophyll data. Marine Ecology Progress Series 354: 245–256. https://doi.org/10.3354/meps07201.
Garrido, S., A. Silva, J. Pastor, R. Dominguez, A. V. Silva & A. M. Santos, 2015. Trophic ecology of pelagic fish species off the Iberian coast: diet overlap, cannibalism and intraguild predation. Marine Ecology Progress Series 539: 271–286. https://doi.org/10.3354/meps11506.
Golikov, A. V., F. R. Ceia, R. M. Sabirov, G. A. Batalin, M. E. Blicher, B. I. Gareev, G. Gudmundsson, L. L. Jørgensen, G. Z. Mingazov, D. V. Zakharov & J. C. Xavier, 2020. Diet and life history reduce interspecific and intraspecific competition among three sympatric Arctic cephalopods. Scientific Reports 10: 1–11. https://doi.org/10.1038/s41598-020-78645-z.
Gonçalves, P., E. Henriques & M. M. Angélico, 2013. Co-occurrence of Trachurus trachurus and Trachurus picturatus spawners in Atlantic Iberian waters and the ability to distinguish their eggs in plankton samples. Fisheries Research 138: 139–145. https://doi.org/10.1016/j.fishres.2012.07.021.
Hardin, G., 1960. The competitive exclusion principle. Science (80-) 131: 1292–1297. https://doi.org/10.1126/science.131.3409.1292.
Helland, I. P., C. Harrod, J. Freyhof & T. Mehner, 2008. Co-existence of a pair of pelagic planktivorous coregonid fishes. Evolutionary Ecology Research 10: 373–390.
Hutchinson, G. E., 1957. Concluding remarks. Cold Spring Harbor Symposia on Quantitative Biology 22: 415–427.
Jackson, A. L., R. Inger, A. C. Parnell & S. Bearhop, 2011. Comparing isotopic niche widths among and within communities: SIBER - Stable Isotope Bayesian Ellipses in R. Journal of Animal Ecology 80: 595–602. https://doi.org/10.1111/j.1365-2656.2011.01806.x.
Jessopp, M., G. E. Arneill, M. Nykänen, A. Bennison & E. Rogan, 2020. Central place foraging drives niche partitioning in seabirds. Oikos 129: 1704–1713. https://doi.org/10.1111/oik.07509.
Knickle, D. C. & G. A. Rose, 2014. Dietary niche partitioning in sympatric gadid species in coastal Newfoundland: evidence from stomachs and C-N isotopes. Environmental Biology of Fishes 97: 343–355. https://doi.org/10.1007/s10641-013-0156-0.
Langton, R. W., 1982. Diet overlap between Atlantic cod, Gadus morhua, silver hake, Merluccius bilinearis, and fifteen other northwest Atlantic finfish. Fishery Bulletin 80: 745–759.
Layman, C. A., D. A. Arrington, C. G. Montaña & D. M. Post, 2007. Can stable isotope ratios provide for community-wide measures of trophic structure? Ecology 88: 42–48.
Leitão, F., V. Baptista, D. Zeller & K. Erzini, 2014. Reconstructed catches and trends for mainland Portugal fisheries between 1938 and 2009: implications for sustainability, domestic fish supply and imports. Fisheries Research 155: 33–50. https://doi.org/10.1016/j.fishres.2014.02.012.
MacKenzie, K. M., M. R. Palmer, A. Moore, A. T. Ibbotson, W. R. C. Beaumont, D. J. S. Poulter & C. N. Trueman, 2011. Locations of marine animals revealed by carbon isotopes. Scientific Reports 1: 21. https://doi.org/10.1038/srep00021.
MacKenzie, K. M., C. Longmore, C. Preece, C. H. Lucas & C. N. Trueman, 2014. Testing the long-term stability of marine isoscapes in shelf seas using jellyfish tissues. Biogeochemistry 121: 441–454. https://doi.org/10.1007/s10533-014-0011-1.
