Abstract
Changes in temperature and CO2 are typically associated with climate change, but they also act on shorter time scales, leading to alterations in phytoplankton physiology and community structure. Interactions among stressors may cause synergistic or antagonistic effects on phytoplankton dynamics. Therefore, the main goal of this work is to understand the short-term isolated and interactive effects of warming and high CO2 on phytoplankton nutrient consumption, growth, production, and community structure in the Ria Formosa coastal lagoon (southern Portugal). We performed microcosm experiments with temperature and CO2 manipulation, and dilution experiments under temperature increase, using winter phytoplankton assemblages. Phytoplankton responses were evaluated using inverted and epifluorescence microscopy. Overall, phytoplankton growth and microzooplankton grazing on phytoplankton decreased with warming. Negative antagonist interactions with CO2 alleviated the negative effect of temperature on phytoplankton and cryptophytes. In contrast, higher temperature benefited smaller-sized phytoplankton, namely cyanobacteria and eukaryotic picophytoplankton. Diatom growth was not affected by temperature, probably due to nutrient limitation, but high CO2 had a positive effect on diatoms, alleviating the effect of nutrient limitation. Results suggest that this winter phytoplankton assemblage is well acclimated to ambient conditions, and short-term increases in temperature are detrimental, but can be alleviated by high CO2.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Phytoplankton are fundamental components of most aquatic communities and have been successfully employed as indicators of ecological status and environmental variability (Platt & Sathyendranath, 2008; Racault et al., 2012). Phytoplankton dynamics have been increasingly used for detecting and forecasting climate change effects on aquatic ecosystems, particularly the long-term effects of increased temperature and carbon dioxide (Chavez et al., 2011; Dutkiewicz et al., 2013, 2015; Cloern et al., 2016; Henson et al., 2016, 2017). However, these climate change variables can also act on shorter time scales, affecting phytoplankton growth and mortality, both directly and indirectly. For instance, large diurnal fluctuations in pH, which can exceed 1 unit, have been observed in coastal waters (Nielsen et al., 2012; Cornwall et al., 2013). These changes are not related with ocean acidification, but rather with shifts in primary and secondary production, river runoff, and upwelling (Eriander et al., 2016 and references therein). Likewise, sea surface temperature may vary significantly on a daily basis (Stuart-Menteth et al., 2003), and diel amplitudes up to 3.5°C have been reported (Stramma et al., 1986). Thus, the study of short-term effects of environmental drivers is critical to understand basic physiological and ecological mechanisms that may modulate phytoplankton acclimation and adaptation to environmental variability.
The effects of high CO2 on phytoplankton dynamics are species-specific and sometimes contradictory (Sobrino et al., 2008; Boyd et al., 2010; Meyer & Riebesell, 2015; Bach & Taucher, 2019). Overall, diffusion of CO2 to the water decreases pH levels and leads to alterations in fundamental biogeochemical processes that depend on seawater CO2 chemistry, potentially affecting marine biodiversity, particularly calcifying organisms (e.g. coccolithophores, Riebesell, 2004; Dutkiewicz et al., 2015). Increases in CO2 may also enhance phytoplankton growth and productivity, due to a downregulation of carbon concentrating mechanisms which increase the energy available for other cellular processes (Giordano et al., 2005; Dutkiewicz et al., 2013). Changes in seawater temperature can have direct effects on phytoplankton metabolic rates and, within defined temperature ranges, cell division rates will increase with temperature and decrease above the upper temperature limit (Eppley, 1972; Collos et al., 2005; Daufresne et al., 2009; Sherman et al., 2016). A gradual shift towards smaller phytoplankton is also expected with ocean warming, driven by increased water column stratification and reduced nutrient availability (Agawin et al., 2000; Daufresne et al., 2009; Morán et al., 2010b; Sommer et al., 2015; Schmidt et al., 2020). In addition, heterotrophic processes, including metabolic and grazing rates of zooplankton (Rose & Caron, 2007; Tsai et al., 2015, 2016), are usually considered more sensitive to temperature than autotrophic processes (López-Urrutia et al., 2006). In this context, the role of microzooplankton, mostly dominated by phagotrophic protists that are able to remove, on average, 62% of phytoplankton daily production (Schmoker et al., 2013), is particularly relevant. Temperature increases have been associated with higher microzooplankton growth rates and grazing rates upon phytoplankton (Aberle et al., 2007, 2012, 2015; O’Connor et al., 2009; Rose et al., 2009; Chen et al., 2012; Horn et al., 2020), eventually leading to a reduction in phytoplankton biomass. The impact of ocean warming on phytoplankton grazing is thus a key topic on climate change research, given that herbivory significantly affects phytoplankton biomass, the efficiency of the biological pump, and the resources available to higher trophic levels (Caron & Hutchins, 2013; Behrenfeld & Boss, 2014). Overall, the impact of warming on phytoplankton and their prevalent mechanisms (i.e. direct metabolic effects driven by warming, indirect effects linked with changes in stratification and nutrient supply, indirect effects driven by increased microzooplankton grazing) are expected to vary in time and space depending on light and nutrient conditions (Sommer et al., 2012; Lewandowska et al., 2014; Dutkiewicz et al., 2015).
Moreover, the occurrence of simultaneous changes in temperature, CO2, and other phytoplankton drivers (e.g. nutrients, light, grazers) may show synergistic and/or antagonistic effects on phytoplankton, thus leading to variable responses of phytoplankton to CO2 (Fu et al., 2012; Dutkiewicz et al., 2015; Liu et al., 2017) and temperature increases (O’Connor et al., 2009; Lewandowska et al., 2014). Addressing the interactions between high CO2 and warming is particularly pertinent, as, overall, CO2 may dampen the growth-enhancing effects of high temperature (Seifert et al., 2020), but the response direction highly depends on the functional group/species (Kroeker et al., 2013). Multi-stressor studies addressing the interactions between temperature and CO2 have mostly used unialgal cultures (e.g. Fu et al., 2007, 2008; De Bodt et al., 2010; Torstensson et al., 2012) to assess effects on phytoplankton physiology. Other studies have analysed the interactive effects of temperature and CO2 on natural phytoplankton assemblages or specific functional types, using a variety of experimental approaches, including continuous cultures (Hare et al., 2007; Feng et al., 2009; Rose et al., 2009), semi-continuous cultures (Tatters et al., 2013; Hoppe et al., 2018), and natural microcosms (Coello-Camba et al., 2014; Holding et al., 2015; Maugendre et al., 2015), and incubation periods ranging between 1 day (Holding et al., 2015) and 30 + days (Keys et al., 2018).
The Ria Formosa coastal lagoon (Southern Portugal), located in an area extremely vulnerable to climate change (IPCC, 2014), is one of the most important confined marine ecosystems in Portugal, from biological and social–economical perspectives. Climate predictions for the southern Portuguese coast include a sea surface temperature increase of 3°C in the winter and 4°C in the summer by 2080–2100 (Santos et al., 2002). Moreover, a significant interannual winter warming trend (0.07–0.10°C yr−1) since 1967, concurrent with a declining trend in phytoplankton biomass, were observed for this ecosystem (Barbosa, 2010). This declining trend could be indirectly related to the stimulatory effect of warming on phytoplankton herbivores, but as recognized by Barbosa (2010), dedicated experiments are needed to conclusively establish direct links between phytoplankton dynamics and climate change. Previous experiments addressing climate change impacts on phytoplankton in the Ria Formosa evaluated short-term interactive effects of high CO2 and exposure to ultraviolet radiation (UVR) during winter (Domingues et al., 2014), and effects of CO2, UVR, and nutrient enrichment during summer (Domingues et al., 2017b). However, no information on the effects of warming on phytoplankton and their dominant grazers is currently available for this climate-sensitive ecosystem.
In this context, this study aimed to understand the short-term isolated and combined effects of warming and high CO2 on phytoplankton growth and mortality due to microzooplankton grazing in the Ria Formosa coastal lagoon during winter, a period commonly characterized by high nutrient availability (Domingues et al., 2017a) and low UVR (Machado, 2010). Our specific objectives were to evaluate: (a) the isolated and combined effects of warming and high CO2 on phytoplankton net growth rate, composition, photosynthetic parameters, and nutrient consumption; and (b) the effects of temperature increase on phytoplankton group-specific instantaneous growth rates and microzooplankton grazing rates. To accomplish these objectives, we used natural phytoplankton assemblages from the Ria Formosa coastal lagoon subjected to (a) a two-stressor microcosm experiment with temperature and CO2 increase, and (b) a dilution experiment under in situ and increased temperature. Based on previous studies (Barbosa, 2010; Morán et al., 2010a; Domingues et al., 2014, 2017b; Sherman et al., 2016), we hypothesized that: (a) the effects of warming and high CO2 on phytoplankton will vary among phytoplankton functional groups, with beneficial effects of temperature for smaller-sized groups, which can be counteracted by CO2, and (b) warming will increase microzooplankton grazing on phytoplankton, particularly on picophytoplankton and nano-sized plastidic flagellates.
Materials and methods
Study site
The Ria Formosa coastal lagoon is a shallow multi-inlet euryhaline system, located on the southern coast of Portugal (Fig. 1). The lagoon has a total wetland area of 105 km2, a mean depth of 2 m, and extends 55 km (E–W) and 6 km (N–S) at its widest point (Andrade et al., 2004). Tides are semidiurnal and mesotidal, with tidal amplitudes ranging between 1.3 and 3 m during neap and spring tides, respectively (Newton et al., 2003). The water column is well mixed, with no persistent or widespread haline or thermal stratification (Newton & Mudge, 2003). Short-term variations in water temperature and pH are not significant in the Ria Formosa (Cravo et al., 2020). Water temperature variability reflects the seasonality of solar radiation, with lower values in January and February (~ 10°C), and higher values during summer months (15–27°C). pH values follow the inverse pattern, with lower values in the summer (7.75) and higher in the autumn/winter (8.30); pH values mirror the variability in dissolved oxygen concentrations and lower values are associated with coastal upwelling events (Cravo et al., 2020).
The Ria Formosa is located in a Mediterranean climate area, with hot and dry summers, and moderate winters. The coastal region adjoining the Ria Formosa is affected by regular upwelling events that extend approximately 6 km upstream the lagoon inlets (Cravo et al., 2014). The lagoon is a breeding and feeding ground for fish and birds, and supports a wide range of human activities, including tourism, fisheries, and shellfish farming. Due to its ecological importance, the Ria Formosa lagoon and adjacent habitat were designated a National Natural Park in 1987, part of Natura 2000 European conservation network. The Ria is a Ramsar wetland and it is included in the Special Bird Protection Area.
Experimental strategy
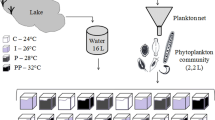
Two experiments were carried out with water samples collected in the Ria Formosa coastal lagoon, under typical winter conditions (February 2012). For both experiments, sub-surface water samples were collected using 10 l polycarbonate bottles at an inner lagoon location (Fig. 1) at low tide, to minimize the influence of adjacent coastal waters. Water temperature and photosynthetically active radiation in the mixed layer were measured with a YSI 30 probe and a LI-COR LI-193 spherical quantum sensor, respectively. Water samples were collected for the determination of inorganic macronutrient concentrations, chlorophyll-a concentration, and phytoplankton composition and abundance, at the beginning of the experiments. Samples were not screened prior to incubation, to avoid damaging delicate microzooplankton (Calbet et al., 2011b; Grinienė et al., 2016) and modifying initial phytoplankton composition (Nogueira et al., 2014). All material used in the experiments and laboratorial analyses was previously washed with HCl 10% and thoroughly rinsed with deionised water.
Experiment 1: effects of warming and high CO2 on nutrient consumption, phytoplankton growth and composition, and photosynthetic parameters
The first experiment tested the effects of warming and high CO2 on the net growth rate and photosynthetic parameters of winter phytoplankton assemblages. Water samples were incubated under two different temperatures: (a) in situ temperature (Tis = 10°C), and (b) in situ + 3°C (Tis+3 = 13°C). The use of a 3°C increase in temperature was based on model predictions for southern coast of Portugal by 2080–2100 (Santos et al., 2002). Due to logistical reasons, treatments subjected to increased temperature were conducted 2 days after treatments subjected to ambient temperature. For each temperature, ambient CO2, and high CO2 treatments were prepared. Carbonate (CO32−, as Na2CO3), bicarbonate (HCO3−, as NaHCO3) and hydrochloric acid (HCl 0.01 N) were added to increase CO2 partial pressure (pCO2) and dissolved inorganic carbon (DIC) to levels expected for the year 2100, according to Gattuso et al. (2010). pCO2 at the beginning and end of the incubation period was estimated with the Seacarb package for R (R Core Team, 2020). Alkalinity, determined by titration (Parsons et al. 1984), and pH were used as input variables. CO2 partial pressure at the beginning of incubation was 389.7 ± 77.0 µatm and 712.3 ± 82.8 µatm in the low and high CO2 treatments, respectively, corresponding to pH values of 8.1 ± 0.1 and 7.9 ± 0.0. After 2-day incubation, pCO2 and pH were 379.4 ± 15.5 µatm and 8.1 ± 0.0 in the low CO2 treatments and 719.0 ± 59.2 µatm and 7.9 ± 0.0 in the high CO2 treatments.
Four different experimental treatments, established in triplicate 2 l polycarbonate bottles, were used to test the effects of warming and CO2: “ambient” (in situ temperature, ambient CO2 levels), “ + CO2” (in situ temperature, high CO2), “ + T” (increased temperature, ambient CO2) and “ + T + CO2” (increased temperature, high CO2). The bottles were incubated in a plant growth chamber (Phytoclima S600) for 48 h, under an ambient light:dark cycle (11 h:13 h), and exposed to photosynthetically active radiation (PAR) intensity of approx. 200 μmol photons m−2 s−1 (equivalent to mean PAR intensity in the mixed layer, estimated according to Domingues et al., 2017a). Bottles were manually homogenized twice daily to avoid settlement of non-motile cells. The experimental treatments were initially prepared in 10 l polycarbonate bottles, from which initial samples were taken; at the end of incubation, final samples were taken from each bottle. Initial and final water samples were used for the determination of pH and alkalinity, concentration of dissolved inorganic macronutrients, chlorophyll-a concentration, phytoplankton composition and abundance, and photosynthetic parameters. Phytoplankton net growth rates (r, d−1) for the whole assemblage or specific functional types were estimated according to Eq. 1, assuming exponential growth:
where Nt and N0 represent chlorophyll-a concentration or phytoplankton abundance at the end and beginning of incubation, respectively (Δt = 2 days). Nutrient net consumption rates or, more accurately, nutrient disappearance rates, that integrate not only nutrient uptake (inward transport through the cell membrane), but also excretion and regeneration (Domingues et al., 2011a), were estimated as the slope of a linear or exponential function adjusted to nutrient concentration over the incubation period.
Experiment 2: effects of temperature increase on phytoplankton growth, and mortality
The second set of experiments tested the effects of temperature on phytoplankton instantaneous growth rates and mortality rates due to microzooplankton herbivory, using the dilution technique (Landry & Hassett, 1982); the water samples were not pre-screened, given that this may significantly alter the initial phytoplankton assemblage (Nogueira et al., 2014). A series of four different dilutions of whole seawater sample were prepared using seawater filtered through glass fibre filters (GF/F, Whatman, particle retention > 0.7 µm). Considering the dilution factor as the proportion of unfiltered seawater sample in each dilution treatment, the following dilutions were prepared: 0.125, 0.25, 0.50, and 1 (undiluted water). In order to avoid differences in nutrient concentration among dilutions, which could lead to the violation of method assumptions, all treatments were enriched with inorganic macronutrients, including silicate (Calbet & Saiz, 2018), at saturating concentrations (+ 50 μM of N and Si, added as nitrate and silicate, respectively, and + 2.5 μM of P, added as phosphate). Additionally, an experimental treatment with undiluted water without nutrient additions (DIL1) was also prepared to account for the potential effects of nutrient addition on the estimation of in situ phytoplankton instantaneous growth rates.
The five experimental treatments (dilutions) were prepared in 10 l polycarbonate bottles, from which the initial samples for the determination of in vivo fluorescence of chlorophyll-a, and phytoplankton composition and abundance were taken. Then, duplicate 1 l polycarbonate bottles were filled, with no headspace, with the respective dilution and incubated for 48 h inside a plant growth chamber, under simulated in situ conditions (ambient light:dark cycle = 11 h:13 h; and PAR intensity of approx. 200 μmol photons m−2 s−1, equivalent to mean PAR intensity in the mixed layer) and controlled temperature. Bottles were manually homogenized twice a day to avoid settlement of non-motile cells. Experiments were carried out under two different temperatures: (a) in situ temperature (Tis = 13°C), and (b) in situ + 3°C (Tis+3 = 16°C). At the end of incubation, aliquots were taken from all experimental treatments for the determination of the above-referenced variables.
For each dilution, net phytoplankton growth rates (r, d−1) during the incubation period (Δt) were calculated according to Eq. 1, using in vivo fluorescence of chlorophyll-a for the whole phytoplankton assemblage and cell abundance for specific phytoplankton groups, at the beginning (N0) and end (Nt) of the incubation, assuming exponential growth. Phytoplankton net growth rates in nutrient-enriched dilutions were then regressed against the exact dilution factor (calculated as the quotient between in vivo fluorescence in each dilution and in vivo fluorescence in undiluted water), and phytoplankton potential instantaneous growth rates (μ0) and microzooplankton grazing rates (g) were estimated as the y-intercept and the slope (when negative, absolute value is considered; for other situations, see below) of the linear regression line, respectively (Landry & Hassett, 1982). In situ instantaneous growth rate of phytoplankton (μ) was estimated according to Eq. 2 (Calbet et al., 2011a):
where NGR.DIL1 represents phytoplankton net growth rate in undiluted samples without added nutrients (DIL1). Dilution experiments were also used to assess phytoplankton potential nutrient limitation, by comparing phytoplankton net growth rates between nutrient-enriched and nutrient-unenriched undiluted treatments (NGR.DIL1 + and NGR.DIL1, respectively). Standard errors (SE) for µ and NGR were estimated using the corresponding error propagation equations (see Calbet et al., 2011a; http://julianibus.de/physik/propagation-of-uncertainty).
Analysis of variance (ANOVA) on the residuals of the linear regression was used to test whether the dilution plots significantly deviated from the linearity assumption of the dilution technique (Worden & Binder, 2003; Morison & Menden-Deuer, 2017). For linear plots with statistically non-significant slopes (i.e. not significantly different from zero), grazing rate was assumed zero, and phytoplankton potential instantaneous growth rate was estimated as the average value of the net growth rates of all nutrient-amended dilution treatments (Chen et al., 2009; Twiss & Smith, 2012; Grinienė et al., 2016; Morison & Menden-Deuer, 2017; Menden-Deuer et al., 2018; Jiang et al., 2021).
For dilution plots that showed significant deviations from linearity, mostly associated with saturated grazing (Gallegos, 1989; Dolan et al., 2000; Teixeira & Figueiras, 2009), data were also tested for non-linearity by polynomial regression analyses. When significant non-linearity was detected (P < 0.05, for the coefficient of quadratic or higher terms in actual dilution factor; (Nejstgaard et al., 1997; Suzuki et al., 2002)), phytoplankton potential instantaneous growth rate (μ0) was estimated as the regression intercept of the linear (non-saturation range) portion of the dilution plot (generally dilution factor ≤ 0.5), and the microzooplankton grazing rate (g) was estimated as μ0 − NGR.DIL1 + . This modified version of the three-point method (Gallegos, 1989) is widely used for analyses of non-linear dilution plots (Strom et al., 2001, 2007; Teixeira & Figueiras, 2009; Teixeira et al., 2011, 2012, 2014; Dix & Hanisak, 2015).
Although dilution is the most commonly used technique for measuring microzooplankton herbivory, the linearity of the dilution plots has been frequently compromised, namely in eutrophic ecosystems, therefore requiring a cautious interpretation (Calbet & Saiz, 2013; Dix & Hanisak, 2015; Horn et al., 2020). Significant negative slopes (one-tailed t-test, P < 0.05) for the whole or part (e.g. L-shaped, V-shaped, and inverted V-shaped dilution plots) of each dilution series were interpreted as a significant grazer effect on phytoplankton. In cases where significant negative slopes were observed, the percentage of particulate net phytoplankton production daily removed by microzooplankton (% pNPP) was estimated as (g/µ × 100) (Calbet et al., 2011a).
Inorganic macronutrients
Samples for determination of inorganic macronutrient concentrations were filtered through 0.2 µm cellulose acetate filters. Soluble reactive phosphorus (SRP), dissolved silicate (DSi), and ammonium (NH4+) were analysed immediately after sampling, using the spectrophotometric methods described by Grasshoff et al. (1999) on a spectrophotometer Hitachi U-2000. Samples for nitrate (NO3−) and nitrite (NO2−) were frozen (− 20°C) until analysis on a Skalar SAN + segmented flow analyser. Dissolved inorganic nitrogen (DIN) concentration was calculated as the sum of NO3−, NO2−, and NH4+ concentrations.
Phytoplankton biomass, abundance, and composition
Chlorophyll-a concentration, used as a proxy for phytoplankton biomass, was determined spectrophotometrically after sample filtration through glass fibre filters (GF/F, Whatman, particle retention > 0.7 µm) and overnight extraction with acetone 90% (Parsons et al., 1984). For dilution experiments, chlorophyll-a in vivo fluorescence was determined using a Turner Designs 10-AU fluorometer.