Macy, W. K., S. J. Sutherland & E. G. Durbin, 1998. Effects of zooplankton size and concentration and light intensity on the feeding behavior of Atlantic mackerel Scomber scombrus. Marine Ecology Progress Series 172: 89–100. https://doi.org/10.3354/meps172089.
Magozzi, S., A. Yool, H. Vander Zanden, M. Wunder & C. N. Trueman, 2017. Using ocean models to predict spatial and temporal variation in marine carbon isotopes. Ecosphere 8: e01763. https://doi.org/10.1002/ecs2.1763.
Martins, M. M., D. Skagen, V. Marques, J. Zwolinski & A. Silva, 2013. Changes in the abundance and spatial distribution of the Atlantic chub mackerel (Scomber colias) in the pelagic ecosystem and fsheries off Portugal. Scientia Marina 77: 551–563. https://doi.org/10.3989/scimar.03861.07B.
Massé, J., A. Uriarte, M. M. Angélico & P. Carrera, 2018. Pelagic survey series for sardine and anchovy in ICES subareas 8 and 9: towards an ecosystem approach. ICES Cooperative Research Report 332: 268. https://doi.org/10.17895/ices.pub.4599.
Maxwell, S. M., K. L. Scales, S. J. Bograd, D. K. Briscoe, H. Dewar, E. L. Hazen, R. L. Lewison, H. Welch & L. B. Crowder, 2019. Seasonal spatial segregation in blue sharks (Prionace glauca) by sex and size class in the Northeast Pacific Ocean. Diversity and Distributions 25: 1304–1317. https://doi.org/10.1111/ddi.12941.
Moreno, T. & J. J. Castro, 1995. Community structure of the juvenile of coastal pelagic fish species in the Canary Islands waters. Scientia Marina 59: 405–413.
Navarro, J., S. C. Votier, J. Aguzzi, J. J. Chiesa, M. G. Forero & R. A. Phillips, 2013. Ecological segregation in space, time and trophic niche of sympatric planktivorous petrels. PLoS ONE 8: e62897. https://doi.org/10.1371/journal.pone.0062897.
Newsome, S. D., C. M. Rio, S. Bearhop & D. L. Phillips, 2007. A niche for isotopic ecology. Frontiers in Ecology and the Environment 5: 429–436. https://doi.org/10.1890/060150.01.
Petalas, C., T. Lazarus, R. A. Lavoie, K. H. Elliott & M. F. Guigueno, 2021. Foraging niche partitioning in sympatric seabird populations. Scientific Reports 11: 2493. https://doi.org/10.1038/s41598-021-81583-z.
Post, D. M., C. A. Layman, D. A. Arrington, G. Takimoto, J. Quattrochi & C. G. Montaña, 2007. Getting to the fat of the matter: models, methods and assumptions for dealing with lipids in stable isotope analyses. Oecologia 152: 179–189. https://doi.org/10.1007/s00442-006-0630-x.
Quevedo, M., R. Svanbäck & P. Eklöv, 2009. Intrapopulation niche partitioning in a generalist predator limits food web connectivity. Ecology 90: 2263–2274. https://doi.org/10.1890/07-1580.1.
R Core Team, 2020. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. [available on internet at https://www.r-project.org/].
Roscales, J. L., E. Gómez-Díaz, V. Neves & J. González-Solís, 2011. Trophic versus geographic structure in stable isotope signatures of pelagic seabirds breeding in the northeast Atlantic. Marine Ecology Progress Series 434: 1–13. https://doi.org/10.3354/meps09211.
Ross, S. T., 1986. Resource partitioning in fish assemblages: a review of field studies. Copeia 2: 352–388. https://doi.org/10.2307/1444996.
Santos, A. M. P., A. Peliz, J. Dubert, P. B. Oliveira, M. M. Angélico & P. Ré, 2004. Impact of a winter upwelling event on the distribution and transport of sardine (Sardina pilchardus) eggs and larvae off western Iberia: a retention mechanism. Continental Shelf Research 24: 149–165. https://doi.org/10.1016/j.csr.2003.10.004.