Phytoplankton composition and abundance were determined using epifluorescence (Haas, 1982) and inverted microscopies (Utermöhl, 1958). Samples for pico- (< 2 µm) and nanophytoplankton (2–20 µm) analysis were preserved with glutardialdehyde (final concentration 2%) immediately after collection, stained with proflavine, and filtered (< 100 mm Hg) onto black polycarbonate membrane filters (Whatman, nominal pore diameter = 0.45 µm). Preparations were made within 24 h of sampling using glass slides and non-fluorescent immersion oil (Cargille type A), and then frozen (− 20°C) until analysis under dark conditions, to minimize loss of fluorescence. Observation was made at 787.5 × magnification using a Leica DM LB epifluorescence microscope. Samples for microphytoplankton (> 20 µm) analysis were preserved with acid Lugol’s solution (final concentration approx. 0.003%) immediately after collection. Acid Lugol’s was chosen given that it is more appropriate than neutral Lugol’s in preserving microflagellates and diatoms (Williams et al., 2016), which are major components of phytoplankton in the Ria Formosa (Barbosa, 2010). Aliquots of 25–50 ml were settled in sedimentation chambers and observed at 400 × magnification using a Zeiss Axio Observer A1 inverted microscope. Cells were identified, whenever possible, to the genus or species level. For both methods, a minimum of 50 random visual fields and at least 400 cells in total, with 50 cells of the most common genus, were enumerated. Assuming that phytoplankton cells were randomly distributed, counting precision was ± 10% (Venrick, 1978). Phytoplankton taxonomic classification followed the online database AlgaeBase (Guiry & Guiry, 2021).
Phytoplankton primary production and photosynthetic parameters
Primary production and photosynthetic parameters were determined using the Steeman-Nielsen (1952) method. 50 ml aliquots of each experimental treatment were collected at the end of incubation into polycarbonate flasks and 100 µl (2 µCi) of 14C-HCO3− were added to each flask. The flasks were incubated in a plant growth chamber, for 2 h, under different PAR intensities (0, 47, 113, 230, and 365 µmol photons m−2 s−1), provided by Philips Cool White fluorescent lamps. Primary production was calculated according to Eq. 3:
where PP is phytoplankton primary production (mg C L−1 h−1), Rs (dpm) is the activity in the sample, Rb (dpm) is the mean activity of the dark flasks, D is the isotopic discrimination (= 1.05), W (mg C L−1) is the amount of dissolved inorganic carbon in the sample, R (dpm) is the total activity of the 14C added to each flask, and \(\Delta t\) is the incubation period (two hours). The concentration of dissolved inorganic carbon was analysed after acidification with HCl (Parsons et al., 1984).
For each experimental treatment and each light level, primary production (PP, mg C L−1 h−1) was converted to biomass-specific primary production (PB = PP/Chla, mg C (mg Chla)−1 h−1). Photosynthetic parameters were estimated according to Eilers & Peeters (1988) model, through non-linear fitting of Eq. 4, using the “nls” function available in R (R Core Team, 2020):
where PB is biomass-specific primary production (mgC mgChla−1 h−1) and E is PAR irradiance during incubation (µmol photons m−2 s−1). Coefficients a, b, and c were used to estimate maximal biomass-specific production rate (\(P_{\max }^{{\text{B}}}\), mgC mgChla−1 h−1), optimal light intensity (Eopt, µmol photons m−2 s−1), and initial slope of the P–E curve, which represents photosynthetic efficiency (α, mg C (mg Chla)−1 h−1 (µmol photons m−2 s−1)−1), according to Eqs. 5, 6, and 7, respectively:
Data analyses
Data normality and homogeneity of variances were tested using Shapiro–Wilk and Levene’s tests, respectively. The isolated and combined effects of warming and high CO2 on nutrient net consumption rates and phytoplankton net growth rates, for the phytoplankton assemblage and specific functional types, were assessed using a two-way analysis of variance (ANOVA), with replication. Effect sizes were assessed by estimating omega-squared statistics, which indicates the percentage of variation in the dependent variable attributable to the independent variable (Olejnik & Algina, 2003; Albers & Lakens, 2018). Differences in phytoplankton photosynthetic parameters and instantaneous growth rates, and microzooplankton grazing rates between in situ and increased temperature were evaluated using one-tailed t-test or Mann–Whitney test, depending on data normality. All statistical analyses were considered at a 0.05 significance level. Data analyses were performed with IBM SPSS® Statistics v.22 software.
Results
Environmental setting
Experiment 1 was used to evaluate the response of a natural plankton assemblage, collected under typical winter conditions (early February 2012, water temperature = 10°C) in the Ria Formosa coastal lagoon, to controlled increases in water temperature and carbon dioxide content, based on four different experimental treatments: ambient (control), + CO2, + T, and + T + CO2. Initial concentrations of dissolved inorganic macronutrients were 0.95 ± 0.18 µM for DIN, 0.18 ± 0.05 µM for SRP, and 3.41 ± 1.38 µM for DSi. Initial chlorophyll-a concentration (Chla) and total phytoplankton abundance varied between 1.11 and 3.88 µg L−1 and 5.26 × 106–5.36 × 106 cells L−1, respectively. In terms of abundance, phytoplankton community was dominated by Synechococcus-like picocyanobacteria (85.1%), with minor contributions of cryptophytes (14.6%) and diatoms (0.3%).
Experiment 2 evaluated the effects of temperature (+ 3°C) on phytoplankton growth and mortality using a natural plankton assemblage collected in late February 2012 (water temperature = 13°C) in the Ria Formosa coastal lagoon. Initial nutrient concentrations were 2.75 ± 2.48 µM DIN, 0.09 ± 0.02 µM DRP and 4.69 ± 1.38 µM SRP. Initial Chla concentration in these experiments was 3.0 ± 0.2 µg L−1 and total phytoplankton abundance was 3.34 × 106 ± 5.17 × 105 cells L−1. Phytoplankton community was dominated, in terms of abundance, by eukaryotic picophytoplankton (38%) and cryptophytes (38%). Other autotrophic nanoflagellates (11%), cyanobacteria (5%), and diatoms (7%) dominated by the colonial centric genus Chaetoceros, were also present in the initial samples. Euglenophytes and dinoflagellates showed minor contributions to total abundance (1%). Initial abundance of phagotrophic protists was 3.90 × 105 ± 5.91 × 104 cells L−1 and the assemblage was mainly composed by aplastidic nanoflagellates (96%) and ciliates (4%).
Effects of warming and high CO2 on nutrient consumption, phytoplankton net growth rate and composition, and photosynthetic parameters
Except for nitrate (Fig. 2A), temperature increase significantly affected net consumption rates of all nutrients (dissolved silicate, dissolved reactive phosphorus, and ammonium), in relation to the control (Fig. 2, Table 1). Under increased temperature, consumption rates of NH4+ (F(1,8) = 28.155; P = 0.001; ω2 = 0.70; Fig. 3B) and DSi (F(1.7) = 114.005; P < 0.001; ω2 = 0.71; Fig. 2D) were significantly higher, but SRP consumption (F(1,8) = 18.778; P = 0.003; ω2 = 0.75; Fig. 2C) was significantly lower. DSi consumption was also significantly higher under high CO2 (F(1,7) = 10.710; P = 0.014; ω2 = 0.06), but the effect size was small. Significant (F(1,7) = 10.710; P = 0.014; ω2 = 0.06) interactive effects between CO2 and temperature increases on DSi consumption rates were also detected, but the magnitude of the interaction was low (Table 1).
Overall, phytoplankton abundance decreased in all experimental treatments during the 2-day incubation period, and changes in phytoplankton composition among experimental treatments were also detected. The relative contribution of cyanobacteria and diatoms to total phytoplankton abundance increased in all experimental treatments in relation to the control (between 111–146%, and 199–271%, respectively). Conversely, the relative contribution of cryptophytes decreased, for all the experimental treatments (58–89%), in relation to the control.
Significant responses of the whole phytoplankton assemblage to warming and high CO2 were observed, in relation to the control treatment (Fig. 3, Table 1). Phytoplankton net growth rates, based on Chla, were negatively affected by warming (F(1,8) = 23.187; P = 0.001; ω2 = 0.45), but were not affected by high CO2 (p > 0.05). Significant negative interactive effects of high CO2 and warming were detected (F(1,8) = 17.489; P = 0.003; ω2 = 0.33) (Table 1, Fig. 3A).
Diatom net growth rates, negative in all treatments, showed no significant responses to warming, but were significantly higher under high CO2 (although still negative), in relation to the control (F(1,8) = 7.699; P = 0.024; ω2 = 0.32) (Fig. 3B, Table 1). No interactions between temperature and CO2 were detected. Cryptophyte net growth rates were negatively affected by both warming (F(1,7) = 157.284; P < 0.001; ω2 = 0.73) and high CO2 levels (F(1,7) = 14.053; P = 0.007; ω2 = 0.06), in relation to the control (Fig. 3C, Table 2). Significant interactive effects of high CO2 and warming on cryptophyte growth rate were also detected (F(1,7) = 65.337; P < 0.001; ω2 = 0.30), leading to lower growth rates in relation to the control. Net growth rates of cyanobacteria were positively affected by warming (F(1,6) = 13.120; P = 0.011; ω2 = 0.60), but high CO2 levels showed no significant effects (Fig. 3D, Table 2).
Phytoplankton photosynthetic parameters, derived from photosynthesis-irradiance (PE) curves (Fig. 4, Table 2), showed only a few significant changes associated with temperature and CO2. Photosynthetic efficiency (0.00574–0.02190 mgC mgChla−1 h−1 (µmol photons m−2 s−1)−1) was significantly higher (P < 0.05) for isolated increases in temperature, in relation to the control, showing a 3.8-fold increase. In the case of maximum biomass-specific production rate, \(P_{\max }^{{\text{B}}}\) (1.95–2.91 mgC mgChla−1 h−1), a significant decline (P < 0.01) was associated with warming and high CO2 (+ T + CO2), in relation to the control.
Photosynthesis-irradiance curves, determined using 14C incubations, in the temperature and CO2 enrichment experiments (experiment 1), depicting biomass-specific production (PB, mgC mgChla−1 h−1) as a function of light (PAR) intensity (E, µmol photons m−2 s−1). Curve fitting was based on the non-linear regression model of Eilers & Peeters (1988) (see Eq. 3)
Effects of warming on phytoplankton instantaneous growth and microzooplankton grazing rates
Dilution experiments were used to evaluate the effects of warming on phytoplankton potential (µo) and in situ (µ) instantaneous growth rates, and microzooplankton grazing rates (g) and impact (%pNPP) and detect potential nutrient limitation of phytoplankton growth. Mean values of μ0, μ, g, and %pNPP, for the whole assemblage or specific phytoplankton functional groups, respective standard errors (± SE), and relevant statistical information are presented in Table 3 (see Fig. 5 for dilution plots). The comparison of phytoplankton net growth rates in undiluted samples with and without added nutrients revealed a significant growth enhancement for diatoms, cryptophytes, and the whole phytoplankton assemblage, for both temperature levels (ambient and + 3°C), and for other plastidic nanoflagellates and eukaryotic picophytoplankton under increased temperature, indicative of nutrient limitation. No apparent effects of nutrient addition on net growth rates of cyanobacteria were detected (see Fig. 5, Table 3). Mean values for μ, g, and grazing impact on daily phytoplankton production ranged between 0.132–0.913 d−1, 0.069–0.876 d−1, and 0–142.6%, respectively, varying according to the phytoplankton group considered and temperature level.
Relationship between dilution factor and net growth rate of (A) natural phytoplankton assemblage, (B) planktonic diatoms, (C) cryptophytes, D other plastidic flagellates, E cyanobacteria, and F eukaryotic picophytoplankton during dilution experiments undertaken under in situ (circles, Tis) and increased (triangles, Tis+3°C) water temperature (experiment 2). Solid symbols (black and grey) represent net growth rates for dilution treatments amended with nutrients; open symbols represent net growth rates for undiluted samples incubated without nutrients. Regression lines were fitted to net growth rates from treatments with added nutrients (black solid symbols); all regression lines shown have statistically significant negative slopes (P < 0.05). Grey bold symbols were not used for linear regressions
Overall, the effects of temperature increase were more marked on phytoplankton growth rates than on microzooplankton grazing rates. At the level of the whole phytoplankton assemblage (Fig. 5A), temperature increase induced a significant decline in both μ0 (P < 0.01), μ, and g (P < 0.05), in comparison with ambient temperature. However, no significant effects of warming were detected on phytoplankton net growth rates in undiluted sample without added nutrients (NGR.DIL1), an experimental treatment that can be considered a proxy for short-term evolution of phytoplankton in nature. Warming was also associated with significant declines in μ0 for both cryptophytes (Fig. 5C) and other plastidic nanoflagellates (Fig. 5D; P < 0.01) but showed no significant effects on g and NGR.DIL1. Regarding planktonic diatoms, no significant effects of warming were detected on μ0, μ, g, or NGR.DIL1 (Fig. 5B), whereas for smaller-sized phytoplanktonic groups (cyanobacteria and eukaryotic picophytoplankton), warming enhanced NGR.DIL1 and NGR.DIL1+, μ0, and μ of eukaryotic picophytoplankton (Fig. 5E, F), but showed no significant effects on g (Table 3). Grazing by microzooplankton upon eukaryotic picophytoplankton under both temperatures was not significantly different from 0.
Discussion
Our two short-term microcosm experiments showed that the effects of warming and acidification varied among phytoplankton functional groups, leading to positive, neutral, and negative effects on phytoplankton growth rates. Overall, phytoplankton growth rates decreased with warming, except for smaller-sized groups. Interactions between CO2 and temperature were negative antagonistic, i.e. the cumulative effects of the two stressors were positive (Piggott et al., 2015), only for the whole assemblage and cryptophytes. Warming was also associated with lower potential and in situ instantaneous growth rates of the phytoplankton assemblage and lower rates of microzooplankton grazing upon phytoplankton.
Diatoms showed a moderate positive response to high CO2 levels, but no effects of temperature or interactions between these two stressors were observed. Positive effects of high CO2 on diatom net growth rates, reported for several marine ecosystems (see review by Bach & Taucher, 2019), were previously referred for a winter phytoplankton assemblage from the Ria Formosa coastal lagoon, sampled approximately one month prior to the present study (Domingues et al., 2014). Our study also revealed a significant increase in silica consumption in relation to the control, not only in high CO2 treatments, but also in the high temperature treatments, without any growth enhancement. This could be attributed to a potential nutrient limitation of the diatom assemblage. Indeed, both the application of nutrient limitation criteria (e.g. N limitation if DIN < 1 M, DIN:P < 10, and Si:DIN > 1: Justíc et al., 1995) and the results from dilution experiments (undiluted samples with and without added nutrients) suggest that nutrients, particularly nitrogen, were a limiting resource to phytoplankton growth. Nitrogen limitation in the Ria Formosa coastal lagoon frequently occurs during summer (Domingues et al., 2015), and was also detected in February 2012 using nutrient enrichment experiments (Domingues et al., 2017a). Nitrate, usually the preferred nitrogen source for diatoms (Domingues et al., 2011b), was equally consumed in all treatments, whereas silica consumption was stimulated in all CO2 and temperature experimental treatments. This uncoupling between Si consumption and diatom growth under warming conditions suggests luxury consumption of Si by diatoms, that only occurs under non-limiting Si levels and that may result in delayed biomass production (Revilla & Weissing, 2008). Si concentration at the beginning of incubation was indeed non-limiting according to different nutrient limitation criteria (Fisher et al., 1988; Justíc et al., 1995). Nutrient enrichment experiments conducted in February 2012 at the same lagoon location also showed that Si was not limiting phytoplankton growth (Domingues et al., 2017a). Therefore, high CO2 and warming probably stimulated luxury consumption of Si by diatoms, but nitrogen limitation prevented the occurrence of warming-induced stimulation of net and in situ instantaneous growth. High CO2 levels probably alleviated the effects of nitrogen limitation, allowing a modest positive increase in net growth rate of diatoms.
Isolated and combined effects of high CO2 and warming on diatoms vary across the literature, ranging from stimulation, no effects, or inhibition of growth rates (Gao & Campbell, 2014; Hoppe et al., 2018). For instance, high CO2 levels may enhance growth of a natural diatom assemblage (Domingues et al., 2014). Stimulatory effects of temperature and CO2, isolated and combined, have been observed for diatom growth (Gao et al., 2012) and for larger-sized diatom species (Wu et al., 2014), although warming may be more influential than CO2, leading to changes in community structure and reduced species richness (Tatters et al., 2013). These variable responses to short-term perturbations indicate that diatom (and phytoplankton) growth is modulated by a complex matrix of environmental variables that interact synergistically and antagonistically. Differences in experimental setups (e.g. cell density, light and temperature levels) and taxon-specific physiology may also lead to differential responses to environmental stressors (Gao & Campbell, 2014).
In contrast to diatoms, net growth rates of cryptophytes decreased under high CO2 levels, and both net growth rates (experiment 1) and potential instantaneous growth rates (experiment 2) were also negatively affected under high temperature. However, the combination of these variables induced a negative antagonistic interaction, where the cumulative effects of the two stressors was positive (Piggott et al., 2015), resulting in the alleviation of the negative effects of temperature. Warming was in fact associated with the lowest net growth rates of cryptophytes, in both experiments. This functional group is indeed associated with colder water temperatures (Mendes et al., 2013; Lv et al., 2014) and is dominant off southern Portuguese coast in the autumn and winter (Goela et al., 2014). In the Ria Formosa, however, maximum abundances of cryptophytes have been observed during summer (Pereira et al., 2007) or spring, and minimum abundances during winter, although their range of seasonal variability is reduced (Barbosa, 2006). Studies addressing the effects of high CO2 and warming on cryptophytes are limited. However, warming was previously reported to have negative effects on cryptophyte growth, when combined with UVR exposure (Halac et al., 2013), even promoting a shift in community composition towards dominance of diatoms and prymnesiophytes, and disappearance of cryptophytes (Vidussi et al., 2011). The reported effects of high CO2 on cryptophytes are variable across the literature. Decreasing pH levels did not affect growth of several species of cryptophytes up to ~ 7 (Berge et al., 2010), and no changes were observed in alloxanthin concentration (specific marker pigment for cryptophytes) with varying CO2 levels in microcosms (Biswas et al., 2015). Furthermore, experiments conducted in the Ria Formosa coastal lagoon during winter (February 2012), with manipulation of UVR and CO2 levels, also showed no significant effects of high CO2, isolated or associated with UVR exposure, on net growth rates of cryptophytes (Domingues et al., 2014). However, increases in the contribution of cryptophytes to total phytoplankton biomass in high CO2 microcosms of natural phytoplankton assemblages (Endo et al., 2016) were also reported.
Other growth-regulating variables, such as nutrients and light, may have played a role limiting cryptophytes growth, but nutrient and light enrichment experiments conducted in the Ria Formosa coastal lagoon at the same time with similar water samples showed that neither nutrients nor light intensity were limiting to cryptophytes (Domingues et al., 2017a). The negative responses of cryptophytes to warming and CO2 could also reflect the beneficial effects of these drivers on grazers, rather than a deleterious direct effect on cryptophyte growth. In addition, microzooplankton grazing pressure on cryptophytes in the Ria Formosa is usually higher than for other phytoplankton groups, particularly diatoms, and the seasonal variability of cryptophytes is also closely associated with grazing by the mixotrophic ciliate Myrionecta rubra (Barbosa, 2006). However, the dilution experiments showed no effects of warming on microzooplankton grazing on cryptophytes.
Net growth rates of cyanobacteria were positively affected by temperature in both experiments, but no significant responses to high CO2 were observed. The latter effect contrast with results derived from an experiment ran two weeks before the present study that reported a reduction in net growth rate of Synechococcus in the Ria Formosa coastal lagoon under increased CO2 levels (Domingues et al., 2014). During summer, growth rate of Synechococcus was also reduced under high CO2 levels; however, nutrient additions (nitrate or ammonium or nitrate + phosphate) counteracted the negative effects of CO2, leading to increased growth of Synechococcus (Domingues et al., 2015). Other studies have reported positive responses of Synechococcus to warming and increased CO2, in terms of cell division and photosynthetic parameters (Fu et al., 2007).
Synechococcus and most cyanobacteria have developed efficient carbon concentrating mechanisms (Rae et al., 2011; Raven & Beardall, 2014), which are downregulated under high CO2 levels; in theory, more energy would become available for other cellular processes, including cell division (Giordano et al., 2005; Raven et al., 2014). Indeed, non-calcifying phytoplankton will probably benefit from high CO2, leading to an increase up to 50% in phytoplankton primary productivity (Schippers et al., 2004). However, in some cases, the responses of cyanobacteria, particularly single-celled forms such as Synechococcus and Prochlorococcus, to high CO2 were modulated by responses to other growth-regulating variables, such as nutrients, light, and temperature (Lomas et al., 2012).
Warming promoted significant increases in cyanobacteria net growth rates in both experiments, indicating that temperature is a major variable regulating cyanobacteria growth. Indeed, the effects of temperature on Synechococcus growth are much more substantial than the effects of CO2 (Fu et al., 2007). Concomitantly, ammonium consumption increased significantly under increased temperature, but not in the high CO2 treatment under in situ temperature, contrasting with nitrate consumption, which responded to neither CO2 nor temperature increases. Cyanobacteria usually prefer ammonium as their N-source (Dokulil & Teubner, 2000; Domingues et al., 2011b), although they can also take up other N-forms, such as nitrate, nitrite, urea, and even atmospheric nitrogen and amino acids (Flores & Herrero, 2005). The simultaneous increase in ammonium consumption and cyanobacteria net growth rates and their preference for ammonium as N-source suggest that higher temperatures stimulate cyanobacteria nutrient uptake and growth. This was also supported by dilution experiments, where cyanobacteria net growth rates under increased temperature were significantly higher than under ambient temperature.
Net growth rates of eukaryotic picophytoplankton (EPP) in dilution experiments were also stimulated by warming. Like cyanobacteria, EPP usually present maximum abundance during summer in the Ria Formosa coastal lagoon, associated with higher temperature (Barbosa, 2006), and in other temperate lagoons (Pequin et al., 2017). Overall, smaller phytoplankton are potential winners under warming conditions, and this reduction in phytoplankton size can lead to profound alterations in carbon transfer to higher trophic levels and to the deep ocean (Maugendre et al., 2015). Ocean acidification, however, may have a detrimental effect on the growth of smaller-sized cells (Richier et al., 2014).