Schoener, T. W., 1974. Resource partitioning in ecological communities. Science (80-) 185: 27–39. https://doi.org/10.1126/science.185.4145.27.
Schulze, T., H. Dörner, U. Baade & F. Hölker, 2012. Dietary niche partitioning in a piscivorous fish guild in response to stocking of an additional competitor: the role of diet specialisation. Limnologica 42: 56–64. https://doi.org/10.1016/j.limno.2011.08.001.
Sousa, F. M., S. Nascimento, H. Casimiro & D. Boutov, 2008. Identification of upwelling areas on sea surface temperature images using fuzzy clustering. Remote Sensing of Environment 112: 2817–2823. https://doi.org/10.1016/j.rse.2008.01.014.
Svanbäck, R. & D. I. Bolnick, 2007. Intraspecific competition drives increased resource use diversity within a natural population. Proceedings of the Royal Society B: Biological Sciences 274: 839–844. https://doi.org/10.1098/rspb.2006.0198.
Swanson, H. K., M. Lysy, M. Power, A. D. Stasko, J. D. Johnson & J. D. Reist, 2014. A new probabilistic method for quantifying n-dimensional ecological niches and niche overlap. Ecology 96: 318–324. https://doi.org/10.1890/14-0235.1.
Tadesse, S. A., 2018. The effects of competition (intra–specific and inter–specific) and predation on the distribution and abundance of guppy fish (Poecilia reticulata). International Journal of Avian & Wildlife Biology 3: 358–365. https://doi.org/10.15406/ijawb.2018.03.00121.
Van Valen, L., 1965. Morphological variation and width of ecological niche. The American Naturalist 99: 377–389.
Watabe, R., H. Tsunoda & M. U. Saito, 2022. Evaluating the temporal and spatio-temporal niche partitioning between carnivores by different analytical method in northeastern Japan. Scientific Reports 12: 11987. https://doi.org/10.1038/s41598-022-16020-w.
Williams, R., 1988. Spatial heterogeneity and niche differentiation in oceanic zooplankton. Hydrobiologia 167–168: 151–159. https://doi.org/10.1007/BF00026301.
Wilson, D. S. & J. Yoshimura, 1994. On the coexistence of specialists and generalists. The American Naturalist 144: 692–707.
Acknowledgements
We would like to thank the Portuguese Institute for the Sea and Atmosphere (IPMA) for fisheries assessment surveys to conduct this work under the EU Data Collection Framework (EU-DCF) through the program for biological data collection in the fisheries sector in Portugal—“Programa Nacional de Amostragem Biológica (PNAB)”.
Funding
Open access funding provided by FCT|FCCN (b-on). This study had the support of national funds through Fundação para a Ciência e Tecnologia, I. P (FCT; Portugal), under the projects UIDB/04292/2020, UIDP/04292/2020, granted to MARE, and LA/P/0069/2020, granted to the Associate Laboratory ARNET, and the transitory norm contract DL57/2016 to Filipe R. Ceia (DL57/2016/CP1370/CT90) at the University of Coimbra. The funding sources had no involvement in this study.
Author information
Authors and Affiliations
Contributions
FRC, YC and JAR conceived and designed the experiments. FRC, AVS, SG and MMA collected the samples. FRC, AVS, SG, JMS and MIL performed the experiments. FRC, YC, SG and JAR analysed the data and performed statistical analyses. FRC wrote the manuscript; other authors provided editorial advice. All authors have read the submitted version of the manuscript and approve its submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Ethical approval
All applicable institutional and/or national guidelines for the care and use of animals were followed.
Consent to participate
Not applicable.
Consent for publication
All authors consented to the publication of this work.
Additional information
Handling editor: Sofie Spatharis
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ceia, F.R., Cherel, Y., Silva, A.V. et al. Drivers of niche partitioning in a community of mid-trophic level epipelagic species in the North Atlantic. Hydrobiologia 850, 1583–1599 (2023). https://doi.org/10.1007/s10750-023-05160-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-023-05160-3