The response of the phytoplankton assemblage (based on Chla) to high CO2 and temperature mirrored the individual responses of cryptophytes, with negative effects of temperature and negative antagonist interactions between the two stressors. Although the abundance of all phytoplankton groups decreased during the 2-day incubation, Chla increased in the control and high CO2 treatment, and declined under high temperature treatments. Cellular chlorophyll content is highly variable, depending on several factors, such as light, nutritional status, and temperature (Behrenfeld et al., 2015). In general, cells subjected to low temperatures usually have higher concentrations of photosynthetic pigments, leading to a more efficient use of light at low temperatures (Valiela, 1995), which can explain the observed decreases in chlorophyll-a concentration under high temperature.
Several studies reported positive correlations between temperature and phytoplankton growth in temperate coastal ecosystems (Montagnes & Franklin, 2001; Collos et al., 2005; Barbosa, 2006; Horn et al., 2020). Temperature raises the substrate-saturated reaction rate of RuBisCO, thus increasing potential phytoplankton growth, providing that growth is not limited by other factors (Beardall & Raven, 2004). However, in both experiments, phytoplankton assemblage, along with cryptophytes and other plastidic nanoflagellates, was negatively affected by warming. Indeed, net growth rates, potential instantaneous growth rates, and in situ instantaneous growth rates were all significantly lower under increased temperature in relation to ambient temperature. In addition, and contrary to our working hypothesis, microzooplankton grazing rates were also lower under increased temperature, so warming did not enhance phytoplankton losses due to herbivory as observed in eutrophic waters (Chen et al., 2012). The exception was for herbivory on pico-sized cells, namely eukaryotic picophytoplankton and cyanobacteria. It is also possible that microzooplankton, mainly composed by nanoflagellates, were feeding on heterotrophic prokaryotes. Overall, results suggest that it is likely that this winter phytoplankton assemblage was well acclimated to winter temperatures and, thus, it was negatively affected by higher temperatures. However, had phytoplankton been given more time to acclimate to a higher temperature, their abundance may have increased, as temperature is the main driver of phytoplankton blooms in shallow productive coastal waters (Trombetta et al., 2019). Despite variable phytoplankton growth responses to high CO2 and temperature and shifts in functional groups, phytoplankton production was not significantly affected by these environmental drivers. The same pattern of changes in growth and community structure and no changes in phytoplankton production were also referred by others (Tortell et al., 2002; Domingues et al., 2014). Reported isolated and combined effects of different drivers on phytoplankton production are indeed contradictory. For instance, warming (Rose et al., 2009) and increased CO2 (Tortell et al., 2008; Song et al., 2014) may lead to increased production, but high CO2 (Feng et al., 2010; Domingues et al., 2014), high temperature, and the combination of both (this study) may also not affect phytoplankton production. In other cases, increases in phytoplankton production due to acidification are only observed at lower temperatures, but this response is constrained by nutrient and light availability (Holding et al., 2015).
Conclusions
Our results showed that short-term variability in different environmental drivers may have varying effects on phytoplankton growth, depending on the functional group. In addition, isolated effects may be counteracted by interactions between those drivers. In general, high temperature and CO2 did not have the beneficial effects on phytoplankton growth that could be expected, suggesting that the winter phytoplankton assemblage from the Ria Formosa coastal lagoon was well acclimated to ambient conditions and even small, short-term changes in specific drivers may cause significant variability in growth and community structure. Our second hypothesis was rejected, given that warming did not increase microzooplankton grazing on phytoplankton, suggesting that microzooplanktonic organisms were also well acclimated to ambient conditions and were apparently not able to increase their metabolic and feeding rates over a short time scale, following rapid increases in temperature. Alternatively, they may have switched to bacterial prey under warmer conditions. In the context of climate change, long-term experiments that allow the development of adaptive responses, in contrast with the plastic responses observed in short-term studies, are the golden standard to evaluate the impacts of future environmental scenarios. However, many climate-related drivers, such as temperature and CO2, also vary on shorter time scales and their study will thus provide a basic understanding of ecological and physiological mechanisms that may further impact phytoplankton acclimation and adaptation to future environmental change.
Data availability
Upon request.
References
Aberle, N., B. Bauer, a. Lewandowska, U. Gaedke & U. Sommer, 2012. Warming induces shifts in microzooplankton phenology and reduces time-lags between phytoplankton and protozoan production. Marine Biology 159: 2441–2453. http://springerlink.bibliotecabuap.elogim.com//10.1007/s00227-012-1947-0.
Aberle, N., K. Lengfellner & U. Sommer, 2007. Spring bloom succession, grazing impact and herbivore selectivity of ciliate communities in response to winter warming. Oecologia 150: 668–681.
Aberle, N., A. Malzahn, A. Lewandowska & U. Sommer, 2015. Some like it hot: the protozooplankton-copepod link in a warming ocean. Marine Ecology Progress Series 519: 103–113. http://www.int-res.com/abstracts/meps/v519/p103-113/.
Agawin, N. S. R., C. M. Duarte & S. Agustí, 2000. Nutrient and temperature control of the contribution of picoplankton to phytoplankton biomass and production. Limnology and Oceanography 45: 591–600.
Albers, C. & D. Lakens, 2018. When power analyses based on pilot data are biased: inaccurate effect size estimators and follow-up bias. Journal of Experimental Social Psychology 74: 187–195.
Andrade, C., M. C. Freitas, J. Moreno & S. C. Craveiro, 2004. Stratigraphical evidence of Late Holocene barrier breaching and extreme storms in lagoonal sediments of Ria Formosa. Marine Geology 210: 339–362.
Bach, L. T. & J. Taucher, 2019. CO2 effects on diatoms: a synthesis of more than a decade of ocean acidification experiments with natural communities. Ocean Science 15: 1159–1175.
Barbosa, A. B., 2006. Estrutura e dinâmica da teia alimentar microbiana na Ria Formosa. PhD Thesis. University of Algarve.
Barbosa, A. B., 2010. Seasonal and interanual variability of planktonic microbes in a mesotidal coastal lagoon (Ria Formosa , SE Portugal). Impact of climatic changes and local human influences In Kennish, M. J. & H. W. Paerl (eds), Coastal Lagoons: Critical Habitats of Environmental Change. CRC Press: 334–366.
Beardall, J. & J. A. Raven, 2004. The potential effects of global climate change on microalgal photosynthesis, growth and ecology. Phycologia The International Phycological Society 43: 26–40. http://www.phycologia.org/doi/abs/10.2216/i0031-8884-43-1-26.1.
Behrenfeld, M. J. & E. S. Boss, 2014. Resurrecting the ecological underpinnings of ocean plankton blooms. Annual review of marine science 6: 167–194. http://www.ncbi.nlm.nih.gov/pubmed/24079309.
Behrenfeld, M. J., R. T. O’Malley, E. S. Boss, T. K. Westberry, J. R. Graff, K. H. Halsey, A. J. Milligan, D. A. Siegel & M. B. Brown, 2015. Revaluating ocean warming impacts on global phytoplankton. Nature Climate Change. http://www.nature.com/doifinder/10.1038/nclimate2838.
Berge, T., N. Daugbjerg, B. Andersen & P. Hansen, 2010. Effect of lowered pH on marine phytoplankton growth rates. Marine Ecology Progress Series 416: 79–91. http://www.int-res.com/abstracts/meps/v416/p79-91/.
Biswas, H., J. Jie, Y. Li, G. Zhang, Z. Y. Zhu, Y. Wu, G. L. Zhang, Y. W. Li, S. M. Liu & J. Zhang, 2015. Response of a natural phytoplankton community from the Qingdao coast (Yellow Sea, China) to variable CO2 levels over a short-term incubation experiment. Current Science 108: 1901–1909.
Boyd, P. W., R. Strzepek, F. Fu & D. A. Hutchins, 2010. Environmental control of open-ocean phytoplankton groups: Now and in the future. Limnology and Oceanography 55: 1353–1376. http://www.aslo.org/lo/toc/vol_55/issue_3/1353.html.
Calbet, A. & E. Saiz, 2013. Effects of trophic cascades in dilution grazing experiments: from artificial saturated feeding responses to positive slopes. Journal of Plankton Research 35: 1183–1191. http://www.plankt.oxfordjournals.org/cgi/doi/10.1093/plankt/fbt067.
Calbet, A. & E. Saiz, 2018. How much is enough for nutrients in microzooplankton dilution grazing experiments?. Journal of Plankton Research 40: 109–117.
Calbet, A., K. Riisgaard, E. Saiz, S. Zamora, S. Stedmon & T. G. Nielsen, 2011a. Phytoplankton growth and microzooplankton grazing along a sub-Arctic fjord (Godthåbsfjord, west Greenland). Marine Ecology Progress Series 442: 11–22.
Calbet, A., E. Saiz, R. Almeda, J. I. Movilla & M. Alcaraz, 2011b. Low microzooplankton grazing rates in the Arctic Ocean during a Phaeocystis pouchetii bloom (Summer 2007): fact or artifact of the dilution technique?. Journal of Plankton Research 33: 687–701.
Caron, D. A. & D. A. Hutchins, 2013. The effects of changing climate on microzooplankton grazing and community structure: drivers, predictions and knowledge gaps. Journal of Plankton Research 35: 235–252. http://www.plankt.oxfordjournals.org/cgi/doi/10.1093/plankt/fbs091.
Chavez, F. P., M. Messié & J. T. Pennington, 2011. Marine primary production in relation to climate variability and change. Annual Review of Marine Science 3: 227–260.
Chen, B., H. Liu, M. R. Landry, M. DaI, B. Huang & J. Sune, 2009. Close coupling between phytoplankton growth and microzooplankton grazing in the western South China Sea. Limnology and Oceanography 54: 1084–1097.
Chen, B., M. R. Landry, B. Huang & H. Liu, 2012. Does warming enhance the effect of microzooplankton grazing on marine phytoplankton in the ocean?. Limnology and Oceanography 57: 519–526. http://www.aslo.org/lo/toc/vol_57/issue_2/0519.html.
Cloern, J. E., P. C. Abreu, J. Carstensen, L. Chauvaud, R. Elmgren, J. Grall, H. Greening, J. O. R. Johansson, M. Kahru, E. T. Sherwood, J. Xu & K. Yin, 2016. Human activities and climate variability drive fast-paced change across the world’s estuarine-coastal ecosystems. Global Change Biology 22: 513–529.
Coello-Camba, A., S. AgustÃ, J. Holding, J. M. Arrieta & C. M. Duarte, 2014. Interactive effect of temperature and CO2 increase in Arctic phytoplankton. Frontiers in Marine Science 1: 1–10, http://journal.frontiersin.org/article/10.3389/fmars.2014.00049/abstract.
Collos, Y., J. Husseini-Ratrema, B. Bec, A. Vaquer, T. L. Hoai, C. Rougier, V. Pons & P. Souchu, 2005. Pheopigment dynamics, zooplankton grazing rates and the autumnal ammonium peak in a Mediterranean lagoon. Hydrobiologia 550: 83–93. http://springerlink.bibliotecabuap.elogim.com/10.1007/s10750-005-4365-1.
Cornwall, C. E., C. D. Hepburn, C. M. Mcgraw, K. I. Currie, C. A. Pilditch, K. A. Hunter, P. W. Boyd & C. L. Hurd, 2013. Diurnal fluctuations in seawater pH influence the response of a calcifying macroalga to ocean acidification. Proceedings of the Royal Society B 280: 20132201. http://rspb.royalsocietypublishing.org.
Cravo, A., S. Cardeira, C. Pereira, M. Rosa, P. Alcântara, M. Madureira, F. Rita, J. Luis & J. Jacob, 2014. Exchanges of nutrients and chlorophyll a through two inlets of Ria Formosa, South of Portugal, during coastal upwelling events. Journal of Sea Research 93: 63–74. http://linkinghub.elsevier.com/retrieve/pii/S1385110114000768.
Cravo, A., A. Rosa, J. Jacob & C. Correia, 2020. Dissolved oxygen dynamics in Ria Formosa Lagoon (South Portugal) – aA real time monitoring station observatory. Marine Chemistry. https://doi.org/10.1016/j.marchem.2020.103806.
Daufresne, M., K. Lengfellner & U. Sommer, 2009. Global warming benefits the small in aquatic ecosystems. Proceedings of the National Academy of Sciences of the United States of America 106: 12788–12793. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2722360&tool=pmcentrez&rendertype=abstract.
De Bodt, C., N. Van Oostende, J. Harlay, K. Sabbe & L. Chou, 2010. Individual and interacting effects of pCO2 and temperature on Emiliania huxleyi calcification: study of the calcite production, the coccolith morphology and the coccosphere size. Biogeosciences 7: 1401–1412.
Dix, N. & M. D. Hanisak, 2015. Microzooplankton grazing experiments in the subtropical Indian River Lagoon, Florida challenge assumptions of the dilution technique. Journal of Experimental Marine Biology and Ecology 465: 1–10. http://linkinghub.elsevier.com/retrieve/pii/S0022098114003256.
Dokulil, M. T. & K. Teubner, 2000. Cyanobacterial dominance in lakes. Hydrobiologia 438: 1–12.
Dolan, J. R., C. L. Gallegos & A. Moigis, 2000. Dilution effects on microzooplankton in dilution grazing experiments. Marine Ecology Progress Series 200: 127–139.
Domingues, R. B., T. P. Anselmo, A. B. Barbosa, U. Sommer & H. M. Galvão, 2011a. Nutrient limitation of phytoplankton growth in the freshwater tidal zone of a turbid, Mediterranean estuary. Estuarine, Coastal and Shelf Science 91: 282–297. http://www.linkinghub.elsevier.com/retrieve/pii/S0272771410003756.
Domingues, R. B., A. B. Barbosa, U. Sommer & H. M. Galvão, 2011b. Ammonium, nitrate and phytoplankton interactions in a freshwater tidal estuarine zone: potential effects of cultural eutrophication. Aquatic Sciences 73: 331–343. http://www.springerlink.bibliotecabuap.elogim.com/10.1007/s00027-011-0180-0.
Domingues, R. B., C. C. Guerra, A. B. Barbosa, V. Brotas & H. M. Galvão, 2014. Effects of ultraviolet radiation and CO2 increase on winter phytoplankton assemblages in a temperate coastal lagoon. Journal of Plankton Research 36: 672–684, http://www.plankt.oxfordjournals.org/cgi/doi/10.1093/plankt/fbt135.
Domingues, R. B., C. C. Guerra, A. B. Barbosa & H. M. Galvão, 2015. Are nutrients and light limiting summer phytoplankton in a temperate coastal lagoon?. Aquatic Ecology 49: 127–146. http://springerlink.bibliotecabuap.elogim.com/10.1007/s10452-015-9512-9.
Domingues, R. B., C. C. Guerra, A. B. Barbosa & H. M. Galvão, 2017a. Will nutrient and light limitation prevent eutrophication in an anthropogenically-impacted coastal lagoon?. Continental Shelf Research 141: 11–25. http://linkinghub.elsevier.com/retrieve/pii/S0278434317300699.
Domingues, R. B., C. C. Guerra, H. M. Galvão, V. Brotas & A. B. Barbosa, 2017b. Short-term interactive effects of ultraviolet radiation, carbon dioxide and nutrient enrichment on phytoplankton in a shallow coastal lagoon. Aquatic Ecology 51: 91–105, http://springerlink.bibliotecabuap.elogim.com/10.1007/s10452-016-9601-4.
Dutkiewicz, S., J. R. Scott & M. J. Follows, 2013. Winners and losers: ecological and biogeochemical changes in a warming ocean. Global Biogeochemical Cycles 27: 463–477.
Dutkiewicz, S., J. J. Morris, M. J. Follows, J. Scott, O. Levitan, S. T. Dyhrman & I. Berman-Frank, 2015. Impact of ocean acidification on the structure of future phytoplankton communities. Nature Climate Change. https://doi.org/10.1038/nclimate2722.
Eilers, P. H. C. & J. C. H. Peeters, 1988. A model for the relationship between light and intensity and the rate of photosynthesis in phytoplankton. Ecological Modelling 42: 199–215.
Endo, H., K. Sugie, T. Yoshimura & K. Suzuki, 2016. Response of spring diatoms to CO2 availability in the western North Pacific as determined by next-generation sequencing. PLoS ONE 11: 1–22.
Eppley, R. W., 1972. Temperature and phytoplankton growth in the sea. Fishery Bulletin 70: 1063–1085.
Eriander, L., A.-L. Wrange & J. N. Havenhand, 2016. Simulated diurnal pH fluctuations radically increase variance in – but not the mean of – growth in the barnacle Balanus improvisus. ICES Journal of Marine Science 73: 596–603.
Feng, Y., C. E. Hare, K. Leblanc, J. M. Rose, Y. Zhang, G. R. DiTullio, P. A. Lee, S. W. Wilhelm, J. M. Rowe, J. Sun, N. Nemcek, C. Gueguen, U. Passow, I. Benner, C. Brown & D. A. Hutchins, 2009. Effects of increased pCO2 and temperature on the north atlantic spring bloom. I. The phytoplankton community and biogeochemical response. Marine Ecology Progress Series 388: 13–25.
Feng, Y., C. E. Hare, J. M. Rose, S. M. Handy, G. R. DiTullio, P. A. Lee, W. O. Smith, J. Peloquin, S. Tozzi, J. Sun, Y. Zhang, R. B. Dunbar, M. C. Long, B. Sohst, M. Lohan & D. A. Hutchins, 2010. Interactive effects of iron, irradiance and CO2 on Ross Sea phytoplankton. Deep-Sea Research Part I: Oceanographic Research Papers 57: 368–383.
Fisher, T. R., L. W. Harding, D. W. Stanley & L. G. Ward, 1988. Phytoplankton, nutrients, and turbidity in the Chesapeake, Delaware, and Hudson estuaries. Estuarine, Coastal and Shelf Science 27: 61–93.
Flores, E. & A. Herrero, 2005. Nitrogen assimilation and nitrogen control in cyanobacteria. Biochemical Society transactions 33: 164–167. http://www.ncbi.nlm.nih.gov/pubmed/15667295.
Fu, F.-X., M. E. Warner, Y. Zhang, Y. Feng & D. A. Hutchins, 2007. Effects of Increased Temperature and CO2 on Photosynthesis, Growth, and Elemental Ratios in Marine Synechococcus and Prochlorococcus (Cyanobacteria). Journal of Phycology 43: 485–496. http://doi.wiley.com/10.1111/j.1529-8817.2007.00355.x.
Fu, F., A. Tatters & D. Hutchins, 2012. Global change and the future of harmful algal blooms in the ocean. Marine Ecology Progress Series 470: 207–233. http://www.int-res.com/abstracts/meps/v470/p207-233/.
Fu, F. X., Y. Zhang, M. E. Warner, Y. Feng, J. Sun & D. A. Hutchins, 2008. A comparison of future increased CO2 and temperature effects on sympatric Heterosigma akashiwo and Prorocentrum minimum. Harmful Algae 7: 76–90.
Gallegos, C., 1989. Microzooplankton grazing on phytoplankton in Rhode River, Maryland: noninear feeding kinetics. Marine Ecology Progress Series 57: 23–33.
Gao, K. & D. A. Campbell, 2014. Photophysiological responses of marine diatoms to elevated CO2 and decreased pH: a review. Functional Plant Biology 41: 449–459.
Gao, K., E. Helbling, D.-P. Häder & D. Hutchins, 2012. Responses of marine primary producers to interactions between ocean acidification, solar radiation, and warming. Marine Ecology Progress Series 470: 167–189. http://www.int-res.com/abstracts/meps/v470/p167-189/.
Gattuso, J.-P., K. Gao, K. Lee, B. Rost & K. G. Schulz, 2010. Approaches and tools to manipulate the carbonate chemistry In Riebesell, U., V. J. Fabry, L. Hansson & J.-P. Gattuso (eds), Guide to Best Practices for Ocean Acidification Research and Data Reporting. Publications Office of the European Union: 41–52.
Giordano, M., J. Beardall & J. a Raven, 2005. CO2 concentrating mechanisms in algae: mechanisms, environmental modulation, and evolution. Annual Review of Plant Biology 56: 99–131. http://www.ncbi.nlm.nih.gov/pubmed/15862091.
Goela, P. C., S. Danchenko, J. D. Icely, L. M. Lubian, S. Cristina & A. Newton, 2014. Using CHEMTAX to evaluate seasonal and interannual dynamics of the phytoplankton community off the South-west coast of Portugal. Estuarine, Coastal and Shelf Science 151: 112–123. http://apps.webofknowledge.com/full_record.do?product=WOS&search_mode=GeneralSearch&qid=12&SID=W1nP4i9eaabP6SGhOzF&page=1&doc=10&cacheurlFromRightClick=no.
Grasshoff, K., M. Ehrhardt & K. Kremling, 1999. Methods of Seawater Analysis. WILEY‐VCH Verlag GmbH, Weinheim. https://onlinelibrary.wiley.com/doi/book/10.1002/9783527613984.
Grinienė, E., S. Šulčius & H. Kuosa, 2016. Size-selective microzooplankton grazing on the phytoplankton in the Curonian Lagoon (SE Baltic Sea). Oceanologia 58: 292–301.
Guiry, M. D. & G. M. Guiry, 2021. AlgaeBase. World-wide electronic publication. National University of Ireland, Galway.
Haas, L. W., 1982. Improved epifluorescence microscopy for observing planktonic micro-organisms. Annales de l’Institut Oceanographique 58: 261–266.
Halac, S. R., S. D. Guendulain-García, V. E. Villafañe, E. W. Helbling & A. T. Banaszak, 2013. Responses of tropical plankton communities from the Mexican Caribbean to solar ultraviolet radiation exposure and increased temperature. Journal of Experimental Marine Biology and Ecology 445: 99–107. http://linkinghub.elsevier.com/retrieve/pii/S0022098113001652.
Hare, C. E., K. Leblanc, G. R. DiTullio, R. M. Kudela, Y. Zhang, P. A. Lee, S. Riseman & D. A. Hutchins, 2007. Consequences of increased temperature and CO2 for phytoplankton community structure in the Bering Sea. Marine Ecology Progress Series 352: 9–16. http://www.int-res.com/abstracts/meps/v352/p9-16/.
Henson, S. A., C. Beaulieu & R. Lampitt, 2016. Observing climate change trends in ocean biogeochemistry: when and where. Global Change Biology 22: 1561–1571.
Henson, S. A., C. Beaulieu, T. Ilyina, J. G. John, M. Long, R. Séférian, J. Tjiputra & J. L. Sarmiento, 2017. Rapid emergence of climate change in environmental drivers of marine ecosystems. Nature Communications 8: 1–9.
Holding, J. M., C. M. Duarte, M. Sanz-Martín, E. Mesa, J. M. Arrieta, M. Chierici, I. E. Hendriks, L. S. García-Corral, A. Regaudie-De-Gioux, A. Delgado, M. Reigstad, P. Wassmann & S. Agustí, 2015. Temperature dependence of CO2-enhanced primary production in the European Arctic Ocean. Nature Climate Change 5: 1079–1082.
Hoppe, C. J. M., K. K. E. Wolf, N. Schuback, P. D. Tortell & B. Rost, 2018. Compensation of ocean acidification effects in Arctic phytoplankton assemblages. Nature Climate Change 8: 529–533.
Horn, H. G., M. Boersma, J. Garzke, U. Sommer & N. Aberle, 2020. High CO2 and warming affect microzooplankton food web dynamics in a Baltic Sea summer plankton community. Marine Biology 167: 1–17.
IPCC, 2014. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. IPCC, Geneva.
Jiang, S., F. Hashihama & H. Saito, 2021. Phytoplankton growth and grazing mortality through the oligotrophic subtropical North Pacific. Journal of Oceanography 77: 505–521.
Justíc, D., N. N. Rabalais, R. E. Turner & Q. Dortch, 1995. Changes in nutrient structure of river dominated coastal waters: stoichiometric nutrient balance and its consequences. Estuarine, Coastal and Shelf Science 40: 339–356.
Keys, M., G. Tilstone, H. S. Findlay, C. E. Widdicombe & T. Lawson, 2018. Effects of elevated CO2 and temperature on phytoplankton community biomass, species composition and photosynthesis during an experimentally induced autumn bloom in the western English Channel. Biogeosciences 15: 3203–3222. https://bg.copernicus.org/articles/15/3203/2018/.
Kroeker, K. J., R. L. Kordas, R. Crim, I. E. Hendriks, L. Ramajo, G. S. Singh, C. M. Duarte & J.-P. Gattuso, 2013. Impacts of ocean acidification on marine organisms: quantifying sensitivities and interaction with warming. Global Change Biology. http://doi.wiley.com/10.1111/gcb.12179.
Landry, M. R. & R. P. Hassett, 1982. Estimating the grazing impact of marine micro-zooplankton. Marine Biology 67: 283–288. http://springerlink.bibliotecabuap.elogim.com/10.1007/BF00397668.
Lewandowska, A. M., D. G. Boyce, M. Hofmann, B. Matthiessen, U. Sommer & B. Worm, 2014. Effects of sea surface warming on marine plankton. Ecology Letters . http://www.ncbi.nlm.nih.gov/pubmed/24575918.
Liu, X., Y. Li, Y. Wu, B. Huang, M. Dai, F. Fu, D. A. Hutchins & K. Gao, 2017. Effects of elevated CO2 on phytoplankton during a mesocosm experiment in the southern eutrophicated coastal water of China. Scientific Reports 7: 1–14.
Lomas, M., B. Hopkinson, J. Losh, D. Ryan, D. Shi, Y. Xu & F. Morel, 2012. Effect of ocean acidification on cyanobacteria in the subtropical North Atlantic. Aquatic Microbial Ecology 66: 211–222.
López-Urrutia, A., E. S. Martin, R. P. Harris & X. Irigoien, 2006. Scaling the metabolic balance of the oceans. Proceedings of the National Academy of Sciences of the United States of America 103: 8739–8744. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1482648&tool=pmcentrez&rendertype=abstract.
Lv, H., J. Yang, L. Liu, X. Yu, Z. Yu & P. Chiang, 2014. Temperature and nutrients are significant drivers of seasonal shift in phytoplankton community from a drinking water reservoir, subtropical China. Environmental Science and Pollution Research International 21: 5917–5928. http://apps.webofknowledge.com/full_record.do?product=UA&search_mode=GeneralSearch&qid=3&SID=X1PxQbuYNMjPY5PkCIh&page=1&doc=3&cacheurlFromRightClick=no.
Machado, L. M., 2010. A radiação UV-B na Ria Formosa: incidência e impactes biológicos. University of Algarve.
Maugendre, L., J.-P. Gattuso, J. Louis, A. de Kluijver, S. Marro, K. Soetaert & F. Gazeau, 2015. Effect of ocean warming and acidification on a plankton community in the NW Mediterranean Sea. ICES Journal of Marine Science 72: 1744–1755.
Menden-Deuer, S., C. Lawrence & G. Franzè, 2018. Herbivorous protist growth and grazing rates at in situ and artificially elevated temperatures during an Arctic phytoplankton spring bloom. PeerJ 6: e5264.
Mendes, C. R. B., V. M. Tavano, M. C. Leal, M. S. Souza, V. Brotas & C. A. E. Garcia, 2013. Shifts in the dominance between diatoms and cryptophytes during three late summers in the Bransfield Strait (Antarctic Peninsula). Polar Biology 36: 537–547. http://springerlink.bibliotecabuap.elogim.com/10.1007/s00300-012-1282-4.
Meyer, J. & U. Riebesell, 2015. Reviews and syntheses: Responses of coccolithophores to ocean acidification: A meta-analysis. Biogeosciences 12: 1671–1682.
Montagnes, D. J. S. & D. J. Franklin, 2001. Effect of temperature on diatom volume, growth rate, and carbon and nitrogen content: reconsidering some paradigms. Limnology and Oceanography 46: 2008–2018. http://www.aslo.org/lo/toc/vol_46/issue_8/2008.html.
Morán, X. A. G., Á. López-Urrutia, A. Calvo-Díaz & W. K. W. Li, 2010a. Increasing importance of small phytoplankton in a warmer ocean. Global Change Biology 16: 1137–1144.
Morán, X. A. G., Á. López-Urrutia, A. Calvo-Díaz & W. K. W. Li, 2010b. Increasing importance of small phytoplankton in a warmer ocean. Global Change Biology 16: 1137–1144.
Morison, F. & S. Menden-Deuer, 2017. Doing more with less? Balancing sampling resolution and effort in measurements of protistan growth and grazing-rates. Limnology and Oceanography: Methods 15: 794–809.
Nejstgaard, J., I. Gismervik & P. Solberg, 1997. Feeding and reproduction by Calanus finmarchicus, and microzooplankton grazing during mesocosm blooms of diatoms and the coccolithophore Emiliania huxleyi. Marine Ecology Progress Series 147: 197–217.
Newton, A. & S. M. Mudge, 2003. Temperature and salinity regimes in a shallow, mesotidal lagoon, the Ria Formosa, Portugal. Estuarine, Coastal and Shelf Science 57: 73–85. http://linkinghub.elsevier.com/retrieve/pii/S0272771402003323.
Newton, A., J. D. Icely, M. Falcao, A. Nobre, J. P. Nunes, J. G. Ferreira & C. Vale, 2003. Evaluation of eutrophication in the Ria Formosa coastal lagoon, Portugal. Continental Shelf Research 23: 1945–1961. http://linkinghub.elsevier.com/retrieve/pii/S0278434303001584.
Nielsen, L., G. Hallegraeff, S. Wright & P. Hansen, 2012. Effects of experimental seawater acidification on an estuarine plankton community. Aquatic Microbial Ecology 65: 271–286. http://www.int-res.com/abstracts/ame/v65/n3/p271-286/.
Nogueira, P., R. B. Domingues & A. B. Barbosa, 2014. Are microcosm volume and sample pre-filtration relevant to evaluate phytoplankton growth?. Journal of Experimental Marine Biology and Ecology 461: 323–330. http://linkinghub.elsevier.com/retrieve/pii/S0022098114002494.
O’Connor, M. I., M. F. Piehler, D. M. Leech, A. Anton & J. F. Bruno, 2009. Warming and resource availability shift food web structure and metabolism. PLoS Biology 7: e1000178. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2723928&tool=pmcentrez&rendertype=abstract.
Olejnik, S. & J. Algina, 2003. Generalized eta and omega squared statistics: measures of effect size for some common research designs. Psychological Methods 8: 434–447.
Parsons, T. R., Y. Maita & C. M. Lalli, 1984. A Manual of Chemical and Biological Methods for Seawater Analysis. Pergamon Press, Oxford.
Pequin, B., V. Mohit, T. Poisot, R. Tremblay & C. Lovejoy, 2017. Wind drives microbial eukaryote communities in a temperate closed lagoon. Aquatic Microbial Ecology 78: 187–200.
Pereira, M. G., J. Icely, S. Mudge, A. Newton & R. Rodrigues, 2007. Temporal and spatial variation of phytoplankton pigments in the Western Part of Ria Formosa Lagoon, Southern Portugal. Environmental Forensics 8: 205–220.
Piggott, J. J., C. R. Townsend & C. D. Matthaei, 2015. Reconceptualizing synergism and antagonism among multiple stressors. Ecology and Evolution 5: 1538–1547.
Platt, T. & S. Sathyendranath, 2008. Ecological indicators for the pelagic zone of the ocean from remote sensing. Remote Sensing of Environment 112: 3426–3436. http://www.sciencedirect.com/science/article/pii/S0034425708001272.
R Core Team, 2020. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna. http://www.r-project.org/.
Racault, M.-F., C. Le Quéré, E. Buitenhuis, S. Sathyendranath & T. Platt, 2012. Phytoplankton phenology in the global ocean. Ecological Indicators 14: 152–163. http://linkinghub.elsevier.com/retrieve/pii/S1470160X11002160.
Rae, B. D., B. Forster, M. R. Badger & G. D. Price, 2011. The CO2-concentrating mechanism of Synechococcus WH5701 is composed of native and horizontally-acquired components. Photosynthesis Research 109: 59–72.
Raven, J. A. & J. Beardall, 2014. CO2 concentrating mechanisms and environmental change. Aquatic Botany 118: 24–37. http://linkinghub.elsevier.com/retrieve/pii/S0304377014000734.
Raven, J. A., J. Beardall & M. Giordano, 2014. Energy costs of carbon dioxide concentrating mechanisms in aquatic organisms. Photosynthesis Research 121: 111–124.
Revilla, T. & F. J. Weissing, 2008. Nonequilibrium coexistence in a competition model with nutrient storage. Ecology 89: 865–877.
Richier, S., E. P. Achterberg, C. Dumousseaud, A. J. Poulton, D. J. Suggett, T. Tyrrell, M. V. Zubkov & C. M. Moore, 2014. Phytoplankton responses and associated carbon cycling during shipboard carbonate chemistry manipulation experiments conducted around Northwest European shelf seas. Biogeosciences 11: 4733–4752.
Riebesell, U., 2004. Effects of CO2 Enrichment on Marine Phytoplankton. Journal of Oceanography 60: 719–729. http://springerlink.bibliotecabuap.elogim.com/10.1007/s10872-004-5764-z.
Rose, J. M. & D. A. Caron, 2007. Does low temperature constrain the growth rates of heterotrophic protists? Evidence and implications for algal blooms in cold waters. Limnology and Oceanography 52: 886–895. http://www.aslo.org/lo/toc/vol_52/issue_2/0886.html.
Rose, J. M., Y. Feng, C. Gobler, R. Gutierrez, C. Hare, K. Leblanc & D. Hutchins, 2009. Effects of increased pCO2 and temperature on the North Atlantic spring bloom. II. Microzooplankton abundance and grazing. Marine Ecology Progress Series 388: 27–40. http://www.int-res.com/abstracts/meps/v388/p27-40/.
Santos, F. D., K. Forbes & R. Moita, 2002. Climate Change in Portugal. Scenarios, impacts and adaptation measures. Gradiva.
Schippers, P., M. Lurling & M. Scheffer, 2004. Increase of atmospheric CO2 promotes phytoplankton productivity. Ecology Letters 7: 446–451. http://www.blackwell-synergy.com/links/doi/10.1111%2Fj.1461-0248.2004.00597.x.
Schmidt, K., A. J. Birchill, A. Atkinson, R. J. W. Brewin, J. R. Clark, A. E. Hickman, D. G. Johns, M. C. Lohan, A. Milne, S. Pardo, L. Polimene, T. J. Smyth, G. A. Tarran, C. E. Widdicombe, E. M. S. Woodward & S. J. Ussher, 2020. Increasing picocyanobacteria success in shelf waters contributes to long-term food web degradation. Global Change Biology 26: 5574–5587.
Schmoker, C., S. Hernandez-Leon & A. Calbet, 2013. Microzooplankton grazing in the oceans: impacts, data variability, knowledge gaps and future directions. Journal of Plankton Research 35: 691–706. http://www.plankt.oxfordjournals.org/cgi/doi/10.1093/plankt/fbt023.
Seifert, M., R. Bjorn, S. Trimborn & J. Hauck, 2020. Meta-analysis of multiple driver effects on marine phytoplankton highlights modulating role of p CO2. Global Change Biology 1–62.
Sherman, E., J. K. Moore, F. Primeau & D. Tanouye, 2016. Temperature influence on phytoplankton community growth rates. Global Biogeochemical Cycles 30: 550–559.
Sobrino, C., M. L. Ward & P. J. Neale, 2008. Acclimation to elevated carbon dioxide and ultraviolet radiation in the diatom Thalassiosira pseudonana: effects on growth, photosynthesis, and spectral sensitivity of photoinhibition. Limnology and Oceanography 53: 494–505.
Sommer, U., N. Aberle, K. Lengfellner & A. Lewandowska, 2012. The Baltic Sea spring phytoplankton bloom in a changing climate: an experimental approach. Marine Biology 159: 2479–2490. http://springerlink.bibliotecabuap.elogim.com/10.1007/s00227-012-1897-6.
Sommer, U., C. Paul & M. Moustaka-Gouni, 2015. Warming and ocean acidification effects on phytoplankton - From species shifts to size shifts within species in a mesocosm experiment. PLoS ONE 10: 1–17.
Song, C., F. Ballantyne IV & V. H. Smith, 2014. Enhanced dissolved organic carbon production in aquatic ecosystems in response to elevated atmospheric CO2. Biogeochemistry 118: 49–60.
Steeman-Nielsen, E., 1952. The use of radio-active carbon (C14) for measuring organic production in the sea. Journal du Counseil International pour l’Exploration de la Mer 18: 117–140.
Stramma, L., P. Cornillon, R. A. Weller, J. F. Price & M. G. Briscoe, 1986. Large diurnal sea surface temperature variability: satellite and in situ measurements. Journal of Physical Oceanography 56: 345–358.
Strom, S. L., M. A. Brainard, J. L. Holmes & M. B. Olson, 2001. Phytoplankton blooms are strongly impacted by microzooplankton grazing in coastal North Pacific waters. Marine Biology 138: 355–368.
Strom, S. L., E. L. Macri & M. B. Olson, 2007. Microzooplankton grazing in the coastal Gulf of Alaska: Variations in top-down control of phytoplankton. Limnology and Oceanography 52: 1480–1494.
Stuart-Menteth, A. C., I. S. Robinson & P. G. Challenor, 2003. A global study of diurnal warming using satellite-derived sea surface temperature. Journal of Geophysical Research 108: 3155. http://onlinelibrary.wiley.com/doi/10.1029/2002JC001534/full.
Suzuki, K., A. Tsuda, H. Kiyosawa, S. Takeda, J. Nishioka, T. Saino, M. Takahashi & C. S. Wong, 2002. Grazing impact of microzooplankton on a diatom bloom in a mesocosm as estimated by pigment-specific dilution technique. Journal of Experimental Marine Biology and Ecology 271: 99–120.
Tatters, A. O., M. Y. Roleda, A. Schnetzer, F. Fu, C. L. Hurd, P. W. Boyd, D. A. Caron, A. A. Y. Lie, L. J. Hoffmann & D. A. Hutchins, 2013. Short- and long-term conditioning of a temperate marine diatom community to acidification and warming. Philosophical transactions of the Royal Society of London. Series B, Biological sciences 368: 20120437. http://www.ncbi.nlm.nih.gov/pubmed/23980240.
Teixeira, I. G. & F. G. Figueiras, 2009. Feeding behaviour and non-linear responses in dilution experiments in a coastal upwelling system. Aquatic Microbial Ecology 55: 53–63.
Teixeira, I. G., F. G. Figueiras, B. G. Crespo & S. Piedracoba, 2011. Microzooplankton feeding impact in a coastal upwelling system on the NW Iberian margin: the Ría de Vigo. Estuarine, Coastal and Shelf Science 91: 110–120.
Teixeira, I. G., B. G. Crespo, T. G. Nielsen & F. G. Figueiras, 2012. Role of microzooplankton during a Phaeocystis sp. bloom in the Oosterschelde (SW Netherlands). Journal of Marine Systems 94: 97–106.
Teixeira, I. G., B. G. Crespo, T. G. Nielsen & F. G. Figueiras, 2014. Stratification-mixing cycles and plankton dynamics in a shallow estuary (Limfjord, Denmark). Journal of Plankton Research 36: 475–489.
Torstensson, A., M. Chierici & A. Wulff, 2012. The influence of increased temperature and carbon dioxide levels on the benthic/sea ice diatom Navicula directa. Polar Biology 35: 205–214.
Tortell, P. D., G. R. DiTullio, D. M. Sigman & F. M. M. Morel, 2002. CO2 effects on taxonomic composition and nutrient utilization in an Equatorial Pacific phytoplankton assemblage. Marine Ecology Progress Series 236: 37–43.
Tortell, P. D., C. D. Payne, Y. Li, S. Trimborn, B. Rost, W. O. Smith, C. Riesselman, R. B. Dunbar, P. Sedwick & G. R. DiTullio, 2008. CO2 sensitivity of Southern Ocean phytoplankton. Geophysical Research Letters 35: 1–5.
Trombetta, T., F. Vidussi, S. Mas, D. Parin, M. Simier & B. Mostajir, 2019. Water temperature drives phytoplankton blooms in coastal waters. PLoS ONE 14.
Tsai, A. Y., G.-C. Gong & W. Shiau, 2015. Viral lysis and nanoflagellate grazing on prokaryotes: effects of short-term warming in a coastal subtropical marine system. Hydrobiologia 751: 43–54. http://springerlink.bibliotecabuap.elogim.com/10.1007/s10750-014-2170-4.
Tsai, A., G. Gong & W. Shiau, 2016. Impact of short-term warming on seasonal variations in bacterial growth, grazing, and viral lysis in coastal waters of Taiwan. Aquatic Microbial Ecology 76: 195–205. http://www.int-res.com/abstracts/ame/v76/n3/p195-205/.
Twiss, M. R. & D. E. Smith, 2012. Size-fractionated phytoplankton growth and microzooplankton grazing rates in the upper St. Lawrence River. River Research and Applications 28: 1047–1053.
Utermöhl, H., 1958. Zur Vervollkommnung der quantitativen Phytoplankton-Methodik. Mitt. Internationale Ver. Theoretische und Angewandte Limnologie 9: 1–38.
Valiela, I., 1995. Marine Ecological Processes. Springer, New York.
Venrick, E. L., 1978. How many cells to count? In Sournia, A. (ed), Phytoplankton Manual. UNESCO, Paris: 167–180.
Vidussi, F., B. Mostajir, E. Fouilland, E. Le Floc’h, J. Nouguier, C. Roques, P. Got, D. Thibault-Botha, T. Bouvier & M. Troussellier, 2011. Effects of experimental warming and increased ultraviolet B radiation on the Mediterranean plankton food web. Limnology and Oceanography 56: 206–218. http://www.aslo.org/lo/toc/vol_56/issue_1/0206.html.
Williams, O. J., R. E. Beckett & D. L. Maxwell, 2016. Marine phytoplankton preservation with Lugol’s: a comparison of solutions. Journal of Applied Phycology Journal of Applied Phycology 28: 1705–1712.
Worden, A. & B. Binder, 2003. Application of dilution experiments for measuring growth and mortality rates among Prochlorococcus and Synechococcus populations in oligotrophic environments. Aquatic Microbial Ecology 30: 159–174.
Wu, Y., D. A. Campbell, A. J. Irwin, D. Suggett & Z. Finkel, 2014. Ocean acidification enhances the growth rate of larger diatoms. Limnology and Oceanography 59: 1027–1034. http://www.aslo.org/lo/toc/vol_59/issue_3/1027.html.
Funding
This work was financially supported by the Portuguese Foundation for Science and Technology (FCT) through projects PTDC/AAC-CLI/103348/2008 and UIDP/00350/2020. FCT provided funding for RBD through a postdoctoral fellowship and a researcher contract (SFRH/BPD/68688/2010, DL57/2016/CP1361/CT0017).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling editor: Daniele Nizzoli.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Domingues, R.B., Barreto, M., Brotas, V. et al. Short-term effects of winter warming and acidification on phytoplankton growth and mortality: more losers than winners in a temperate coastal lagoon. Hydrobiologia 848, 4763–4785 (2021). https://doi.org/10.1007/s10750-021-04672-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-021-04672-0